Abstract
Background:
Sphenopalatine ganglion (SPG) blocks may be effective and affordable treatments for acute migraine but are rarely used in the primary care setting, making them inaccessible treatment for those suffering from migraine.
Objective:
To outline evidence for the use of SPG block in the treatment of acute migraine. To determine the feasibility and effectiveness of primary care in-office and at-home SPG block administration.
Methods:
A retrospective chart review of patients receiving in-office or at-home SPG block to treat acute migraine was completed. Pre- and post-treatment pain measurements were the primary outcome, along with patient-reported adverse effects.
Results:
Results from this study revealed that 87% of SPG block administrations (via catheter and atomizer) resulted in pain relief with minimal adverse effects. Methods and materials for catheter and atomizer administration of SPG blocks are outlined, in addition to tips for integrating this service into primary care practices.
Conclusions:
This study showed that SPG blocks may be administered effectively in primary care settings or at home with proper patient education and are an effective treatment modality to decrease pain associated with acute migraine.
Introduction
Transnasal sphenopalatine ganglion (SPG) blocks, while predominantly used as preventive migraine treatment in specialty care settings such as neurology, may also be a successful but infrequently used acute treatment for migraine and can be utilized in primary care or emergency settings.1 –4 While the mechanism of SPG blocks remains debated, the SPG may be a key actor in the trigeminovascular etiology of migraines, making SPG blocks an important therapeutic procedure to consider.5 –7 Most widely used acute migraine treatments target calcitonin gene–related peptide (CGRP) and other neuropeptides after release from trigeminal neurons associated with meningeal vasculature.8,9 Some scientists hypothesize that SPG blocks may limit vasodilation and minimize trigeminal activation, therefore potentially preventing the release of neuropeptides such as CGRP. 10 Blocking activation of the trigeminovascular system before triggering the neuropeptide cascade makes SPG blocks a particularly useful, if underutilized, acute migraine treatment.
The use of injected anesthetic blocks at the branches of the trigeminal nerve (V1/V2/V3) or occipital nerve for migraine and other headache treatment has been described in the literature.11 –13 SPG blocks target the same trigeminal fibers but may be applied topically rather than using an injection due to the proximity of the SPG to the upper posterior nasal mucosa. Additional benefits of SPG blocks include limited adverse effects due to the lack of systemic absorption of anesthetic and readministration potential without increased symptom sensitivity.14 –18
While there is some evidence showing the success of SPG blocks as acute migraine treatment, they are predominantly used as preventive treatment due to the logistical difficulty that comes with specialty clinic administration. However, SPG blocks can be performed in primary care settings, where the majority of patients with acute migraine are encountered. 16 In addition, it has been suggested that patients can be educated to perform SPG blocks at home and may be supplied the tools they need to successfully use SPG blocks as acute treatment. 19 Although there are limited studies on the safety of patient-administered SPG blocks, there is ample evidence for the safety of nasal lidocaine, including one double-blind trial comparing patient-administered nasal lidocaine to placebo saline.16,17,20
Objectives
Due to the limited data on primary care and at-home use of SPG blocks for acute migraine treatment, 21 a retrospective review of clinical records was conducted to assess overall treatment efficacy and patient-reported adverse effects from a cohort of patients who received in-clinic or at-home SPG block treatment for acute migraine symptoms from a primary care clinic in the southeastern US. This article will describe the results of this review and provide information on methods, materials, and billing of in-clinic and at-home SPG blocks for acute migraine treatment in the primary care setting.
Methodology
Methods and materials of SPG block in primary care settings
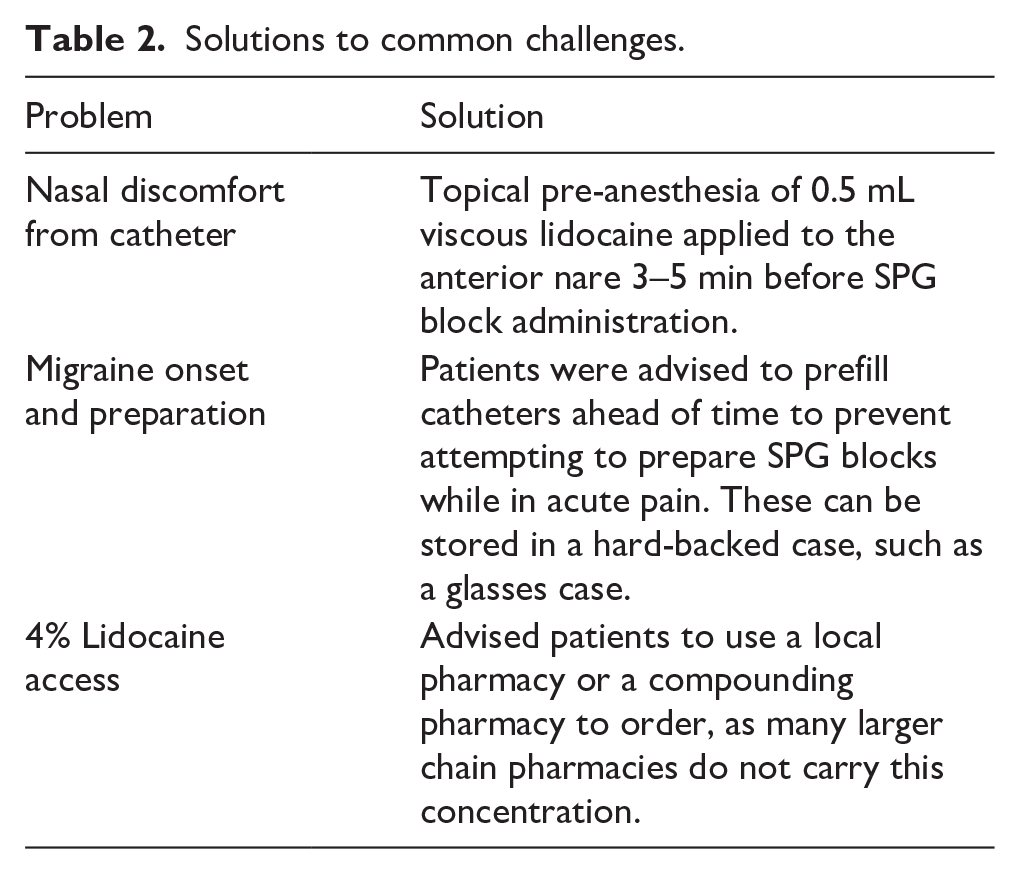
This study collected and analyzed three different sets of retrospective data using chart review for patients receiving SPG blocks from 2017 to 2020 at a primary care clinic in the southeastern United States. This included pre-post pain and symptom data on patients who received in-office catheter SPG blocks for acute migraine treatment, as well as patients who had self-administered catheter SPG blocks and in-office atomizer SPG blocks. Clinical methods for these three different modalities are outlined below. Materials required for the three modalities studied are listed in Table 1. Solutions to common challenges are also presented in Table 2. Institutional Review Board (IRB) approval (IRB# FHC-20211206-001) for retrospective chart review was obtained prior to data abstraction and analysis.
Catheter and atomizer SPG block materials.

Solutions to common challenges.
In-office catheter SPG blocks
Patients presenting to the primary care clinic with acute migraine were offered SPG block as a treatment option. A syringe with 2 mL of 2% viscous lidocaine was attached to a 2-inch, 14-gauge Angiocath. The patient lay supine on the exam table with the neck extended. The Angiocath was inserted into the nare at an angle to allow it to pass between the middle and inferior turbinates. The catheter was fully inserted to the hub or otherwise until slight resistance was felt, and 1 mL of lidocaine was deposited into the posterior nasal cavity in the region of the SPG. The patient remained supine for 15 min.
Self-administered catheter SPG blocks
Patients who responded to in-office SPG block were offered self-administration teaching for use at home as a primary abortive option for acute migraine. Self-administration materials were the same as in-office materials, except a shorter 1.25-inch, 16-gauge Angiocath was used. Patients were instructed in syringe preparation and dosing, and insertion technique was demonstrated in front of a mirror. They were sent home with instructions to administer the block for an acute migraine and were given a log to record their responses.
In-office atomizer SPG blocks
After some experience with self-administered catheter SPG blocks, we repeatedly received feedback that some patients were limited by the necessity of lying supine for 15 minutes and were often unable to use the treatment at the onset of pain due to being out in public or at work. For these patients, we used the Mucosal Atomizer Device (MAD) attached to a 3cc syringe to administer an SPG block while remaining upright. We used 4% oral/topical lidocaine solution which was the most concentrated solution that was readily available without compounding. Further patients presenting to the clinic with acute migraine who requested SPG block were given the option of the atomizer or catheter procedure. The atomizer tip syringe was inserted into the nare with the patient seated upright, and 0.3 mL of 4% lidocaine was sprayed in a short burst while the patient gently inhaled nasally.
Study design
Administration and outcome data were obtained through chart review and were deidentified for aggregate analysis. All patients were treated by family medicine physicians at a primary care clinic in the southeastern US and received in-clinic SPG blocks using a catheter or atomizer or at-home SPG blocks using a catheter. Patients included in the study were those that had a CPT code for SPG block in their medical record. Patients with other headache diagnoses who did not meet the ICHD-3 (International Classification of Headache Disorders, 3rd Edition) criteria for migraine diagnosis were excluded. Patients under the age of 18 years were also excluded. A total of 218 patients were initially enrolled in the study, and 21 were excluded due to age (n = 4) or non-migraine headache diagnosis (n = 17).
Data extraction and analysis
After eligible patient charts were identified, data extraction included reviewing encounter notes for the day of the procedure and follow-up appointments. Each individual treatment, defined as one use of the SPG block, was assessed as an independent data point, with the primary outcome defined as patient-reported pain relief. Pain relief was defined as improving pain from patient-reported baseline. No change in pain and increased pain were also assessed. For the at-home procedure only, pain relief and pain freedom were assessed. Pain freedom was defined as the absence of pain or total improvement in pain from patient-reported baseline. Following the at-home procedures, a brief qualitative survey was administered at a follow-up appointment to determine administration preferences and patient-reported adverse effects. Patient comfort with the procedure was assessed with a yes/no question. Outcome measurements were collected at 20 min post-procedure in-office and 20 min, 1 h, and 4 h for at-home procedures. Other data collected included patient-reported adverse effects, migraine type (acute or chronic), co-medications, administration method, as well as demographic information (age and gender).
Result data were coded based on response (decrease pain = 1, no change = 0, worsening pain = −1) and descriptive statics were run using SPSS for all sets of data.
Results
The accumulated results from all three application methods are reported in Table 3.
Pain relief outcomes of in-office, at-home, and atomizer SPG blocks.

In-office SPG blocks
A total of 161 patients ages 20–81 (mean = 52, STD = 14, 87% female, 13% male) who were treated with in-office SPG block for migraine between 2017 and 2020, for a total of 378 individual treatments. The average number of procedures per participant was 2.3. The average response was 0.8 (STD = 0.36). The most common patient-reported adverse effects included unpleasant taste, nasal discomfort, and temporary throat numbness from swallowed lidocaine. These were classified as mild effects and did not prevent patients from wanting to repeat the procedure.
Self-administered catheter SPG blocks
A total of 16 patients aged 32–68 (mean 49, STD = 11, 93% female, 7% male) performed SPG block procedures at home between 2017 and 2018. Among these 16 patients, a total number of 116 instances of migraine pain were reported and treated with a self-administered SPG block. The average number of migraines and subsequent treatments per participant was 7.2. The average response after 20 min was 0.82 (STD = 0.34). Pain relief and pain freedom over time is shown in Figure 1. Reported adverse effects were similar to those reported with in-office SPG blocks and included unpleasant taste, nasal stinging, and facial tingling. All patients reported being comfortable with self-administration. Most patients reported preferring at-home administration to in-clinic administration (76%), while 3 (18%) patients were neutral, and 1 (6%) preferred in-clinic administration. The most commonly reported reason for preferring self-administration was lifestyle convenience and ability to administer at the start of symptoms.

Time to Pain Relief and Freedom for At-Home SPG Blocks.
In-office atomizer SPG blocks
A total of 20 patients aging from 24 to 82 (mean = 49, STD = 16.8, 90% female, 10% male) who presented in-office with acute migraine had an SPG block administered with an atomizer between August 2021 and March 2022. Among these 20 patients, a total of 26 procedures with the atomizer were completed. The average response was 0.81 (STD = 0.48). Reported adverse effects were the same as reported in the in-office catheter and at-home catheter data sets.
Discussion
This review outlined evidence for the effectiveness and practical application of transnasal SPG blocks for acute migraine treatment in primary care settings and at home. Ample data have shown that the SPG may play a critical role in migraine pathogenesis; however, SPG blocks are not commonly used in the primary care setting. In our retrospective study, 87% of administrations resulted in pain relief with minimal adverse effects, and administration via the catheter and atomizer methods were both successful at treating acute migraine. While the consistent pain relief experienced across all administration types may be counterintuitive due to the assumed limited precision of lidocaine delivery with patient-delivered and atomizer SPG block methods, this review may provide evidence of a broader mechanism of action for SPG blocks than previously understood.
Although SPG block’s proposed method of action is anesthetizing the sphenopalatine ganglion via precise delivery of lidocaine to the posterior nasal cavity, the success of patient-delivered and atomizer methods may suggest that there are nerve endings more easily accessible in the nasal cavity that may be contributing to the analgesic effect of SPG blocks. We hypothesize that these blocks may actually be anesthetizing two nerve branches from the SPG that terminate at the sphenopalatine foramen, rather than the SPG itself. These nerve branches are likely the posterior superior lateral nasal nerve which lies on the middle and superior nasal turbinates and the nasopalatine nerve which lies on the posterior aspect of the nasal septum. Therefore, SPG blocks may be effective even if delivered more anteriorly in the nasal cavity. While our data affirm that this may be the case, further research should continue to evaluate the specific method of action for this important pain-relieving procedure.
This study has several limitations. As a retrospective cohort study at a single site, it may have limited generalizability. Statistical power was not taken into consideration when completing the chart review, and so it was not calculated prior to data extraction. Furthermore, no control group was utilized; however, our results show rates of response well above traditional placebo effects for migraine. Future research could consider comparison to placebo for both in-office and at-home SPG block procedures. Due to the limitations of retrospective chart review, this study did not analyze information on the onset of migraine pain, the presence of aura, delayed symptomatology often associated with migraine treatment including rebound migraine, nor the temporality of the few adverse effects experienced by some participants. Pain freedom was only analyzed for the at-home data but would be an important measurement for future studies to compare in-office to at-home procedure effectiveness. The study also did not analyze 2-h post-procedure response. VAS, which is commonly used in prospective and controlled trials on migraine, was not used, but future studies should consider replicating our study using this tool. Many of these limitations are due to the nature of our study, which was conceived only after all procedures were complete and only uses the limited information available to us in the chart.
Putting into practice
Despite this study’s positive results, primary care providers may still face challenges in providing SPG blocks to their patients. Many physicians have reported the high cost of dedicated SPG catheters such as Sphenocath and Tx360 as the main reason they do not use SPG blocks in their practice. These catheters have a retail cost of $60-$90 per one-time-use device, whereas the materials for our methods typically cost less than $3 for the catheter method and less than $10 for the atomizer method. For home use, both the catheter and atomizer may be used multiple times with proper sanitation between uses.
Reimbursement is another reason primary care physicians may not commonly provide SPG blocks. The CPT code most commonly used for transnasal SPG block is 64505. There is some argument about whether this is an appropriate code for this procedure since it describes “injection” of the sphenopalatine ganglion. In our experience, Medicare plans generally reimburse 64505, limited to three instances every 6 months. We advise providers to consider obtaining preauthorization for commercially insured patients.
Conclusion
In summary, this review shows that SPG blocks are an effective, affordable, and practical treatment for acute migraine pain and should be considered for use in primary care offices and at-home following simple patient education. Methods for three different administration techniques are outlined and practical tips for their implementation in primary care practices are included. In our clinical practice, we have found that patients greatly benefit from this procedure’s convenience, effectiveness, and minimal adverse effects. Therefore, SPG blocks should be considered for primary care providers interested in alternative therapies for their patients with migraine.
Footnotes
Acknowledgements
The authors would like to thank Jonathan Beckwith for his leadership in this project and assistance with manuscript editing.
Ethical considerations
IRB approval was granted by BRANY IRB on 12/06/2021 (IRB# FHC-20211206-001).
Consent to participate
Not applicable since the informed consent portion of the declarations section is not applicable due to the retrospective nature of this study. All data were extracted via chart review.
Consent for publication
Not applicable.
Author contributions
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported, in part, by the National Institute of Neurological Disorders and Stroke of the National Institutes of Health under Award Number R43NS125643. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.C. is a partner and currently practices at The Family Health Centers, where data reported in this study were collected. J.C. is also the co-founder and Chief Medical Officer of Olfax Medical and receives financial compensation as a speaker and consultant for AbbVie Inc.
Data availability statement
Data may be made available on a case-by-case basis. Please contact the corresponding authors.
