Abstract
Introduction:
Children and adolescents with type 1 diabetes (T1D) often experience glucose fluctuations during dynamic activities such as summer camps. Evidence on the clinical accuracy and usability of intermittently scanned CGM (isCGM; FreeStyle Libre 2) in these real-world pediatric settings is limited.
Aims:
To evaluate within-participant changes in continuous glucose monitoring (CGM)–derived glycemic metrics during a pediatric T1D summer camp compared with a pre-camp period under health care professional (HCP)–supervised use of FreeStyle Libre 2, an isCGM system. Secondary aims were to assess sensor accuracy and user satisfaction.
Methods:
This prospective single-arm observational study enrolled children and adolescents with T1D attending a 3-day residential summer camp. CGM-derived metrics during the on-camp period were compared with those from a pre-camp period within the same participants under structured HCP supervision. Sensor accuracy was assessed using paired capillary blood glucose–sensor glucose measurements, and user satisfaction was evaluated using post-camp questionnaires.
Results:
Twenty-one participants were analyzed. The on-camp period showed a significant increase in time in range (54.5%–64.3%, P = 0.031) and a reduction in time above range > 250 mg/dL (17.7%–4.7%, P = 0.049), while time below range remained unchanged. Sensor accuracy, evaluated by mean absolute relative difference, was acceptable within ranges (13.6% ± 10.5% on-camp, 12.7% ± 10.5% pre-camp) reported in pediatric studies with variability.
Conclusions:
Glycemic metrics were improved during a pediatric T1D summer camp conducted under a structured, HCP-supervised setting incorporating isCGM, without an increase in hypoglycemia. These findings support the feasibility of supervised, camp-based CGM–supported glucose management in pediatric populations.
Keywords

Introduction
Type 1 diabetes (T1D) management in children and adolescents requires continuous attention to glycemic management while minimizing the risk of hypoglycemia, particularly in settings characterized by increased physical activity and irregular daily routines (e.g., International Society for Pediatric and Adolescent Diabetes (ISPAD) guidelines on glycemic targets). 1 Pediatric diabetes summer camps provide a unique educational and clinical environment in which participants engage in structured activities under medical supervision, offering opportunities to support both safe glucose management and diabetes self-care education.
Continuous glucose monitoring (CGM) has become an integral component of pediatric diabetes care. International consensus guidelines emphasize CGM-derived metrics, including time in range (TIR), time above range (TAR), and time below range (TBR), as clinically meaningful indicators of glycemic management beyond glycated hemoglobin alone.1,2 Previous studies have demonstrated that CGM use in pediatric camp settings, particularly with real-time CGM systems, can be associated with improvements in glycemic outcomes on-camp participation.1,2 However, evidence remains limited regarding the performance of intermittently scanned CGM (isCGM) systems in such structured, short-term educational environments.3,4
FreeStyle Libre 2 is a widely used isCGM system that provides factory-calibrated glucose measurements and optional glucose alarms, offering potential advantages in usability and feasibility in pediatric populations. While its accuracy and acceptability have been reported in outpatient settings, data on its use during pediatric diabetes camps—where intensive supervision, frequent glucose review, and environmental factors coexist—are scarce.4,5 Moreover, few studies have examined within-participant changes in CGM-derived glycemic metrics on-camp participation using isCGM systems.
Therefore, the primary aim of this prospective observational study was to evaluate within-participant changes in CGM-derived glycemic metrics, including TIR, TAR, and TBR, during a pediatric T1D summer camp compared with a pre-camp period under health care professional (HCP)–supervised use of FreeStyle Libre 2. Secondary aims included the assessment of sensor accuracy and user satisfaction, as well as exploratory historical comparisons with previous camps using other CGM systems.
Methods
Study population
Baseline Characteristics of Study Participants (Campers; n = 21)

BMI, body mass index; IQR, interquartile range; isCGM, intermittently scanned continuous glucose monitoring; rtCGM, real time continuous glucose monitoring; SDS, standard deviation score; SMBG, self-monitoring of blood glucose.
Study design
This prospective single-arm observational study was conducted during the 55th Higokko Smile Summer Camp in Misato Town, Kumamoto, Japan (August 16–18, 2025). This research was approved by the Ethics Review Committee at Aso Medical Center (Ethics No. 2025-002). The study was conducted in accordance with the principles of the Declaration of Helsinki. Before enrollment, campers and their parents/legal representatives received both oral and written information about the study. Written informed consent was obtained from parents/legal representatives, and assent was received from all campers.
At the end of the camp, the campers and HCPs completed a tailored questionnaire rating (5-point Likert) the satisfaction and usability of the isCGM (Supplementary Fig. S1), partly adopted from a previous report. 6
The isCGM, Libre 2 system is capable of operating as an rtCGM when connected to a smartphone; however, in the present camp, all participants were supervised through centralized monitor-based management, and the device was intentionally used in isCGM mode. Libre 2 sensors were applied 6 days before the camp, providing interstitial glucose data every minute. Capillary blood glucose (BG) was simultaneously measured using Medisafe-FIT (Terumo Corporation, Tokyo, Japan) for calibration-free accuracy comparison. CGM-derived metrics included TAR level 2 (>250 mg/dL), TAR level 1 (181–250 mg/dL), TIR (70–180 mg/dL), time in tight range (TITR; 70–140 mg/dL), TBR level 1 (54–69 mg/dL), TBR level 2 (<54 mg/dL), % coefficient of variation (CV), glucose management indicator (GMI), and mean absolute relative difference (MARD).
Paired BG–sensor glucose (SG) readings were analyzed using regression, Bland–Altman, and Parkes and Clarke Error Grid analyses.
Structured HCP supervision and camp procedures
HCP-supervised glucose management
During the camp, glucose management was conducted under structured supervision by HCPs, including experienced diabetologists, specialized diabetes nurses, pharmacists, clinical laboratory technologists, and nationally registered dietitians. Compared with usual outpatient care, HCP involvement during the camp was more frequent and proactive. CGM data were reviewed at least four times daily (before breakfast, before lunch, before dinner, and at bedtime), as well as ad hoc in response to alarms or abnormal glucose trends.
Insulin dose adjustments were guided by predefined targets and standardized principles. The target glucose range was set at 70–180 mg/dL, with particular attention to the prevention of hypoglycemia. Correction boluses for hyperglycemia were administered according to each participant’s preexisting insulin sensitivity factor, while hypoglycemia was treated using standardized carbohydrate supplementation (typically 5–15 g of fast-acting carbohydrates, depending on age and body size). Capillary blood glucose measurements were additionally obtained 30 min after correction for hyperglycemia or hypoglycemia to confirm recovery and sensor accuracy.
Carbohydrate counting was performed using standardized meal plans, and HCPs supported participants in estimating carbohydrate intake and selecting appropriate insulin doses. Insulin regimens (multiple daily injections or pump therapy) were not changed during the camp.
Use of FreeStyle Libre 2 pre- and on-camp
Before the camp, participants used FreeStyle Libre 2 in their usual home environment, with glucose data primarily reviewed by the participants and their families during routine daily care. HCP review of CGM data pre-camp was limited to regular outpatient visits. The pre-camp period was selected as a pragmatic comparator reflecting usual daily life under routine care. We further acknowledge that alternative designs, such as supervised run-in periods or parallel comparator groups, would offer stronger internal validity but were not feasible in this real-world camp setting.
During the camp, FreeStyle Libre 2 was used continuously with frequent scanning by both participants and HCPs. Glucose trend arrows and optional alarms were actively utilized to anticipate glycemic excursions and guide timely interventions. CGM data were centrally reviewed by HCPs to support real-time decision-making on-camp, in contrast to the predominantly self-managed use in pre-camp.
Camp schedule and daily activities
The camp followed a standardized daily schedule to minimize variability in external factors. Meals were provided at fixed times (approximately 08:00, 12:00, and 18:00), with planned snacks as needed to prevent hypoglycemia. Physical activity was scheduled in defined blocks, including morning recreational activities, afternoon outdoor or sports activities, and evening group programs, including “Carbohydrate Counting Lecture,” “How to Use Glucagon Intra-nasally,” and “You’re the Nurse Today—Take Care of Your Patient!”, with activity intensity monitored by HCPs.
Glucose levels were checked before and after major physical activity sessions, and preventive carbohydrate intake or insulin dose adjustments were implemented when appropriate. Overnight glucose monitoring included scheduled checks (almost every hour) and alarm-based responses to reduce the risk of nocturnal hypoglycemia.
Interpretation regarding potential confounding
Because the camp environment involved structured meals, scheduled physical activity, peer support, and intensive HCP supervision, the observed changes in glycemic metrics reflect the combined effects of these factors together with isCGM use. The study design does not allow isolation of the independent contribution of each component.
Primary outcome
The primary outcome was to evaluate within-participant changes in glycemic metrics between periods in the context of structured HCP-supervised camp participation.
Glycemic indices such as TAR, TIR, TITR, and TBR on-camp were compared with those obtained during the pre-camp period.
Secondary outcomes
Secondary outcomes included (1) analytical accuracy of the isCGM system compared with capillary BG values assessed by MARD, regression, Bland–Altman, and error grid analyses and (2) subjective evaluations of usability, satisfaction, and recommendation scores obtained from questionnaires completed by campers and HCPs.
Statistical analysis
All statistical analyses were performed using SPSS version 16.0 (IBM Corp., Armonk, NY, USA). A two-sided P value < 0.05 was considered statistically significant. Continuous variables are presented as mean ± standard deviation (SD) for approximately normally distributed data and as median with interquartile range (IQR) for non-normally distributed data. Categorical variables are presented as counts and percentages.
For the primary within-participant comparisons between the pre-camp and on-camp periods, normality of paired differences for each CGM-derived metric (TIR, TITR, TAR level 1, TAR level 2, TBR level 1, and TBR level 2) was assessed using the Shapiro–Wilk test, complemented by visual inspection of histograms and Q–Q plots.
When paired differences were normally distributed, two-tailed paired Student’s t-tests were applied. When normality assumptions were not met, the nonparametric Wilcoxon signed-rank test was used. The specific statistical test applied for each metric is indicated in the corresponding table footnotes.
Effect sizes for paired comparisons were calculated as Cohen’s d for normally distributed differences and as rank-biserial correlation (r) for Wilcoxon tests to provide complementary information on the magnitude of change beyond p values.
No formal adjustment for multiple comparisons was performed for the primary CGM metrics because the analyses were predefined and hypothesis-driven; however, results for secondary and exploratory outcomes should be interpreted cautiously.
Participants were included in the primary analysis if valid CGM data were available for both periods, defined as ≥70% of possible CGM readings during each assessment period, consistent with international CGM consensus recommendations.
Sensor accuracy analyses
Sensor accuracy was evaluated using paired capillary BG and SG measurements collected during both periods.
MARD was calculated as:

MARD values are presented as mean ± SD. Between-period comparisons of MARD were performed using paired t-tests or Wilcoxon signed-rank tests as appropriate based on normality of paired differences.
Agreement between BG and SG was further assessed using:
Linear regression analysis (Pearson correlation coefficient for normally distributed values), Bland–Altman analysis with mean bias and 95% limits of agreement, Parkes and Clarke error grid analyses with proportions of paired values within zones A and A + B.
Exploratory subgroup analyses according to glycemic range (<70 mg/dL, 70–180 mg/dL, >180 mg/dL) were descriptive only and not powered for formal statistical inference.
Paired BG–SG values obtained during sensor warm-up periods or with physiologically implausible values were excluded according to predefined criteria.
Questionnaire analyses
Likert-scale questionnaire responses were treated as ordinal variables and are presented as median (IQR).
Comparisons between campers and HCPs were performed using the Mann–Whitney U test. Within-device comparisons across periods were conducted using the Wilcoxon signed-rank test where applicable.
Given the exploratory nature of satisfaction analyses and the absence of prespecified hypotheses, no correction for multiple testing was applied; findings should therefore be interpreted as descriptive.
Exploratory analyses
Day-by-day changes in CGM-derived metrics during the camp were analyzed descriptively. No formal statistical testing was performed for these temporal analyses due to limited statistical power and the short observation period.
Exploratory historical comparisons with previous camp years using other CGM systems were descriptive only and were not adjusted for potential confounders.
No formal sample size calculation was performed because this study was designed as an exploratory feasibility investigation conducted within a single pediatric diabetes camp. Consequently, the study was not statistically powered to detect small or moderate differences in secondary outcomes, including sensor accuracy metrics and hypoglycemia exposure.
Results
Participant characteristics and CGM data availability
A total of 21 children and adolescents with T1D participated in the summer camp. Of these, all participants were included in the primary analysis based on the availability of valid CGM data for both the pre-camp and on-camp periods. The mean age was 9.9 ± 4.0 years, with 42.9% being female; the mean BMI was 18.3 ± 2.3 kg/m2 (+0.3 SD), and the median diabetes duration was 3.0 years (IQR: 0.25–10) (Table 1). Insulin therapy consisted of multiple daily injections in 33.3% and insulin pump therapy in 66.7% of participants (Table 1). The average HbA1c level was 7.6% ± 0.9%. Prior to camp participation, 61.9% of the participants were using real-time (rt)CGM, 33.3% isCGM, and one participant (4.8%) used SMBG alone (Table 1).
The median CGM wear time was 93.3% during the pre-camp period and 96.2% during the on-camp period. All included participants had sufficient CGM data coverage (≥70% of possible readings) for both periods and were therefore eligible for paired analyses.
Primary outcome: within-participant changes in CGM-derived glycemic metrics
The primary outcome was the within-participant change in CGM-derived glycemic metrics between the pre-camp and on-camp periods. Compared with the pre-camp period, the on-camp period was associated with a significant improvement in overall glycemic management. TIR (70–180 mg/dL) increased significantly from 54.5% (IQR: 43.9–62.7) during the pre-camp period to 64.3% (IQR: 54.8–75.6) on-camp (P = 0.031) (Fig. 1, Table 2). TITR (70–140 mg/dL) showed a modest but nonsignificant increase from 36.7% (IQR: 25.3–45.2) to 40.0% (IQR: 33.9–59.3) (P = 0.081) (Fig. 1, Table 2).

Within-participant changes in CGM-derived glycemic metrics between the pre-camp and on-camp periods. Paired comparisons of CGM-derived glycemic metrics between the pre-camp and on-camp periods are shown for each participant: time above range (TAR) level 2 (>250 mg/dL), TAR level 1 (181–250 mg/dL), time in range (TIR; 70–180 mg/dL), time in tighter range (TITR; 70–140 mg/dL), time below range (TBR) level 1 (54–69 mg/dL), and TBR level 2 (<54 mg/dL). Box plots indicate the median and interquartile range, with whiskers showing the minimum and maximum values; individual data points represent participants. Paired statistical tests were applied as appropriate. CGM, continuous glucose monitoring.
Changes in CGM-Derived Metrics Pre- and on-Camp Periods
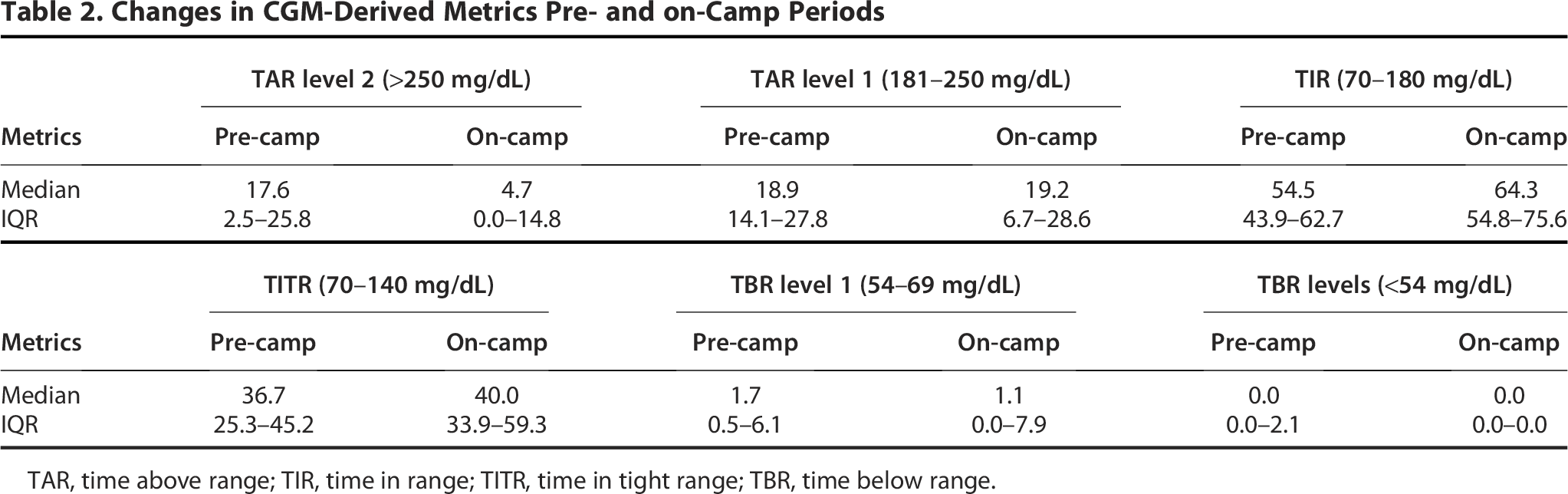
TAR, time above range; TIR, time in range; TITR, time in tight range; TBR, time below range.
Correspondingly, TAR level 2 (>250 mg/dL) significantly decreased from 17.6% (IQR: 2.5–25.8) to 4.7% (IQR: 0.0–14.8) (P = 0.049), and TAR levels 1 (181–250 mg/dL) was comparable between pre- and on-camp period (P = 0.938) (Fig. 1, Table 2). Based on normality assessment, TIR and TAR level 2 were analyzed using the Wilcoxon signed-rank test.
In contrast, TBR level 1 (54–69 mg/dL) and TBR level 2 (<54 mg/dL) remained stable (1.7% [IQR: 0.5–6.1] vs. 1.1% [IQR: 0.0–7.9], P = 0.289; 0.0% [IQR: 0.0–2.1] vs. 0.0% [IQR: 0.0–0.0], respectively. P = 0.443) (Fig. 1, Table 2), indicating that the improvement in TIR was achieved without an increase in hypoglycemia. These improvements were observed consistently across participants, as illustrated in Figure 1, which displays paired pre-camp and on-camp values for each CGM metric. These changes reflect the combined effects of structured camp participation and intensive HCP supervision with isCGM data review.
Exploratory analysis: day-by-day changes in glycemic metrics during the camp
To explore short-term temporal trends during the camp, day-by-day changes in CGM-derived metrics were analyzed descriptively. TIR showed a gradual increase from day 1 to day 3 of the camp (54.5—58.6—62.5%—64.3%), whereas TAR level 2 showed a corresponding decrease (17.6—11.4—9.8%—4.7%). TBR remained stable throughout the camp period. These analyses were exploratory, and no formal statistical comparisons were performed for day-by-day changes.
Safety outcome
Hypoglycemia exposure remained low during the camp. There was no significant increase in time spent in hypoglycemia (<70 mg/dL) on-camp compared with the pre-camp period, and time spent below 54 mg/dL was minimal in both periods. No episodes of severe hypoglycemia requiring third-party assistance or diabetic ketoacidosis occurred during the camp.
Secondary outcome: sensor accuracy of isCGM
As a secondary outcome, sensor accuracy was evaluated using paired capillary BG and SG measurements obtained during the camp. A total of 293 paired BG–SG values (111; pre-camp, 182; on-camp) were analyzed. No significant difference was observed between average SG during the camp (122.5 mg/dL [IQR: 88.0–177.8] on-camp vs. 118.0 mg/dL [IQR: 95.0–163.0] pre-camp, P = 0.749) (Fig. 2). The values of BG and SG on- or pre-camp were also comparable (Fig. 2).

Comparison of CGM-derived metrics and agreement between blood glucose and sensor glucose. CGM-derived metrics and agreement between capillary blood glucose (BG) and sensor glucose (SG) values during the pre-camp and on-camp periods. Panels show average SG, mean absolute relative difference (MARD), coefficient of variation (%CV), average BG and SG during the on-camp period, average BG and SG during the pre-camp period, and glucose management indicator (GMI). Data are presented as box plots with individual data points; comparisons between periods were performed using paired tests.
The MARD between SG and BG was 13.6% ± 10.5% on-camp and 12.7% ± 10.5% pre-camp (P = 0.641). The coefficient of variation (%CV) and GMI values were comparable between on-camp and pre-camp periods (%CV: 37.9% ± 11.6% vs. 34.5% ± 10.6%, P = 0.844; GMI: 6.2% [IQR 5.4–7.6] vs. 6.4% [IQR 5.7–7.2], Wilcoxon signed-rank test; P = 0.636. Figure 2).
Single regression analysis demonstrated a strong linear correlation between BG and SG values both on and pre-camp (Fig. 3A and B).
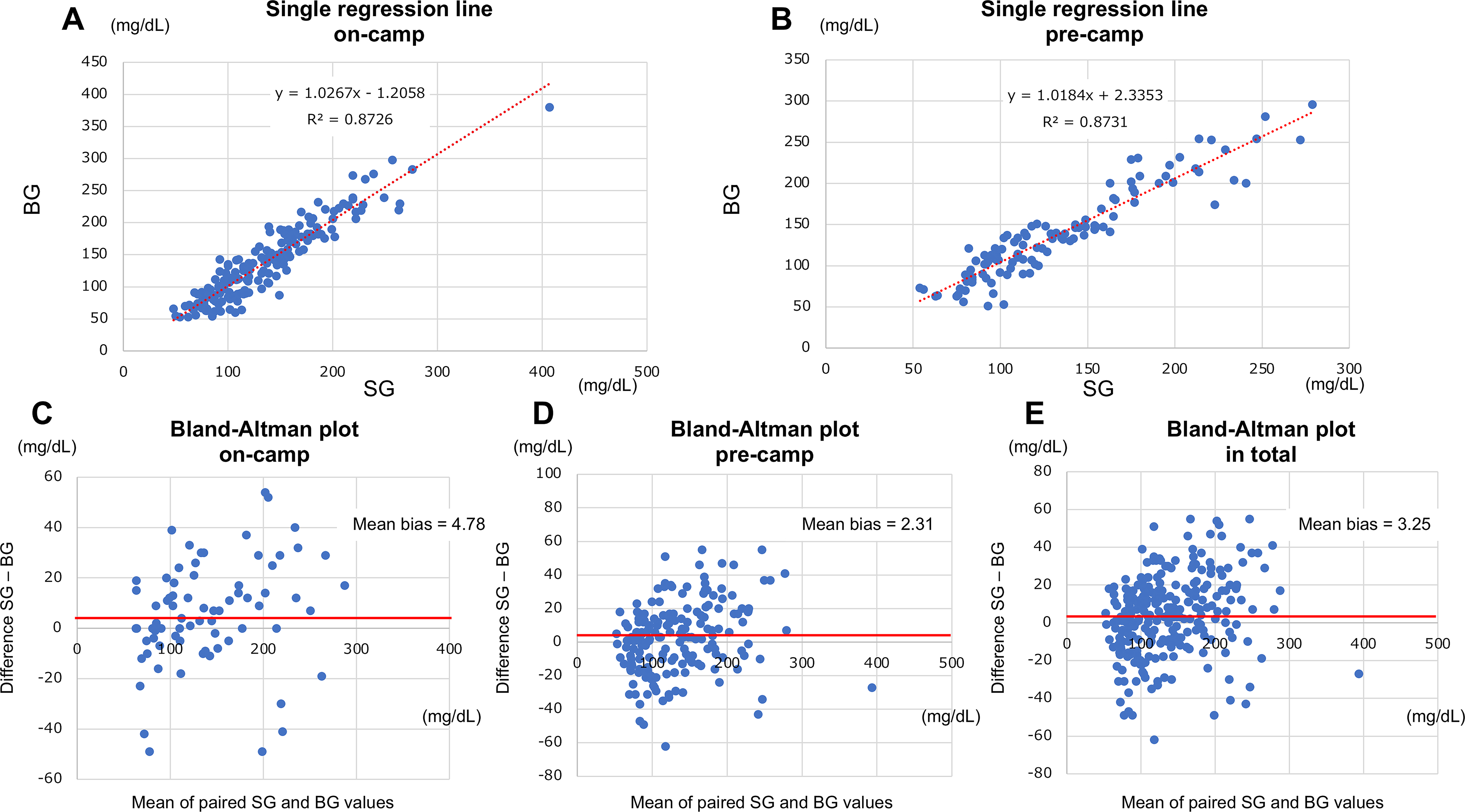
Regression and Bland–Altman analyses of paired sensor glucose and blood glucose values. Agreement between paired SG and BG measurements obtained pre-camp and on-camp periods.
Bland–Altman analysis revealed small mean biases of 4.78 mg/dL on-camp, 2.31 mg/dL pre-camp, and 3.25 mg/dL overall, suggesting clinically usable agreement between SG and BG (Fig. 3C–E). Although a certain degree of variability was noted, these findings support that the isCGM system maintained consistent accuracy across different environmental and activity conditions.
Furthermore, Parkes error grid analyses demonstrated that 82.4%–88.3% of paired values were within zone A and 100% within zones A + B, both on- and pre-camp (Fig. 4A). Clarke error grid analyses demonstrated that 76.4%–81.1% of paired values were within zone A and 100% within zones A + B, both on- and pre-camp (Fig. 4B). These findings support the clinical accuracy and reliability of the isCGM system under camp conditions.

Parkes and Clarke error grid analyses of sensor glucose accuracy. Clinical accuracy of SG measurements assessed using
Secondary outcome: user satisfaction and usability
User satisfaction with FreeStyle Libre 2 during the camp was evaluated using post-camp questionnaires completed by campers and HCPs.
Among campers, key strengths included ease of monitor use, understanding of glucose trend curves, and interpretation of trend arrows, indicating favorable usability in the camp setting.
HCPs reported significantly higher scores than campers regarding sensor insertion difficulty (P = 0.0012, Mann–Whitney U test), reflecting differences in technical experience (Supplementary Fig S1A).
Overall, the questionnaire results indicate good acceptability and usability of FreeStyle Libre 2 in a supervised pediatric camp environment.
Detailed questionnaire data and exploratory descriptive comparisons with previous camps using other CGM systems are provided in the Supplementary Materials.
Exploratory historical comparison with previous CGM-based camps
These historical comparisons across CGM systems are purely descriptive and were not designed to compare device performance. No formal statistical comparisons across devices were performed. As an exploratory analysis, CGM-derived glycemic outcomes during the current Libre 2 camp were descriptively compared with those from previous summer camps conducted using Dexcom G6 (2023) and Dexcom G7 (2024). Although improvements in TIR were observed across all camp years, these comparisons were not adjusted for differences in participant characteristics, camp conditions, or CGM systems, and no formal statistical comparisons were performed. The results of these exploratory historical comparisons are presented in Supplementary Figure S3.
These consistent findings across the three consecutive years suggest the potential reproducibility of CGM-based diabetes education programs in improving real-world glycemic outcomes and user engagement among children and adolescents with T1D.
Discussion
In this prospective single-arm observational study, we evaluated within-participant changes in CGM-derived glycemic metrics during a pediatric T1D summer camp under HCP-supervised use of isCGM, FreeStyle Libre 2. The primary finding was a significant improvement in TIR accompanied by a reduction in TAR level 2 on-camp compared with the pre-camp period, without an increase in hypoglycemia. Although TIR improved significantly during the camp, glycemic variability, as reflected by %CV, remained statistically unchanged (34.5% vs. 37.9%, P = 0.844), suggesting that improved overall glucose control was achieved without a clinically meaningful increase in variability. These results suggest the feasibility of a HCP-supervised, camp-based glucose management strategy incorporating isCGM for short-term glycemic management in pediatric camp settings.7,8
Interpretation of the primary outcome
The observed improvement in glycemic metrics was based on paired within-participant analyses, minimizing the influence of interindividual variability. Importantly, TBR did not increase during the camp, suggesting that improved glycemic management was achieved without compromising safety.1,9 This finding is particularly relevant in pediatric camp environments, where increased physical activity and irregular schedules may increase the risk of hypoglycemia.
However, the intervention evaluated in this study was inherently composite. Improvements in glycemic outcomes likely reflect the combined effects of structured HCP supervision, the use of isCGM for frequent glucose review, and the controlled camp environment, including standardized meals, scheduled physical activity, and peer support.10,11 As such, the present design does not allow isolation of the independent contribution of FreeStyle Libre 2 from that of intensive supervision or the camp setting itself. Rather than attributing causality to a single component, our findings should be interpreted as evidence of the effectiveness of a supervised isCGM-based management strategy in this specific context.
Comparison with previous CGM-based camp studies
Previous pediatric diabetes camp studies, including those using real-time CGM systems, have reported improvements in glycemic management on-camp participation. 12 In exploratory historical comparisons with our prior camps using Dexcom G6 and G7, similar improvements in TIR were observed.13,14 However, these comparisons were not randomized and were subject to differences in participant characteristics, device features, and camp conditions across years. 15 Therefore, no conclusions regarding equivalence or superiority among CGM systems can be drawn. The consistency of directional improvements across devices nevertheless suggests that structured supervision and frequent CGM-informed decision-making may be key drivers of improved glycemic outcomes in camp settings.
Sensor accuracy and usability
Sensor accuracy should be interpreted with appropriate caution. In pediatric CGM studies, MARD values around 10%–13% have commonly been reported under real-world conditions, 6 while accuracy can vary depending on study design and glucose variability.
In our camp setting, the mean MARD of approximately 13% falls within these reported pediatric ranges; however, the wide IQR suggests appreciable variability, indicating that accuracy may be closer to the upper end of what is typically observed in some real-world pediatric settings, 11 particularly during periods of rapid glucose change related to meals and activity.
Furthermore, clinical accuracy may differ at glycemic extremes. 9 Although Parkes and Clarke error grid analyses showed that all paired values were within clinically acceptable zones (A + B), the distribution across zones and the observed scatter suggest that sensor performance may be less stable during hypoglycemia or marked hyperglycemia, and therefore confirmatory capillary BG remains important when symptoms and sensor readings are discordant or when glucose is rapidly changing.
As secondary outcomes, sensor accuracy and user satisfaction with FreeStyle Libre 2 were evaluated. Accuracy analyses demonstrated acceptable agreement between sensor glucose and capillary blood glucose measurements, consistent with previous reports of Libre 2 performance in pediatric populations.16,17 High levels of user satisfaction and usability were also observed, supporting the practicality of isCGM use in residential camp settings where ease of use and minimal device burden are particularly important.
Clinical and educational implications
The present findings have practical implications for pediatric diabetes education and camp-based care. 18 Supervised use of isCGM may facilitate timely glucose review, structured insulin adjustment, and real-time educational feedback for children and adolescents with T1D.19,20 Such settings may also provide an opportunity for HCPs to reinforce self-management skills and for participants to gain confidence in CGM-guided glucose management in a supportive environment.
Limitations
Several limitations should be acknowledged. First, the single-arm observational design without a control group precludes causal inference. Second, the short duration of the camp limits conclusions regarding longer-term glycemic outcomes after camp participation. Third, the composite nature of the intervention prevents disentanglement of the effects of isCGM use, HCP supervision, and the structured camp environment.
Fourth, the pre-camp period differed substantially in supervision intensity, meal composition, and activity structure; therefore, observed differences between pre-camp and camp periods should not be interpreted as device effects.
Fifth, the modest sample size limited statistical power, particularly for secondary outcomes such as sensor accuracy, hypoglycemia exposure, and subgroup analyses. No a priori power calculation was performed, increasing the risk of type II error; accordingly, nonsignificant findings should not be interpreted as evidence of equivalence.
Finally, exploratory comparisons with previous camp cohorts using Dexcom G6/G7 systems were purely descriptive and were neither prospectively designed nor statistically powered to evaluate differences in device performance, accuracy, or clinical effectiveness. Substantial heterogeneity in study design, participant characteristics, supervision protocols, and camp structure across years precludes direct comparative interpretation. No formal statistical comparisons were conducted across devices, and device superiority or equivalence cannot be inferred from these data. To minimize potential misinterpretation, these historical data have been relocated to the Supplementary Materials and should be considered contextual rather than inferential. Accordingly, the present findings should be regarded as hypothesis-generating and warrant confirmation in larger, adequately powered prospective studies.
In conclusion, HCP-supervised use of FreeStyle Libre 2 during a pediatric T1D summer camp was associated with improved CGM-derived glycemic metrics compared with a pre-camp period, without an increase in hypoglycemia. Although the independent effects of individual intervention components cannot be isolated, these findings provide preliminary evidence supporting the feasibility and potential short-term benefits of supervised isCGM-based glucose management strategies in pediatric diabetes camp settings.
Footnotes
Acknowledgments
The authors would like to thank all study participants and their guardians for their participation as well as diabetologists, specialized diabetes nurses, pharmacists, clinical laboratory technologists, and national registered dieticians affiliated with the Kumamoto University Hospital, Morinoki Clinic, Misato Rehabilitation Clinic, Kumamoto Chuo Hospital, and others for the medical support all over the camp. We also want to thank the members in “Kumamoto Children’s Type 1 Diabetes Summer Camp Executive Committee,” including alumni of the past camps in charge of the camps organization, as well as all the camp staff.
Authors’ Contributions
T.K., M.Y., H.T., and K.N. researched data. T.K. wrote the article. T.S. reviewed/edited the article. Y.K. and N.K. contributed to discussion and reviewed/edited the article.
Data Availability Statement
Tatsuya Kondo is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Raw CGM data are available from the corresponding author upon reasonable request.
Generative AI Disclosure
The authors used generative artificial intelligence–assisted tools for language editing and grammatical improvement of the article. The authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
Author Disclosure Statement
Approval of the Research Protocol: This research was approved by the Ethics Review Committee at Aso Medical Center (Ethics No. 2025-002). The study was conducted in accordance with the principles of the Declaration of Helsinki.
Informed Consent: Before enrollment, campers and their parents/legal representatives received both oral and written information about the study. Written informed consent was obtained from parents/legal representatives and assent was received from all campers.
Approval date of Registry and the Registration No. of the study/trial: N/A.
Animal Studies: N/A.
Naoto Kubota has received lecture fees from Kowa Pharmaceutical Co., Ltd.; Novo Nordisk Pharma Ltd.; Sumitomo Pharma Co., Ltd.; Teijin Pharma Ltd.; Nippon Boehringer Ingelheim Co., Ltd.; Taisho Pharmaceutical Co., Ltd.; and Mitsubishi Tanabe Pharma Corporation. He has received research grants from Provigate Inc.; the Manpei Suzuki Diabetes Foundation; Shimadzu Corporation; and Kowa Pharmaceutical Co., Ltd. He has received scholarship grants from Sumitomo Pharma Co., Ltd.; Takeda Pharmaceutical Co., Ltd.; Mitsubishi Tanabe Pharma Corporation; Novartis Pharma K.K.; Novo Nordisk Pharma Ltd.; and Roche Diagnostics K.K. Endowed department is sponsored by Terumo Corporation.
Funding Information
This study was supported by The Japan Diabetes Foundation/Costco Research Grant No. 2023-04. The sponsor of this study had no role in the study design, data collection, data analysis, data interpretation, or writing of the report.
Supplemental Material
Supplemental Material
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
