Abstract
Metabolic acidosis is a frequent complication when chronic kidney disease (CKD) develops, plus it is linked to adverse outcomes such as muscle wasting, inflammation, and progression of kidney disease. This article is intended to evaluate how correcting metabolic acidosis affects adult CKD patients via summarizing evidence from systematic reviews with meta-analyses. We conducted an umbrella review. Systematic reviews with meta-analyses assessing alkali therapy in CKD patients were included. Methodological quality was assessed using a Measurement Tool to Assess Systematic Reviews (AMSTAR 2). The Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) certainty of evidence approach was used for rating the certainty of evidence. Twelve meta-analyses that were published between 2007 and 2024 were analyzed, and these included 169 associations, out of which 128 remained after excluding single-study outcomes. Only 8 (6.2%) of these associations were supported by high-certainty evidence, which included a slower decline in glomerular filtration rate, reduced hospitalization rates, lower diastolic blood pressure, increased serum bicarbonate after short-term therapy, and reduced use of β-blockers and vasodilators. However, most associations (52.3%) were rated as having very low certainty. Less than 10% of the associations analyzed in the selected systematic reviews have high certainty of evidence, which underscores a significant gap between common clinical practices and the strength of the supporting evidence.
Trial registration:
PROSPERO CRD42024548458.
Introduction
In patients with chronic kidney disease (CKD), as the Glomerular Filtration Rate (GFR) declines, so does the kidney’s ability to excrete hydrogen ions and generate bicarbonate, leading to the development of chronic metabolic acidosis. 1 This typically occurs when GFR falls below 60 mL/min/1.73 m2, with more pronounced reductions when it is less than 30 and 15 mL/min, that is, in CKD stages G4 and G5. The adjusted prevalence in adults with serum bicarbonate less than 22 mmol/L is 7.7% and 6.7% in those with and without diabetes in stages G3 and A1, respectively, increasing to 38.3% and 35.9% in CKD stages G5 and A3. 1
Metabolic acidosis is associated with an increased risk of protein catabolism, muscle wasting, inflammation, and other complications, such as impaired cardiac function and mortality, which are also associated with decreased GFR.1–3 Although causality between metabolic acidosis and these outcomes has not been determined, treatment of acidosis with bicarbonate has been recommended. 1 The KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease recommends that, in individuals with CKD, pharmacologic therapy with or without dietary intervention should be considered to prevent the development of acidosis with potential clinical implications (e.g., serum bicarbonate <18 mmol/L in adults). 1 However, this recommendation is not without adverse effects, which is why monitoring treatment for metabolic acidosis is also recommended to ensure that it does not cause serum bicarbonate concentrations above the upper limit of normal or adversely affect blood pressure control, serum potassium, or fluid status. 1
The working group that developed this Clinical Practice Guideline did not issue a graded recommendation for the treatment of acidosis due to the lack of large-scale randomized clinical trials supporting its use, so the overall evidence remains limited. 1 The results of small- to medium-sized clinical trials in people with subclinical metabolic acidosis have also been inconclusive, so well-designed clinical trials are needed to determine the efficacy and safety of sodium bicarbonate and determine whether this intervention improves patient outcomes.1,2 To summarize the evidence from these primary studies, several systematic reviews have been published.4–8 However, to the best of our knowledge, there is no umbrella review on this topic. 9 Accordingly, the objective of this study was to conduct an umbrella review evaluating the effects of correcting metabolic acidosis in patients with CKD.
Methods
Protocol registration
This umbrella review was conducted in accordance with the recommendations outlined in the Cochrane Handbook 10 and is reported following the Preferred Reporting Items for Overviews of Reviews (PRIOR) statement (Table S1). 11 The protocol for this review was registered in the International Prospective Register of Systematic Reviews (PROSPERO) under the code CRD42024548458.
Search strategy
Three authors independently reviewed published meta-analytic systematic reviews by searching PubMed, Scopus, Embase, Web of Science, and Ovid Medline databases from their inception to December 2024.
The search strategy is presented in Table S2. We reviewed the studies of bibliographies to verify referenced sources and ensure the inclusion of relevant studies.
Eligibility criteria
Eligible studies for this umbrella review included systematic reviews with meta-analyses published in peer-reviewed journals. The target population comprised adults diagnosed with CKD at any stage and presenting with metabolic acidosis. Studies were considered if they evaluated the effects of correcting metabolic acidosis, including interventions with oral or dialysis bicarbonate supplementation, compared to no correction or other treatment strategies. If multiple systematic reviews addressed the exact same intervention(s) and outcome(s), we prioritized the inclusion of the review with the largest number of primary studies and highest methodological quality, as assessed by A Measurement Tool to Assess Systematic Reviews (AMSTAR 2) 12 assessment tool. Additionally, we performed an overlap analysis of the primary studies included in the meta-analyses using the Corrected Covered Area (CCA) method to identify potential duplication of evidence that could bias the results. 13
Eligible outcomes included primary parameters such as nutritional status, bone turnover, blood pressure, quality of life, hospitalization rates, and mortality. Additional outcomes considered were uremia, antihypertensive agent use, GFR, proteinuria, and the development of cardiovascular disease. There were no restrictions on language, sex, or CKD stage for inclusion in this review. Studies were excluded if they did not include a meta-analytical component or if the population did not consist of CKD patients with metabolic acidosis.
Data extraction
Two reviewers independently extracted data using a predefined protocol, with any discrepancies resolved by a third reviewer. Information collected included study details (author, year, number of primary studies, sample size), type of intervention, outcomes assessed, and effect measures (risk ratio (RR), odds ratio (OR), mean difference (MD), or Standardized Mean Difference (SMD) with 95% confidence intervals (CIs)).
Outcomes were classified as primary (blood pressure, bone turnover, hospitalization, mortality) and secondary (uremia, use of antihypertensives, GFR, proteinuria, and cardiovascular disease). Statistical methods used, such as random-effects models, heterogeneity (I2), and the use of subgroup analyses, were also recorded to ensure analytical consistency.
Quality assessment of evidence and certainty of evidence
The methodological quality of the included meta-analyses was assessed using the AMSTAR 2 tool. 12 The certainty of the evidence was evaluated using the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) framework, 14 which classifies confidence in effect estimates into four levels: high, moderate, low, and very low.
Results
Selection of studies
A total of 2822 articles were retrieved from the PubMed, Scopus, Embase, Web of Science, and OVID-Medline databases. After eliminating duplicates, 2328 unique articles remained. Screening titles and abstracts led to the exclusion of 2314 records. Two additional articles15,16 were removed after full-text evaluation because they did not include meta-analysis. Ultimately, 12 articles4,5,7,17–25 met the criteria for inclusion in this umbrella review (Figure 1).
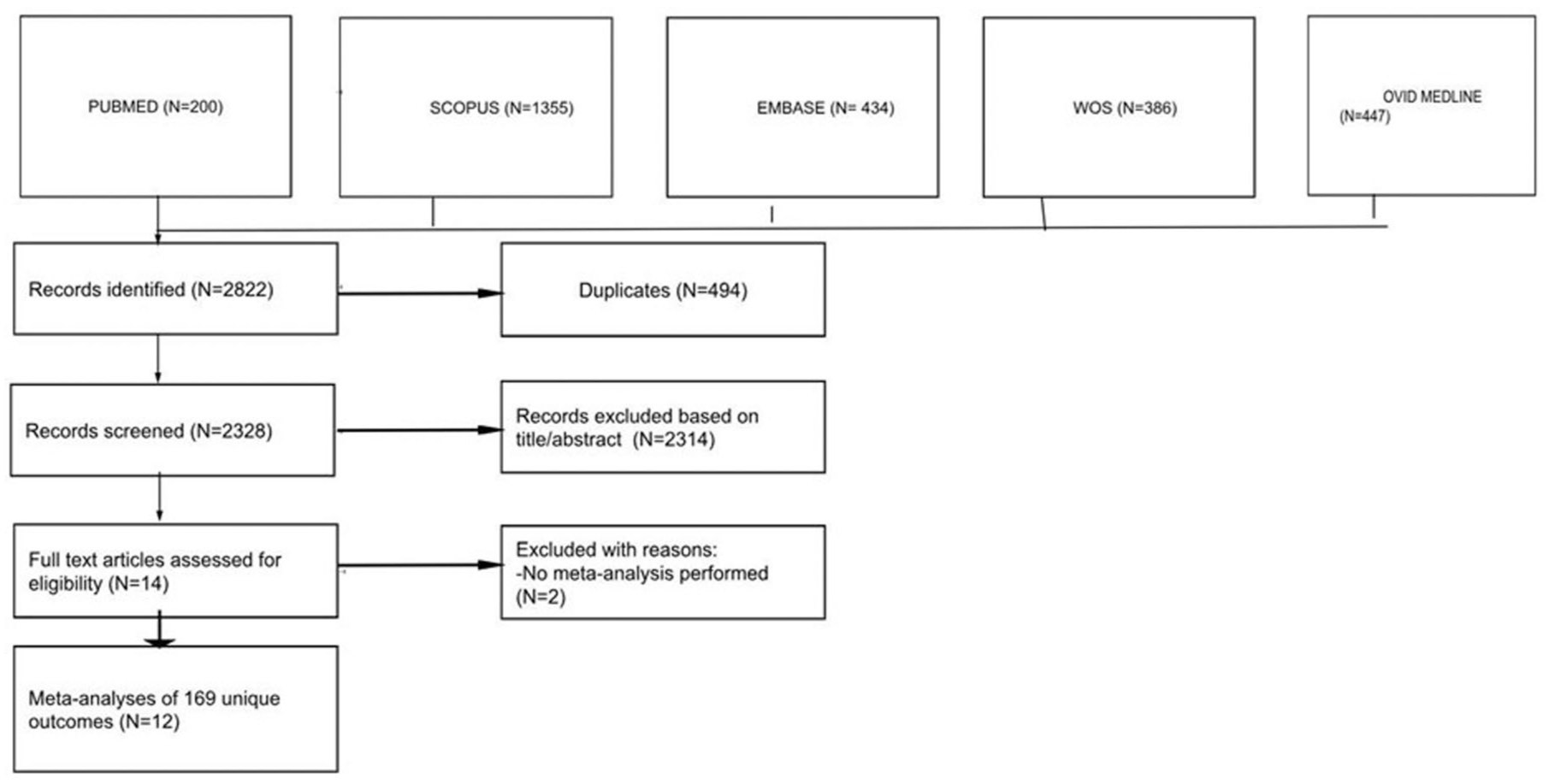
PRISMA flow diagram of the identification, screening, eligibility, and inclusion of systematic reviews and meta-analyses in this umbrella review.
Characteristics of the included meta-analyses
The 12 eligible articles, published between 2007 and 2024, evaluated 169 associations examining the correction of metabolic acidosis in relation to both clinical outcomes (e.g., systolic blood pressure) and laboratory parameters (e.g., proteinuria). The general characteristics of the included studies are summarized in Table 1. The number of outcomes assessed per study ranged from 4 to 30, with a median of 562 participants per outcome (range: 12–2746). Notably, aside from Hultin et al., 17 none of the studies conducted a GRADE assessment of their findings. Although some outcomes were similar across studies, differences in intervention types and/or participant populations ensured that no identical outcomes had to be excluded in this review. Additionally, the CCA overlap analysis yielded a value of 0.18%, indicating a slight degree of overlap. 13
Summary characteristics of included systematic reviews and meta-analyses.

GRADE: Grading of Recommendations Assessment, Development, and Evaluation; NR: not reported.
Methodological quality of included meta-analyses
The AMSTAR 2 (Table S3) confidence assessment indicated that all 12 articles were rated as having critically low confidence. Among the critical domains, questions 2 and 4 most frequently received “No” or “Partial Yes” responses (10 out of 12 articles), whereas question 11 received these responses less frequently (2 out of 12 articles).
Summary findings and heterogeneity of the included meta-analyses
The summary of findings is displayed in Table 2. Among the 169 associations, the summary of random-effects estimates ranged from −1872 to 77. A total of 56 associations (33%) were statistically significant at p < 0.05 based on the random-effects model. Regarding heterogeneity, 59 associations (34.9%) had low heterogeneity (I2 < 30%); 26 (15.3%) showed high heterogeneity (I2 = 30%–60%); and 43 (25.4%) exhibited very high heterogeneity (I2 > 60%). The remaining 41 associations (24.2%) included only one or no studies, preventing the calculation of statistical heterogeneity.
Summary of meta-analysis results and certainty of evidence (GRADE).

GFR: glomerular filtration rate; eGFR: estimated glomerular filtration rate; PTH: parathyroid hormone; BP, blood pressure; BUN: blood urea nitrogen; ACR: albumin-to-creatinine ratio; SBP: systolic blood pressure; BMI: body mass index; ESKD: end stage kidney disease; CV: cardiovascular; CKD: chronic kidney disease; MA: metabolic acidosis; MD: mean difference; SMD: standardized mean difference; RR: risk ratio; OR: odds ratio; NA: not applicable; NR: not reported; RCT: randomized control trial; GRADE score: Grading of Recommendations Assessment, Development and Evaluation score; RRT: renal replacement therapy; BW: body weight; HGS: handgrip strength; LBM: lean body mass; MAMC: mid-arm muscle circumference; NPNA: normalized protein nitrogen appearance; PCR: protein catabolic rate; SGA: subjective global assessment; STS: sit-to-stand time.
NA, as those studies had less than two primary studies included in their analysis, so no association could be made.
The authors divided the studies into two groups: 13 studies plus Raphael et al. with high-dose IV bicarbonate and 13 studies plus Raphael et al. with low-dose IV bicarbonate, since Raphael et al. compared high and low doses versus a control group.
Certainty of evidence
As previously mentioned, 41 of the 169 associations analyzed in this review were based on a single study or none. The GRADE framework assessment (Table S4) of the remaining 128 associations found that only 8 (6.2%) were supported by high-quality evidence. These included a reduced use of β-blockers (intervention vs control, RR = 0.39, 95% CI: 0.18–0.84) and a reduced use of vasodilators (intervention vs control, RR = 0.64, 95% CI: 0.45–0.92); no change in mean systolic blood pressure in 14 primary studies after medium-term (24–28 weeks) sodium bicarbonate use (intervention vs control, MD = −2.25, 95% CI: −6.00 to 1.50); no change in mean systolic blood pressure in 14 primary studies in CKD stage 4 patients (intervention vs control, MD = 1.86, 95% CI: −1.21 to 4.93); a slower decline in GFR (mL/min/1.73 m2; intervention vs control, MD = −4.44, 95% CI: −4.92 to −3.96); a reduction in diastolic blood pressure (intervention vs control, MD = −1.26, 95% CI: −2.33 to −0.19); lower hospitalization rates (intervention vs control, OR = 0.37, 95% CI: 0.25–0.55); and increased serum bicarbonate levels in patients receiving short-term alkali therapy (⩽7 days; intervention vs control, MD = 5.4, 95% CI: 4.0–6.7).
All comparisons were made against placebo, no intervention, or standard therapy. The remaining associations were classified as moderate-quality evidence (n = 23; 17.9%), low-quality evidence (n = 30; 23.4%), and very low-quality evidence (n = 67; 52.3%).
Discussion
Our main results show that less than 10% of the associations evaluated in the included systematic reviews were supported by high certainty of evidence according to our independent GRADE assessment. These include that bicarbonate use is not associated with changes in mean systolic blood pressure, that its use is associated with a reduction in diastolic blood pressure compared to the control group, and that the control group has greater use of β-blockers and vasodilators than patients using bicarbonate. Likewise, there is high-quality evidence regarding the association of bicarbonate use with a slower decline in GFR, higher serum bicarbonate levels, and lower hospitalization rates.
Metabolic acidosis develops as CKD progresses due to a decreased acid excretory capacity and high daily endogenous and exogenous acid loads, resulting in a positive H+ balance. 26 As nephron mass decreases in CKD patients, the single-nephron GFR increases, which, in turn, increases acid excretion, especially ammoniagenesis. 27 However, net renal acid excretion decreases because the remaining nephrons do not excrete the acid load sufficiently.26,27 Furthermore, there is a decreased renal capacity to produce ammonia to neutralize the daily acid load, resulting in a normal anion gap metabolic acidosis characteristic of patients with moderate CKD. 28 Despite this, titratable acid excretion is relatively preserved, in contrast to the reduced renal ammonia production, until the GFR falls below 15 mL/min/1.73 m2 (G5). 28 When titratable acid excretion decreases, a high anion gap metabolic acidosis occurs secondary to the accumulation of phosphate and other anions. 28 Additionally, as CKD progresses, proximal tubule bicarbonate reabsorption also decreases, which is associated with an increase in the bicarbonate load per nephron that exceeds its reabsorption capacity. 29
The first data on the beneficial effects of alkalizing therapy on CKD progression were published in 1931. 30 Currently, sodium bicarbonate is the main pharmacological treatment for metabolic acidosis in patients with CKD, with a value below 22 mmol/L established as the diagnostic threshold, after some observational studies showed that plasma or venous blood concentrations below 21–23 mmol/L are characterized by greater CKD progression and higher mortality. 31 However, despite numerous clinical trials testing its efficacy and systematic reviews attempting to synthesize their evidence, only 8 of the 169 associations evaluated in the systematic reviews that met the pre-established selection criteria were supported by high-quality evidence, including aspects related to blood pressure, CKD progression, and safety.
The effects of sodium bicarbonate use on blood pressure were supported by conflicting evidence; however, there was a theoretical risk that its use could increase blood pressure. The KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease recommends monitoring its use due to the possibility that it may negatively affect blood pressure control. 1 This theoretical risk is related to the fact that sodium loading increases fluid retention; however, the evidence calls this claim into question. In a study that aimed to profile the fluid retention characteristics of sodium bicarbonate and sodium citrate to determine the efficacy of these buffer media as hyperhydrating agents, it was found that although plasma volume expansion was greater in both bicarbonate and citrate users compared to the control group using water alone, no increase in body mass was observed. 32 Additionally, in animal models of hypertension, blood pressure was observed to increase only if sodium was coadministered with chloride, but not with other anions (including bicarbonate), suggesting that the effects of sodium on blood pressure only occur when chloride is the coadministered anion. 2 Indeed, human studies suggest that in patients with and without CKD, ingested sodium in the form of bicarbonate is completely excreted in the urine, while sodium in the form of chloride is retained, which potentiates its effect on systolic blood pressure.33,34
Another result with high-quality evidence is that the use of beta-blockers and vasodilators is higher in the control group compared to patients who use bicarbonate. However, it is necessary to highlight the uncertainty in the medication findings due to the difficulty in comparing dose changes of antihypertensive medication and diuretics within and between the clinical trials included in the systematic review that found this association. This also diminishes the quality of the evidence that sodium bicarbonate treatment is not associated with an increase in the use of antihypertensives or diuretics 24 (Table 2). This is relevant because increased antihypertensive treatment was the argument used to explain the dramatic decrease in diastolic blood pressure in the systematic review that supports the high quality of evidence for this association. 18
Regarding the high-quality evidence that bicarbonate use is associated with a slowing of the decline in GFR, this may be related to the fact that correcting metabolic acidosis reverses its effects on CKD progression. 31 Indeed, metabolic acidosis contributes to the development of the inflammatory process in CKD patients and the vicious cycle initiated by acid retention leads to a cascade of events that contribute to the progression of CKD. 3 Metabolic acidosis triggers increased ammoniagenesis in the remaining nephrons, the production of endothelin-1, and the activation of the renin–angiotensin system, which subsequently promotes complement activation by ammonium as well as the generation of proinflammatory and profibrotic mediators. 3 As a result, interstitial fibrosis, tubular atrophy, and loss of podocyte integrity occur, culminating in the loss of functional nephrons. 3 Despite this, the effect of sodium bicarbonate on renal fibrosis is not fully understood. In a study of diabetic patients, sodium bicarbonate use did not significantly reduce TGF-β1, KIM-1, fibronectin, NGAL, or urinary albuminuria during 6 months of treatment. 35
The CRIC observational study found that elevated serum bicarbonate levels above 24 mmol/L are associated with an increased risk of congestive heart failure; specifically, for every 1 mmol/L increase above this threshold, the risk increases by 14%. 36 Following this association, several studies evaluating bicarbonate use have considered hospitalizations as a surrogate measure of safety. Our findings show high-quality evidence that bicarbonate treatment successfully increases serum bicarbonate levels in patients who receive it. We also found high-quality evidence suggesting that bicarbonate use is associated with a lower rate of hospitalizations. However, it is important to note that this conclusion comes from a systematic review 22 that, after excluding one study with a high risk of bias but with considerable statistical weight, 37 did not find a significant reduction in hospitalizations. This finding is consistent with evidence indicating that adverse effects related to fluid retention, such as weight gain, occur primarily when sodium is administered as chloride,33,34 and not necessarily when it is administered as bicarbonate.
The present study is the first umbrella review to synthesize the published evidence on correcting metabolic acidosis in patients with CKD. The high certainty of evidence observed for some outcomes has important clinical implications. First, bicarbonate therapy effectively increases serum bicarbonate levels, supporting its use in treating metabolic acidosis. Second, it may slow CKD progression, although the exact mechanisms remain unclear. Third, no significant variations in blood pressure were observed, though monitoring systolic blood pressure is advisable due to potential unverified changes in concomitant medications. These results may guide physicians in using bicarbonate therapy to correct metabolic acidosis and mitigate CKD progression with relative safety.
Our findings should be interpreted in light of several limitations. The clinical and methodological heterogeneity of the primary studies included may have influenced the results. For example, trials enrolled patients with CKD at different stages, varying prevalence of metabolic acidosis, and baseline bicarbonate levels above 22 mmol/L. Variability in follow-up duration, intervention doses, and study designs—such as open-label, single-center trials with small sample sizes—may have affected the observed effects on key outcomes.
Despite these limitations, our GRADE evaluation indicated high-certainty evidence for several outcomes, even though most systematic reviews were rated as low or critically low in methodological quality according to AMSTAR 2. This apparent discrepancy reflects the different focus of these tools: AMSTAR 2 evaluates the methodological rigor of systematic reviews, whereas GRADE assesses the certainty of the underlying evidence. Nevertheless, the low AMSTAR 2 ratings caution against overinterpreting the comprehensiveness and reproducibility of the included reviews.
Conclusion
In conclusion, less than 10% of the associations analyzed in the included systematic reviews were supported by high certainty of evidence, particularly those related to the effects of sodium bicarbonate on the treatment of metabolic acidosis, blood pressure, CKD progression, and safety outcomes. However, our review highlights the need for new, well-designed clinical trials to address the methodological limitations observed in the existing studies. Future research should include patients with CKD stages in which metabolic acidosis is clinically relevant, enroll individuals with bicarbonate levels for which bicarbonate therapy is recommended, and recruit sufficiently large samples to yield robust and generalizable results, among other key aspects.
Supplemental Material
sj-docx-1-taj-10.1177_27558428251411463 – Supplemental material for Effects of correcting metabolic acidosis in chronic kidney disease patients: An umbrella review
Supplemental material, sj-docx-1-taj-10.1177_27558428251411463 for Effects of correcting metabolic acidosis in chronic kidney disease patients: An umbrella review by Percy Herrera-Añazco, Miguel A. Huayta-Cortez, Juan R. Ulloque-Badaracco, Christian N. Rojas, Nella Altamirano-Cabezas and Vicente A. Benites-Zapata in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
None to report.
ORCID iDs
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data generated or used during our research are included in this published article and its Supporting Information Files.
Supplemental material
Supplemental material for this article is available online.
