Abstract
Background:
Thyroid disease is a type of endocrinologic disease that should be treated early to avoid thyroid disease-related comorbidities, especially cardiovascular complications secondary to hyperthyroidism, which requires medication therapy to achieve an euthyroid state. However, patients are exposed to medication-related adverse drug reactions (ADRs) while taking drugs to treat thyroid disease. ADRs are unpredictable and unintended medication effects on the patient.
Objective:
This study aimed to determine the prevalence and severity of ADRs and the contributing factors of medications used for treating thyroid disease.
Design:
A cross-sectional study.
Method:
A multicenter, hospital-based study was conducted through patient interviews at public hospitals in Northwest Ethiopia. A simple random sampling technique was used to identify patients for the interview. The ADR was assessed using the Naranjo algorithm of the ADR probability scale, and the Shamrock severity of ADR assessment tool was used to identify ADR. STATA version 17 was used for data analysis. The results were summarized using descriptive statistics, including frequency, mean, median, and standard deviation. To identify the independent predictors of ADRs, logistic regression analysis was conducted, and a p-value <0.05 is considered statistically significant.
Result:
Among 330 study participants, 59 were experiencing ADRs. Regarding the severity of ADR, 30 (50.85%) adverse drug events were mild, 28 (47.46%) were moderate, and 1 (1.69%) was severe. Factors independently associated with ADRs were substance use (AOR = 3.48 (1.40–8.68), p = 0.007), traditional medicine users (AOR = 3.77, 95% CI: (1.13–12.54), p = 0.031), disease comorbidity (AOR = 3.58, 95% CI: (1.42–9.04), p = 0.007), and drug interaction (AOR = 2.63, 95% CI: (1.07–6.47), p = 0.035).
Conclusion:
The findings revealed that the incidence of ADRs among thyroid disease patients was 17.88%. Substance use, traditional medicine use, disease comorbidity, and drug interaction were the significant factors associated with ADR.
Plain language summary
Treatment of thyroid patients are important but patients are exposed to medication-related adverse drug reactions (ADR) while taking drugs to treat thyroid disease. ADRs are unpredictable and unintended medication effects on the patient. A multicenter hospital-based cross-sectional study design was conducted through patient interviews at public Hospitals, northwest Ethiopia. A simple random sampling technique was used to identify patients for interview. The adverse drug reaction was assessed by using the Naranjo algorithm of the adverse drug reaction probability scale, and the Shamrock severity of ADR assessment tool was used to identify ADR. STATA version 17 was used for data analysis. To identify the independent predictors of ADRs, logistic regression analysis was conducted, and a p-value <0.05 is considered statistically significant association with ADRs. Among 330 study participants, 59 were experiencing ADRs. On their severity of ADR, 30 (50.85%) ADEs were mild, 28 (47.46%) ADEs were moderate, and 1 (1.69%) ADE was severe. Factors independently associated with ADRs were substance use, traditional medicine user, disease comorbidity, and drug interaction. This finding concluded that the incidence of ADRs among thyroid disease patients was 17.88% and it was found that Substance use, traditional users, comorbidity and drug interaction were associated with ADRs.
Keywords
Introduction
Thyroid disease is one of the different endocrinologic diseases that should be treated early to avoid thyroid disease-related comorbidities, especially cardiovascular complications secondary to hyperthyroidism.1,2 Therefore, thyroid supplementation and antithyroid drugs have a greater impact on maintaining euthyroidism and decreasing thyroid-related complications, including arrhythmia caused by hyperthyroidism and cardiomyopathy caused by hypothyroidism.3,4 However, patients are exposed to medication-related reactions known as adverse drug reactions (ADRs) that impair patient treatment outcomes, increasing the cost of treatment-related harm5,6 and also decreasing patients’ treatment satisfaction, and these ADRs result in patients experiencing medication non-adherence that also affects patients’ desire for treatment.7,8 ADR is included in a broad term known as adverse drug events (ADEs) that are unintended medication-related reactions causing harm or injuries to the medication users, and they can also lead to patient ADR-related hospitalization.9,10
WHO defines an ADR as any noxious or unintended response to a drug that occurs at doses usually used for prophylaxis, diagnosis, or treatment of disease or the modification of psychological function, and the Food and Drug Administration (FDA) classifies an ADR as any adverse event related to the use of a drug in humans, whether or not it is thought to be a drug-related reaction.11–14 This includes any adverse event that arises during the use of a drug product in professional practice, accidental or intentional drug overdose, drug abuse, drug withdrawal, adverse event that occurs from drug abuse, and any significant failure of expected pharmacologic action. 15
ADR is also one of the types of ADEs, including drug-related allergic or idiosyncratic reactions.16,17 These reactions increase patient burden and length of hospital stay, correlated with medication misadventure, including ADRs that are undesired, unexpected, and/or excessive medication response of patients.14,18,19 Among these ADRs, a few are unpredictable and unique. 20 Nevertheless, many of these ADRs are expected and can be avoided as their pharmacological mechanism of action makes them predictable. 21 ADRs are undesired patient responses to a noxious drug that occur in a normal dose range used for a certain disease prevention and/or treatment. 22 ADEs are recognized while the drug is produced and stated by the manufacturers, 23 and drugs causing ADRs are reported by patients or consumers, accompanied by post-marketing surveillance 24 that leads the drug to be withdrawn from the market by responsibility of the FDA or by the manufacturer voluntarily. 25
ADRs, including rash, hypersensitivity, agranulocytosis, and others, caused by thyroid drug formulation and antithyroid drugs are frequently reported in different studies,26–29 and also hepatotoxicity is reported in patients using thionamides of propylthiouracil antithyroid drugs (ATDs).30–32 Based on different studies, ADRs were reported in patients using levothyroxine.33–36 However, there is scarce availability of medication-related ADRs and their determinants among thyroid disorder patients in Ethiopia. Therefore, this study was aimed at and conducted to assess ADRs and their determinants among thyroid disease patients on the thyroid formulation for hypothyroidism and antithyroid drugs for the treatment of hyperthyroidism in Ethiopian hospitals.
Methods
Study area and period
The study was conducted at the University of Gondar Comprehensive and Specialized Hospital (UoGCSH), Feleghiwot Comprehensive and Specialized Hospital (FHCSH), and Debre Tabor Comprehensive and Specialized Hospital (DTCSH), which are located in Northwest Ethiopia, from May 30 to July 30, 2024.
Serving more than seven million people, UoGCSH is a significant teaching hospital situated 738 km from Addis Ababa. It provides treatments like internal medicine, surgery, and endocrinology care, especially for thyroid and diabetic disorders.
DTCSH, located in Debre Tabor Town, some 667 km from Addis Ababa, was founded in 1920. It serves more than 101,500 patient visits a year and has about 110 inpatient beds, making it a model for Ethiopia’s sophisticated medical treatment.
FHCSH serves seven million people and is located in Bahir Dar, 565 km from Addis Ababa. It was founded in 1963 and offers an extensive variety of therapeutic services and referrals, including radiotherapy and chemotherapy. Each of these hospitals serves a large population in the Amhara region with a variety of medical needs.
Study design
The study was conducted using a multicenter hospital-based cross-sectional study.
Study population, inclusion, and exclusion criteria
The source population comprised all adult thyroid disease patients receiving follow-up care at the ambulatory clinics of Northwest Ethiopian hospitals. The study population included all eligible thyroid patients who were on follow-up between May 30 and July 30, 2024. Patients aged 18 years or older diagnosed with hypothyroidism or hyperthyroidism, including toxic multinodular goiter, Graves’ disease, and thyrotoxicosis, who had been on treatment and attending follow-up for at least three months, and who provided informed consent, were included. Patients with speech or hearing impairments that interfered with communication and those with incomplete or poor medical documentation were excluded from the study.
Sample size determination and sampling technique
The sample size was calculated using a single population proportion formula as follows: n = Zα/22 p(1 − p)/d2, where n = is the desired sample size; Zα/2 = is the standard normal distribution at 95% confidence level usually 1.96, p = proportion of thyroid patients who were exposed for ADR since there is no study conducted in ADR of ATDs 50% prevalence were used (p = 0.5), and d = marginal error (5% = 0.05) n = 1.962 0.5(1–0.5)/0.052 = 384.16 = ~384. Since the study population was less than 10,000, a correction formula was used to obtain the final sample size (nf). nf = n/(1 + (n/N)) = 384/(1 + (384/1600)) = 310. Where of the final sample size and the total study population (1600). After considering a 10% contingency, 341 study participants were included in the study. Eleven patients were not willing to participate and were excluded from the study, and a final analysis was performed on 330 patients.
A systematic random sampling technique was employed to select the study participants. Then, the total sample size was proportionally allocated for each selected hospital. The proportional allocation of samples to the total population of each hospital was applied using the formula as follows: n = n × Ni/N, where n = required sample size, Ni = total number of patients in each hospital, and N = total number of patients of selected hospitals. The number of participants who visited each hospital was determined as follows. Study subjects recruited from UoGCSH, having 600 patients = 341*600/1600 = 128, study subjects recruited from DTCSH, having 350 patients = 34*350/1600 = 75, and study subjects recruited from FHCSH, having 650 patients = 341*650/1600 = 138 (Figure 1).

Diagrammatic representation of the sampling procedure at selected hospitals in Northwest Ethiopia.
Study variable
Adverse drug reactions were the dependent variable, and socio-demographic characteristics, social drug use, traditional medicine use, the history of thyroidectomy, the duration of thyroid disease, the presence of comorbidity, the number of comorbidities, and the source of the drug were the independent variables.
Data collection technique and instrument
Data collection procedure
Data was collected by a clinical pharmacist using a structured questionnaire and data extraction format. The data collection tool was developed after reviewing published literature and from the thionamides and levothyroxine article, and the ADR assessment tool of the Naranjo Adverse Drug Reaction Probability Scale (NADRPS), 37 causality and probability scale, 38 and Hartwig and Siegel severity scale 39 were used to assess ADR. The data abstraction format consists of the patient’s demographics and medication experience, diagnosis, disease duration, dosage regimen, ADRs, history of thyroidectomy, laboratory results, and duration of treatment.
Data quality assurance
Data quality control and data analysis
To ensure the quality of the data, training was given to the data collectors, and an English version of the data collection questionnaire was translated into Amharic and back-translated to English. A structured questionnaire was designed by reviewing different works of literature to include important variables. The collected data were cleared, entered into EPI Data version 4.6, checked for correctness, and exported to STATA version 17 for analysis. Descriptive analysis was used to compute the frequency, mean, and standard deviation (SD) for the continuous variables and categorical data. Logistic regression analysis was conducted to identify the independent predictors of ADR. Independent variables having a p-value <0.25 in the bivariate logistic regression analysis were entered into the multivariable logistic regression analysis to control the confounding effect, and a p-value <0.05 was considered significant.
Operational definitions and definitions of terms
The adverse drug reaction: Unpleasant and unintended effect of the medication.
ADE: Is defined as a harm brought on by taking medication at dosages intended for treatment, diagnosis, or prophylaxis. 40
Medication adherence: Ability and willingness of patients to take medications as prescribed by physicians, as intended, and/or the patient’s behavior for medication. 41
Substance use history: Refers to using khat, cigarettes, and tobacco within 3 months. 42 Participants were categorized based on their substance use as follows: coffee and khat users were divided into current users, who consumed these substances within the past 30 days, and non-users, who did not use them in the last month. Smokers were classified as such if they had smoked for at least 1 month without discontinuation, while alcohol drinkers were defined as those who consumed alcohol for at least 6 months. Ultimately, participants were identified as substance users if they used coffee, khat, alcohol, or smoked.
Disease co-morbidity: The presence of two or more diseases. 43
Polypharmacy: It is recognized as the use of five or more medications. 44
Drug interaction: Defined as the pharmacological activity of one drug being altered by the concomitant use of another drug. 45
Results
Socio-demographic and clinical data of the patients
In this study, 341 participants were included; 11 patients were withdrawn from the study, and a final analysis was performed on 330 patients. The mean age of the participants was 44.71 (43.21–46.22) years. Among the study participants, females were more than half, 281 (85.15%) of the total participants. Among the participants, 241 (73.03%) were married and 226 (68.48%) were living in a rural area. In addition, 175 (53.03%) of the study participants could not read or write. Most of the participants, 292 (88.48%) were not employed in their occupation and 38 (11.52%) were employed. Most of the study participants, 250 (75.76%) had a substance use habit, of which 260 (88.74%) drank coffee, and almost all of the study participants, 305 (92.99%) were living with their family (Table 1).
Sociodemographic characteristics of patients with adverse drug reaction among thyroid disease patients at an ambulatory clinic in three hospitals of Northwest, Ethiopia, from May 30 to July 30, 2024 (n = 330).
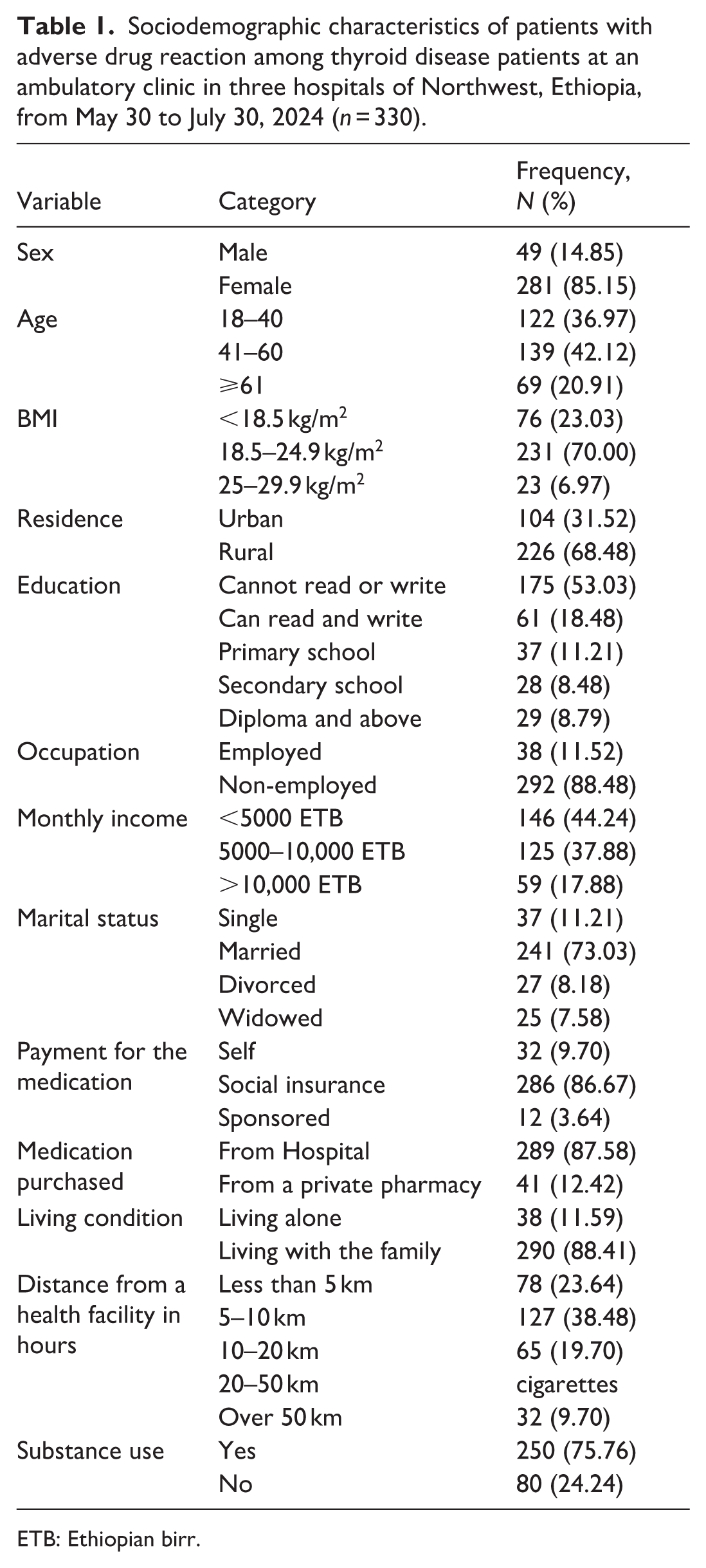
ETB: Ethiopian birr.
The Ethiopian birr (ETB). Substance use includes the use of coffee, khat, alcohol, and cigarettes.
Clinical characteristics of the study participants
Most of the study participants were diagnosed with hyperthyroidism (285; 86.36%) and 45 (13.64%) were diagnosed with hypothyroidism. Of the total 330 study participants, 27 (8.18%) received non-pharmacologic treatment with thyroid surgery. More than half of the study participants, 181 (54.85%) had a treatment duration of between 1 and 3 years. Of the total study participants, one-third of the patients, 109 (33.03%), had disease comorbidity, and more than half of the study participants had more than three disease comorbidities (Table 2).
Clinical characteristics of thyroid disease among patients at the ambulatory clinic in three hospitals of Northwest Ethiopia, from May 30 to July 30, 2024 (n = 330).

Adverse drug reaction incidence, causality, probability, and severity
A total of 59 ADRs were reported in this study. Among 330 study thyroid patients, the overall prevalence of ADRs was found to be 17.88% (95% CI: 13.72–22.03) (Figure 2).

Prevalence of ADR among thyroid disease patients at the ambulatory clinic in three hospitals of Northwest Ethiopia, from May 30 to July 30, 2024 (n = 330).
Using the Naranjo causality assessment algorithm for ADR, 8 (13.56%) of ADRs were definite, 31 (52.54%) of ADRs were probable, 14 (23.73%) of ADRs were possible, and 6 (10.173%) of ADRs were Doubtful. Based on the modified Hartwig ADR severity assessment scale, 30 (50.85%) ADRs were mild, 28 (47.46%) were moderate, and 1 (1.69%) was severe. Assessing ADE’s preventability was done using the modified Shamrock and Thornton preventability criteria, which showed that 15 (25.42%) ADRs were definitely preventable, 19 (32.20%) ADRs were probably preventable, and 25 (42.37%) of the ADRs were non-preventable (Table 3).
Severity of ADRs based on the modified Hartwig ADRs Severity Assessment Scale, and ADR preventability among thyroid disease patients at the ambulatory clinic in three hospitals of Northwest Ethiopia, from May 30 to July 30, 2024 (n = 330).

ADR: adverse drug reaction.
Level: 1&2 = Mild, 3&4 = Moderate, 5–7 = Severe.
Potent iatrogenic offending medication class associated with ADR
There were 59 ADRs among patients with thyroid disease receiving antithyroid drugs, levothyroxine, and medication for disease comorbidity. More than half of the ADRs were seen with antithyroid disease medications 37 (62.70%), followed by anticoagulants 8 (13.60%) (Table 4).
Potential offending agents for the experienced ADRs (N = 59) among thyroid disease patients at the ambulatory clinic in three hospitals of Northwest Ethiopia, from May 30 to July 30, 2024 (n = 330).

ADR: adverse drug reaction.
Determinants of medication-related ADRs
The findings from multivariate logistic regression indicated the presence of a significant association between medication ADR and four independent variables (substance use, traditional medicine use, the presence of disease comorbidity, and the presence of drug interaction). Therefore, patients who used substances were 3.48 times more likely to have ADR than those who were not substance users (AOR = 3.48 (1.40–8.68), p = 0.007). Those who were traditional medicine users were 3.77 times more likely to experience ADR than those who were not traditional medicine users (AOR = 3.77, 95% CI: (1.13–12.54), p = 0.031). Meanwhile, those who had comorbid disease were 3.58 times more likely to have ADR than those who did not have disease comorbidity (AOR = 3.58, 95% CI: (1.42–9.04), p = 0.007). On the other hand, those who have a drug interaction were 2.63 times exposed to ADR than those who did not have a drug interaction (AOR = 2.63, 95% CI: (1.07–6.47), p = 0.035) (Table 5).
Bivariate and multivariate analysis of factors associated with adverse drug reaction among thyroid disease patients at an ambulatory clinic in three hospitals of Northwest Ethiopia, from May 30 to July 30, 2024 (n = 330).

ETB: Ethiopian birr.
Significance.
Discussion
This study aimed to determine the prevalence and identify the determinants associated with the adverse effects of thyroid hormone supplementation and antithyroid medication among patients who underwent follow-up at three hospitals in the Northwest Ethiopia. Medication adverse effects are unwanted and unintended medication-related reactions that result in patients harm and hesitate patients to take their medication, which resulted in decreament of patient treatment outcome that needs the intervention to stop medication or to adjust medication dosage and or treating that unpleasant reaction of medication using another medication for treatment or as prevention of such unpleasant reaction before occurring 22 and ADEs causes patient dissatisfaction of their medication as a result of medication-related reaction and direct toxicity of the treatment used for another medical condition. 46
A study was conducted on the determinants of medication adverse drug effects among patients with thyroid disorders in a multicenter hospital-based cross-sectional study that interviewed 330 patients who attended the ambulatory clinic of three selected hospitals in Northwest Ethiopia. The NADRPS was used for the assessment of the adverse drug reaction of thyroid disease patients’ likelihood of experiencing ADR related with thyroid drugs using Naranjo, which comprises of a certain questions including the correlation between the time of medication administration and the patients’ ADR experience, the possible causes of the event, previous patient ADR experience, the therapeutic concentration of the drug and dose range of the medication. 37 We used NADRPS because it is consistent and reliable to evaluate ADRs of patients treated with complex medication.
The findings of the present study state that most of the patients were females (85.15%), as stated in other research findings, thyroid disorders are more prevalent in females, and the mean age of participants was 45 (95% CI, 43.21–46.22) years. This study revealed that the prevalence of thyroid drug supplementation and antithyroid medication-related ADR is 17.90%, and this study shows that substance use (75.76%), traditional medicine use (6.36%), the presence of comorbid disease (33.94%), and patients having drug interaction (20.00%) were significantly associated with thyroid medication ADR.
In this study, the prevalence of ADRs of thyroid formulation and antithyroid drugs was found to be 17.90%, which is in line with a study conducted in Germany, which showed that the overall ADR of ATDs was 14.3%. 47 This finding is higher than a study conducted in the United Kingdom in which the thyroid medication-related ADRs were 6% and 4% for carbimazole and propylthiouracil, respectively. 26 This discrepancy may be due to the difference in the reporting of ADR in this study; all ADRs of antithyroid medication-taking patients were reported, whereas in the study conducted in the United Kingdom, ADRs of thionamides were separately reported.
According to this study, substance use was strongly associated with the development of antithyroid medication adverse drug reactions. Those patients having a history of substance use were 3.48 times exposed to medication ADRs than those who are not substance users, which is similar to a study conducted on the impact of alcohol and patient safety related to ADRs. 48 The possible justification for this might be that substance use, particularly alcohol and cigarette smoking, can interact with antithyroid drugs and result in ADRs through several mechanisms. Both alcohol and tobacco can interfere with the metabolism of antithyroid medications, leading to altered drug levels that may increase toxicity or reduce efficacy. Additionally, alcohol and tobacco use can intensify side effects, such as liver toxicity. Furthermore, both substances can weaken the immune system, making patients more vulnerable to infections. Smoking, in particular, can also worsen the symptoms of Graves’ disease, which induces antithyroid drug exposure and increases their ADRs.49–52
Based on this finding, traditional medicine users were strongly associated with the development of medication adverse drug reactions. Patients who used traditional medicine were 3.77 times exposed to medication adverse drug reactions than those who did not use traditional medicine. This is because, despite the importance of traditional medicine in treating the symptoms of thyroid diseases, the appropriate use and preparation of these remedies are often not well understood. This lack of knowledge can lead to herb-drug interactions, as the dosages are typically not established, potentially resulting in patients developing adverse drug reactions to antithyroid medications.53–57
Based on this finding, the presence of comorbid disease was strongly associated with the development of antithyroid medication adverse drug reactions. Patients who used traditional medicine were 3.58 times exposed to medication adverse drug reactions than those without disease comorbidity. This finding is comparable with the findings of a prospective observational study 58 conducted among the inpatients of the general medicine department of Justice K.S. Hegde Charitable Hospital showed that the presence of polypharmacy due to several comorbid diseases, a retrospective cohort study conducted in Australia and a study conducted in Nigeria and other different countries comorbidity impact on ADR respectively were associated with exposure of medication ADRs.59–61
According to this study, the presence of drug interaction was strongly associated with the development of antithyroid medication adverse drug reactions. Those patients who had drug interactions were 2.63 times exposed to medication adverse drug reactions than those without a drug interaction. This finding is comparable with the findings of a prospective study conducted among admitted patients to the internal medicine ward, which showed that drug interaction was associated with exposure of medication ADRs 62 and also similar with a retrospective study conducted in Jirshian tertiary hospital, which showed that ADR were reported with patients having potential drug interactions (PDDIs), 63 and also in line with spontaneous reporting of reports of ADR caused by PDDIs 64 and also similar with a study conducted in Washington. 65
Strengths and limitations of the study
The assessment of causality using standard scales given and has employed a standard trigger tool to identify ADEs reported not only by the patient, this increases the probability of detecting ADEs and its multicenter study is the strength of the study, but this study have also limitations including being a cross-sectional study design and not able to know the impact of ADRs on patient treatment outcome, and intervention were not given for ADRs so further prospective and interventional study is important.
Conclusion
The findings revealed that the incidence of ADRs among patients with thyroid disease was 17.88%. Substance use, traditional medicine use, disease comorbidity, and drug interaction were the significant factors associated with ADR. It is necessary to report ADR to provide intervention and to promote patient safety, and the authors recommend a prospective study addressing the limitations of this study.
Footnotes
Acknowledgements
The authors would like to thank the data collectors, and we would like to extend our sincere thanks to all study participants.
Ethical considerations
Ethical approval and an ethical clearance certificate were obtained from the Debre Tabor University Ethical Review Committee with reference number CBE 305/2016 and used as a centralized ethical clearance that covers all the included study sites.
Consent to participate
Written informed consent was then obtained from the study participants for the interview, and the ethical review committee of Debre Tabor University approved all the informed consents. This study was conducted in compliance with the Declaration of Helsinki. Consent from participants who could not read or write was obtained in the presence of the witness on behalf of the participant to document their voluntary participation, and the witness has signed the consent documents for participants who cannot read or write. Study participants were also told they had the full right to refuse to interview. Moreover, the personality identifiers were kept to ensure the patient’s confidentiality and the privacy of the study participants.
Consent for publication
Consent for the publication of this result was obtained directly from the patient.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All the data supporting the findings of this research are found in this manuscript.
