Abstract
There has been an unusual increase in the incidence of anti-glomerular basement membrane disease, anti-neutrophil cytoplasm antibody (ANCA)-associated vasculitis, and double-positive cases since the coronavirus disease 2019 (COVID-19) pandemic. Rare cases have been consistently reported, including anti-glomerular basement membrane antibody and ANCA double-positive crescentic glomerulonephritis after severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection as well as coexistent membranous nephropathy and proteinase 3 ANCA-associated crescentic glomerulonephritis following vaccination. We described both anti-phospholipase A2 receptor (PLA2R) antibody-positive and -negative membranous nephropathy superimposed on anti-glomerular basement membrane disease. Upon and beyond these findings, we herein reported three cases of biopsy-confirmed membranous nephropathy concurrent with anti-glomerular basement membrane antibody and ANCA double-negative crescentic formation, which manifested as acute kidney injury clinically. In one case, repeated renal biopsies confirmed de novo occurrence of crescents in the setting of PLA2R-associated primary membranous nephropathy immediately after SARS-CoV-2 infection. Subsequent use of anti-CD20 agents led to substantial recovery of renal function in two patients. Our report indicated that an indirect process resulting from downstream implications of this viral infection may be responsible for a plethora of unusual entities in glomerular diseases since the COVID-19 pandemic. As such, emerging insights may help decipher the complexity of these increasingly recognized yet equally detrimental clinicopathologic combinations, particularly in the post–COVID-19 era.
Keywords
Introduction
There has been an unusual increase in the incidence of anti-glomerular basement membrane (GBM) disease, anti-neutrophil cytoplasm antibody (ANCA)-associated vasculitis, and double-positive cases since the coronavirus disease 2019 (COVID-19) pandemic. 1 Rare cases of patients with both ANCA- and anti-GBM antibody-positive crescentic glomerulonephritis (GN) were consistently reported after the viral infection, 2 and a case of coexistent membranous nephropathy (MN) with proteinase 3 ANCA-associated crescentic GN was reported following vaccination. 3 In 2022, we also described both anti-phospholipase A2 receptor (PLA2R) antibody-positive and -negative MN superimposed on anti-GBM disease.4,5
In addition to these findings, we recently encountered exceptionally rare cases of MN concurrent with anti-GBM antibody and ANCA double-negative crescentic formation, which manifested as acute kidney injury (AKI) clinically. In one of the cases, de novo occurrence of crescents was observed on repeated renal biopsies in the setting of PLA2R-associated primary MN immediately after severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. As these distinct and challenging clinicopathologic combinations were left uncovered in a recent kidney international report focusing on COVID-19 and related glomerular diseases, 6 we decided to share our findings and highlight the importance of deciphering the complexity of these unusual entities. The study acquired proper institutional approval and necessary consent from the patients. The reporting of this case study adheres to the Case Report (CARE) guidelines, 7 and all patient details have been de-identified.
Case presentation
Three patients were admitted to and treated at the Department of Nephrology, the First Hospital of Hebei Medical University, located in ShiJiaZhuang in north China. The time points of their respective admission were, in the order of their case presentation, May 2023, October 2024, and April 2024.
The first case was that of a previously healthy male in his early 70s who complained of edema in the lower extremities for 3 months, had urine volume reduction for 20 days, and was vaccinated twice with Sinovac-CoronaVac (inactivated, Vero cell, Sinovac Research and Development Co., Ltd., Beijing, China); he had an episode of COVID-19 2 months before edema onset. On arrival, he had a blood pressure of 162/98 mmHg with moderate pretibial pitting edema. Laboratory tests revealed anemia, hypoalbuminemia, and a major surge in serum creatinine (Scr) level (Table 1). Urinalysis showed protein 2+ and occult blood 3+, whereas the 24-h urinary protein level was 3.7 g/0.6 L. SARS-CoV-2 RNA was negative on reverse-transcriptase polymerase chain reaction assay. There was no evidence for autoimmune diseases, chronic infection, or malignancy. In particular, anti-GBM antibody and ANCA remained negative in repeated tests that were performed 3 days apart. Chest computed tomography (CT) was unremarkable, and ultrasonography showed normal-sized kidneys with increased cortical echogenicity. Renal pathology subsequently confirmed MN concurrent with anti-GBM antibody and ANCA double-negative crescentic formation (Figure 1, upper panel). Precisely, there were 4 cellular, 14 fibrocellular, 5 fibrotic, and 5 small fibrocellular crescents among the 31 visible glomeruli on light microscopy, without any discernible spike formation upon extensive damage to the Bowman’s capsule. Furthermore, immunofluorescence (IF) detected granular staining of immunoglobin G (IgG), (2+), and C3 (+) deposits along the capillary wall, whereas PLA2R, NCAM-1, NELL-1, THSD7A, and EXT1/2 were all negative (Figure 2). Nevertheless, the diagnosis was derived from the findings of electron microscopy (Figure 1). Treatment with cyclophosphamide and steroids did not salvage his renal function, and he remained oliguric during his hospital stay of 22 days. He was on maintenance hemodialysis for 2 years without any evidence of secondary factor; since then, his plasma albumin, predialysis Scr level, and residual daily urine output in May 2025 were 40.2 g/L, 549.6 µmol/L, and <50 mL, respectively.
Results of the patients from clinical workup on admission.

CRP: C-reactive protein; ANCA: anti-neutrophil cytoplasmic antibody; GBM: glomerular basement membrane; PLA2R: phospholipase A2 receptor; ANA: anti-nuclear antibody; dsDNA: double-stranded DNA; IFE: immunofixation electrophoresis; COVID-19: coronavirus disease 2019; HBV: hepatitis B virus; HCV: hepatitis C virus.
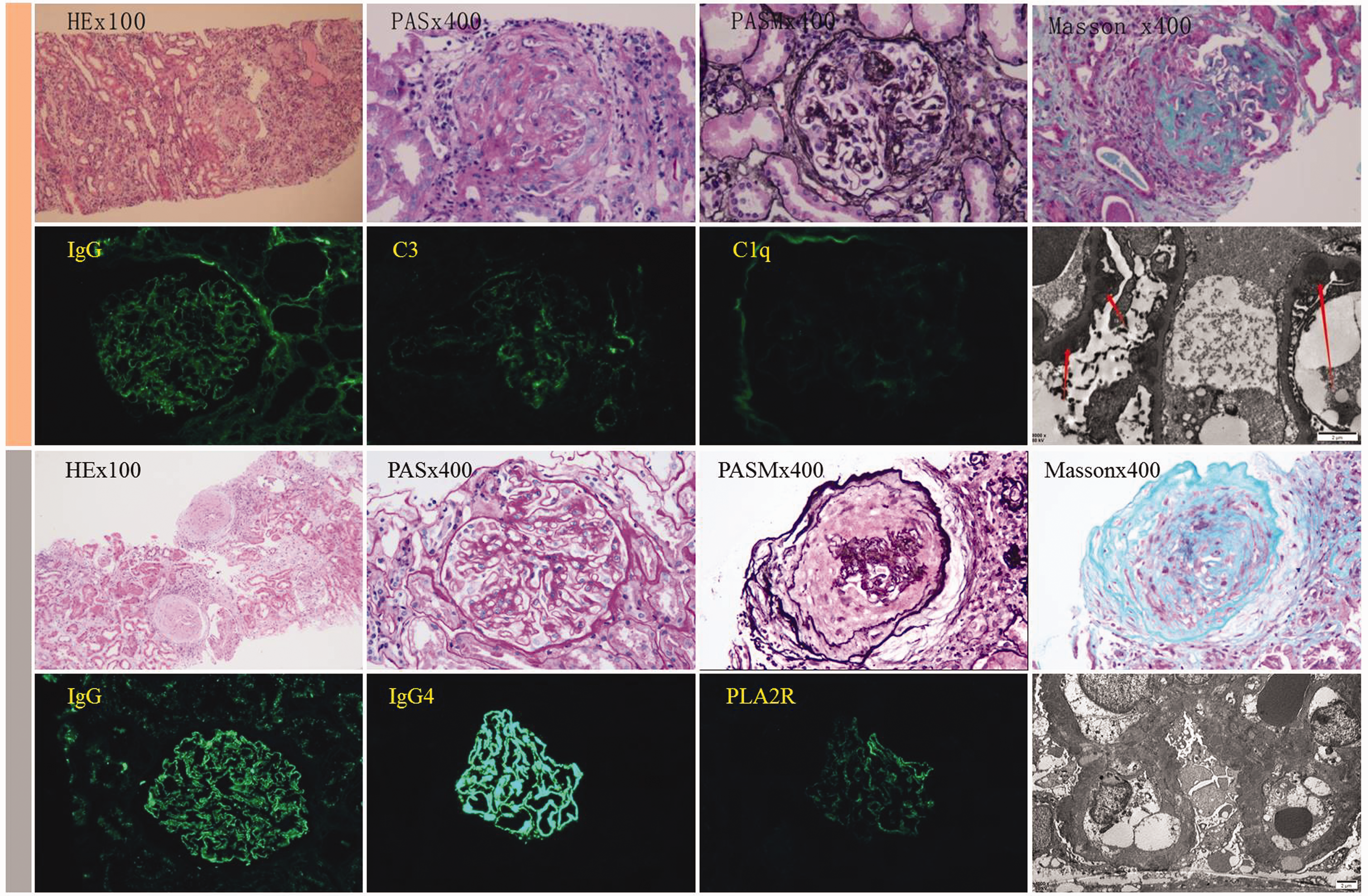
Pathological findings in the first and second cases of anti-GBM antibody and ANCA double-negative crescentic formation superimposed on membranous nephropathy. The yellow strip shows representative light microscopy, immunofluorescence, and electron microscopy findings from the biopsy of the first patient. Light microscopy results showed cellular crescent, fibroid necrosis, and infiltration of inflammatory cells. Thickened GBM was also visible. Immunofluorescence revealed global granular staining along the capillary loop for IgG (2+), C3 (+), and C1q (±). Electron microscopy showed irregular thickening of the GBM with a maximum read of 900 nm and diffuse podocyte foot-process effacement. In particular, subepithelial and subendothelial electron-dense deposits are clearly observed (arrows, original magnification: 8000×). The gray strip shows representative light microscopy, immunofluorescence, and electron microscopy results of the biopsy specimen in the second patient. Light microscopy revealed thickening of the basement membrane with formation of “spike” and epithelial deposits of erythrophilic substance. Furthermore, the existence of crescents was visible. Immunofluorescence revealed granular IgG (3+), IgG4 (3+), and PLA2R (+) deposits along the capillary loop. Electron microscopy showed diffuse foot-process effacement with electron-dense deposits within the basement membrane under the epithelial space (original magnification: 6000×). GBM: glomerular basement membrane; ANCA: anti-neutrophil cytoplasm antibody; IgG: immunoglobulin G; PLA2R: phospholipase A2 receptor.

Immunofluorescence depicting negative results for PLA2R, NCAM-1, NELL-1, THSD7A, and EXT1/2 along the glomerular capillary wall in the first patient. PLA2R: phospholipase A2 receptor.
The second case was that of a male in his late 50s who was referred to us for edema of the lower extremities that had persisted for 2 months and elevated Scr level for 2 weeks. He had received three shots of Sinovac-CoronaVac vaccine and developed COVID-19 3 months before the edema. Major abnormalities in his laboratory tests were as follows: proteinuria, hypoalbuminemia, elevated Scr level, and positive antibody against PLA2R (Table 1); anti-GBM antibody and ANCA were negative. Chest CT showed no evidence of pulmonary lesion. Renal pathology revealed glomerular lesions consistent with MN accompanied with crescent formation (Figure 1, lower panel), showing 2 fibrocellular and 1 fibrotic crescents among the 13 visible glomeruli with intact Bowman’s capsule. The patient experienced no oliguria during his AKI phase and eventually had an Scr level of 96.8 μmol/L and proteinuria of 1.2 g/2.2 L 2 months after two intravenous infusions of 1000 mg rituximab (RituxanTM, Roche Shanghai), each separated by 14 days. 8 Seven months after completion of the rituximab treatment, his plasma albumin level, Scr level, and 24-h urinary protein excretion in May 2025 were 39.8 g/L, 92.6 µmol/L, and 0.86 g/1.7 L, respectively.
The third case was that of a previously healthy male in his early 30s who had a history of three vaccinations (inactivated, Vero cell, National Vaccine and Serum Institute, Beijing, China) and one episode of COVID-19 thereafter. Particularly, he had undergone renal biopsies repeatedly between the episodes of viral infection. The first biopsy yielded PLA2R-associated MN with a normal Scr level 9 months ago (Figure 3, upper panel), which was treated using rituximab and began to mitigate. However, the remission was interrupted after a positive COVID-19 test 5 months ago, and there was an unexpected rise in the Scr level; the condition remained unattended until his referral to our department. Initial workup on admission is presented in Table 1. Of note, his serum anti-PLA2R antibody titer, 24-h proteinuria, and Scr level were 18.1 RU/mL, 19.6 g/2.1 L, and 323.3 µmol/L, respectively. Causes for rapid decline in the kidney function, including renal vein thrombosis, were carefully ruled out. A repeat renal biopsy revealed crescentic formation upon stage II MN (Figure 3, lower panel), and there were 3 small fibrocellular, 2 fibrocellular, and 1 fibrotic crescents among the 15 visible glomeruli with indiscernible damage to the Bowman’s capsule. Obinutuzumab was given along with 12 mg daily methylprednisolone. Serum antibody against PLA2R, 24-h proteinuria, and Scr level 3 months later were undetectable, 2.5 g, and 119.7 µmol/L, respectively, and the patient had experienced no oliguria ad interim. At 1 year after his second biopsy, the patient’s laboratory parameters in May 2025 were as follows: plasma albumin level, Scr level, and 24-h urinary protein excretion of 42.8 g/L, 139.1 µmol/L, and 0.92 g/1.6 L, respectively.

Pathological findings in the third case of anti-GBM antibody and ANCA double-negative crescentic formation superimposed on membranous nephropathy upon repeated biopsies. The first biopsy performed in October 2023 (deep blue strip) showed glomerular features consistent with membranous nephropathy. Immunofluorescence microscopy demonstrated segmental IgG/IgG4 and PLA2R staining along the glomerular capillary wall. Electron microscopy showed electron-dense deposits along the subepithelial space and diffuse podocyte foot-process effacement (original magnification: 6000×). Repeated biopsy performed after 9 months (light blue strip) showed glomerular features consistent with stage II membranous nephropathy, along with crescent formation (marked by asterisk in the figure), fibroid necrosis, and inflammatory cell infiltration. Tubulointerstitial lesions were also visible. IgG and IgG4 were detected using immunofluorescence along the glomerular capillary wall, in addition to PLA2R staining by immunohistochemistry. Electron microscopy demonstrated irregular thickening of the GBM with a maximum read of 2000 nm and diffuse podocyte foot-process effacement. In particular, electron-dense deposits were clearly visible in the subepithelial space and within the basement membrane (original magnification: 6000×). GBM: glomerular basement membrane; ANCA: anti-neutrophil cytoplasm antibody; IgG: immunoglobulin G; PLA2R: phospholipase A2 receptor.
Discussion
We present the cases of three patients with ANCA and anti-GBM antibody double-negative crescentic formation superimposed on underlying MN, which, in the absence of systemic autoimmune disease or chronic infection, is believed to be a rare variant of MN.9,10 Our findings are based on the results of 92 native kidney biopsies of MN within 2 years, from October 2022 to October 2024. In comparison, the corresponding values reported by Mayo Clinic prior to the COVID-19 pandemic were 19 patients of the 5108 biopsy-confirmed MN patients over the period of 20 years. 9 In terms of incidence/100 patients/year, it was 1.63 against 0.18, which may seemingly suggest an increased probability of encountering this unique clinical entity.
Crescentic formation is considered one of the pathogenetic mechanisms for AKI in MN. 11 However, the patients in this study exclusively had anti-GBM GN or ANCA-associated GN superimposed on MN, which literally differed from those of our study in pathogenesis and immunological perturbation. Irrespective of the origin of the crescent, rapid diagnosis and early induction of immunosuppressive therapy are fundamental because MN with crescentic formation may follow a progressive course and lead to end-stage renal histopathology. More likely, the absence of antibody may likely blunt the clinical awareness of crescent formation. As such, our case series virtually provided a unique opportunity to dynamically observe the pathologic lesions, response to immunosuppression, and renal outcomes in this rare glomerular pathophenotype at different time points. Clearly, our first and second patients had late and timely diagnosis/treatment, respectively, resulting in the need for hemodialysis and recovery of renal function.
With the most extensive and irreversible damage to the glomerular structure, our first case showed similarity (global granular IgG/C3 deposit on the IF) to and differences from the early report, 9 which had, on an average, 25% glomerular crescents or necrosis and a more advanced stage of MN (stage II or higher) in comparison to a corresponding value of 72% and multisite electron-dense deposits in our patient. Ubiquitous electron-dense deposits at subepithelial, intra-GBM, and subendothelial sites and within the mesangial area could initiate cross-reactive T cell responses 12 and contribute to extensive crescent formation. 10 Intriguingly, there was no PLA2R and five other antigens on IF in this patient; this “absence” may be attributed to the fact that the six tested markers accounted for 76% of the possible antigens most often associated with primary MN and thus left certain room in the so-called “antigen void.” 13 Congruently, we recently found positive PCDH7 on fluorescence in primary MN. 14
We found no direct evidence supporting participation of the viral infection in crescentic formation in the third patient. However, a rapid decline in renal function immediately after COVID-19 and de novo formation of crescents in the setting of primary MN essentially implicated possible involvement of the virus. 15 In this regard, mechanisms underlying AKI in the wake of the COVID-19 pandemic included molecular mimicry, vaccine adjuvants, and polyclonal activation. 16 Immunopathologic perturbations underlying glomerular injury are speculated as one of the major causative factors. 6 Moreover, severe glomerular damage owing to immune complex deposition may elicit crescents, which then cause further structural damage. 17 Hence, glomerular lesions in MN were exaggerated by tissue inflammation, local immune cell infiltration, endothelial injury, and crescent formation. However, the possibility of coincidence cannot be excluded in the absence of direct histopathologic evidence. Nevertheless, we did not observe any such case over the two decades before the COVID-19 pandemic, and there is lack of reported cases in the literature. This speculation was further supported by the fact that more immune-mediated unusual combinations have sprouted during the COVID-19 pandemic.6,14 Indeed, the mRNA COVID-19 vaccines–related immune-mediated adverse events are characterized by de novo or relapsing glomerular diseases. 18
In view of these findings and a review of the literature, we intended to adhere to the prepandemic treatment protocol, which focused on MN predominantly with oral prednisolone and cyclophosphamide. 9 Alternatively, treatment could include rituximab, especially considering ongoing or recent infection with SARS-CoV-2, with obinutuzumab as a rescue therapy. 19 Obviously, the expeditious use of anti-CD20 agents may contribute to utmost preservation of renal function, as in our second and third cases.
Limitations and prospects for future research
We reported three cases of anti-GBM antibody and ANCA double-negative crescentic formation in MN that occurred within a short period since the COVID-19 pandemic. More cases are needed to confirm the “higher incidence” and explore new etiologic factors in the post–COVID-19 era. Nonetheless, it is essential to maintain a high level of vigilance for MN patients with an unexplained rapid decline in renal function. This heightened level of suspicion leads to earlier detection, better understanding, and optimal management of this rare clinical entity. Arguably, our purpose is to raise awareness regarding early detection of novel and inexorable glomerular diseases in the post–COVID-19 era and preserve kidney function. This is of relevance in the face of the reported pleiotropic adverse effects of COVID-19 vaccines. 18
Conclusion
Our report may help consolidate current knowledge on this rare variant of MN and meet the ever-emerging challenges derived from these rare but serious clinicopathologic combinations, especially in the post–COVID-19 era.
Footnotes
Acknowledgements
The authors would like to thank the patients for providing informed consent for publishing their case details.
CRediT authorship contribution statement
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Ethics statement
This study was conducted ethically in accordance with the World Medical Association Declaration of Helsinki and approved by the Ethics Committee of the First Hospital of Hebei Medical University (2023S00383). Written informed consent to treatment and publication of this study was obtained from the patients.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
