Abstract

Keywords
Obesity is increasingly prevalent among individuals with type 1 diabetes (T1D), affecting up to 37% of adults with this condition. 1 This trend introduces additional cardiovascular risks, including hypertension, dyslipidemia, and an elevated risk of coronary heart disease. 2 With the global T1D population expected to rise from 3.7 million in 2021 to approximately 13.5 to 17.4 million by 2040, effective interventions to manage obesity are urgently needed. 3 Although glucagon-like peptide 1 receptor agonists (GLP-1RAs) like semaglutide are effective for weight reduction in individuals without diabetes, research on their impact in patients with T1D is limited. 4
We conducted a retrospective chart review of 23 patients with T1D, 86% of whom were overweight or obese, to evaluate the impact of semaglutide on weight loss and glycemic control. The predominantly non-Hispanic white (91%), female (83%) cohort had a mean age of 45 and a mean diabetes duration of 25 years. Treated at Keck Medicine of USC Beverly Hills, participants received primarily telemedicine-based care during and after the COVID-19 pandemic, over a 7- to 50-month follow-up. This period limited hemoglobin A1c (HbA1c) data capture. All patients were on semaglutide therapy for at least six months, with 78% using continuous glucose monitors (CGMs) and 61% utilizing automatic insulin delivery (AID) systems.
As shown in Figure 1, semaglutide treatment led to significant weight loss, with patients losing an average of 5% of their baseline weight (about 3.75 kg) over a 22-month follow-up. At baseline, 43% of participants were classified as obese (body mass index [BMI] ≥ 30). By the final follow-up (after 9+ months), two participants had transitioned from obese to overweight, and one from overweight to normal weight. Most participants achieved weight loss on lower semaglutide doses: six on 0.25 mg, five on 0.5 mg, eight on 1.0 mg, and only four on 2.0 mg.
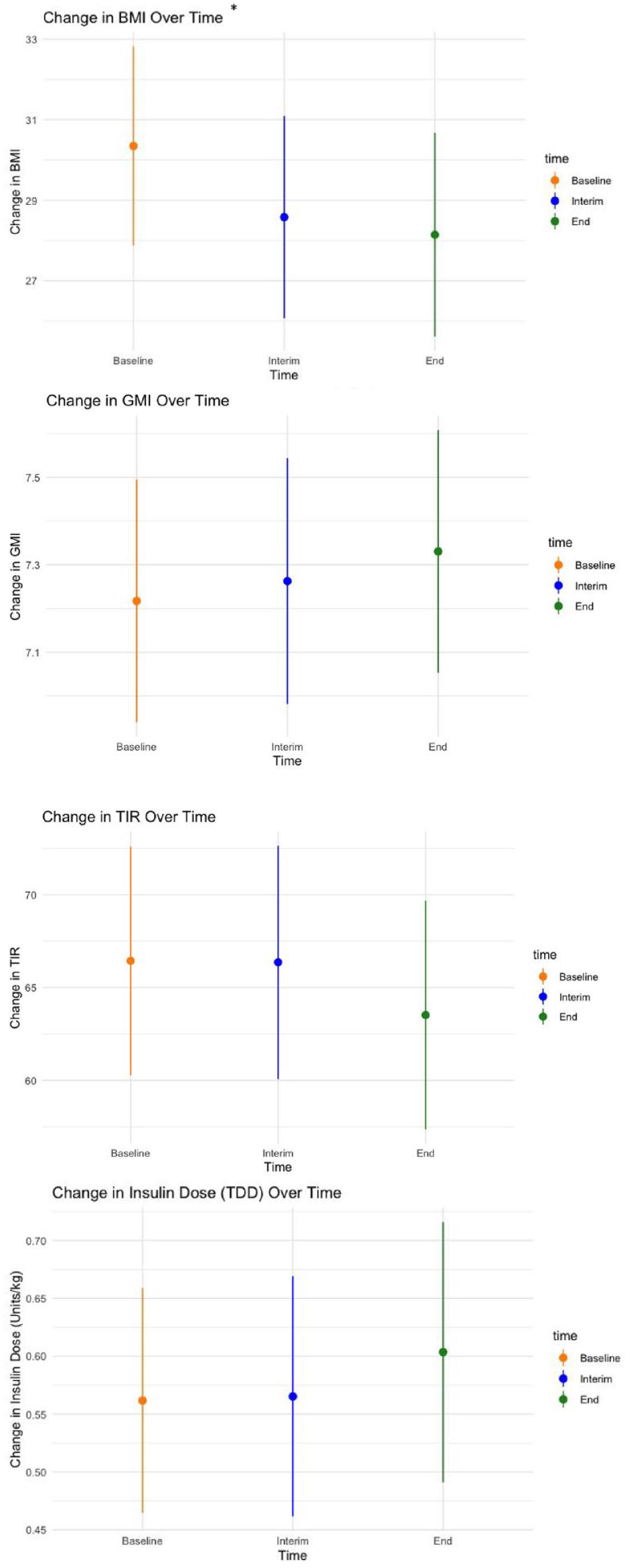
Changes in weight and continuous glucose monitoring (CGM) metrics over time during semaglutide treatment.
Despite this notable weight loss, semaglutide did not lead to significant improvements in glycemic control. Over the study period, no statistically significant changes were observed in glucose management indicators (GMIs) or time in range (TIR). The average total daily insulin dose (TDD) remained relatively unchanged, decreasing slightly from a mean of 47.7 ± 18 units/day (0.56 units/kg) at baseline to 46.9 ± 21.7 units/day (0.61 units/kg) by the conclusion, with no significant changes noted during follow-up.
Our results suggest that the primary therapeutic benefit of semaglutide in patients with T1D is modest weight loss rather than glycemic control. While semaglutide was effective in reducing weight, it does not appear to significantly impact glycemic control or insulin dosing requirements. This finding aligns with other smaller studies on semaglutide in T1D populations, which reported weight reduction but inconsistent improvements in glycemic outcomes.5,6
Although limited by a small sample size and observational design, our results suggest that semaglutide may be a promising option for addressing obesity in individuals with T1D. However, its impact on improving glycemic control remains unclear. Larger, randomized controlled trials are needed to investigate the broader implications of GLP-1RAs in this population, especially considering the cardiovascular and inflammatory risks associated with obesity in T1D.
Footnotes
Abbreviations
AID, automatic insulin delivery; BMI, body mass index; CGM, continuous glucose monitor; GLP-1RA, glucagon-like peptide 1 receptor agonist; GMI, glucose management indicator; HbA1c, hemoglobin A1c; TDD, total daily insulin dose; TIR, time in range; USC, University of Southern California.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
