Abstract
Objective
This study aimed to evaluate the safety profile of progesterone by analyzing adverse event data from the Food and Drug Administration Adverse Event Reporting System (FAERS) between 2004 and 2024.
Materials and methods
This retrospective, observational pharmacovigilance study was based on data from the FAERS database. A total of 1827 adverse event reports associated with progesterone were retrieved. Disproportionality analysis methods, including the reporting odds ratio, proportional reporting ratio, Bayesian confidence propagation neural network, and multi-item gamma Poisson shrinker, were used to evaluate the frequency, signal strength, and time-to-onset of adverse events.
Results
Among 24,589,239 adverse event reports in the reporting system, 1827 were associated with progesterone, covering 22 system organ classes and 152 preferred terms. The most frequently reported preferred terms were maternal exposure during pregnancy (151 cases), spontaneous abortion (144 cases), and abnormal product odor (114 cases). The top three preferred terms showing the strongest signals were decidual cast (reporting odds ratio: 2825.23), chondrodermatitis nodularis chronica helicis (reporting odds ratio: 3897.61), and autoimmune dermatitis (reporting odds ratio: 1519.29). Most adverse events occurred within 30 to 180 days after exposure. Newly identified preferred terms associated with progesterone included acute eosinophilic pneumonia, meningioma, and autoimmune dermatitis.
Conclusions
This study identified notable safety concerns associated with progesterone use and detected several rare adverse events. These findings underscore the need for continued monitoring, updated prescribing guidelines, and further investigation into progesterone formulations and adverse event mechanisms.
Keywords
Introduction
Progesterone is a pivotal steroid hormone orchestrating key reproductive functions, including endometrial preparation for implantation, menstrual cycle regulation, and pregnancy maintenance. Clinically, it is indicated for managing progesterone-deficiency disorders (e.g. anovulatory bleeding and dysmenorrhea) and perimenopausal syndrome when combined with estrogen. In obstetrics, progesterone supplementation remains a cornerstone for treating threatened or recurrent miscarriage associated with luteal insufficiency. Although evidence supports its efficacy in reducing miscarriage risk in cases of recurrent pregnancy loss, 1 its routine use in early pregnancy remains debated due to inconsistent effects on live birth rates. 2
Adverse events (AEs) associated with progesterone use have been reported, ranging from mild to severe complications. Common AEs associated with progesterone use include breakthrough bleeding, 3 headache, dizziness, 4 depression, 5 breast tenderness, 6 nausea, vomiting, 7 arrhythmia, and anaphylaxis. 8 These AEs, particularly those affecting the cardiovascular system, may elevate health risks, lead to severe consequences, and negatively impact patients’ quality of life and clinical outcomes. 9 Healthcare professionals should closely monitor patient responses to progesterone to ensure both safety and efficacy.
The Food and Drug Administration (FDA) Adverse Event Reporting System (FAERS) is a comprehensive database that collects and analyzes reports of AEs associated with drugs and therapeutic biologics. 10 It plays a crucial role in post-marketing surveillance by detecting safety signals that may not emerge during pre-marketing clinical trials. 11 This study aimed to evaluate the safety profile of progesterone by analyzing FAERS AE reports from 2004 Q1 to 2024 Q3. By assessing the frequency, distribution, and strength of AE signals, we provide a comprehensive overview of safety concerns associated with progesterone use in clinical practice. This analysis contributes to the current understanding of progesterone safety and supports informed clinical decision-making, ultimately improving patient care and outcomes.
Methods
Ethical statement
This pharmacovigilance study was conducted in accordance with the ethical principles of the Declaration of Helsinki, as revised in 2024.
Data source
This study analyzed AE reports from the publicly available FAERS database, covering the period from 2004 Q1 to 2024 Q3. Adverse drug reactions associated with progesterone were comprehensively compiled, and all preferred terms (PTs) across system organ classes were carefully extracted. In this study, “progesterone” refers to the naturally occurring hormone and its pharmaceutical preparations. Reports explicitly mentioning synthetic progestins, such as medroxyprogesterone acetate, were excluded during data cleaning to focus on the safety profile of progesterone. Progesterone was designated as the primary suspect drug (PS) in FAERS, allowing an in-depth examination of its association with AEs. FAERS accepts AE reports from various sources, including physicians, pharmacists, patients, pharmaceutical companies, and other healthcare professionals. 12 AEs were coded using version 25.0 of the Medical Dictionary for Regulatory Activities (MedDRA). 13 The FAERS dataset comprises seven distinct databases, including demographic and administrative information, adverse drug reaction data, drug details, therapy start and end dates, report sources, and indications for use or diagnoses. To improve data reliability, a deduplication process was applied: for records with matching case unique IDs (CASE_ID), the most recent FDA received date (FDA_DT) was retained. If both CASE_ID and FDA_DT were identical, the record with the highest reporting ID (PRIMARY_ID) was selected. Only cases in which progesterone was identified as the primary suspected drug were included in the analysis.
All data used in this study were derived from the publicly available FAERS database, in which patient details are deidentified by the FDA. Furthermore, no personal identifiers were accessed, processed, or reported in our analysis. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 14
Signal detection and statistical analysis
Disproportionality analysis is widely applied in pharmacovigilance research as a method to compare the proportion of specific AEs associated with a particular drug to the proportion of the same AEs reported for all drugs in the database. Based on this approach, disproportionality measures are used to identify potential associations between drugs and AEs. 15
Four key metrics were employed to assess the signals of progesterone-related AEs: reporting odds ratio (ROR), proportional reporting ratio (PRR), Bayesian confidence propagation neural network (BCPNN), and multi-item gamma Poisson shrinker (MGPS). 16 These methods, which rely on 2 × 2 contingency table-based statistical analyses (Supplementary Table S1), are used to estimate the relative frequency of specific AEs attributed to a target drug over a defined period within the database. By calculating the disproportionality of AE reporting, these methods provide statistical insights into the association between a drug and specific AEs.
The ROR method assesses the likelihood of reporting a specific AE linked to progesterone compared with its reporting for all other drugs in the database. 17 PRR is calculated by dividing the proportion of AE reports for progesterone by that for other drugs. 18 The BCPNN method, based on Bayesian statistics, estimates the strength of the association between progesterone and specific AEs. 19 The MGPS method adjusts for multiple comparisons, providing a more conservative estimate of signal strength. 20 Specific formulas and signal detection criteria for the four algorithms are presented in Supplementary Table S2. Data analysis was performed using R software (version 4.4.0).
Data presentation
The SOC-level analysis provided an overview of AE categories, while the PT-level analysis identified specific clinical reactions with significant signals. Descriptive statistics were used to summarize the data, and results were presented in tables and charts to facilitate a detailed evaluation of progesterone-related AEs. This comprehensive approach offered valuable insights into the safety profile of progesterone in real-world clinical practice.
Results
Basic information on AEs
The flowchart of the data selection process is shown in Figure 1. As presented in Table 1, a total of 1827 AE cases associated with progesterone were reported in the FAERS database from 2004 Q1 to 2024 Q3. The majority of reports originated from consumers (42.64%), followed by physicians/healthcare professionals (25.72%), and medical device manufacturers (14.29%), with other reporters contributing smaller proportions. Regarding age distribution, over half of the cases (54.73%) had unknown age information. The most commonly reported age group was 18–44 years (25.73%), followed by 45–64 years (15.33%). Females accounted for 78.82% of cases, while sex was unspecified in 21.18% of cases. Weight data were largely unavailable, representing 81.23% of cases; among known cases, 13.63% accounted for individuals weighing less than 73 kg. Outcomes showed that 40% of cases had unknown results, while 37.71% were categorized as “other outcomes.” Hospitalizations occurred in 14.67% of cases, and severe outcomes, including death (1.46%) and life-threatening events (2.28%), were less frequently reported. Over the 20-year study period, the highest number of reports was recorded in 2024 (21.13%), followed by a steady decrease in earlier years. These findings provide a comprehensive overview of the characteristics of progesterone-related AEs in real-world settings.

Flow diagram of progesterone-related adverse event selection from the FAERS. FAERS: Food and Drug Administration Adverse Event Reporting System.
Characteristics of adverse events related to progesterone in the FAERS database (2004 Q1 to 2024 Q3).

CN: consumer; PH: physician/healthcare professional; MD: medical device manufacturer; OT: other; RN: regulatory authority; LW: lawyer; CA: case resolved; LT: life-threatening; DE: death; DS: disability; RI: recovery/resolved with sequelae.
As shown in Figure 2, most progesterone-related AE reports originated from the United States (1164, 63.71%), followed by Canada (125, 6.84%) and Germany (96, 5.26%). This geographic distribution highlights the predominance of reports from North America and Europe.

Country-wise distribution of AEs for progesterone. AE: adverse event.
Distribution and signal strength of progesterone AEs at the SOC level
A total of 152 positive risk signal AEs associated with progesterone were identified, involving 22 SOCs. The distribution of progesterone-related AEs across SOCs, measured by the number of PTs, is shown in Figure 3. Progesterone-related AEs were predominantly associated with pregnancy, puerperium, and perinatal conditions; reproductive system and breast disorders; injury, poisoning, and procedural complications.

Distribution of SOCs by quantity. SOC: system organ class.
In addition, we calculated the signal strength of progesterone-related AEs at the SOC level. As shown in Table 2, the top three SOCs with the strongest signals were pregnancy, puerperium, and perinatal conditions; reproductive system and breast disorders; and product issues.
AE for progesterone at the SOC level in the FAERS database.
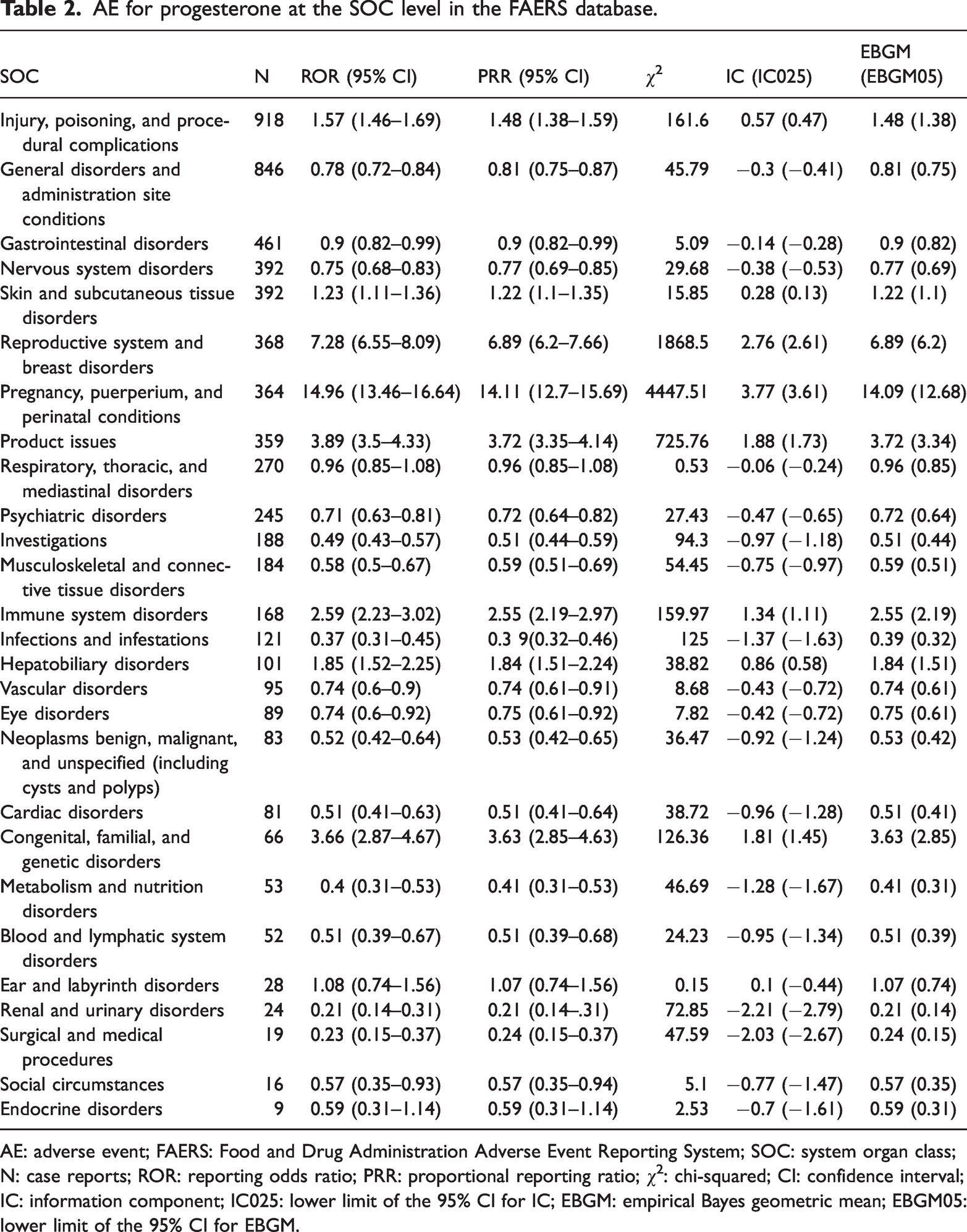
AE: adverse event; FAERS: Food and Drug Administration Adverse Event Reporting System; SOC: system organ class; N: case reports; ROR: reporting odds ratio; PRR: proportional reporting ratio; χ2: chi-squared; CI: confidence interval; IC: information component; IC025: lower limit of the 95% CI for IC; EBGM: empirical Bayes geometric mean; EBGM05: lower limit of the 95% CI for EBGM.
Signal strength and the frequency of progesterone-related AEs reported at the PT level
Table 3 and Figure 4 present the frequency of positive signals for the top 30 PTs reported for progesterone. The top three PTs were maternal exposure during pregnancy (n = 151), abortion spontaneous (n = 144), and product odor abnormal (n = 114). Similarly, Table 4 and Figure 5 show the signal strength of the top 30 PTs. The PTs with the highest signal strength were decidual cast, chondrodermatitis nodularis chronica helicis, and autoimmune dermatitis, with corresponding RORs (95% confidence intervals (CIs)) of 2825.23 (1336.81, 5970.87), 3897.61 (1007.63, 15076.37), and 1519.29 (908.61, 2540.41), respectively. These findings are further illustrated in Figure 6, highlighting their strong associations and statistical significance.
The positive signal frequency of reports in the top 30 PTs of progesterone.

FAERS: Food and Drug Administration Adverse Event Reporting System; PT: preferred term; N: case reports; ROR: reporting odds ratio; PRR: proportional reporting ratio; χ2: chi-squared; CI: confidence interval; IC: information component; IC025: lower limit of 95% CI for IC; EBGM: empirical Bayes geometric mean; EBGM05, lower limit of the 95% CI for EBGM.

Positive signal frequency of reports in the top 30 PTs of progesterone. PT: preferred term; FAERS: Food and Drug Administration Adverse Event Reporting System; ROR: reporting odds ratio; PRR: proportional reporting ratio; χ2: chi-squared; IC: information component; EBGM: empirical Bayes geometric mean.
The positive signal strength of reports in the top 30 PTs of progesterone.
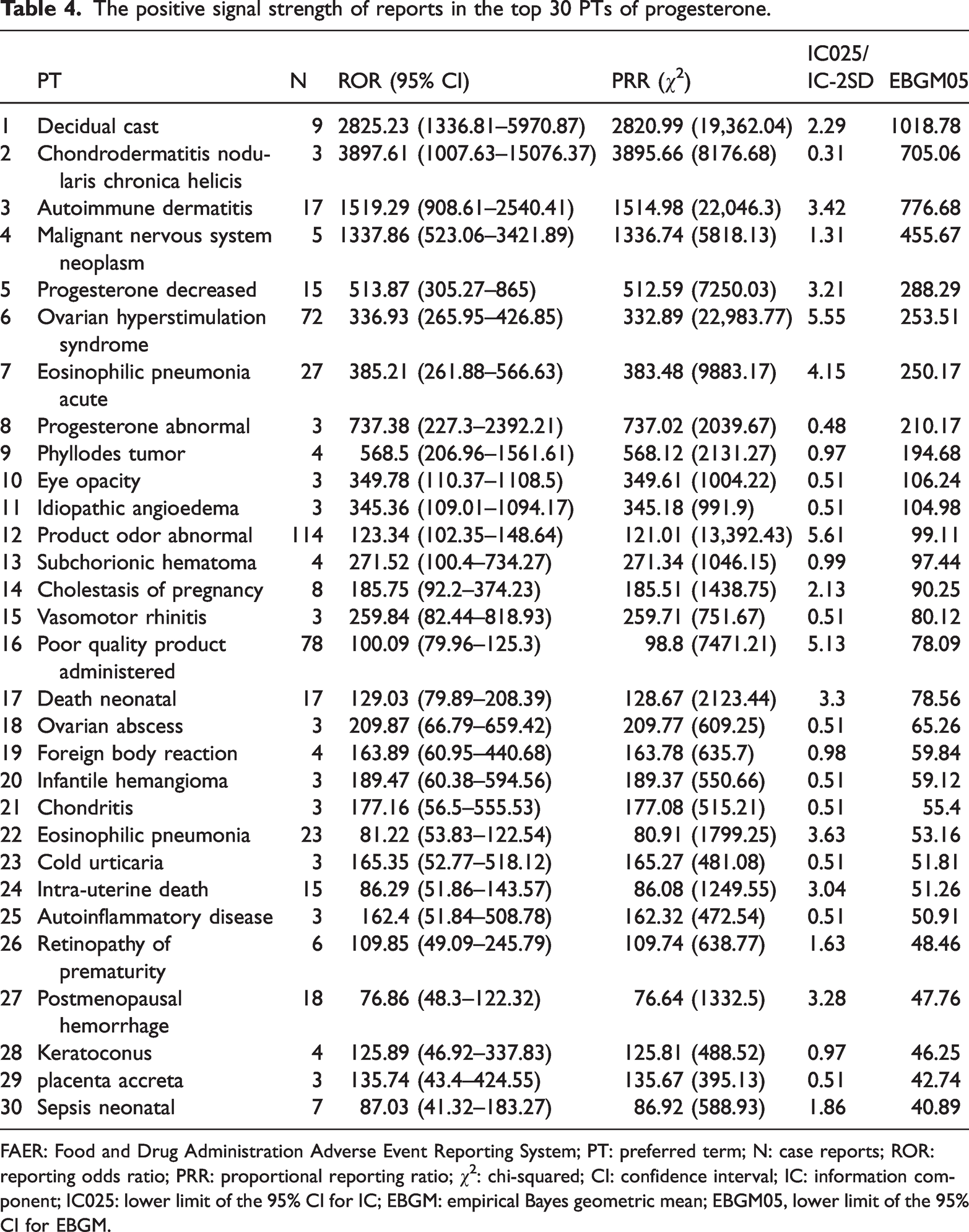
FAER: Food and Drug Administration Adverse Event Reporting System; PT: preferred term; N: case reports; ROR: reporting odds ratio; PRR: proportional reporting ratio; χ2: chi-squared; CI: confidence interval; IC: information component; IC025: lower limit of the 95% CI for IC; EBGM: empirical Bayes geometric mean; EBGM05, lower limit of the 95% CI for EBGM.

Positive signal strength of reports in the top 30 PTs of progesterone. PT: preferred term; FAERS: Food and Drug Administration Adverse Event Reporting System; ROR: reporting odds ratio; PRR: proportional reporting ratio; χ2: chi-squared; IC: information component; EBGM: empirical Bayes geometric mean.

Risk signal volcano plot for progesterone. The horizontal axis shows the log2 ROR value, and the vertical axis indicates the chi-squared (χ2) value. ROR: reporting odds ratio.
Time-to-onset of events
Figure 7 illustrates the distribution of time-to-onset (in days) for AEs associated with progesterone use. The majority of AEs occurred within 30–180 days (n = 192, 43.0%), followed by those occurring within less than 30 days (n = 100, 22.4%). AEs reported between 180–360 days and 360–720 days accounted for 12.8% (n = 57) and 12.1% (n = 54), respectively. This distribution indicates that most progesterone-related AEs manifest within the first 6 months of exposure. Notably, 9.8% (n = 43) of AEs occurred after 720 days.

Time-to-onset distribution (in days) for progesterone-related AEs. AE: adverse event.
Discussion
This study analyzed AE reports related to progesterone in FAERS from 2004 to 2024. Several important findings emerged, providing valuable information for a deeper understanding of progesterone safety. The analysis also highlighted issues that warrant attention in clinical use.
Regarding trends in AE reporting, the number of progesterone-related AE reports has steadily increased in recent years, peaking in 2024. This rise may reflect heightened awareness and improved reporting practices among healthcare professionals and consumers. Enhanced pharmacovigilance systems and better education on reporting procedures likely contributed to more frequent and comprehensive documentation of AEs. 21
In terms of report sources, consumers accounted for the largest proportion. This trend reflects growing patient awareness and engagement in drug safety. Recent efforts to encourage patient participation in pharmacovigilance, including initiatives promoting direct reporting in databases such as FAERS, have further supported this trend. 22 The increasing availability of online platforms and mobile health applications has also facilitated consumer reporting, leading to a higher number of submissions by nonprofessionals. 23 A significant proportion of reports originated from countries such as the United States, Canada, and Germany, reflecting the robust pharmacovigilance systems and proactive monitoring mechanisms in these regions. Healthcare professionals in these countries are generally more aware of the importance of AE reporting, contributing to higher reporting rates. 24
The study also found that progesterone-related AEs predominantly occurred in female patients and were concentrated in the 18–44 years age group. This pattern is closely related to the primary use of progesterone in the treatment of reproductive health–related conditions. Women in this age group use progesterone more frequently for reasons such as irregular menstruation, luteal support in assisted reproductive technology, and prevention of habitual miscarriage, making them more likely to be exposed to progesterone-related risks. These findings suggest that clinicians should carefully assess the risks and benefits when prescribing progesterone for this population.
Regarding the timing of AEs, most occurred within 6 months of exposure, with the 30–180 day period being the most common. This may reflect the body’s adaptation to hormonal changes induced by progesterone. As a hormone, progesterone affects the endocrine system upon administration, and the body requires time to adjust to these changes. During this adaptation period, various AEs may occur. 25 Although life-threatening and fatal AEs are relatively rare, their severity warrants careful attention. Clinicians should maintain high vigilance and strengthen patient monitoring and management.
Consistent with previous findings, this study observed common side effects of progesterone use, such as headache, dizziness, and nausea.26,27 From 2004 Q1 to 2024 Q3, frequently reported AEs primarily involved injury, poisoning, general and site-related disorders, and gastrointestinal problems. Progesterone can interact with other drugs, potentially causing abnormal metabolism or toxicity. 28 Overdose may result in symptoms such as dizziness and, in severe cases, confusion or hypotension. Progesterone injections can cause injection-site inflammation or infection. 29 Its smooth-muscle relaxing effect may delay gastric emptying, leading to bloating and indigestion. 30 Long-term oral use may result in cholestatic reactions and right upper abdominal pain. 31
Our study also identified several novel progesterone-related AEs, including acute eosinophilic pneumonia and meningioma, as well as rare but high signal-strength events, including chronic nodular chondrodermatitis of the helix, autoimmune dermatitis, and malignant nervous system tumors. These findings highlight the need for further investigation into their underlying mechanisms. Eosinophilic pneumonia is characterized by eosinophilic infiltration of lung tissue and can be triggered by drugs through immune-mediated hypersensitivity reactions. 32 Progesterone, as a steroid hormone, has been implicated in modulating immune responses, potentially shifting the balance toward Th2-mediated hypersensitivity pathways. 33 The high signal strength indicates an idiosyncratic reaction linked to genetic or epigenetic predispositions, such as polymorphisms in cytokine or eosinophil regulatory genes. 34 Meningiomas are primarily associated with genetic mutations or hormonal influences. Progesterone receptors (PR) are frequently expressed in meningioma tissues, suggesting that progesterone may promote tumor growth. 35 Prior studies have shown that high levels of exogenous progesterone can enhance the proliferation of meningioma cells in vitro, particularly in hormone-sensitive tumors. 5 Although causality remains speculative, these findings may explain the observed association and warrant further investigation. Autoimmune dermatitis may result from progesterone’s immune-modulating effects, as the hormone can influence regulatory T cell function and cytokine profiles. 33 Dysregulated immune homeostasis can lead to loss of self-tolerance, precipitating autoimmune skin reactions. This is consistent with reports of progesterone exacerbating autoimmune diseases, such as lupus erythematosus, through interactions with estrogen receptors and immune pathways. 36
Given these findings, clinicians should pay close attention to these rare but serious AEs, particularly in high-risk populations such as patients with comorbid autoimmune diseases or a history of allergies. Prior to treatment, risk stratification should be conducted through baseline assessments, including eosinophil counts and imaging examinations, along with individualized risk–benefit evaluations. During treatment, structured monitoring within the first 6 months is critical, as 43% of AEs occur between 30 and 180 days after exposure. Symptom checklists and targeted laboratory tests can facilitate timely detection of AEs, allowing appropriate interventions. Regulatory authorities should also consider updating progesterone product labels to clearly indicate potential risks associated with long-term and high-dose use of progesterone. Clinical guidelines should emphasize selecting appropriate administration methods according to the characteristics of different formulations. 37
Although informative, this study has several limitations. The FAERS database relies on spontaneous reporting, which inevitably introduces reporting bias. Some AEs may remain unreported for various reasons, or there may be inaccurate reports. To mitigate reporting bias, healthcare professionals should receive regular training to enhance awareness of the importance of AE reporting. Additionally, more convenient electronic reporting systems, including digital and automated tools, could simplify the reporting process. Encouraging patients to directly report AEs through accessible online platforms or applications may further reduce the reporting gap. Data incompleteness is another limitation. Key information, such as patient age, sex, weight, outcomes, comorbidities, or concurrent medications that may influence AEs, is often missing. This hampers in-depth analysis of relationships between these factors and AEs and increases the difficulty of controlling for confounding factors. Future studies should aim to include complete key information or adopt prospective research methods for comprehensive data collection, thereby improving evidence quality and providing more reliable guidance for clinical decision-making. Due to data constraints, this study was unable to stratify or compare safety signals across different routes of progesterone administration (oral vs. vaginal vs. intramuscular). Different administration routes may lead to varying systemic exposure levels and thus divergent adverse-event profiles; the absence of such information is an additional limitation. Moreover, this study could not distinguish the effects of different progesterone formulations, doses, or treatment durations on the occurrence and severity of AEs. Although major synthetic progestins were excluded, FAERS records often lack detailed information on drug subtypes, such as micronization processes or excipient composition, which may further impede precise interpretation of our findings. Given the diversity of progesterone formulations and regimens, future research should focus on collecting more detailed data to clarify the safety characteristics of different formulations.
Conclusion
This pharmacovigilance study identified significant signals for both known and potentially novel AEs associated with progesterone, including rare but strongly associated reactions. The findings underscore the importance of continued vigilance, particularly in high-risk patients, and suggest that proactive monitoring and further investigation into the underlying mechanisms of these events are warranted. The inherent limitations of spontaneous reporting data should be considered when interpreting these results.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605261417447 - Supplemental material for Safety profile of progesterone: Insights from an FDA Adverse Event Reporting System (FAERS)–based pharmacovigilance study
Supplemental material, sj-pdf-1-imr-10.1177_03000605261417447 for Safety profile of progesterone: Insights from an FDA Adverse Event Reporting System (FAERS)–based pharmacovigilance study by Ren Xu, Xiaona Wang, Junqin Zhang, Yanan Ren, Luyang Su, Lu Sang and Jianzhi Su in Journal of International Medical Research
Footnotes
Acknowledgments
Not applicable.
Authors’ contributions
Ren Xu contributed to the conception and design of the study, data analysis, and interpretation of the results.
Xiaona Wang assisted with data analysis and manuscript preparation.
Junqin Zhang contributed to data collection and provided critical revisions to the manuscript.
Luyang Su performed statistical analyses and reviewed the manuscript.
Yanan Ren contributed to the study design and critical revision of the manuscript.
Lu Sang contributed to the interpretation of the results and manuscript editing.
Jianzhi Su supervised the study, provided overall guidance, and reviewed the final manuscript.
Availability of data and materials
Declaration of conflicting interests
The authors declare no potential conflicts of interest regarding the research, authorship, or publication of this article.
Ethics approval and consent to participate
This retrospective pharmacovigilance analysis used exclusively deidentified, publicly available data from the FAERS database. The study protocol was reviewed and designated as exempt from formal ethics approval by Hebei General Hospital. As no human participants were directly involved and all data were anonymized, the requirement for informed consent was waived.
Funding
This study was supported by the Medical Science Research Project of Hebei (NO. 20230273, NO. 20240719, NO. 20230328, NO. 2026042).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
