Abstract
Background:
This study investigated unique burdens experienced by parents of young children with type 1 diabetes in the context of contemporary diabetes management.
Methods:
Self-report surveys and medical record information from the T1D Exchange clinic registry were used. Parental burden and family impact scores were tabulated across demographic and clinical characteristics, overall and according to age group (<4, 4-<6, and 6-<7 years).
Results:
The mean age of the 549 children was 5.2 ± 1.2 years (n = 102 <4 years, n = 266 4-<6 years, and n = 181 6-<7 years) and mean duration of diabetes was 2.4 ± 1.0 years. Mean hemoglobin A1c was 8.2% ± 1.1%. Approximately one-third (32%) reported their child was currently using CGM and over half (58%) reported using insulin pumps. The most frequently endorsed parent-reported burdens of diabetes were feeling discouraged by the treatment plan (85%), child being deprived regarding food (76%), child being excluded from activities or events (73%), and having difficulty dealing with school staff (72%). Areas endorsed for negative family impact were diminished amount or quality of sleep for family members (59%) and need for flexible working arrangements to help care for their child (55%).
Conclusions:
Substantial burden remains for parents of young children with type 1 diabetes despite the availability of advanced technologies for diabetes management.
Keywords
Parents of young children with type 1 diabetes face significant stress and burden.1,2 A majority, if not all, of diabetes management is the responsibility of the parents, and there are many challenges to diabetes management in children aged 7 and under. Young children are rapidly emerging in the realms of physical, cognitive, social, and emotional development. These changes impact the interaction between children and their caregivers as they navigate diabetes management. In addition, primary caregivers of young children need to trust others with diabetes management responsibilities as the child is introduced to child care and school settings. 3 Each developmental stage of young childhood has unique challenges and issues pertaining to diabetes management. Given this, it is important to consider the experience of parents caring for children with type 1 diabetes at different developmental stages.
There is ample evidence that suggests parents of young children with type 1 diabetes experience emotional distress while caring for their child.4-7 There have been calls for research to refine our knowledge of parental burden and distress to inform the design of interventions for families of young children with type 1 diabetes.8,9 Parental coping strategies and distress have implications for treatment adherence, quality of life, and family functioning. Interventions have been developed to help parents cope with caring for young children with type 1 diabetes.10-14 However, few of these psychosocial interventions have been focused on the adoption of advanced diabetes technology in very young children.
Diabetes technologies have improved substantially in the past several decades, with tools such as smart insulin pumps with bolus calculators and continuous glucose monitors (CGMs). Along with these technical advancements come new challenges and possible burdens, especially for young children and their families. For example, it can be difficult for parents to find “real estate” on their small children’s bodies to implant devices, and other caregivers need to be educated on these devices. Thus, it is important to ascertain typical burdens and stresses of parents of young children with diabetes in the contemporary era of diabetes management. It is also important to understand how the burdens reported by parents are impacted by use or nonuse of different diabetes technologies, as well as by other demographic factors such as socioeconomic status.
The aim of this study is to understand present-day burdens experienced by parents caring for their young child with type 1 diabetes. We plan to investigate the burden reported by families with children in different age groups, toddlers (under 4 years), preschoolers (4-<6 years), and school age children (6-<7 years), to understand the unique burdens and family impacts at different developmental stages. Children in toddlerhood have limited verbal abilities which make it difficult to convey symptoms, preschool children are entering the school environment for the first time, and school age children and their parents need to establish new relationships with caregivers and rely on them more due to the longer days spent at school. All of these factors may have an impact on reported diabetes burdens. We anticipate that parents of young children with type 1 diabetes will report significant burdens and distress, highlighting the need to explore the factors related to burden and distress and ultimately identifying approaches to overcome the challenges reported by parents of young children with type 1 diabetes.
Methods
The T1D Exchange Clinic Registry (T1D Exchange) includes 74 endocrinology practices based in the United States, including 58 centers serving youth with type 1 diabetes (see Supplementary Material). To date, over 30 000 participants have been enrolled in the registry, and details of the enrollment process, eligibility criteria, and baseline data collection have been previously reported. 15 Data were collected for the clinic registry central database from medical records in addition to questionnaires completed by parents of the young children enrolled in the registry. 15
This report includes 549 young children enrolled in the registry from February 1, 2015, to May 2, 2016, aged less than 7 years with a clinical diagnosis of type 1 diabetes for at least 1 year, whose parent/guardian completed the two surveys assessing the main constructs of interest in this study: parental burden and family impact, respectively (described below). Demographic, socioeconomic, and diabetes management factors were obtained from parent/guardian questionnaires. Insulin pump and CGM use were reported by parent/guardian and confirmed by clinic report. The most recent hemoglobin A1c (HbA1c) measurement within 6 months prior to registry enrollment (mean duration of HbA1c measurement prior to enrollment was 8 days) was obtained from the clinic medical record. HbA1c values were measured by point-of-care device or local laboratory. Occurrences of diabetic ketoacidosis (DKA) and severe hypoglycemia (SH) during the 3 months prior to enrollment were reported by parent/guardian. DKA was defined by occurrence of ketoacidosis diagnosed by a doctor requiring a visit to hospital, emergency room, or other health care facility. SH was defined as hypoglycemia resulting in seizure or loss of consciousness.
The Problem Areas in Diabetes Survey–Parent Revised version (PAID-PR) 16 measures the degree of burden experienced by the parents while managing their child with type 1 diabetes. Parents/guardians were asked to rate their agreement with 18 statements regarding the burden of diabetes-related issues using a 0 (disagree) to 4 (agree) Likert-type scale. Total score for each participant was the mean of all nonmissing responses multiplied by 25, with a total possible of 100. Higher PAID scores represent greater parental burden related to the child’s diabetes.
The Family Impact Survey (FIS) 17 measures a different kind of burden, how often diabetes impacted family life in the past year. Parents were asked to rate how often diabetes negatively affected areas such as school, work, finances, and well-being using a 0 (almost never) to 3 (almost always) Likert scale, which was scored for each participant as the mean of all nonmissing responses multiplied by 33.3 for a total score out of 100. Higher FIS scores represent more negative impact of diabetes on the family. Parents had the option of selecting “not applicable” for any of the responses on the FIS. These values were excluded from tabulations and calculations of the total score.
Statistical Analyses
For both the PAID-PR and FIS, the mean total score was tabulated according to various demographic and clinical characteristics. The amount of burden and the degree of negative family impact for the individual responses to the PAID-PR and FIS surveys were tabulated overall and according to age group (<4, 4-<6, and 6-<7 years). The associations between each mean total score and demographic and clinical characteristics were assessed overall and according to age group (<4, 4-<6, and 6-<7 years) using t-tests or the Wilcoxon rank-sum procedure (or Kruskal-Wallis if >2 levels) as appropriate. Data analyses were performed using SAS version 9.4 (2011 SAS Institute Inc, Cary, NC). All P values are two-sided, and due to multiple comparisons only P values <.01 were considered statistically significant.
Results
Participant Characteristics
The mean age of the 549 children was 5.2 ± 1.2 years (n = 102 <4 years, n = 266 4-<6 years, and n = 181 6-<7 years) and mean duration of diabetes was 2.4 ± 1.0 years. Mean HbA1c was 8.2% ± 1.1%. Approximately one-third (32%) were identified as current CGM users and more than half (58%) were using insulin pumps. Additional cohort characteristics are shown in Table 1.
Cohort Characteristics.
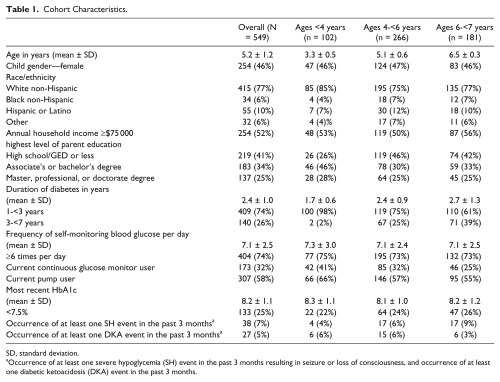
SD, standard deviation.
Occurrence of at least one severe hypoglycemia (SH) event in the past 3 months resulting in seizure or loss of consciousness, and occurrence of at least one diabetic ketoacidosis (DKA) event in the past 3 months.
Survey Tabulations
Figures 1 and 2 show the areas in which parents feel most burdened by or experience high negative family impact from their young child’s diabetes for each age group. Overall, most parents reported the largest burden of diabetes related to feeling discouraged by the treatment plan (85%). Other areas in which parents felt most burdened by diabetes involved their child being deprived regarding food (76%), child being excluded from activities or events (73%), and having difficulty dealing with school staff (72%). Parents felt least burdened about worrying that their child would have low blood sugars (11%) or about the future and possibility of serious complications for the child (14%), and feeling upset when child’s diabetes management is off track (16%).
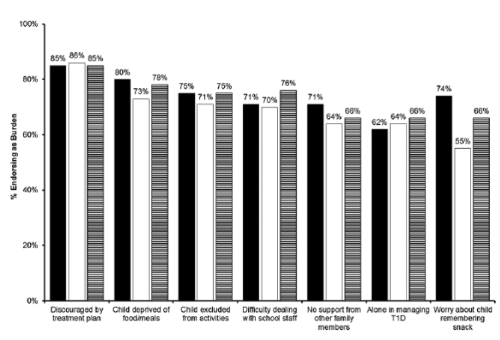
Areas parents report most burden. Solid black bars represent <4 years old. Solid white bars represents 4-<6 years old. Black-and-white striped bars represents 6-<7 years old.
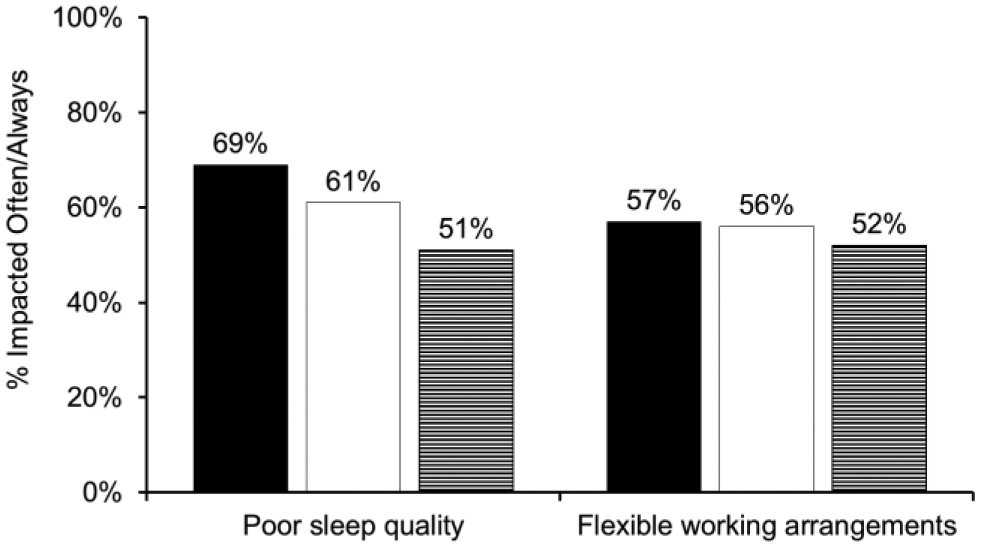
Areas parents report greatest negative family impact. Solid black bars represent <4 years old. Solid white bars represents 4-<6 years old. Black-and-white striped bars represents 6-<7 years old.
Areas in which parents experienced frequent negative family impact were diminished amount or quality of sleep for family members (59%) and the need for flexible working arrangements to help care for their child (55%). Problems with regular school attendance (7%), negative impact of diabetes on school performance (5%), and the inability to participate in activities or trips (5%) were the areas in which parents reported the lowest impact on their family life.
Parental Burden Results
The mean total PAID score was 53.8 ± 18.4 overall, 53.8 ± 18.2 for parents of children <4 years, 52.2 ± 18.0 for parents of children 4-<6 years, and 56.1 ± 18.9 for parents of children aged 6-<7 years (P = .16; Table 2a). The mean total PAID score was not significantly different between parents of females and parents of males (PAID 55.0 ± 17.5 vs 52.8 ± 19.1, P = .19) but when stratified by age group there was a trend toward higher PAID scores in parents of male toddlers compared with parents of female toddlers (58.4 ± 17.4 vs 49.9 ± 18.4, P = .04; Table 2a).
Mean Problem Areas In Diabetes (PAID) Score According to Demographic and Clinical Characteristics.
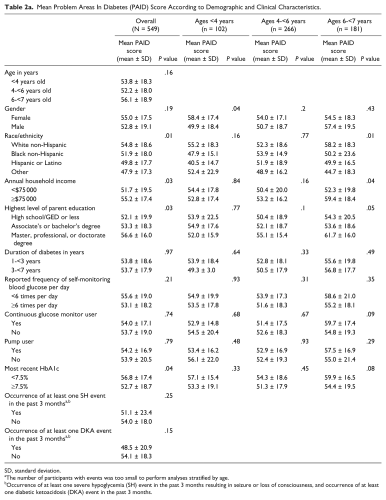
SD, standard deviation.
The number of participants with events was too small to perform analyses stratified by age.
Occurrence of at least one severe hypoglycemia (SH) event in the past 3 months resulting in seizure or loss of consciousness, and occurrence of at least one diabetic ketoacidosis (DKA) event in the past 3 months.
Mean Family Impact Survey (FIS) Score According to Demographic and Clinical Characteristics.
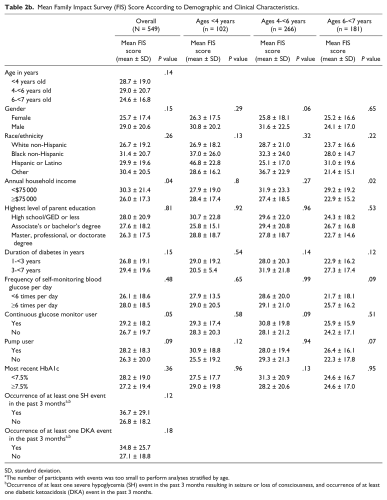
SD, standard deviation.
The number of participants with events was too small to perform analyses stratified by age.
Occurrence of at least one severe hypoglycemia (SH) event in the past 3 months resulting in seizure or loss of consciousness, and occurrence of at least one diabetic ketoacidosis (DKA) event in the past 3 months.
Overall, there was a trend toward higher PAID scores in parents with higher education and earning $75 000 or more annually (P = .03 for both), but this trend was not seen when stratified by age group (Table 2a). A trend toward higher PAID scores was seen among parents of white non-Hispanic children overall and in the 6-<7 year age group (P = .01 for both).
Mean total PAID scores did not differ among parents of participants using a pump versus injections for insulin delivery (P = .79) or among CGM users versus nonusers (P = .74) when assessed for the overall cohort or when stratified by age group.
No significant difference in mean PAID scores were seen among parents of children achieving glycemic target of HbA1c <7.5% versus HbA1c ≥7.5% (56.8 ± 17.4 vs 52.7 ± 18.7, P = .04), who had at least one SH event versus no event (51.1 ± 23.4 vs 54.0 ± 18.0, P = .25), or who had at least one DKA event versus no event (48.5 ± 20.9 vs 54.1 ± 18.3, P = .15). Results were similar when stratified by age group.
Family Impact Results
The mean total FIS score was 27.5 ± 19.3 overall, 28.7 ± 19.0 for parents of children <4 years, 29.0 ± 20.7 for parents of children 4-<6 years, and 24.6 ± 16.8 for parents of children aged 6-<7 years (P = .14, Table 2b). The mean total FIS score was not significantly different between parents of females and parents of males (25.7 ± 17.4 vs 29.0 ± 20.6 for females and males, respectively; P = .15, Table 2b).
A trend toward higher family impact of diabetes among households earning <$75 000 annually was seen overall (P = .04) and in the 6-<7 year age group (P = .02) (Table 2b).
The mean total FIS score was slightly higher for parents of children using a CGM compared to non-CGM users (29.2 ± 18.2 vs 26.7 ± 19.7, P = .05) but this was not statistically significant. Results were similar when stratified by age group.
No significant difference in mean FIS scores were seen among parents of children achieving glycemic target HbA1c of <7.5% versus HbA1c ≥7.5% (28.2 ± 19.0 vs 27.2 ± 19.4, P = .36), who had at least one SH event versus no event (36.7 ± 29.1 vs 26.8 ± 18.2, P = .12), or who had at least one DKA event versus no event (34.8 ± 25.7 vs 27.1 ± 18.8, P = .18). Results were similar when stratified by age group.
Conclusions
Data for this study were collected from the largest published national (United States) sample of young children under age 7 using previously validated surveys, and the sample was drawn from multiple diabetes treatment centers in geographically diverse areas, which contribute to both the validity and the generalizability of the results. Parents endorsed substantial burdens in three main areas: collaborating with others, worry about the child feeling excluded, and feeling alone with diabetes management. In terms of family impact, parents reported sleep issues and the need for flexible work arrangements as significant factors. The age span of 3-<7 encompasses many dynamic changes and developmental stages and so it was originally thought burden may be different in each age group however our results suggest there is no significant difference in terms of type and frequency of burden between the age periods of toddler, preschoolers, and school aged children.
The vast majority (85%) of parents reported feeling discouraged by their treatment plan; this could have multiple meanings. For example, the parents may feel that they exert substantial efforts and the results do not match their expectations with respect to optimizing or normalizing glycemic control. It could also mean that parents do not think that current treatment tools are adequate to manage their child’s diabetes and glycemic variability. To address this very common concern, it may be beneficial to families if health care team members discuss expectations around treatment plans with parents of young children with type 1 diabetes. Certainly, future research would benefit by more in depth exploration of this theme using qualitative methods.
Other themes that emerged suggest parents of young children with diabetes may benefit from learning specific skills to help support them in caring for their child’s diabetes. For example, parents reported not getting support from other family members. These parents may experience less burden if they learn strategies to help bolster their support system and manage the emotional experience of entrusting others with their child’s diabetes care. Parents also reported having difficulty working with school staff, and the need for flexible work arrangements from their employers. For many families during this time period, the child with diabetes will transition to care environments outside of the home (school, daycare, etc). These transitions are introducing a new level in the family ecosystem beyond the nuclear family and interventions that acknowledge and target these changes in contexts for diabetes care may minimize burdens or negative impacts. Coaching parents on how to collaborate with school staff and advocate for their child would be a timely intervention during this developmental stage. Interventions that target advocacy skills and communication skills may have direct benefit on the burdens identified by parents.
Two themes that emerged from the data were worry that the child will feel excluded socially and worry that child will feel deprived of food due to their diabetes. Research has documented that significant stress is often associated with mealtime in young children with diabetes.18-21 Parental worry that their child is feeling deprived or excluded regarding food may make it difficult for parents to adhere to the healthy dietary choices they want to make for their child and family, which may play a role in mealtime stress. Given how often young children eat, parental distress around food can quickly mount to very frequent feelings of burden throughout the day, potentially detracting substantially from parents’ quality of life and optimal diabetes management.
Sleep interference is endorsed as an issue and has been a consistent theme in the literature.22-25 Parent’s and children’s sleep quality can be compromised due to the need for diabetes care in the night. Remote monitoring technology that accompanies CGM use may help parents so they do not need to rise out of bed and disturb the child to obtain a glucose value. This type of technology may also help manage fear of hypoglycemia, however, parents using remote monitoring still need to disrupt their sleep and possibly their child’s sleep if a corrective dose or low glucose treatment is needed.
There were two trends in regards to demographic data for both diabetes burden and family impact. There was a trend toward higher parent-reported burden among parents of non-Hispanic white children and among parents with higher income or education. It is possible that diabetes-related burdens may represent a greater proportion of their stressors compared to families from racial/ethnic minorities or those with fewer resources, who may be exposed to more stressors overall from a variety of sources. In addition, there was a trend toward higher parent-reported negative family impact among parents with lower income. Parents who have less income may report diabetes having a more negative impact because of financial stress due to missed work time and additional medical expenses.
We found that parents of children who use CGM reported more diabetes burden than parents of children who do not use CGM. The same trend was found among parents whose children wear pumps, where they reported more burden than parents whose children do not wear pump. However, given the cross-sectional nature of these data, we cannot determine causality. In other words, we cannot determine whether families who find diabetes more burdensome turn to technology to alleviate the burden, or if wearing advanced diabetes technology creates more burden. Future longitudinal research can help assess these associations.
There are a few limitations to consider when interpreting the results of this study. First, the design of this study was cross sectional so no causal inferences can be drawn from the results. Second, self-reported surveys were used for acquisition. It is possible that parents who enrolled their child in the registry and completed the entry survey may feel more burden or negative impact from their child’s diabetes than parents who chose not to enroll or complete the survey. Further studies using multiple methods of reporting such as survey and qualitative methodology may help to clarify findings with numerous possible interpretations. Third, we chose to conduct the analysis in three different age groups due to the diversity in developmental skills observed across this young age range. A major goal of this study was to understand parents’ perceptions of burden, and subgrouping the analysis helped us to accomplish this goal. However, the subgroupings contained relatively small numbers or respondents, therefore, detecting statistical significance within the different subgroups becomes more challenging. Finally, advanced diabetes technologies are evolving at a rapid pace, and findings based on parents’ exposure to earlier models may not apply to current or future technologies. Thus, this can be a challenge to the generalizability of results when technology has already evolved and helpful new features are available but not yet utilized and assessed by parents.
This investigation highlights that all families of young children with diabetes experience diabetes related burden and report that diabetes has impact on their family life. Behavioral interventions need to be developed and available to all families that focus on helping the parents of young children understand how usual developmental challenges at different stages impact diabetes management of a young child. Intervention components need to provide strategies, skills, and support for integration into routine care to support parental quality of life related to diabetes management. These interventions can also provide guidance to parents implementing advanced diabetes treatments and technologies. Such interventions may have a positive impact on parents and ultimately on diabetes outcomes for young children with type 1 diabetes.
Footnotes
Abbreviations
CGM, continuous glucose monitor; DKA, diabetic ketoacidosis; FIS, Family Impact Survey; HbA1c, hemoglobin A1c; PAID-PR, Problem Areas in Diabetes Survey–Parent Revised version; SH, severe hypoglycemia; T1D Exchange, T1D Exchange Clinic Registry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported through the Leona M. and Harry B. Helmsley Charitable Trust. MV received support from the National Institutes of Health grant K12-DK094714. MEH and BJA also received support from the National Institute of Diabetes and Digestive and Kidney Disease, K12097696.
