Abstract
Insomnia, including insomnia disorder, is a common but often overlooked complaint in primary care settings. It is a risk factor for various medical and psychiatric diagnoses and is associated with substantial health care costs. While cognitive behavioral therapy for insomnia (CBT-I) is the first-line treatment for insomnia disorder, access to CBT-I is limited. This article provides a pragmatic approach to screening, assessment, and treatment of insomnia in the primary care setting, promoting a population health approach. The authors review the role of CBT-I, treatment of comorbid conditions, and pharmacologic recommendations in working with primary care patients with insomnia. In addition, the authors highlight the potential utility of technology in improving access to insomnia care.
Keywords
Introduction
Sleep complaints are highly and increasingly prevalent in the primary care setting.1–3 Despite this, insomnia and insomnia symptoms are often not addressed in primary care, in part due to inconsistent approaches to screening and treatment.4,5 Primary care providers may feel that they do not have sufficient training in the management of insomnia, that treatment options for insomnia are unsatisfactory, or that their patients have limited access to the full range of treatments for insomnia.5,6
The goal of this article is to provide pragmatic guidance for primary care and mental health care providers in addressing insomnia disorder and insomnia symptoms in the primary care setting. This review highlights tools for rapid assessment, key questions for clinical interview, and recommendations for pharmacotherapy and behavioral treatment, including therapist-delivered and technology-based options for cognitive behavioral therapy for insomnia (CBT-I).
Background
Insomnia disorder is defined by the Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5) as difficulty initiating sleep, difficulty maintaining sleep, or experiencing early morning awakenings. Symptoms must be present for at least 3 months, occur at least three nights per week, and cause clinically significant distress or impairment. 7 In addition, it cannot be adequately explained by coexisting medical or psychiatric conditions, by the physiologic effects of a drug of abuse or medication, or by a current sleep-wake disorder (such as narcolepsy or parasomnia). In this article, we use insomnia disorder interchangeably with the term “chronic insomnia.” We also refer to insomnia-related sleep concerns as “insomnia symptoms.”
Many individuals may experience insomnia symptoms without meeting criteria for insomnia disorder. The prevalence of insomnia symptoms is as high as 50% in the general population, while prevalence estimates of insomnia disorder range from 5% to 20%.8–13 Women have been noted to experience insomnia at higher rates than men. 14 Compared to younger adults, older adults report more sleep difficulty and more frequent nocturnal awakenings.14–17 Rates of insomnia in patients with dementia have been reported between 25% and 35%.18,19
Insomnia symptoms and insomnia disorder are associated with medical conditions (including hypertension, hyperlipidemia, migraine and tension headaches, cognitive impairment, persistent pain, and obesity), adverse events (including mortality, myocardial infarction, and other cardiac events), and workplace and non-workplace injuries.8,20–22 Insomnia also has substantial economic impact. A study of US Medicare claims data 23 found that patients with insomnia disorder had higher health care costs than those without insomnia, regardless of whether the insomnia was treated. Furthermore, greater health care utilization and costs were noted in the 12 months prior to insomnia diagnosis, with the majority of costs being attributed to inpatient care. Estimated direct and indirect costs for insomnia disorder have been calculated to exceed US$100 billion annually.24,25
Sleep disruption is a common criterion of psychiatric disorders and has a suggested bidirectional relationship with depression, anxiety, and bipolar disorder.26,27 Sleep disruption is also a risk factor for the development of psychiatric conditions including depression and mania28–31 and is associated with worse psychiatric outcomes.32–34 Insomnia has been associated with higher risk for suicidal behaviors among psychiatric populations 35 and is an independent risk factor for suicide attempts. 36
Screening, evaluation, and diagnosis
Despite the prevalence of insomnia symptoms in the general population, primary care providers do not routinely screen for sleep complaints.4,5 Brief screening questions feasible in the primary care setting may include “Are you having any problems with sleep?” Item 3 on the Patient Health Questionnaire-9 (PHQ-9), 37 which is routinely used for depression screening, asks if the individual has had “trouble falling or staying asleep, or sleeping too much” and could be an efficient way to screen for sleep concerns in addition to depressive symptoms. While there are no widely accepted screening tools for the assessment of insomnia, we recommend use of the Insomnia Severity Index (ISI), which is described in more detail below.38–40 It is important to note that while a positive screen may identify patients with potential insomnia disorder, it is not sufficient for an insomnia diagnosis.
Insomnia disorder is a clinical diagnosis that is based on patient report and history. The purpose of the clinical evaluation is to characterize the sleep problem, identify factors that impact sleep, and assess level of impairment and distress.
When characterizing the sleep problem, the evaluation should focus on several elements: (1) nature of the sleep problem, (2) sleep schedule, (3) sleep hygiene, and (4) chronicity of the sleep problem. Patients typically report difficulty with falling asleep, staying asleep, or both. To make a diagnosis of insomnia disorder these symptoms must be present for at least 3 months and occur at least three nights per week. Inquiring about the patient’s sleep schedule over the prior 2 weeks can often provide illuminating information. This includes inquiries into the hour which the patient goes to bed, length of time it takes to fall asleep, number of awakenings and length of time it takes to fall back asleep, hour at which patient gets out of bed to start the day, and timing and duration of naps. When assessing sleep hygiene, it is important to gather information about pre-bedtime activities (e.g. screentime, exercise, or heavy meals), environmental qualities of the bedroom, and activities the bed is used for (e.g. sleep, reading, and screentime). It can also be helpful to inquire about the estimated total sleep time versus expected total sleep time, sleep aids or substances before bed, and feelings about sleep (e.g. how anxious the patient is about sleep difficulty).41,42
In the clinical evaluation of insomnia disorder, it is important to identify factors that impact sleep. This can be accomplished by exploring sleep hygiene, as described above, as well as evaluating medical and psychiatric comorbidities, exploring the impact psychosocial factors have on symptoms, and including a review of prescription medications and quantity/timing of substance use (including caffeine). When evaluating level of impairment caused by the insomnia symptoms, the clinician should inquire about how poor sleep has impacted the patient’s well-being and ability to function. This can be compared to the patient’s premorbid functional state. The degree of impairment is integral to making the diagnosis of insomnia disorder.
Differential diagnoses
Mental health and substance use disorders
For a diagnosis of insomnia disorder, the patient’s sleep problems cannot be completely explained by a psychiatric condition. This can be challenging to determine as sleep disruption is a common symptom of several psychiatric diagnoses. The DSM-5 includes sleep disruption as part of the diagnostic criteria for depressive disorders, bipolar spectrum disorders, anxiety disorders, post-traumatic stress disorder (PTSD), adjustment disorder, neurocognitive disorders, and acute stress disorder. 7 Sleep disruption is also common in autism spectrum disorder, psychotic disorders, and in situations of abuse, neglect, or other social distress (e.g. housing instability, economic instability, food instability, and legal problems).
Thus, diagnostic assessment to differentiate insomnia from underlying mental health or substance use disorders is warranted. The PHQ-9 37 and Generalized Anxiety Disorder 7-Item Scale (GAD-7)43,44 are helpful tools in screening for depressive and anxiety disorders in the primary care setting. The Alcohol Use Disorders Identification Test–Consumption (AUDIT-C) can be utilized for rapid screening of alcohol use disorder. Inquiring about caffeine, nicotine, and illicit substance use is also critical; when doing so, it is important to ask quantity and timing.
Medical conditions
Although insomnia has been reported to be 10 times more likely to be a symptom of a mental health disorder than a medical illness, 45 when evaluating differential diagnoses for sleep complaints one must consider medical causes in addition to mental health and substance use disorders.
An exhaustive list of medical conditions potentially comorbid with insomnia is too numerous to list, however some categories to consider include cardiovascular, pulmonary, gastroenterological, genitourinary, endocrine, rheumatologic, reproductive, neurological, infectious, and otolaryngological disorders. 46
When evaluating insomnia symptoms in the patient with known medical conditions, it is important to keep in mind that an insomnia disorder can co-occur with medical conditions,19,47 be exacerbated by medical conditions, 48 and increase risk for medical conditions.49,50 For example, insomnia is reported in 55%–84% of patients with obstructive sleep apnea, 51 23%–50% of patients with chronic obstructive pulmonary disease,52,53 more than 25% of women during perimenopause,54,55 and in more than 50% of patients with chronic pain. 56
Many neurological disorders are associated with insomnia: up to 60% of patients with Parkinson’s disease, 58% of patients with epilepsy, 56% of patients with stroke, 50% of patients with traumatic brain injury, and 37% of patients with multiple sclerosis report sleep disruption. 19 This may be either a direct result of central nervous system dysfunction in pathways that regulate sleep or an indirect result of symptoms due to the neurological disorder (e.g. pain, weakness, immobility, and respirator dysfunction).
Given the prevalence of insomnia symptoms in medical conditions, it is therefore crucial to obtain both a thorough medical history and a medical review of symptoms. We also highlight the need to rule out other major sleep disorders, namely disordered breathing, narcolepsy/hypersomnia, parasomnias, periodic limb movement/restless leg syndrome, bruxism, and circadian rhythm disorders.57,58 Pavlova and Latreille 57 provide a practical review of the main diagnostic features of the major sleep disorders. Providers in primary care may use the below mentioned screening tools to aid in determining need for further assessment by a sleep specialist, namely the ISI for insomnia;40,59 STOP-Bang Questionnaire for obstructive sleep apnea; 60 the Epworth Sleepiness Scale (ESS) for daytime sleepiness common in obstructive sleep apnea, narcolepsy, periodic limb movement disorder, and insomnia;61–63 and the International Restless Leg Syndrome Study Group Rating Scale (IRLS) for restless leg syndrome. 64 In situations in which the sleep-related symptoms represent a sleep disorder that falls outside of the scope of primary care, a referral to a sleep specialist should be made. 40
Screening tools
Screening tools may help identify insomnia disorder and other underlying sleep-related conditions. The following may be useful in the primary care setting.
Sleep diary/log
A sleep log completed by patients can monitor at-home subjective sleep patterns. Sleep logs estimate time in bed, total sleep time, time in bed before first onset of sleep, total time awake in the middle of the night, and time in bed after the morning wakening.8,65 Many sleep diaries are available in the CBT-I literature8,66 and in the public domain. In addition, there are increasing mobile app resources available, which include sleep diary capabilities and are user friendly for patients and clinicians.67,68
Insomnia Severity Index (ISI)
The ISI is a seven-item self-report questionnaire that asks patients to rate quality of sleep disturbance (difficulty falling asleep, staying asleep, and waking up too early), sleep satisfaction, and functional impairment due to sleep concerns over the past 2 weeks.8,40 It is easily administered and scored in the office, and score cut-offs range from no clinically significant insomnia; scores of 0–7, subthreshold insomnia; scores of 8–14, clinical insomnia (moderate severity); scores of 15–21, clinical insomnia (moderate severity); and scores of 22–28 (severe). A cut-off score of 10 has an 86.1% sensitivity and 87.7% specificity for detecting insomnia. 59
Epworth Sleepiness Scale (ESS)
The ESS is an eight-item scale designed to assess general daytime sleepiness, which is often associated with obstructive sleep apnea (OSA), narcolepsy, periodic limb movement disorder, and insomnia disorder.61–63 Patients are asked to rate the likelihood of dozing in different contexts (e.g. sitting and reading, in a car, and while stopped for a few minutes in traffic). The endorsed items are added together for a total score ranging from 0 to 24, with cut-offs ranging from 0–5 (lower normal daytime sleepiness), 6–10 (higher normal daytime sleepiness), 11–12 (mild excessive daytime sleepiness), 13–15 (moderate excessive daytime sleepiness), and 16–24 (excessive daytime sleepiness). 69
STOP-Bang Questionnaire
The STOP-Bang Questionnaire is an eight-item “Yes” or “No” questionnaire that assesses for common symptoms of OSA. 60 “Yes” items are added together for a total score that stratifies patients by risk for OSA. Scores of 0–2 are classified as low risk for OSA, scores of 3–4 require further classification, and scores of 5–8 are classified as high risk for OSA. For a more nuanced discussion of scoring, see Chung et al. 60
International Restless Leg Syndrome Study Group Rating Scale (IRLS)
The IRLS is a 10-item self-report scale to assess the severity of restless leg symptoms. 64 Item scores are added together for a total score and higher scores suggest higher symptom severity. Cut-off ranges are mild (scores 1–10), moderate (scores 11–20), severe (21–30), and very severe (31–40).
Patient Health Questionnaire-9 (PHQ-9)
The PHQ-9 is a nine-item self-report depression screening questionnaire commonly used in primary care settings and is recommended by the U.S. Preventive Services Task Force. 37 Patients rate frequency of common depression symptoms over the past 2 weeks, including the presence of suicidal ideation. Score cut-offs range from minimal depression (scores of 0–4), mild depression (scores 5–9), moderate depression (scores 10–14), moderately severe depression (scores 15–19), and severe depression (scores 20–27).
Generalized Anxiety Disorder Scale 7-item Scale (GAD-7)
The GAD-7 is a seven-item self-report anxiety scale.43,44 Patients rate frequency of common anxiety symptoms over the past 2 weeks. Score cut-offs range from no anxiety (scores 0–4), mild anxiety (scores 5–9), moderate anxiety (scores 10–14), and severe anxiety (scores 15–21).
Alcohol Use Disorders Identification Test-Consumption (AUDIT-C)
The AUDIT-C is a three-item self-report scale, a subset of the 10-item AUDIT, developed by the World Health Organization to screen for alcohol use disorders. 70 Questions assess frequency and amount of alcohol consumption over the past year. Scores range from 0 to 12 with scores of 4 or more or 3 or more suggestive of potentially risky drinking behavior in men and women, respectively.71–73 These scores may be associated with adverse health outcomes.
Treatment and management
Once the factors impacting a patient’s sleep have been identified, treatment of insomnia symptoms should address each of these components in a stepwise manner; in some cases, concurrent treatment may be prudent.
Treatment for medical and psychiatric comorbidities
As noted above, the relationship between insomnia and medical and psychiatric conditions can be complex. Insomnia may be a symptom of, may exacerbate, or may be comorbid with medical or psychiatric conditions. Numerous guidelines and meta-analyses support CBT for insomnia for management of insomnia when comorbid conditions are present.3,4,74–78
Treatment of comorbid medical and psychiatric conditions should follow standard guidelines, while being mindful of whether such treatments have the potential to exacerbate sleep-related symptoms. For example, corticosteroids (systemic or inhaled), decongestants, thyroid hormone supplementation, stimulants, certain antidepressants (e.g. venlafaxine and bupropion), and opioids can disrupt sleep. 79
Cognitive Behavioral Therapy for Insomnia (CBT-I)
CBT-I is well-established as the first-line treatment for insomnia disorder80–83 and is recommended by the American Academy of Sleep Medicine,80,82 the American College of Physicians, 83 and the European Sleep Research Society. 84 CBT-I has been shown to improve remission rates, onset to sleep latency, sleep efficiency, wake after sleep onset, and sleep quality. It is efficacious in individual, group, telephonic, web-based, and self-help bibliotherapy contexts.85,86 There is some evidence that stimulus control therapies alone improve sleep; however, evidence is limited.82,83,87
CBT-I consists of three main components: (1) stimulus control, 87 (2) sleep restriction to promote sleep consolidation, 88 and (3) cognitive therapy that targets thoughts and worries interfering with sleep. CBT-I can be tailored to the individual; any or all the three CBT-I components can be utilized in a 6-week or shorter treatment. 89
Stimulus control and sleep restriction both work to optimize the sleep drive and circadian rhythm. Stimulus control is a set of instructions that address conditioned arousal, where the aim is to strengthen the bed and bedroom’s association with sleep. Through classical conditioning, patients with insomnia may have come to associate the bed and bedroom with wakefulness. This can occur through several mechanisms: (1) they may have engaged in non-sleeping behaviors in the bed and bedroom (e.g. talking on the phone, eating, reading, watching television, or listening to music); (2) if bedtime has been the first quiet moment of the day, they may have found themselves reflecting on the day’s activities or planning for the next day, resulting in worry and a more aroused state;39,88 or (3) they may have experienced frustration and anxiety related to difficulty sleeping. Within a stimulus control framework, patients are coached on reserving the bed and bedroom solely for sleep. They are encouraged to engage in all other activities outside of the bed, with the exception of sexual activity.
Sleep hygiene strategies are often utilized in stimulus control and can reinforce sleep restriction goals. Recommendations include keeping a consistent bedtime (with an emphasis on not going to bed before a set bedtime or only when feeling sleepy) and wake time (reinforcing getting up regardless of how well one slept the night before), getting up when one cannot sleep, using the bed solely for sleeping, avoiding daytime napping, creating a buffer zone before bed, and working to eliminate worry in bed (e.g. getting up if worrying). Furthermore, recommendations include getting sunlight exposure in the eyes (without looking directly at the sun) upon waking, limiting alcohol and nicotine before bed, avoiding caffeine ingestion 10 h before bedtime, avoiding heavy meals or exercise before bed, avoiding bright artificial lights in the evening (or consider use of blue light blocking glasses), and keeping the bedroom environment conducive to sleeping (cool, dark, and quiet). 38 Sleep hygiene further emphasizes not “trying too hard” to sleep, and giving CBT-I time to produce change, acknowledging that most sleep problems develop over time.
Every individual has a unique drive for sleep and wakefulness. Those with a strong sleep drive fall asleep readily, and those with a strong wake drive rise and start their day easily. Sleep restriction seeks to balance those drives in individuals struggling with insomnia by increasing the nocturnal sleep drive. Reducing the excessive time in bed increases the drive to sleep, generally promotes sleep consolidation, and minimizes unwanted wakefulness at the beginning, middle, or end of the night.87,88
In order to get more sleep, patients with insomnia may spend more time in bed by going to bed earlier, staying in bed later in the morning, or taking naps. These behaviors often decrease the sleep drive. For instance, naps disrupt the sleep drive much the same way that snacking decreases the hunger drive. Late afternoon naps, early evening naps, and long naps (30 min or longer) are especially likely to impact a patient’s ability to fall asleep at bedtime. Going to bed early typically results in one of the two situations: (1) if the patient has a strong sleep drive, they may fall asleep quickly, only to experience frequent nocturnal awakenings or early morning awakening; or (2) if the patient does not have a strong sleep drive, they may lie awake tossing and turning.
Sleep restriction consolidates sleep and is based upon the principle that for an individual who ideally sleeps 7–8 h, bedtime should occur 7–8 h prior to the desired wake time. Therefore, in sleep restriction, a sleep schedule is identified for the patient wherein bedtime is 7–8 h prior to the desired wake time or wake time is set 7–8 h after the desired bedtime. In some circumstances, this interval is first restricted to shorter durations as determined by how much time a patient is initially sleeping (not to be less than 5 h) and gradually increased by 15–30 min weekly as sleep improves. 39
As part of sleep restriction, naps are eliminated. If naps are unavoidable, patients are counseled to take naps as early as possible (e.g. before 1 p.m. or 2 p.m.) and to keep them shorter than 30–40 min. Naps later than or longer than these recommendations may delay the natural peak of the body’s sleep drive.
The cognitive therapy component of CBT-I provides a framework to help patients challenge dysfunctional thoughts and beliefs about sleep that may be contributing to sleep anxiety and arousal. The cognitive model of insomnia by Harvey 90 posits that individuals with insomnia spend more thought energy worrying about their ability to sleep and the impact of limited sleep on day-to-day functioning. This worry triggers more “autonomic arousal and emotional distress” which in turn contributes to more selective attention toward sleep-related threats/cues both internally and externally. 90 A systematic review of cognitive treatment components found specific empirical support for paradoxical intention and cognitive therapy to address these concerns. 91 Paradoxical intention hypothesizes that insomnia is in part maintained by an individual’s attempt to control sleep. In paradoxical intention, the individual is told to focus on staying awake. Cognitive therapy focuses on addressing cognitions that perpetuate sleep anxiety and sleep disruption. Specifically, cognitive therapy targets unhelpful cognitions related to beliefs about sleep, worry and rumination about sleep, attentional bias focusing on “sleep-related threats,” 91 inaccurate perceptions of sleep deficits, and addressing safety behaviors that perpetuate unhelpful sleep beliefs. 91 Identifying unhelpful cognitions and behaviors create opportunity for behavioral experiments to test and challenge beliefs and address maladaptive behaviors. CBT-I also includes relaxation skills such as diaphragmatic breathing and mindfulness to promote stress management and reduced sleep-related anxiety and arousal.
Clinical considerations in specific populations
While CBT-I is specifically recommended for insomnia disorder, CBT-I can benefit populations with comorbid conditions that impact sleep, including depression, anxiety, PTSD, substance use disorders, and chronic pain. 92 Here, we highlight a few clinical considerations.
For patients with comorbid depression, it is helpful to think of the interaction of sleep with the depression. Such patients may go to bed early due to anhedonia or to avoid negative thoughts, may wake up earlier than desired, or may spend substantial time in bed (therefore interfering with stimulus control). Patients with depression may also report negative beliefs about sleep, which can be targeted through cognitive therapy. 39
For patients with comorbid anxiety, targeting any cognitive component of anxiety that is contributing to insomnia is important. Engaging in stimulus control and breaking the association of the bed with anxiety may be a focus of the intervention. Finally, skills such as mindfulness may be helpful for calming thoughts and reducing arousal.
PTSD comorbid with insomnia may best be treated by first targeting PTSD symptoms. However, CBT-I may still be beneficial, albeit with several clinical considerations. Sleep may be avoided due to nightmares, hypervigilance, or trauma-related associations. 39 It can be helpful for patients to understand that sleep avoidance may result in more nightmares due to rapid eye movement (REM) rebound; although sleep avoidance initially reduces REM and nightmares, it often results thereafter in greater REM sleep. Highlighting tools to reduce rumination and intrusive memories when trying to fall asleep may also be helpful.
Chronic pain is often associated with sleep disruption. Helping patients understand factors that increase and decrease pain using an intervention like cognitive behavioral therapy for chronic pain may be beneficial. Furthermore, focusing on functional goals related to sleep and pain, as opposed to striving for the elimination of pain or the elimination of sleep disruption, may be helpful. Helping patients better manage their pain will likely aid in improving sleep.
With respect to substance use disorders, many recreational substances disrupt sleep. Substance use is therefore a contraindication to CBT-I. It is recommended that patients with alcohol or other substance misuse first address substance use and then target insomnia. 39
Alternatives to therapist-delivered CBT-I
Although CBT-I is the first-line treatment for insomnia disorder, access is often limited due to practice setting constraints, lack of trained clinicians to administer the intervention, or lack of provider knowledge about the intervention. 5 Primary care settings with integrated behavioral health providers may provide CBT-I. While much of the CBT-I literature has targeted changing provider behavior and increasing access to CBT-I, several studies have assessed training non-behavioral health providers to administer CBT-I93,94 and Internet- and mobile app–based treatments.95–97
To date, online and mobile app–based CBT-I has shown improvement in sleep quality, severity of insomnia, daytime fatigue, functional health and well-being, and sleep-related quality of life.95,96,98,99 Sustained improvement at 1-year follow-up has been demonstrated as well. 97 Most online programs or mobile apps include the core CBT-I components and vary in cost and operating system availability. CBT-I coach, a collaboration between the VA National Center for Post-Traumatic Stress Disorder, Stanford University School of Medicine, and the Department of Defense’s National Center for Telehealth and Technology, is free and has evidence to support use as an adjunctive tool to CBT-I 67 as well as a self-management tool. 68
There is also burgeoning literature on consumer sleep tracking devices, which can capture sleep-related data for patients.100,101 The use of these devices may be considered for tracking the timing and duration of sleep which may be more convenient and accurate than logging such data by hand in a sleep diary. 102 Using wearables for this purpose may also reduce the stress/worry of keeping an accurate sleep diary while trying to get sufficient sleep.
Pharmacologic treatment
While the American Academy of Sleep Medicine, 82 the American College of Physicians, 83 and the European Sleep Research Society 84 recommend CBT-I as the first-line treatment for insomnia disorder, in routine clinical practice sleep medications (including the off-label use of prescription medication such as trazodone, mirtazapine, and hydroxyzine) are often used without an endpoint (sometimes for decades). This is despite the fact that primary care providers often wish to reduce the use of pharmacologic treatments for insomnia. 103 Medication strategies are considered second-line treatment for insomnia disorder, utilized when CBT-I is either unsuccessful or not available. When prescribed, sleep medication should be used on a time-limited basis.83,84
A range of medications have been approved by the U.S. Food & Drug Administration for insomnia. The American College of Physicians, the European Sleep Research Society, and the American Academy of Sleep Medicine each have recommended certain medications for short-term use based on reviews of the existing literature; recommended medication includes benzodiazepines, benzodiazepine receptor agonists (also referred to as “Z drugs”), suvorexant, ramelteon, and certain sedating antidepressants including doxepin.85,104,105 However, many of these medications, including benzodiazepines and benzodiazepine receptor agonists, have substantial adverse effects, including tolerance, dependence, risk of misuse, and adverse cognitive and behavioral effects. 106 In addition, benzodiazepines and Z drugs have been found to be associated with an increased risk for falls and fractures among older adults as well as morbidity/mortality.107–109
Conclusion
Insomnia is well-established as a population health issue associated with various deleterious outcomes. We recommend a stepwise approach within the primary care setting to address insomnia (see Figure 1). First, we recommend general routine screening for sleep complaints. For patients who endorse sleep-related symptoms, further assessment of insomnia may include an assessment for comorbid medical or psychiatric illness and, when indicated, referral to a sleep specialist. We have provided questions for assessment and considerations for differential diagnosis and treatment planning.
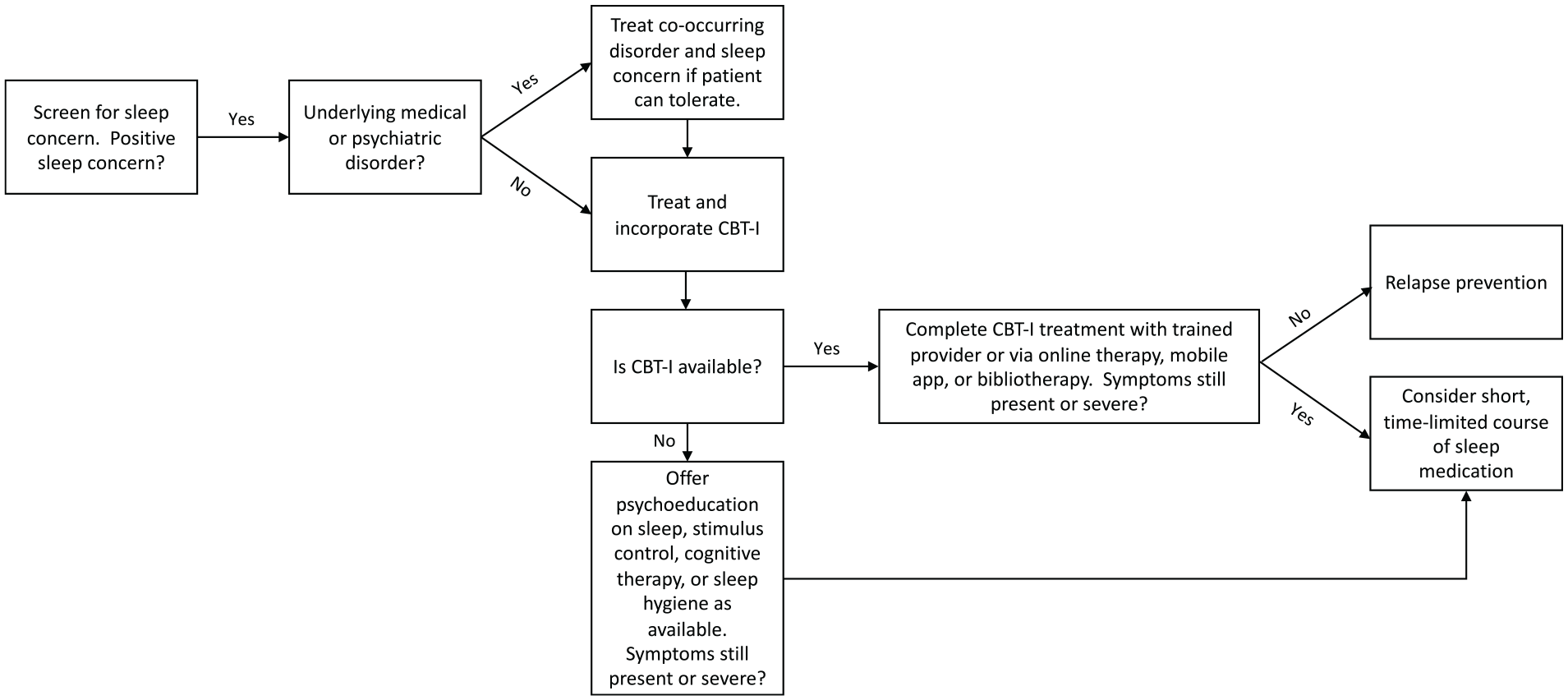
Insomnia treatment algorithm.
CBT-I is the first-line treatment for insomnia disorder, which may be administered in-person or via Internet or mobile app. We recommend applying the tactics and strategies described above to educate patients and promote good sleep. If medications are indicated (namely, sleep disruption is so severe that the patient is unable to productively engage in the intervention), then medications are recommended only in the short term (4–5 weeks).
We recognize that, in a busy primary care visit, addressing sleep may not feel like a priority. However, based on the prevalence, associations with adverse outcomes, social and economic costs, and multitude of treatment options, we support an approach that routinely screens for insomnia and connects patients to evidence-based treatment.
