Abstract
Background:
The management of idiopathic membranous nephropathy (IMN) continues to be a significant challenge to date. A limited number of observational studies have compared Obinutuzumab’s (OBZ) therapeutic potential, with OBZ appearing to be more effective than rituximab (RTX) in achieving clinical remission.
Objective:
This study was designed to evaluate the effectiveness and safety profile of OBZ for IMN treatment.
Design:
Clinical studies regarding the application of OBZ for IMN were included, and the effector sizes were pooled by meta-analysis. Sensitivity analysis and subgroup analysis were adopted to evaluate the robustness and reliability of the findings.
Data sources and methods:
Several electronic databases were systematically searched for relevant studies. Effectors, including the rates of total remission (TR) and complete remission (CR), reported in these studies were extracted and pooled as non-comparative binary variables. The rates of immunologic remission, B-cell depletion, and adverse events (AEs) were recorded.
Results:
Among the studies retrieved in the initial search, 9 eligible studies with 344 participants were included in this meta-analysis. OBZ therapy increased the TR rates at 6 months (0.78 (0.71, 0.85)) and 12 months (0.88 (0.82, 0.93)) and improved the CR rates at 6 months (0.13 (0.03, 0.23)) and 12 months (0.27 (0.16, 0.39)) compared to RTX. No evident heterogeneity or publication bias was observed. The results persisted in the subgroup analysis in terms of OBZ, as the initial treatment for membranous nephropathy (MN), for refractory MN, or for RTX-refractory MN. Interestingly, OBZ treatment was linked to a greater probability of achieving TR than RTX treatment (At 6 months, odds ratio was 4.07 (1.94, 8.53)) and (At 12 months, odds ratio was 10.79 (3.38, 34.39)). OBZ therapy consistently led to a relatively high rate of immunological remission and effective B-cell clearance and did not increase the incidence of AEs or severe AEs.
Conclusion:
OBZ therapy is associated with improved clinical remission and might be an attractive option for patients with IMN, especially those with refractory or RTX-refractory IMN. High-quality clinical studies are, however, warranted to validate these results further.
Trial registration:
This study was registered as a meta-analysis in PROSPERO (Number: CRD42025635162).
Plain language summary
Idiopathic membranous nephropathy (IMN) is a type of kidney disease, and its treatment can be quite challenging. Recent research showes that obinutuzumab (OBZ) may be more effective than rituximab (RTX) in helping patients achieve kidney remission, which indicates their proteinuria decreases greatly or returns to normal. This study analyzed data from recent clinical studies involving a total of 344 patients to assess how well OBZ works and how safe it is. The results showed that OBZ treatment increased the likelihood of both overall and complete remission at 6 and 12 months. Patients treated with OBZ were more likely to experience kidney improvement compared to those receiving RTX. Additionally, OBZ efficiently cleared harmful immune cells without raising the risk of side effects or serious adverse events. Overall, OBZ appears to be a promising treatment option for IMN, especially for patients who do not respond well to other therapies like RTX. However, more high-quality clinical studies are needed to confirm these findings. Still, OBZ could have the potential to become an alternative agent in managing this challenging kidney disease.
Introduction
Idiopathic membranous nephropathy (IMN) is believed to develop from autoimmune conditions, such as antibodies developing against the M-type phospholipase A2 receptor (PLA2R) and other novel target antigens, which represent more than 80% of cases. 1 Secondary membranous nephropathy (MN) is caused by preexisting disease conditions, such as those arising due to drugs, malignancies, and infectious diseases. 2 IMN remains the primary cause of nephrotic syndrome (NS) in adults currently. Approximately 20%–30% of patients with IMN experience spontaneous remission with supportive therapy, 3 whereas 40%–50% of NS patients progress to chronic kidney failure within 10–15 years, particularly those with increased anti-PLA2R antibody (anti-PLA2Rab) levels.4,5
The latest KDIGO guidelines recommend that patients with IMN who are at low risk of disease progression should receive optimized supportive therapy, and those with a high or very high risk of disease progression should be considered for immunosuppressive therapy. 6 Traditional immunosuppressants, including glucocorticoids, cyclophosphamide (CTX), and calcineurin inhibitors (CNIs), are the mainstream treatment agents for IMN, which achieve relatively high rates of overall clinical remission. 7 However, substantial side effects have been associated with these immunosuppressors, such as hyperglycemia, obesity, infections, and tumors, raising considerable concerns.8,9 In recent years, B lymphocytes, which produce pathogenic antibodies, such as the anti-PLA2Rab, have been confirmed to exert a vital role during IMN pathogenesis and progression. Accordingly, monoclonal antibodies with selective B-cell depletion were developed to treat IMN. Rituximab (RTX) is a chimeric anti-CD20 antibody that can effectively deplete CD19+ or CD20+ B lymphocytes to significantly decrease anti-PLA2Rab production, thereby achieving clinical remission for IMN with few side effects.10–12 Therefore, RTX is recommended as one of the first-line treatments for IMN in the updated guidelines. 6 However, about 30%-40% of patients with IMN remain unresponsive or resistant to RTX and experience treatment failure.7,10 This challenge for clinicians demands the development of novel therapies, including newer monoclonal antibodies targeting the CD20 molecule. 13
Obinutuzumab (OBZ) is a newly developed and fully humanized type II anti-CD20 antibody that was originally designed for RTX-refractory B-cell malignancies. 14 Numerous studies show OBZ is superior to RTX in inducing more profound B-cell clearance in blood samples or tissues.14,15 As a recently developed anti-CD20 antibody with more efficacy in B-cell clearance, OBZ provides a rationale for potential use in patients with IMN. 16 Recently, several observational studies have been carried out to observe and assess the effectiveness and potential application of OBZ, and the findings have demonstrated that OBZ is associated with good clinical remission in the treatment of IMN.17–21 However, such studies have been small-scale, non-randomized controlled investigations, and no systematic reviews or meta-analyses to date have validated these study findings. In this context, the present study, designed as a meta-analysis, was conducted to systematically assess the efficacy and safety profile of OBZ for IMN by synthesizing the effectors from these clinical studies.
Methods
This study was registered as a meta-analysis in PROSPERO (CRD42025635162), performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Statement 22 and the guidelines for Meta-analysis of Observational Studies in Epidemiology (MOOSE) reporting.
Search strategy
The electronic databases, including MEDLINE, EMBASE, Web of Science, the Cochrane Library, and China National Knowledge Infrastructure, were systematically searched for relevant studies from database inception until April 21, 2025. Furthermore, the citations of the eligible studies were also searched and reviewed. The search strategy developed according to the PICOM was listed as follows:
The keyword-based search strategy was listed in Supplemental Material.
Study selection
All clinical studies that evaluated the efficacy and/or safety of OBZ for MN were initially chosen for analysis. The specified inclusion criteria were as follows: (a) studies that included participants aged above 18 years and (b) studies that evaluated OBZ’s efficacy and safety in patients with MN. The studies that had a sample size of <10 participants, were conducted on secondary MN or another glomerulonephritis, or had inadequate data for analysis, were excluded. Two authors performed the study selection process.
Primary and secondary outcomes
The total remission (TR) rate and complete remission (CR) rate were used as the primary outcomes. CR was characterized by either achieving proteinuria levels below 0.3 g/day or demonstrating a reduction in the urine protein to creatinine ratio (UPCR) from baseline values to under 0.3 g/g accompanied by stable estimated glomerular filtration rate (eGFR). Partial remission (PR) was characterized by either achieving proteinuria levels below 3.5 g/day or demonstrating a reduction in the UPCR by over 50% from baseline value to below 3.5 g/g, accompanied by stable eGFR. The TR rate was CR plus PR. Relapses were characterized by the recurrence of proteinuria exceeding 3.5 g or a UPCR exceeding 3.5 g/g. Immunologic response was defined as an anti-PLA2Rab titer <5 RU/mL. B-cell depletion was defined as counts of CD19+ or CD20+ cells <5/μL. Refractory MN was defined as resistance to immunosuppressive treatment for more than 6 months.6,23 RTX-refractory or resistant MN was defined as the inability to achieve either CR or PR more than 6 months following the final dose of RTX. 20 Immunologic response, relapse, adverse events (AEs) and severe AEs (SAEs) were considered secondary outcomes.
Data extraction and quality assessment
Basic characteristics, including first author, publication year, OBZ dose, and period of follow-up, were extracted from each of the included studies. The outcome variables, including the eGFR and the number of CR, PR, immunologic response, disease relapse, and SAE cases, were also collected. Other interesting variables, such as the counts of CD20+ or CD19+ B lymphocytes and the anti-PLA2Rab levels, were recorded. The included studies were subjected to an assessment of the risk of bias. Specifically, randomized controlled trials (RCTs) were evaluated using the risk of bias tool, and one-arm trials or cohort studies with a control were evaluated using the methodological index for non-randomized studies (MINORS) criteria. 24 Two reviewers independently extracted the relevant information and assessed the study quality. Potential disputes should be settled through consultation with another author.
Statistical analysis
Review Manager 5.3 (Cochrane Collaboration, Copenhagen, Denmark) and Stata12.0 (StataCorp LP, College Station, TX, USA) were employed for data analysis. Clinical parameters, including TR, CR, relapses, and AEs, were extracted or calculated, and the final values were pooled. Among the nine studies included in the meta-analysis, six are single-arm studies, and the remaining three are case-control studies with unbalanced control groups. We performed a pre-post analysis to compare outcomes within the OBZ-treated cohort. Thus, the effect sizes in these studies are treated as non-comparative binary variables. Consequently, the parameters, such as TR and CR, were first transformed on the basis of the formula presented in the previous reports25–27 (Supplemental Material), following which the final values were pooled to obtain the preliminary effector size. The obtained preliminary effector size was transformed to derive the final effector size using the following conversion formulas presented in the Supplemental Material. The heterogeneity of the meta-analysis was examined by the I2 statistic. Specifically, if I2 is less than 50%, the fixed effects model was used; and if I2 is more than 50%, the random effects model was used to combine effect sizes. Sensitivity analysis and subgroup analysis were performed to determine the robustness and reliability of the findings. Begg’s and Egger’s tests were selected for the evaluation of publication bias. A p-value less than 0.05 (two-tailed) was deemed to indicate statistical significance.
Results
Study selection
A total of 2757 citations were retrieved from the 5 databases. A total of 1002 duplicates and 1667 irrelevant records were removed after screening. Eleven studies were subsequently identified in the qualitative analysis, and 9 articles enrolling 344 participants were deemed eligible for the quantitative analysis20,21,28–34 after excluding 2 articles because of duplication 35 and ineligible participants. 36 The selection process of this meta-analysis is depicted in Figure 1.

Study screening flow.
Characteristics of the eligible studies
All included studies, except for Teisseyre’s study, were conducted in China and were published from 2024 to 2025. The treatment protocols in these studies included OBZ monotherapy or in combination with the concurrent use of low-dose or tapered immunosuppressants.20,21,29,30,32 Among the studies, six one-arm studies were conducted to assess the efficacy and safety of OBZ,20,29–32,34 and the remaining three RTX-controlled studies were conducted to compare the efficacy of OBZ with that of RTX.21,28,33 None of the studies were RCTs. The average quality score of the six one-arm studies was 10.6 points, while that of the other three studies was 18.7 points. The baseline characteristics and MINORS scores of the eligible studies are listed in Table 1.
Characteristics of the included studies.
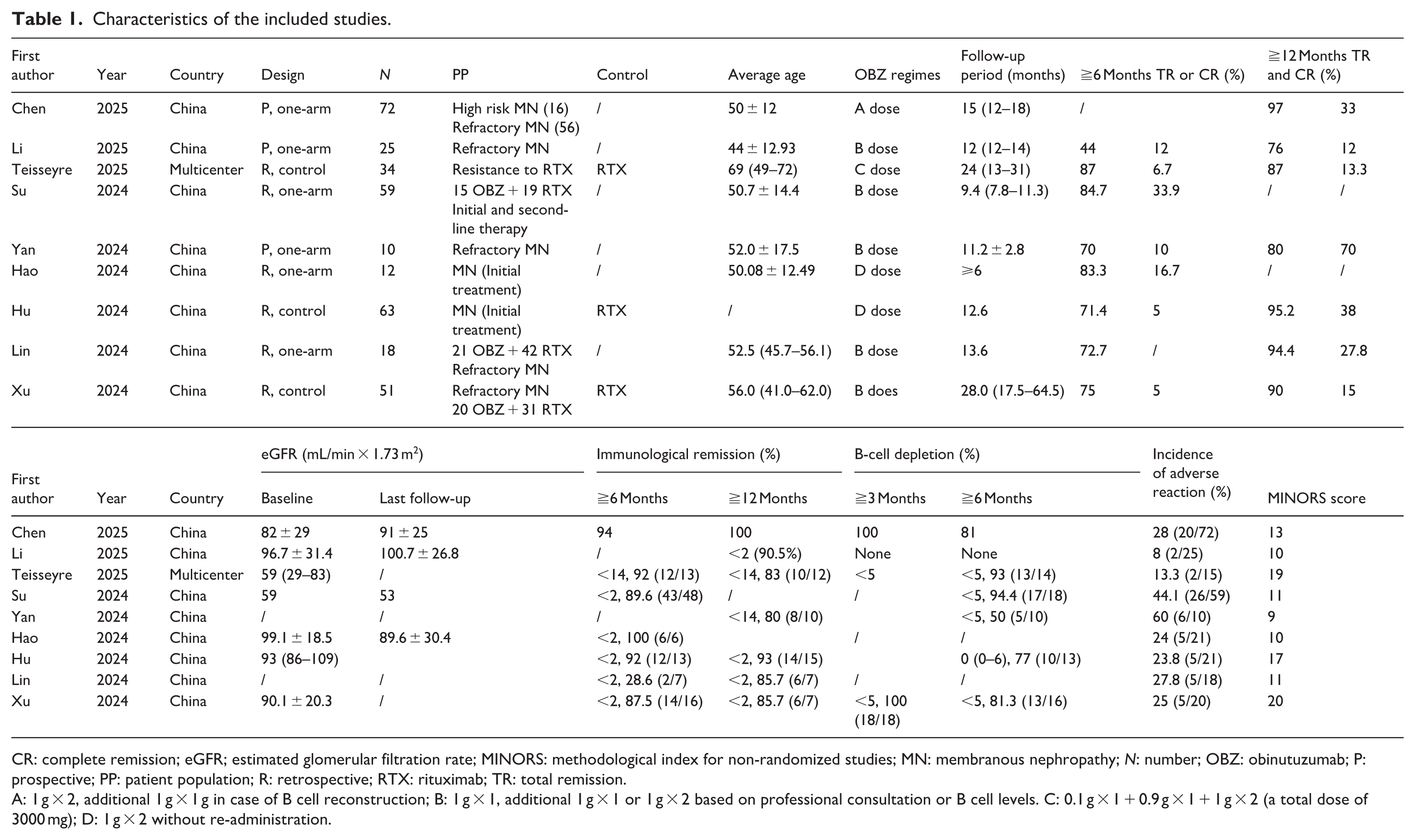
CR: complete remission; eGFR; estimated glomerular filtration rate; MINORS: methodological index for non-randomized studies; MN: membranous nephropathy; N: number; OBZ: obinutuzumab; P: prospective; PP: patient population; R: retrospective; RTX: rituximab; TR: total remission.
A: 1 g × 2, additional 1 g × 1 g in case of B cell reconstruction; B: 1 g × 1, additional 1 g × 1 or 1 g × 2 based on professional consultation or B cell levels. C: 0.1 g × 1 + 0.9 g × 1 + 1 g × 2 (a total dose of 3000 mg); D: 1 g × 2 without re-administration.
Primary outcomes
TR rate
Seven studies reported the TR rate at 6 months,20,21,28,31–34 while seven other studies reported the TR rate at 12 months,21,28–33 with stable eGFRs after OBZ treatment. The pooled odds ratio was 3.68 (2.48, 5.46) at 6 months (in Figure 2) and 7.41 (4.35, 12.63) (in Figure 4) at 12 months. The calculated TR rate was 0.78 (0.71, 0.85) at 6 months and 0.88 (0.82, 0.93) at 12 months. Heterogeneity was not present among studies (p = 0.72, I2 = 0% at 6 months or p = 0.11, I2 = 42% at 12 months), and a fixed model was applied. The sensitivity analysis shows that the results are robust. No publication bias was observed at 6 months (Figure 3, Begg’s Test p = 0.764; Egger’s test p = 0.698) or 12 months (Figure 5, Begg’s Test p = 0.764; Egger’s Test p = 0.115).

The pooled TR of OBZ in MN at 6 months.

The Funnel plot of pooled TR of OBZ in MN at 6 months.

The pooled TR of OBZ in MN at 12 months.

The Funnel plot of pooled TR of OBZ in MN at 12 months.
CR rate
Six studies reported CR rates at 6 months,20,21,28,31,33,34 and seven studies reported CR rates at 12 months.21,28–31,33 The pooled CR rate was 0.13 (0.03, 0.23) (in Figure 6) at 6 months and 0.27 (0.16, 0.39) (in Figure 8) at 12 months. Heterogeneity was present across studies ((p = 0.003, I2 = 73%, at 6 months) or (p = 0.02, I2 = 71%, at 12 months)), and a random effects model was applied. No publication bias was present at 6 months (Figure 7, Begg’s Test p = 0.133; Egger’s Test p = 0.481) or 12 months (Figure 9, Begg’s Test p = 0.230; Egger’s Test p = 0.347).

The pooled CR rate of OBZ in MN at 6 months.

The Funnel plot of pooled CR rate of OBZ in MN at 6 months.

The pooled CR rate of OBZ in MN at 12 months.
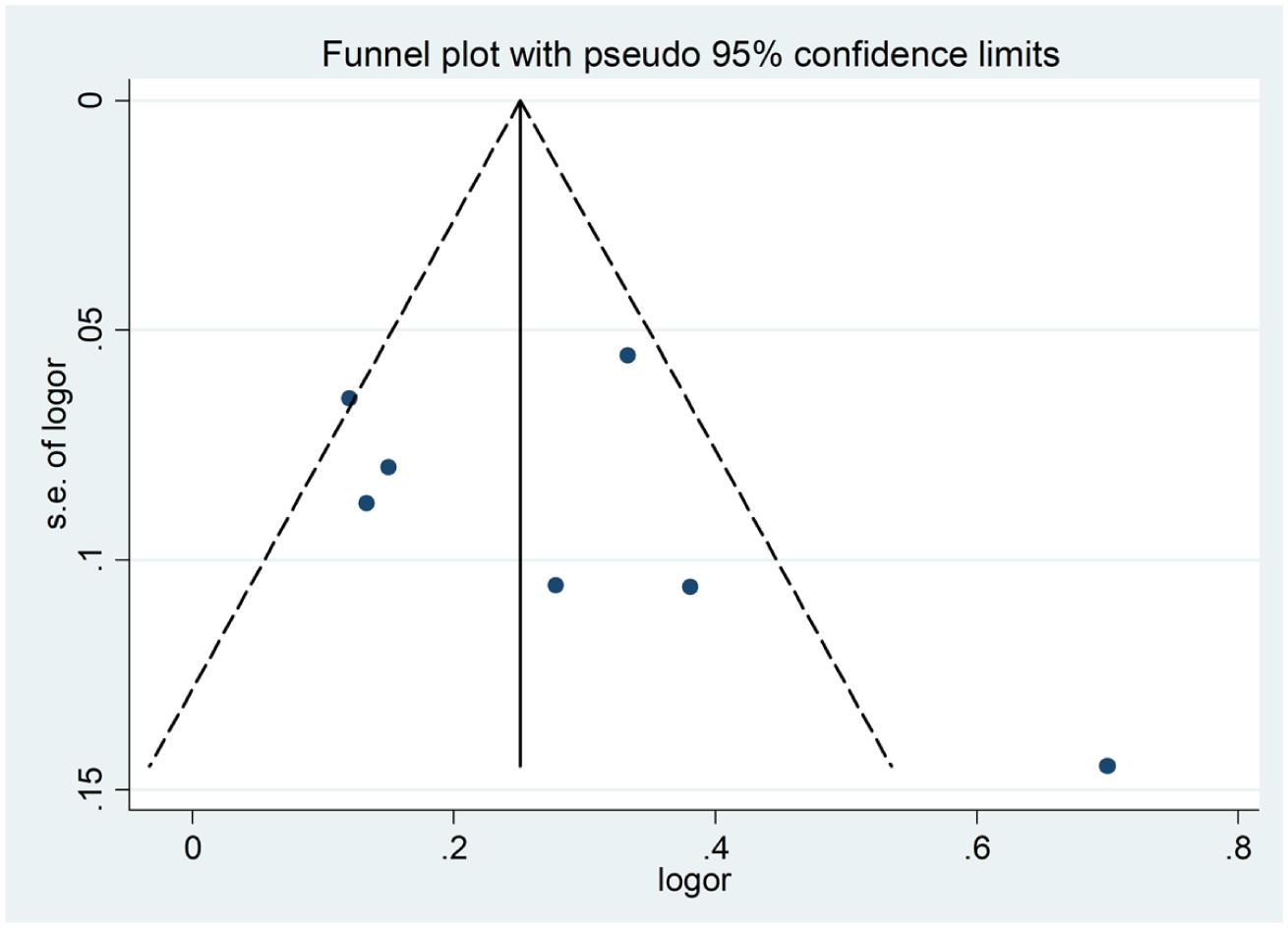
The Funnel plot of pooled CR rate OBZ in MN at 12 months.
Secondary outcomes
TR rate when OBZ was used as initial monotherapy for IMN
Four studies reported TR rates when using OBZ as the initial monotherapy for MN.20,30,33,34 At 6 months, the calculated TR rate was 0.52 (0.37, 0.63) (data not shown). At 12 months, the pooled odds ratio was 2.85 (1.43, 4.28), with no heterogeneity (p = 0.84; I2 = 0%) in two studies,30,34 and the calculated TR rate increased to 0.74 (0.59, 0.81), as shown in Figure 10.

The results of subgroup analysis.
TR rate when OBZ was used for refractory MN
Six studies reported TR rates using OBZ for refractory MN patients.20,21,29–32 The pooled odds ratio was 0.72 (0.36, 1.09) at 6 months (data not shown), and the estimated TR rate was 0.42 (0.26, 0.52) (data not shown). At 12 months, the pooled odds ratio was 1.76 (1.28, 2.24), with no heterogeneity (p = 0.14; I2 = 39%), as shown in Figure 10, and the calculated TR rate increased to 0.64 (0.37, 0.45).
TR rate when OBZ was used for RTX-refractory MN
Two studies reported TR rates of OBZ for RTX-refractory MN.20,28 The pooled odds ratio was 1.04 (0.16, 1.92) at 6 months, with no heterogeneity (p = 0.17; I2 = 46%), as shown in Figure 10, and after conversion, the estimated TR rate was 0.51 (0.14, 0.66).
Comparison of OBZ and RTX in terms of efficacy
Three studies were conducted to compare the efficacy of OBZ with that of RTX at both 6 and 12 months.21,28,33 The pooled odds ratio was 4.07 (1.94, 8.53), with no heterogeneity (p = 0.24; I2 = 30%) at 6 months, as shown in Figure 11. Interestingly, the effect increased to 10.79 (3.38, 34.39), with no heterogeneity (p = 0.93; I2 = 0%) at 12 months, as shown in Figure 12.
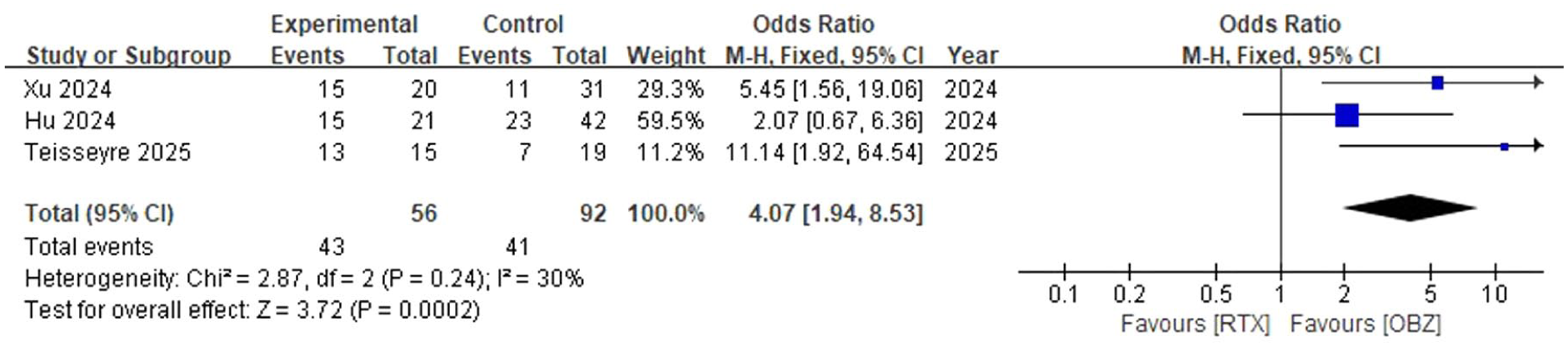
Comparison of the efficacy of OBZ with RTX at 6 months.

Comparison of the efficacy of OBZ with RTX at 12 months.
Rates of immunological remission and B-cell depletion
All nine studies reported the rates of immunological remission and B-cell clearance after OBZ treatment. 34 The immunological remission rates ranged from 87.5% to 94% at 6 months and from 80% to 100% at 12 months. Similarly, the rates of B-cell depletion ranged from 54.5% to 94.4% at 6 months (Table 1).
Kidney function, AEs, and SAEs
Seven studies reported stable kidney function in terms of eGFR,20,21,28–30,33,34 and the remaining two reported stable kidney function in terms of creatinine level31,32 throughout the follow-up period, as shown in Table 1. All the studies reported AEs. In brief, the incidence of AEs ranged from 8% to 60%, mainly infusion-related reactions, and no SAEs were reported during drug infusion or follow-up, as shown in Table 1. Compared to RTX, OBZ does not increase the risk of AEs or SAEs.21,28,33
Discussion
The available studies on the clinical application of OBZ for the treatment of IMN patients were limited. It is, therefore, necessary to systematically evaluate this novel anti-CD20 antibody’s efficiency and safety by pooling the current data. In this study, such data were aggregated, and it was understood that OBZ significantly increased the TR and CR rates for the treatment of IMN, including initial therapy, refractory MN, and RTX-refractory MN.10,37 The results of this study provide preliminary evidence supporting the potential application of OBZ in IMN patients in the future.
MN is currently the primary form of NS in adults, and about one-third of these patients are resistant or intolerant to standard therapy, with this patient population being associated with a poor prognosis.3,6 The discovery and identification of novel nephritogenic antigens have significantly increased our understanding of MN pathogenesis. 38 The identification of the PLA2Rab has provided a theoretical foundation for the development of therapeutic interventions that target B-cell lineages to remove pathogenic immune complexes. 39 RTX is a first-generation chimeric monoclonal antibody targeting the CD20 molecule and has been recommended as one of the first-line therapies for IMN, especially for those with a high risk of progression.6,10 However, 30%–40% of IMN patients develop resistance to RTX or relapse after RTX-based regimens.9,40 Therefore, it is imperative to explore alternative therapies and develop new monoclonal antibodies for the treatment of refractory MN. 40
Therefore, OBZ was engineered, developed, and introduced to address this issue. OBZ is a second-generation monoclonal antibody against the CD20 antigen expressed in B cells and exerts a profound and sustained ability to eliminate pathogenic B cells in clinical settings. 41 Currently, OBZ is known to be more effective in treating autoimmune diseases.42–46 Moreover, emerging clinical evidence indicates OBZ is an efficacious and safe therapy for patients with proteinuria diseases such as refractory NS.47,48 Furthermore, recent evidence has demonstrated OBZ can be used effectively for patients with refractory IMN who are resistant to standard treatment or RTX.19–21,34,49 For example, Sethi et al. first assessed the therapeutic effectiveness of OBZ in refractory MN in 2020 49 using 10 MN patients (1 secondary MN patient and 1 MN patient in the allograft) with resistance to RTX or suppressive agents and reported that 9 of these patients achieved CR and PR after OBZ treatment. Recently, Su et al. retrospectively analyzed 59 IMN patients with OBZ treatment, 20 with 20 patients receiving OBZ as the first-line therapy (not receiving any immunosuppressants) while the remaining patients received OBZ as the second-line therapy (resistant to other immunosuppressants, including RTX). The authors reported that 90% and 82% overall remission rates were observed for these patients, respectively. Interestingly, for patients who were resistant to RTX treatment, the TR rate reached 64.3%. Similar results were also reported in certain other recent studies.21,32
Although these studies have preliminarily confirmed the good efficacy of OBZ for IMN, the studies were small, non-randomized clinical works with numerous inherent limitations. Therefore, the present meta-analysis was performed to address this issue. Nine studies enrolling 344 participants with IMN were included in this study. The meta-analysis revealed that at 6 months, the rates of TR and CR for IMN were 78% and 13%, respectively, and at 12 months, these rates increased further to 88% and 27%, respectively, both higher than RTX in the MENTOR study. 10 Moreover, in the subgroup analysis, the clinical efficacy of OBZ either as initial treatment for MN or OBZ for refractory MN or RTX-refractory MN was consistently demonstrated at both 6 and 12 months. Besides, it is observed that during OBZ treatment, other concomitant immunosuppressants in refractory MN could be successfully withdrawn or tapered, thereby minimizing their adverse side effects in the long term. The findings showed that OBZ treatment is more likely to achieve clinical remission, and these beneficial effects become more pronounced over time.10,26 Interestingly, compared to RTX, OBZ treatment was found to be associated with a greater likelihood of clinical remission (At 6 months, odds ratio: 4.07 (1.94, 8.53)) versus (At 12 months, odds ratio: 10.79 (3.38, 34.39)), demonstrating the possible superiority of OBZ over RTX. Moreover, OBZ treatment was associated with relatively high rates of immunologic remission and B-cell depletion and did not increase the risk of AEs or SAEs. Taken together, these results verified the potential clinical efficacy and safety of OBZ therapy for IMN. However, our results stem from a limited set of observational data, and interpretation should be cautious; future head-to-head RCTs are essential to establish its efficacy and superiority.
There are some explanations for the improved efficacy of OBZ. First, RTX is a type I anti-CD20 antibody that clusters CD20 within lipid rafts, which leads to enhanced complement-dependent cytotoxicity (CDC) and antibody-dependent cellular cytotoxicity (ADCC) and weakened direct cell death (DCD). 50 In contrast, OBZ is a type II anti-CD20 antibody that is modified through glycosylation, 51 and this structural feature contributed to increased binding affinity to the FcγRIII receptor and augmentation of its DCD, ADCC, and antibody-dependent phagocytosis (ADP). 52 Second, the upregulation of complement inhibitory proteins and exhaustion of complement molecules weaken the RTX-induced CDC and contribute to RTX resistance.53,54 With little or no CDC, the influence of the complements on OBZ biological activity is negligible.52,55 Third, in terms of pharmacokinetics, the binding of RTX to B cells results in an antibody saturation concentration, with a level nearly twice that of OBZ. 14 Furthermore, OBZ removal was 4.3 times lower than that of RTX, and its half-maximal effective value was 15-fold lower, while exhibiting a 4.7 times higher cell-kill coefficient. 14 More importantly, OBZ has been demonstrated to rapidly reduce memory B cells or plasma blasts/plasma cells in peripheral blood and tissue 56 or CD19+ B cells in lymphoid tissue. 57 After 2 OBZ doses, the comprehensive B-cell removal persisted until 24 weeks, which was significantly longer than that of RTX. 57 Thus, OBZ may be associated with increased effective and sustained clearance of B cells. The superiority of OBZ over RTX may be translated to better clinical improvement in B-cell malignancies.58,59 Similarly, this study provides a pathophysiological rationale for the treatment of refractory IMN or RTX-refractory IMN and the possible reasons for the achieved better therapeutic effectiveness.
Limitations
First, the studies incorporated in this research were observational studies and not RCTs, and no studies were conducted to compare OBZ and standard treatment. Thus, the quality of the included studies was relatively poor. Second, the sample size of the studies was small, and the follow-up interval was short, which may have led to an overestimation or underestimation of clinical efficacy and long-term side effects. Third, the participants enrolled in this study were primarily from Chinese backgrounds and those with refractory IMN or RTX-refractory IMN; thus, the results do not apply to non-Chinese participants or the general IMN population. Fourth, heterogeneity is still considerable (I2 > 70%) in CR rate estimates despite no obvious heterogeneity in TR rate estimates. It is noted that CR rates varied significantly, ranging from 5% to 70%, which possibly contributed to this substantial heterogeneity. Furthermore, heterogeneity disappeared in the secondary outcomes within certain subgroups, indicating that other factors such as disease model, study design, and treatment modality also contributed to the observed heterogeneity. Fifth, although Begg’s and Egger’s tests were not statistically significant, publication year (primarily 2024 and 2025) and country (predominantly China) could contribute to residual publication bias. Finally, B-cell counts and anti-PLA2Rab levels were not monitored in several of the included studies because of high costs or health insurance coverage. These limitations should be considered during the interpretation of the results, and high-quality RCTs evaluating the therapeutic effect of OBZ (NCT04629248, NCT05050214) must be conducted.
Conclusions
This meta-analysis evaluated the efficacy and safety of OBZ in treating IMN as reported in the 9 included studies. The findings demonstrated that OBZ therapy is associated with good clinical remission and that OBZ might be a more effective and attractive option for patients with IMN, especially those with refractory or RTX-refractory IMN. Owing to the study limitations, well-designed clinical studies, such as head-to-head RCTs, are warranted to confirm OBZ’s efficacy and safety profile.
Supplemental Material
sj-docx-1-taj-10.1177_27558428261421704 – Supplemental material for A meta-analysis on the efficacy and safety of obinuzumab for membranous nephropathy
Supplemental material, sj-docx-1-taj-10.1177_27558428261421704 for A meta-analysis on the efficacy and safety of obinuzumab for membranous nephropathy by Li-Xia Yu, Chuan-Li Qian, Jing-Rong Qian, Qi-Zhang, Xu-Li Yang and Qi-Feng Liu in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-docx-2-taj-10.1177_27558428261421704 – Supplemental material for A meta-analysis on the efficacy and safety of obinuzumab for membranous nephropathy
Supplemental material, sj-docx-2-taj-10.1177_27558428261421704 for A meta-analysis on the efficacy and safety of obinuzumab for membranous nephropathy by Li-Xia Yu, Chuan-Li Qian, Jing-Rong Qian, Qi-Zhang, Xu-Li Yang and Qi-Feng Liu in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-docx-3-taj-10.1177_27558428261421704 – Supplemental material for A meta-analysis on the efficacy and safety of obinuzumab for membranous nephropathy
Supplemental material, sj-docx-3-taj-10.1177_27558428261421704 for A meta-analysis on the efficacy and safety of obinuzumab for membranous nephropathy by Li-Xia Yu, Chuan-Li Qian, Jing-Rong Qian, Qi-Zhang, Xu-Li Yang and Qi-Feng Liu in Therapeutic Advances in Chronic Disease
Footnotes
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the “National Tutorial System” Training Program for Key Young Health Professionals in Suzhou (Qngg2023048); Medical Education Collaborative Innovation Fund, Jiangsu University (JDY2023016); Medical Research Project, Jiangsu Provincial Health Commission (Z2023039).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data can be provided upon request.
Supplemental material
Supplemental material for this article is available online.
