Abstract
Background:
One fundamental element of strengthening maternal health initiatives and outcomes involves improving medication access. However, most of the people in many low- and middle-income countries do not have access to these essential medicines for maternal and child health.
Objectives:
This study aimed to determine the availability of necessary medications for maternal health in public health facilities in Mwanza, Tanzania.
Methods:
A retrospective cross-sectional study was conducted in April 2024 in public health facilities in Ilemela and Nyamagana districts. Availability of 13 essential medicines was collected from 36 public health facilities. Data was collected from the public health facilities’ store records (ledgers and bin cards) using a checklist (data collection tool) adapted and modified from the World Health Organization (WHO) Health Action International tool.
Results:
The overall median point availability of essential medicines for maternal health was 94.4% (interquartile range (IQR), 80.6–97.2). The overall public health facility median number of stock-outs (periods when medicines were unavailable) was 3, and the median number of stock-out days was 94 in the past 3 months. The most stocked-out medicines during the period were calcium gluconate (93.4%), misoprostol (32.9%), gentamicin (16.1%) and methyldopa (10.6%). Generally, medicines used only for the management of maternal health conditions were most stocked out compared to medicines with broader indications, 33.3% and 13.9%.
Conclusion:
Despite a relatively high overall median point availability, frequent stock-outs were observed, particularly for critical medicines like calcium gluconate, misoprostol, gentamicin and methyldopa. Medicines used solely for maternal health were more likely to experience stock-outs compared to those with broader indications. These findings highlight the need for improved supply chain management and prioritization of maternal health-specific medicines to enhance the quality of maternal healthcare services in the region.
Plain Language Summary
This study explored the availability of essential medicines for maternal health in public health facilities in Ilemela and Nyamagana districts, Mwanza, Tanzania. Access to these medicines is crucial for improving maternal health outcomes, but many low- and middle-income countries face challenges in ensuring availability. Data on 13 essential maternal health medicines were collected from 36 public health facilities, using records like ledgers and bin cards. The study found that 94.4% of the essential medicines were generally available. However, stock-outs – periods when medicines were unavailable – were frequent. Some of the most commonly stocked-out medicines included calcium gluconate, which was unavailable 93.4% of the time, misoprostol (32.9%), gentamicin (16.1%) and methyldopa (10.6%). Medicines used exclusively for maternal health, such as those for pregnancy and childbirth-related conditions, were more prone to stock-outs (33.3%) compared to medicines with broader uses (13.9%). These shortages of critical maternal health medicines emphasize the need for improved supply chain management and better prioritization of these drugs. By addressing these gaps, the quality of maternal healthcare services could be significantly enhanced, ensuring better health outcomes for mothers in the region.
Introduction
Globally, maternal mortality is still a major problem. Reducing the global maternal mortality rate (MMR) to less than 70 deaths per 100,000 live births by 2030 is one of the sustainable development goals (SDGs) of the 2030 Agenda. 1 With proven, cost-effective treatments for infectious illnesses and maternal complications, the majority of maternal deaths can be avoided or treated. 2 The MMR is still high in low- and middle-income countries (LMIC) despite several worldwide efforts to reduce it. 3 Indeed, the global MMR decreased by 34.3% between 2000 and 2020, from 339 to 223 per 100,000 live births. 4 Approximately 95% of the 287,000 maternal deaths that were reported in 2020 took place in LMICs; nevertheless, 69% of all maternal deaths globally in 2020 took place in Africa, with an MMR of 531 deaths per 100,000 live births. 5 Therefore, lowering maternal mortality is still a major global health concern, particularly in LMICs.
The availability of essential medicines in African countries is hampered by a number of factors, including a lack of human resources, financial limitations, the high cost of currently available medications on the market, inadequate inventory management, manual consumption forecasting and inefficiencies in drug registration. 6 One fundamental element of strengthening maternal health initiatives and outcomes involves improving medication access. The WHO released a list of ‘priority medicines’ for maternal and child health in April 2011 to assist nations in selecting medications for women’s treatment. Medications for the treatment of postpartum haemorrhage, severe pre-eclampsia and eclampsia, maternal sepsis, sexually transmitted infections, premature birth, human immunodeficiency virus/acquired immune deficiency syndrome (HIV/AIDS) and malaria were prioritized for maternal health. 7 However, the majority of the people in many LMICs do not have access to these essential medicines for maternal and child health (MCH), which continues to be a significant problem.8,9 This ultimately limits initiatives to lower mortality and improve maternal health. The unpredictable supply of pharmaceuticals, particularly in public health facilities, is the primary cause of the problem with MCH medication availability.10,11
In sub-Saharan Africa, the availability of medications for maternal health care has been reported in very few studies.12,13 Research on the availability of key life-saving medications can assist in identifying policy analysis gaps and increasing accessibility. 14 This study aimed to determine the availability of necessary medications for maternal health in public health facilities in Mwanza, Tanzania. Mwanza Region was selected due to its urban demographic structure and its role as a referral hub for northwestern Tanzania. The study focuses on public facilities in urban districts to assess whether supply chain challenges persist even in relatively better-resourced settings.
Methodology
Study design and area
In April 2024, we conducted a retrospective cross-sectional study in the Mwanza region of Tanzania, in Ilemela and Nyamagana districts, encompassing the Mwanza urban area. According to the 2022 Population and Housing Census data, Ilemela district has a population of 509,687 people, while Nyamagana district has a population of 594,834 people. 15 The healthcare system in these districts comprises 40 public health facilities, including hospitals, health centres and dispensaries that serve as primary access points for maternal healthcare services. All public health facilities that offer maternal healthcare services located in Ilemela and Nyamagana districts of Mwanza were included in this study. The zonal referral hospital, the regional referral hospital and the military hospital were excluded. These hospitals were excluded because they operate under different administrative structures, funding mechanisms and procurement systems compared to standard public health facilities.
Sample size and sampling procedure
The sample size (n) was calculated using the Taro Yamane formula:
Data collection procedure
Data on 13 essential medicines were collected from the store records of 36 public health facilities (ledgers and bin cards) using a checklist (data collection tool) adapted and modified from the WHO-HAI (Health Action International) tool. 16 These 13 medicines were selected based on the WHO’s list of priority maternal health medicines and their inclusion in Tanzania’s national essential medicines list, targeting life-threatening maternal conditions such as postpartum haemorrhage, pre-eclampsia/eclampsia and sepsis. 17 Point availability was calculated as the percentage of facilities that had stock of the respective medicine at the time of the visit, irrespective of the amount or pack size available, as recommended by the WHO-HAI methodology. Period availability was calculated as the total number of days in which the medicine was available divided by 90 and multiplied by 100. Maximum stock out days for 36 facilities were 3240 (36 × 90 days). Medicine stock-outs for the past 3 months for all 36 public health facilities were calculated as the total number of days the respective medicine was out of stock divided by 3240, then multiplied by 100.
Data analysis
Data for descriptive statistics were entered and cleaned in Microsoft Excel before being exported to STATA version 15 (Stata Corp, College Station, TX) for further analysis. In the statistical analysis, frequencies and percentages were calculated. The results were presented in frequency distribution tables and figures. Medicine point and period availability were calculated as the percent availability of individual medicines and the median average percent. The availability of medicines in public healthcare facilities and the ranges were described as < 30%, 30%–49%, 50%–80% and >80% for very low, low, fairly high and high availability, respectively. 18
Results
The study was carried out in 36 public health facilities (1 hospital, 7 health centres, 28 dispensaries).
Availability of essential medicines for maternal health
Point availability
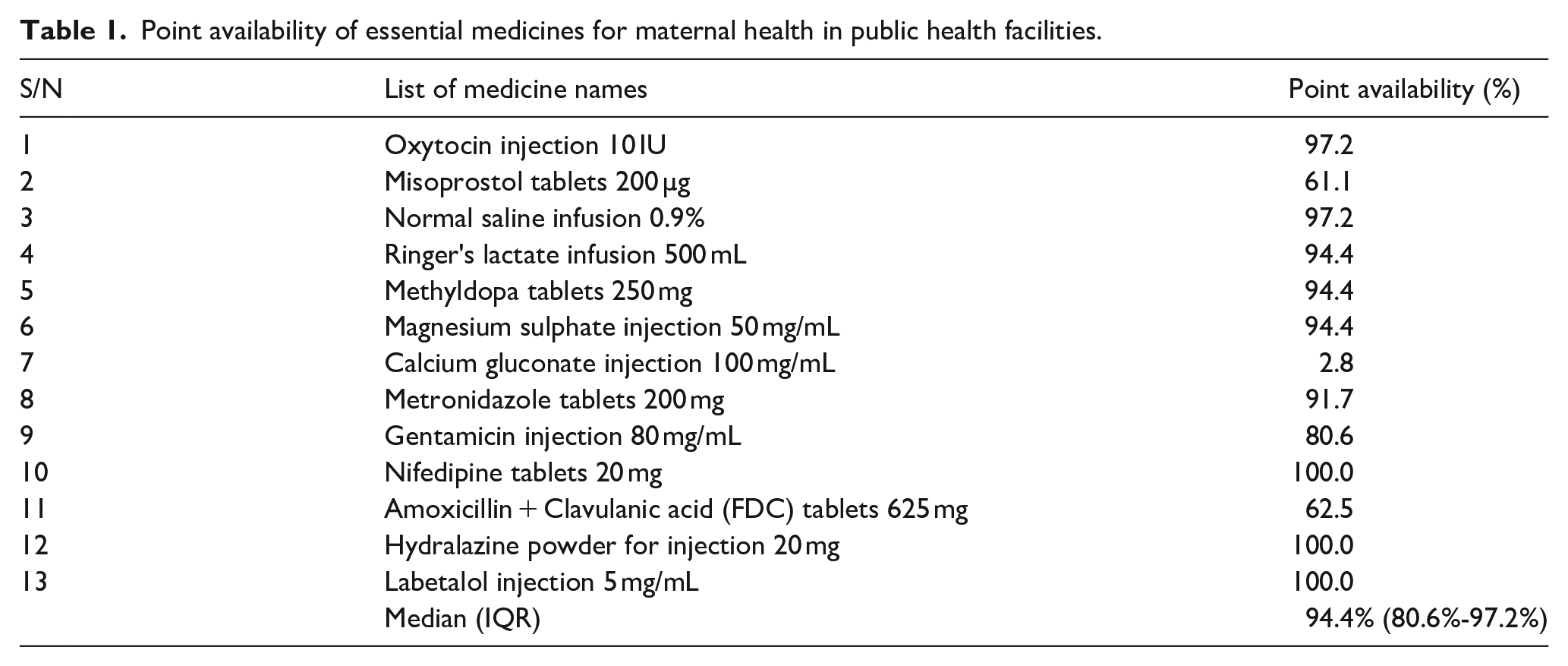
The overall median point availability of essential medicines for maternal health was 94.4% (IQR: 80.6%–97.2%). In terms of individual medicine availability, Labetalol, Hydralazine and Nifedipine had the highest availability (100.0%), followed by Oxytocin and Normal Saline (97.2%). On the other hand, Calcium Gluconate, Misoprostol, and Amoxicillin + Clavulanic acid were found in only 2.8% (very low), 61.1% (fairly high) and 62.5% (fairly high) of the facilities visited, respectively, as shown in Table 1.
Point availability of essential medicines for maternal health in public health facilities.
Period availability
The overall median period availability of essential medicines for maternal health in the past 3 months was 83.3% (IQR, 69.4%–86.1%). Of the medicines that are used only for maternal health conditions, Magnesium sulphate (86.1%) and Oxytocin (83.3%) had the highest overall availability in the past 3 months. The period availabilities were fairly high for Gentamicin (69.4%), Methyldopa (72.2%), and Misoprostol (50.0%). Calcium Gluconate (2.8%) had the lowest overall availability in the past 3 months, as shown in Figure 1.

Period availability of essential medicines for maternal health in public health facilities.
Stock-outs of essential medicines for maternal health
The overall public health facility median number of stock-outs was 3, and the median number of stock-out days was 94 days in the past 3 months. Thus, the percentage of days out of stock was 104% (94/90 × 100). The most stocked-out medicines during the period (the recent 3 months) were Calcium Gluconate (93.4%), Misoprostol (32.9%), Gentamicin (16.1%) and Methyldopa (10.6%). Generally, medicines used only for the management of maternal health conditions were most stocked out compared to medicines with broader indications, 33.3% and 13.9%, respectively, as shown in Table 2.
Stock-outs of essential medicines for maternal health in public health facilities.

Discussion
The overall availability of essential medicines for maternal health in this study was high (94.4%) compared to other previous studies conducted in other Tanzanian regions and several African countries. A study conducted at the Kongwa district of Dodoma region reported shortages of essential medicines for maternal health in most of the facilities on the day of the survey. Oxytocin was found in very few facilities (28.0%). 19 Intravenous (IV) infusions (Ringer Lactate and Normal Saline) were present in only half of the facilities (50.0%). 19 Another study conducted in six districts of Lindi and Mtwara regions reported poor availability of essential medicines for maternal health in the health facilities. These were Oxytocin (18.0%), Misoprostol (7.0%), Normal saline (36.0%), Ringer lactate (39.0%), Hydralazine (3.0%) and Magnesium sulphate (52.0%). 20 One more study conducted in Zanzibar showed that medicines such as Methyldopa, Hydralazine, Labetalol and Gentamicin were available in only 27.5%, 21.6%, 2.0% and 29.4% of the facilities visited, respectively. 21 The difference between our study and previous studies suggests that the government’s efforts to reduce maternal mortality may be having a positive impact on the supply chains and distribution of these medicines. The government’s strategy to improve access to essential medicines for maternal health is a promising development, as the reliable availability of these medicines is crucial for delivering quality maternal healthcare, especially in resource-limited settings. 22
Calcium gluconate, used as an antidote for the management of toxicity of Magnesium sulphate and treatment of hypocalcemia, was available in only 2.8% of public health facilities. The availability of Calcium gluconate in this study was very low compared to previous studies conducted in Addis Ababa (33.0%), 10 Dessie (40.0%) 12 and North Karnataka (86.4%). 23 This gap in the provision of Calcium Gluconate points to the need for the government to prioritize improving the procurement, distribution and stock management of Calcium Gluconate. Strengthening the supply chain for this essential medicine should be a focal point in efforts to enhance maternal healthcare service delivery and ensure that all pregnant women have access to the life-saving treatments they require. 24
The median number of stock-outs was reported as 3 per public health facility over the past 3 months, indicating that essential medicines for maternal health were unavailable on multiple occasions during this period. The median number of stock-out days was 94 days in the past 3 months, meaning that medicines were unavailable for approximately 31 days per month, on average. The stock-out reported in this study was lower than a study that was conducted in Dessie, northeast Ethiopia, where the mean number of stock-outs was 3.9, and the mean number of stock-out days was 128.9 days. 12 A study conducted in Uganda revealed that the stock-out days for maternal health commodities ranged from 7 to 20 days in the public sector. 25 This extended duration without essential medicines can severely hinder the provision of adequate maternal care, increasing the risk of complications during pregnancy, childbirth, and the postpartum period. The medicines with the highest stock-out rates during the recent 3 months were Calcium Gluconate (92.4%), Misoprostol (32.9%), Gentamicin (16.1%) and Methyldopa (10.6%). Misoprostol is used to prevent and treat postpartum haemorrhage (PPH). PPH is a leading cause of maternal mortality, and the lack of misoprostol could have severe consequences for maternal health outcomes. 26 Gentamicin, an antibiotic crucial for treating infections, including sepsis during pregnancy, 27 compromises infection control when it is out of stock. Methyldopa, a key antihypertensive drug for managing high blood pressure during pregnancy, 28 being out of stock could contribute to poor management of pre-eclampsia and other hypertensive disorders of pregnancy. The stock-out rate was higher for medicines used solely for maternal health (33.3%) compared to those with broader indications (13.9%). This is in agreement with the findings from a previous study, medicines used only for the management of maternal health conditions had the lowest availability compared to those with broader indications, 47% and 65%, respectively. 10 This indicates that medicines specifically used for maternal health conditions were more likely to be unavailable, while medicines with multiple uses across different health conditions were better stocked. The lack of these critical medications could result in poor maternal health outcomes, increased morbidity and even maternal deaths.26–28 Strengthening the supply chain, improving procurement practices and prioritizing maternal health-specific medications are key steps to reducing stock-outs and enhancing the quality of obstetric care in the region. The cornerstone of inventory management practices for the availability of essential medicines for maternal health in Africa is maintaining up-to-date logistics management tools. The study done in Rwandan public hospitals revealed that hospitals with up-to-date logistics tools for their pharmaceutical management were 33.25 times more likely to have their emergency obstetric drugs in stock at all times compared to those that do not regularly update their logistics tools. 29
Limitations
The main limitation of this study was the potential inaccuracies or errors that were present in some store records. Some store records were subjected to human error, data entry mistakes or incomplete information, which could introduce biases or inaccuracies in the data collected. This study was conducted in urban districts that reflect relatively better-resourced settings, and the findings may not be generalizable to rural or remote districts, where supply chain challenges are often more marked.
Conclusion
This study revealed a concerning gap in the availability of essential medicines for maternal health in public health facilities in Mwanza, Tanzania. Despite a relatively high overall median point availability, frequent stock-outs were observed, particularly for critical medicines like Calcium gluconate, Misoprostol, Gentamicin and Methyldopa. Medicines used solely for maternal health were more likely to experience stock-outs compared to those with broader indications. These findings highlight the need for improved supply chain management and prioritization of maternal health-specific medicines to enhance the quality of maternal healthcare services in the region. Future studies are recommended to include rural districts for comparison.
Footnotes
Acknowledgements
The authors are grateful to the staff of public health facilities in Nyamagana and Ilemela districts for their cooperation and support in providing the necessary data.
Ethical considerations
This study was approved by the Joint Bugando Medical Centre (BMC)/Catholic University of Health and Allied Sciences (CUHAS) ethics & review committee and was granted an ethical clearance certificate number 2830-2024. Permission to conduct the study in Ilemela and Nyamagana districts was given by the Mwanza Regional Administrative Secretary (RAS). Written consent to visit public health facilities was obtained from the District Medical Officer’s (DMO) office.
Consent for publication
Not applicable.
Author contributions
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Availability of data and materials
The dataset generated and/or analysed during the current study is available from the corresponding author upon reasonable request.
