Abstract
This study examined the short-term effects of a structured exercise prescription (ExerciseRx), combining afternoon interval exercise and postmeal walking, on meal-related glycemia using the Stelo glucose biosensor (GB; Dexcom) that displays real-time glucose data. Thirteen adults with type 2 diabetes, not on insulin, completed two successive ∼10-day sensor wear periods (blinded and unblinded to GB). Each wear period included a structured 4-h in-clinic visit under sedentary or ExerciseRx conditions. During a 10-day unblinded wear period, participants had higher time-in-range (74% ± 21% vs. 57% ± 29%, P < 0.001) and more total daily steps than the blinded period. During in-clinic visits, regardless of blinding, ExerciseRx lowered the 4-h mean (150 ± 31 vs. 168 ± 45 mg/dL, P < 0.01) and postmeal peak (213 ± 47 vs. 241 ± 63 mg/dL, P = 0.01) glucose compared with being sedentary. These findings support the value of structured ExerciseRx with a real-time GB to improve lifestyle habits in adults with type 2 diabetes not on insulin.
Introduction
Postprandial hyperglycemia and physical inactivity are major contributors to dysglycemia in individuals with type 2 diabetes (T2D) who are not on insulin therapy. 1 Meal-related glucose excursions are often pronounced after dinners with high caloric density and sedentary behavior. 1 Replacing sedentary behavior before dinner with a structured exercise prescription (ExerciseRx), as well as some light activity post dinner may help improve glycemia, particularly when paired with real-time continuous glucose monitoring (rtCGM) to help reinforce behavior change.
Glucose biosensors (GBs) are new rtCGM systems designed specifically for individuals without diabetes, with prediabetes, or with diabetes but not on insulin therapy. These systems offer improved visualizations to highlight personalized glucose trends and help users with T2D not on insulin to better understand the effects of food and exercise on their glycemia. In addition to possible glycemic improvement,2–4 thematic analyses from clinical trials suggest that rtCGM reinforces positive lifestyle and behavior changes and reduces diabetes stress and burden.2,5,6
While walking after meals can blunt postprandial excursions in T2D,7–9 interval-based activities (which are more intensive) also improve glycemia 10 while offering superior health and fitness benefits over light activity.11,12 More specifically, high-intensity interval training in the afternoon has been shown to result in better glucose management throughout the day than morning training in men with T2D. 13 An ExerciseRx consisting of predinner high-intensity interval walking followed by postdinner light walking, supported by a GB that visualizes glucose patterns, may help individuals better understand, and potentially adapt to, the effects of more intense exercise on meal-related glycemia.
The primary objective of this prospective study was to assess the feasibility and short-term glycemic effects of an ExerciseRx in CGM-naïve adults with noninsulin-treated T2D. As a secondary objective, we examined the additional influence of unblinded versus blinded GB use, recognizing that visibility of real-time glucose data is likely to independently support behavior change and glycemic improvements.
Methods
Study design and population
The study was performed at a single center (Centricity Research, Toronto), in accordance with the Declaration of Helsinki, and was approved by an independent ethic committee (WCG Institutional Review Board). Both written and verbal informed consent were obtained from all study subjects. Eligible participants with T2D (not on insulin) were aged ≥18 years with an HbA1c of ≥6.7% (58 mmol/mol), free of cardiovascular, renal, neurological, or psychological disease and injuries, naïve to CGM, and willing to wear a GB (Dexcom Stelo Glucose Biosensor) for the full study duration. Exclusion criteria included known allergies to adhesives, the presence of skin changes at the sensor wear site, pregnancy, end-stage renal disease, adherence to carbohydrate-restrictive diets, or immunosuppression.
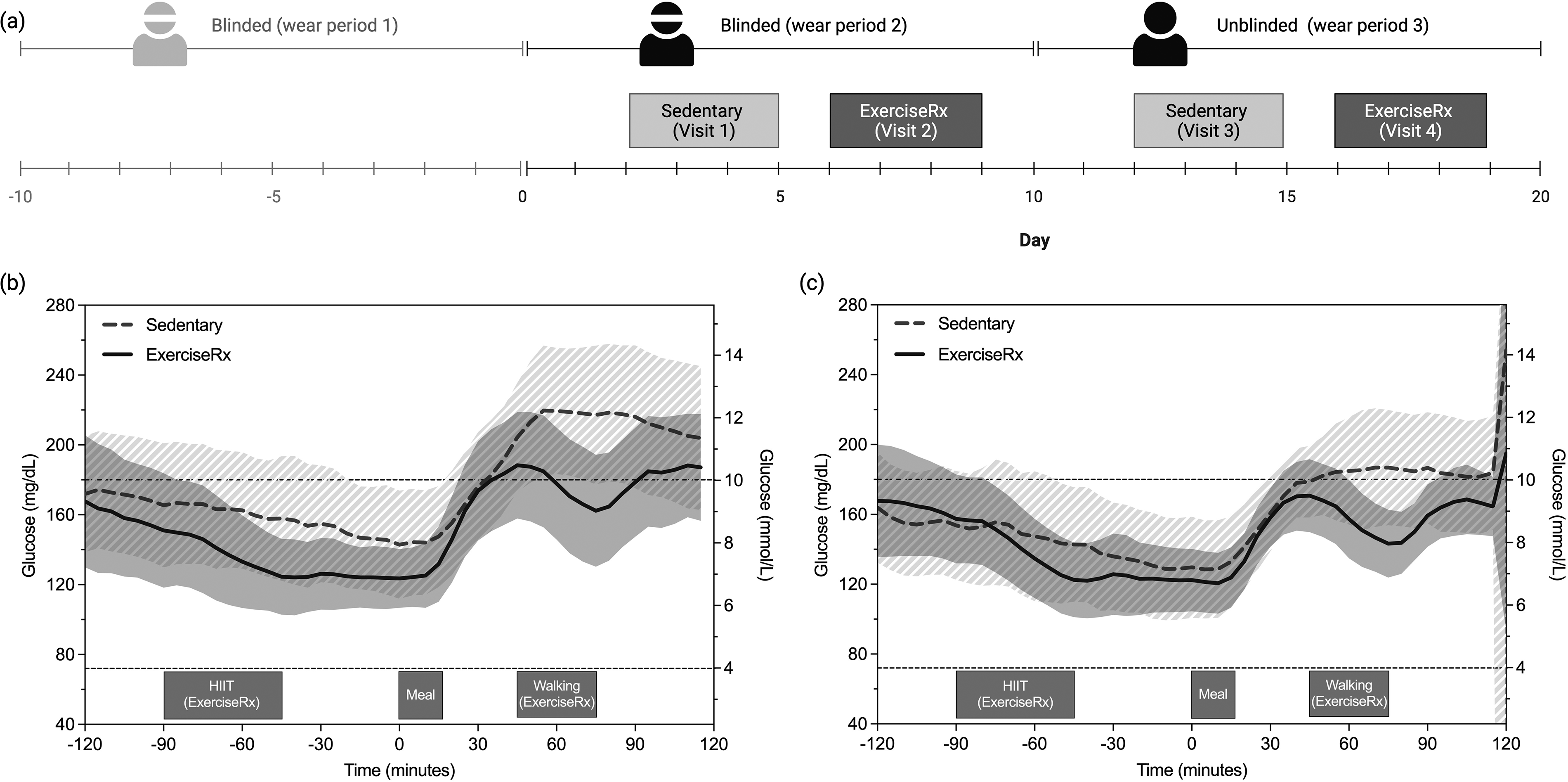
This feasibility study comprised three consecutive GB wear periods (Fig. 1a), each lasting up to 10.5 days. During the first and second wear periods, participants’ data were not visible to them (blinded). During the third wear period, participants’ test smartphone application displayed glucose and activity data in real time (unblinded). Only wear periods two (blinded) and three (unblinded), which included in-clinic visits were included in the present analysis. Hereafter, wear period two will be referred to as the blinded period and wear period three as the unblinded period. Throughout the study, participants logged their food intake using the MyFitnessPal app and wore a wrist-based activity tracker (Apple Watch) to monitor physical activity levels. Figure 2 shows the Stelo GB and some of its features available to the user.
A graphic of

A visual representation of the Stelo Glucose Biosensor with the personalized smartphone application and wrist-based activity tracker. Note, images are not to scale.
In-clinic visits
During the screening visit, participants underwent a modified Ebbeling submaximal treadmill walking test 14 to evaluate exercise-related peak heart rate (HRpeak) and maximal exercise capacity.
During the blinded and unblinded GB wear periods, participants attended two in-clinic visits each (i.e., one sedentary and one with an ExerciseRx) that featured a standardized dinner meal (∼60 g carbohydrate) at 17:30 (hh:mm) (Fig. 1a). The ExerciseRx visit included a 45-min premeal interval-based treadmill walking session (16:00–16:45) and a 30-min postdinner walk (18:15–18:45). The order of the visits was not randomized.
Data collection and analysis
CGM metrics were averaged by participant as: (1) Sedentary versus ExerciseRx (regardless of GB visibility); and (2) blinded versus unblinded (regardless of ExerciseRx). In-clinic CGM data were analyzed 120 min before to 120 min after meal initiation. To assess short-term effects over the ∼10 day blinded and unblinded wear periods, aggregated glycemic metrics and behavioral outcomes (step count and dietary intake, where available) were also compared.
Data normality was assessed using the Anderson–Darling test. In-clinic and short-term glycemic within-subject comparisons were analyzed using paired t-tests (normal distribution) or Wilcoxon matched-pairs signed-rank tests (non-normal distribution). Behavioral outcomes were also assessed for a subset of participants with available data. All analyses were exploratory and conducted using Prism version 10.4.2 (GraphPad, Boston, MA, USA), with significance defined as P < 0.05.
Results
Participant characteristics
Thirteen participants (4 females, 9 males) completed all four in-clinic visits and were included in the analysis. The cohort had a mean ± SD age of 52 ± 14 years, body mass index of 30.8 ± 7.8 kg/m2, HbA1c of 7.8 ± 0.9% (61 ± 10 mmol/mol), and diabetes duration of 7.5 ± 6.8 years. At enrollment, 6/13 participants were using noninsulin antihyperglycemic agents (2 ± 0.6 medications).
In-clinic glycemic effects of ExerciseRx
GB metrics stratified by the 2 h before and following meal initiation are shown in Table 1. Mean glucose concentration for the 4-h in-clinic period was significantly lower with ExerciseRx (150 ± 31 mg/dL vs.168 ± 45 mg/dL, P < 0.01, Fig. 1b–c). In the postmeal period, ExerciseRx significantly reduced peak glucose (213 ± 47 vs. 241 ± 63 mg/dL, P = 0.01) and net glucose area-under-the-curve (netAUC: 4281 ± 1789 vs. 5182 ± 2209 mg*min/dL, P = 0.04) relative to sedentary.
Glycemic Outcomes for (1) Sedentary Versus ExerciseRx and (2) Blinded Versus Unblinded Glucose Biosensor Conditions for the In-Clinic Visits (N = 13)
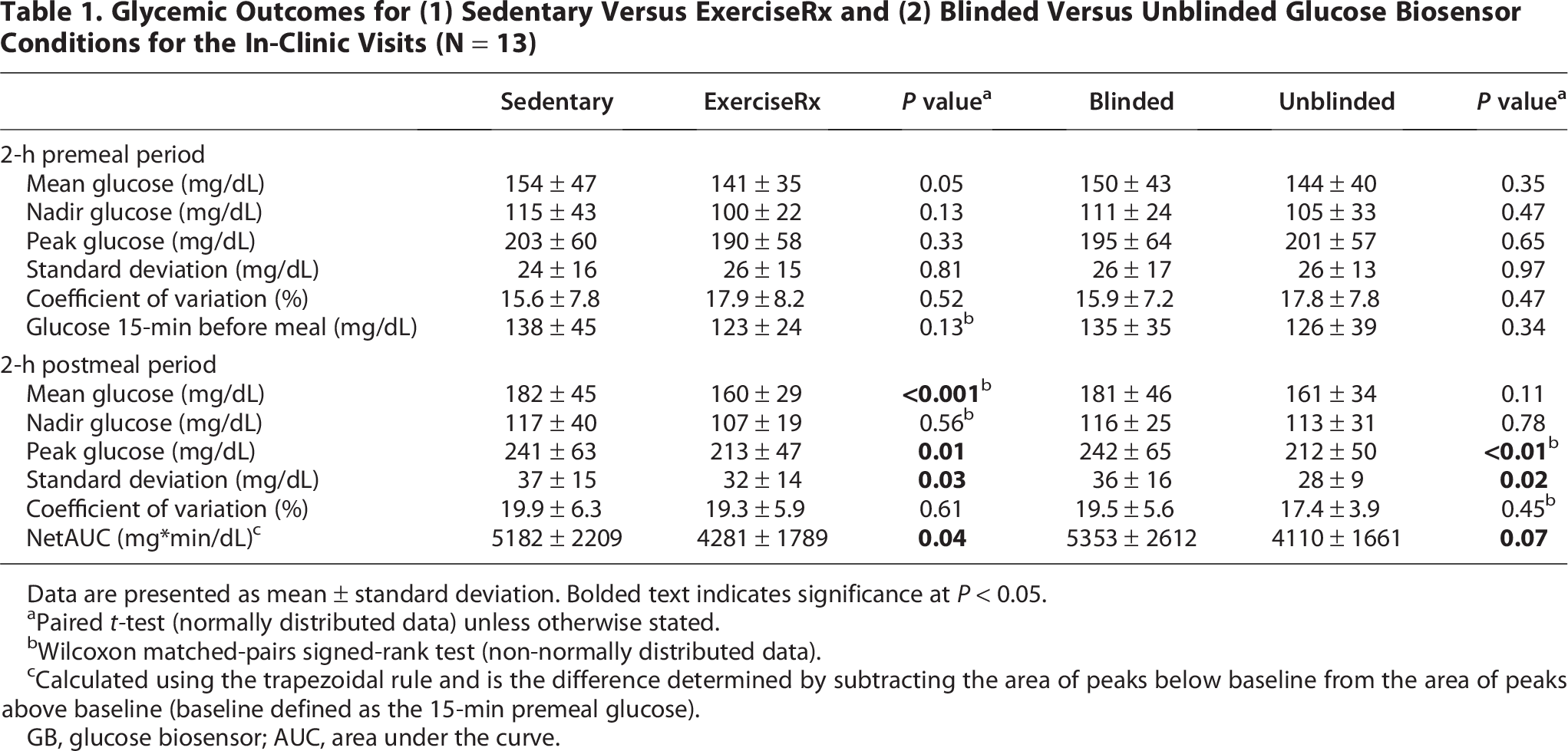
Data are presented as mean ± standard deviation. Bolded text indicates significance at P < 0.05.
Paired t-test (normally distributed data) unless otherwise stated.
Wilcoxon matched-pairs signed-rank test (non-normally distributed data).
Calculated using the trapezoidal rule and is the difference determined by subtracting the area of peaks below baseline from the area of peaks above baseline (baseline defined as the 15-min premeal glucose).
GB, glucose biosensor; AUC, area under the curve.
In-clinic glycemic effects of GB visibility
The 2-h premeal glucose metrics were similar between the blinded and unblinded GB wear periods (Table 1), however mean glucose concentration over the entire 4-h in-clinic period was lower with the unblinded versus blinded GB wear (153 ± 36 vs. 166 ± 44 mg/dL, P = 0.16, Fig. 1b–c). In the postmeal period, the unblinded visits were associated with a lower peak glucose level relative to the blinded visits (212 ± 50 vs. 242 ± 65 mg/dL, P < 0.01). Other glucose metrics also trended lower in the postmeal period with unblinded visits, with varying significance (Table 1).
Short-term effects of GB visibility
Over the ∼10-day wear periods, time in range (TIR: 70–180 mg/dL) was significantly higher with unblinded compared with blinded (74.0% ± 21.1% vs. 57.2% ± 29.4%, P < 0.001), as was time in tight range (TITR: 70–140 mg/dL; 39.4% ± 30.7% vs. 29.7% ± 31.7%, P = 0.01). Time above range >180 mg/dL was reduced with unblinded (25.8% ± 21.4% vs. 42.0% ± 30.5%, P < 0.001). Time <70 mg/dL was negligible (≤1%) across all wear periods.
In a subset of 5 participants with sufficient step count data for analyses, there was no difference in step count between the blinded and unblinded wear periods (6471 ± 2635 steps/day vs. 6617 ± 5406, P = 0.94). Among those with sufficient dietary data (n = 9), mean daily calorie intake was 52 ± 256 kcal/day lower during the unblinded than the blinded wear period (1464 ± 572 kcal/day, P = 0.56), with modest reductions in carbohydrate (−10 ± 44 g/day, P = 0.53) and protein (−5 ± 13 g/day, P = 0.24), and increase in fat (1 ± 14 g/day, P = 0.86) intake.
Discussion
This feasibility study demonstrated that a combined ExerciseRx consisting of predinner high-intensity interval walking followed by postdinner light walking, supported with unblinded GB wear is achievable and beneficial for short-term improvements in various glycemic metrics in CGM-naïve adults with T2D not on insulin. Over the full ∼10-day wear period, there was also a ∼17% increase in TIR with unblinded GB compared with blinded GB. These findings contribute to the growing interest in individualized lifestyle strategies that leverage rtCGM data to reinforce behavior change and improve glycemia in this population. 15 Although current physical activity guidelines for T2D emphasize total weekly activity of 150 min per week of accumulated moderate-to-vigorous intensity physical activity, or 75 min of very vigorous exercise, 16 they offer limited direction on how exercise timing around meals may influence glycemia. Our findings add to emerging literature and recommendations that support activity timed around meals for glycemic management in T2D. 17
The main aim of this study was to assess the feasibility and short-term glycemic impact of a meal-timed ExerciseRx in CGM-naïve adults with noninsulin-treated T2D. Prior studies have demonstrated that both interval exercise 10 and postprandial walking7–9 improve glycemia in T2D. More recently, real-world CGM data in individuals with newly diagnosed T2D demonstrated that days including structured exercise bouts ≥10 min (aerobic, interval, or strength training) were associated with a 0.2 mmol/L (3.6 mg/dL) lower mean 24-h glucose compared with nonexercise days. 18 While these findings highlight the general benefits of physical activity, our study was unique in being the first, to our knowledge, to test the sequential use of interval and walking exercise around dinner, a time typically associated with large glycemic excursions. 1 Combining interval exercise with postmeal walking was associated with a mean glucose reduction of ∼20 mg/dL compared with the sedentary condition during the 4-h in-clinic observation period. This effect appeared to be driven by reductions in postprandial glucose, as only modest differences were observed before the meal. This supports the findings of others that high-intensity exercise may not be associated with immediate reductions in glycemia in individuals with T2D, but that glucose can be lower in the hours following the activity, including around meals.10,13,19 High-intensity interval training can increase circulating catecholamines, at least transiently, which can promote hepatic glucose output and acutely promote hyperglycemia. 20 However, these hormonal responses return to baseline within an hour after intensive activity, 20 and were therefore unlikely to have largely influenced glucose during the postprandial window in this study. In contrast, the glycemic improvements observed after dinner likely reflect a synergistic effect of both exercise types used for the intervention. Of note, premeal high-intensity exercise can enhance skeletal muscle glucose uptake by stimulating contraction-mediated glucose uptake into muscle and increasing muscle microvascular perfusion. 21 When combined with insulin-mediated glucose uptake from the meal, and the additional glucose clearance stimulated by post-meal walking, the exercise sequence of the present study may have potentiated postprandial glycemic benefits.
While an ExerciseRx should not be deemed as essential for the adoption of a physically active lifestyle, some evidence suggests that exercise prescriptions which detail activity type, intensity, frequency, and duration are more effective than vague advice from providers. 22 Independent of exercise, a secondary aim of this analysis was to explore whether an unblinded real-time GB with activity and meal-related feedback appeared to support both at-home and in-clinic glycemic improvements. Modest improvements in pre- and postmeal glucose were observed during unblinded in-clinic visits, but given that meals and exercise were standardized, these differences should be interpreted with caution. It is possible that real-time GB feedback influenced behavior through heightened motivation or altered effort in-clinic, but other factors (such as previsit activity, food intake, or stress) may have also contributed to these differences. This study and others4,5 highlight that short-term CGM use may benefit individuals with noninsulin-treated T2D by increasing awareness on the effects of meals and exercise on glycemia.23,24 An earlier feasibility and pilot study by Bailey et al. 25 found that individuals with prediabetes or diabetes who used rtCGM to self-monitor glucose and activity during an 8-week exercise intervention reported greater self-efficacy and more frequent exercise compared with those receiving standard care, benefits which were largely attributed to the motivational effect of seeing real-time glucose trends. Collectively, these findings underscore the potential for GB to function as a behavioral feedback tool, which may help enhance self-awareness and promote sustained exercise and lifestyle improvements in this population.
The strengths of this study include the use of a exercise prescription paired with a GB smartphone application that integrates both sensor glucose and activity data, the evaluation of glycemic effects in-clinic, and focus on a population often underrepresented in CGM and exercise research. However, the small sample size and the short study duration limit the study generalizability. Moreover, the sustainability of the observed glycemic benefits associated with exercise commitment (45-min high-intensity and 30-min postmeal walking) remains uncertain in this population where behavior change is acknowledged to be challenging. 26 Additionally, the observational design does not allow for comparisons across different activity timing strategies (e.g., pre- vs. postmeal exercise alone) or time-of-day effects and we could not randomize the order of visits, since all the subjects were CGM- and exercise-naïve which precluded randomization for logistical reasons (i.e., difficult to “wash out” possible behavior modifications caused by first wearing unblinded GB). In conclusion, this feasibility study suggests that an ExerciseRx combining high-intensity interval training before dinner with postmeal walking may help reduce postprandial hyperglycemia in individuals with noninsulin-treated T2D. The use of a real-time GB with exercise integration and a structured ExerciseRx may further enhance engagement by highlighting the immediate impact of behavior change on glucose levels. Larger and longer-term studies with different exercise timing and types are needed to confirm these findings and explore their practicality.
Authors’ Contributions
L.V.T.: Formal analysis (equal); writing—original draft (equal); writing—review and editing (equal). C.C.: Formal analysis (equal); writing—review and editing (equal). P.G.: Formal analysis (equal); writing—review and editing (equal). S.E.B.: writing—review and editing (equal). M.L.J.: Conceptualization (equal); writing—review and editing (equal). M.C.R.: Conceptualization (equal); writing—original draft (equal); writing—review and editing (equal). M.C.R. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
Acknowledgments
The authors thank all the study site staff and participants for their contribution to this study.
Author Disclosure Statement
L.V.T. has received in kind research support from Dexcom, Inc. C.C., P.G., and M.L.J. are employees of Dexcom, Inc. M.C.R. reports speaker fees from Novo Nordisk, Sanofi, and Dexcom, Inc.; receives consultant fees from Insulet Corporation, Eli Lilly and Company, and Dexcom, Inc.; and is a stock and shareholder of Zucara Therapeutics.
Funding Information
Funding for the study was provided by Dexcom, Inc.
