Abstract
Introduction:
Anticholinergic medications which are commonly prescribed to older adults can produce more pronounced adverse effects compared to the younger population.
Objectives:
To investigate the prevalence of polypharmacy, describe the prescription patterns of anticholinergic medications, determine the prevalence of exposure to high-risk anticholinergic cognitive burden (ACB) medications, and identify the potential predictors of exposure to high-risk ACB medications in older adults in a Nigerian secondary hospital.
Methods:
This cross-sectional study that included older adults prescribed at least one anticholinergic medication was conducted at a secondary care hospital in Nigeria. The study data were initially summarized using descriptive statistics, whereas multivariable logistic regression analysis was used to identify the potential predictors of exposure to high-risk ACB medications. In this study, the cumulative ACB scores were dichotomized into low risk (score 1–2) and high risk (score ⩾ 3). A p value less than 0.05 was considered to be statistically significant.
Results:
A total of 256 older adults were included in this study; 113 (44.1%) had polypharmacy, whereas 26.6% (188/256) were exposed to high-risk ACB medications. Of a total of 391 anticholinergic medications used by the population, furosemide (36.57%) was the most commonly prescribed. The adjusted multivariable logistic regression analysis indicated that patients who had acute diseases were 2.5 times (adjusted odds ratio (AOR) = 2.49, 95% confidence interval (CI): 1.40–4.45) more likely to be exposed to high-risk ACB medications than those with chronic diseases.
Conclusion:
The study demonstrates a high prevalence of polypharmacy and exposure to high-risk ACB medications suggesting the need for pharmaceutical care in this high-risk population. Older adults’ exposure to high-risk ACB medications was significantly associated with prescriptions for acute diseases.
Keywords
Introduction
Available evidence has shown that older adults are more vulnerable to adverse drug reactions events (ADEs) than younger adults due to polypharmacy, and a range of age-related alterations in pharmacokinetics and pharmacodynamics.1,2 Exposure of older adults to some medication classes has been linked to an increased risk of ADEs. 3 Medication classes such as benzodiazepines, selective serotonin reuptake inhibitors, tricyclic antidepressants, and antipsychotics have been implicated as “fall risk drugs” in older adults.4,5 This is due to their more pronounced effects in this population compared with younger populations. These medications are most frequently used to treat urinary incontinence, insomnia, anxiety, allergies, and chronic obstructive pulmonary disease in older population. 6 The adverse effects of anticholinergic medications include dry mouth, constipation, blurred vision, increased heart rate, and confusion. 6 Their pronounced effects in older adults could lead to difficulties in communication, constipation, and falls. 7
Direct positive relationships between increasing anticholinergic medication exposure and poor physical function measures have previously been found in older adults.8,9 Several studies have also found a link between anticholinergic medication exposure and mortality, as well as an increased risk of hospitalization.10–12 The cumulative impact of taking one or more anticholinergic medications is known as the anticholinergic burden. 13
Anticholinergic burden is a tool for assessing the impact of cumulative anticholinergic medication exposure on physical and cognitive function in older adults. Despite the presence of evidence that increased anticholinergic burden is associated with deficits in physical and cognitive function in older adults,14,15 anticholinergic medications for older adults are not routinely evaluated for anticholinergic burden. 16
Previous studies from various countries have shown that older adults were exposed to anticholinergic medications.14,15,17,18 To our knowledge, only one study has investigated Nigerian older adults’ exposure to anticholinergic medications. 19 This research was carried out in southwest Nigeria. Thus, the paucity of data from Africa, especially Nigeria, as well as the gap revealed in the previous Nigerian study 19 underscored the need for the present study. Therefore, the objectives of the study were to investigate the prevalence of polypharmacy, describe the prescription patterns of anticholinergic medications, determine the prevalence of exposure to high-risk anticholinergic cognitive burden (ACB) medications, and identify the potential predictors of exposure to high-risk ACB medications in older adults in a Nigerian secondary hospital.
Method
Study design and setting
This cross-sectional, retrospective study was conducted at a public secondary hospital in Maiduguri, Nigeria. Currently, this hospital is a 460-bed healthcare facility. It serves as a main service and referral hospital for Maiduguri Metropolis and the entire Borno State, northeast Nigeria. The study setting was the General Out-patients’ Department (GOPD) of the hospital. This department serves as the primary care unit of the hospital. In other words, it is the first port of call for general medical care in the study setting. Also, referrals for specialist care are made here when necessary.
Sample size and patients’ selection
All patients aged ⩾65 years that were prescribed at least one anticholinergic medication from 1 January 2019 through 31 December 2019, at the GOPD of the study hospital were identified by the second author and included in the study.
Inclusion criteria
Older adults aged ⩾65 years that visited the GOPD of the study hospital in 2019 and received a prescription of at least one anticholinergic medication were included in the study.
Exclusion criteria
Older adults aged <65 years, those that received care outside the GOPD of the study hospital in 2019, and those that had no anticholinergic medications prescription were excluded from the study.
Ethical considerations
Ethics approval (SSH/GEN/641) for the study was obtained from the Ethics and Research Committee of the study hospital before the commencement of data collection. Due to the retrospective nature of the study, the need for informed consent for inclusion in and/or collection/use of data for the present study was waived by the ethics committee. The confidentiality of patients’ information was maintained throughout the study duration by using anonymous codes.
Data collection and measurements
A master list of medications with potential anticholinergic effects available in Nigeria was developed (see Appendix 1) by reviewing several previously published studies.8,9,20–26 Data were taken at one time-point for each participant. For those who had repeated visits, only data on their last visit were extracted. Data extracted by the second author from the patients’ medical records included age, sex, documented chief complaints, primary diagnosis, comorbidities, prescribed medications, and dosages. Accuracy checks were done on the extracted data by the first author.
Definition of variables and data management processing
In the present study, five or more medications including anticholinergic medications in a prescription were used to define polypharmacy in agreement with previous studies.27–32 The ACB online calculator was used to measure exposure to medications with anticholinergic properties. 33 The ACB calculator ranks medications with anticholinergic potential on a 3-point scale (no risk = 0 score, low risk = 1–2 scores, and high risk = 3 scores). The ACB score for a patient is the sum of points for his or her number of medications. In other words, for each patient, the ACB score for each medication with anticholinergic effects in a prescription was calculated and added together. In this study, the cumulative ACB scores were dichotomized into low risk (score 1–2) and high risk (score ⩾3). Furthermore, for multivariable logistics regression analysis cumulative ACB score of 1–2 (low risk) was scored zero point, whereas a score ⩾3 (high risk) was scored one point.
Statistical analysis
Descriptive statistics for continuous data, such as number of prescribed medications, and ACB scores were reported as mean ± standard deviation (SD). Categorical data were expressed as frequencies and percentages. Pearson chi-square test was used to investigate the association between categorical variables. Multivariable logistic regression analysis was carried out to identify potential independent predictors of exposure to high-risk ACB medications. Statistical analysis was done using IBM SPSS Statistics for Windows, Version 20.0 (Armonk, NY, USA: IBM Corp.). A p value less than 0.05 was considered to be statistically significant.
Results
Background characteristics of the study population
A total of 256 patients aged ⩾65 years and prescribed at least one anticholinergic medication in 2019 were included in the study. Ninety-three (36.3%) patients were between 65 and 69 years old. The majority of patients are males (52.7%, n = 135/256), while 52.0% (133/256) were married. The most predominant chronic disease in the study population was hypertension (37.5%, n = 108/288) followed by arthritis (6.9%, n = 20/288), as shown in Table 1.
Background characteristics of the study population.

Some patients had multiple chronic diseases.
Others = osteoporosis, chronic liver disease, angina, stroke, pyelonephritis, and cancer.
Prevalence of polypharmacy, exposure to high ACB score medications, and prescribed anticholinergic medication class
The prevalence of polypharmacy was 44.1% (113/256), while the mean ACB score over the study period was 1.72 ± 0.98. A considerable proportion (26.6%, n = 188/256) of older adults were exposed to high-risk ACB medications during the study period. Of the 1396 medications prescribed during the study period, 391 were anticholinergic medications. Figure 1 shows the frequency of anticholinergic medication classes prescribed to the patients in the study setting during the year under review. Cardiovascular drugs (56.8%, n = 222/391) and antihistamines (20.2%, n = 79/391) were the most frequently prescribed.
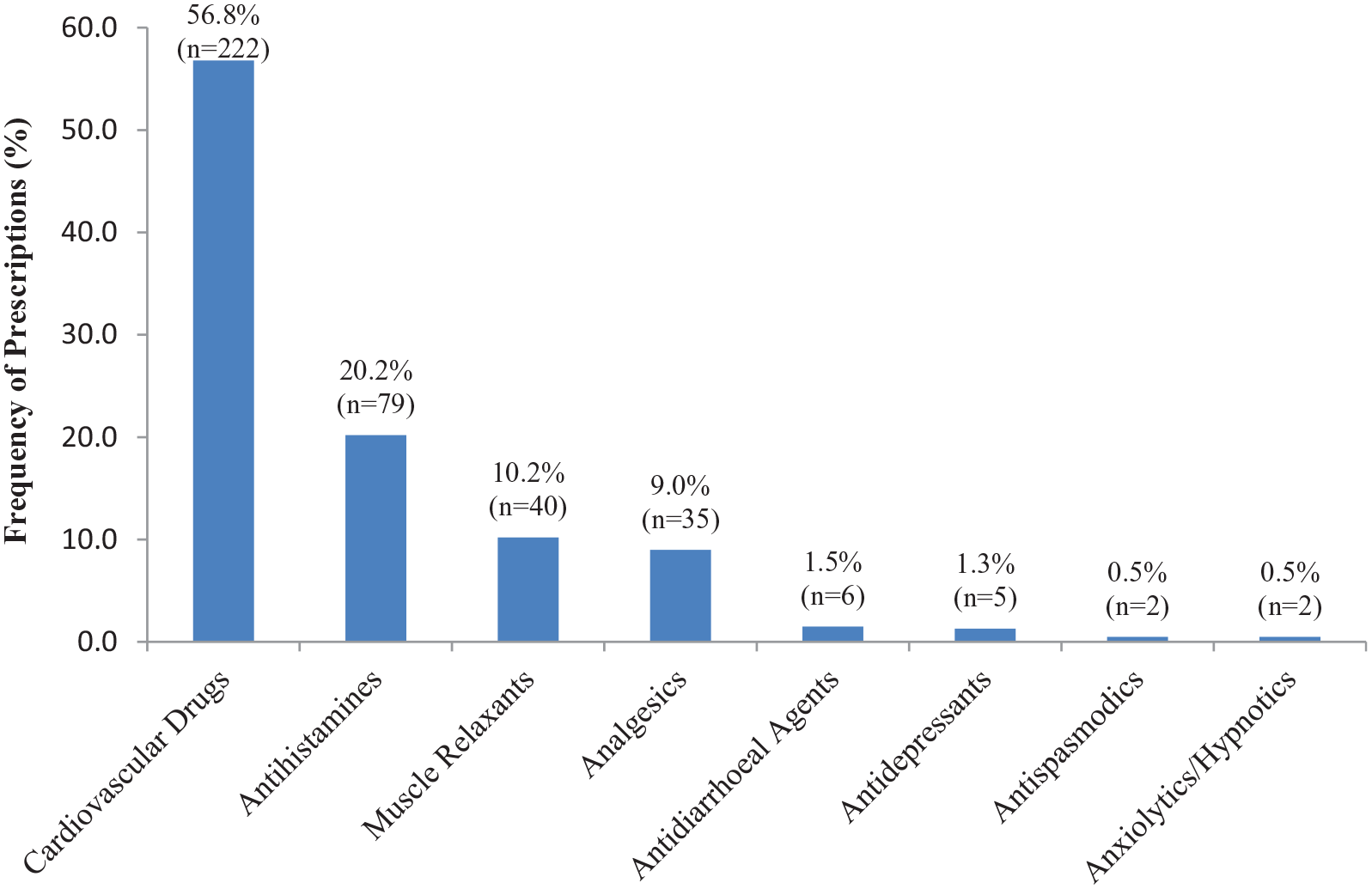
The Proportion of anticholinergic medication prescribed by drug class (N = 391).
Prescribed individual anticholinergic medications
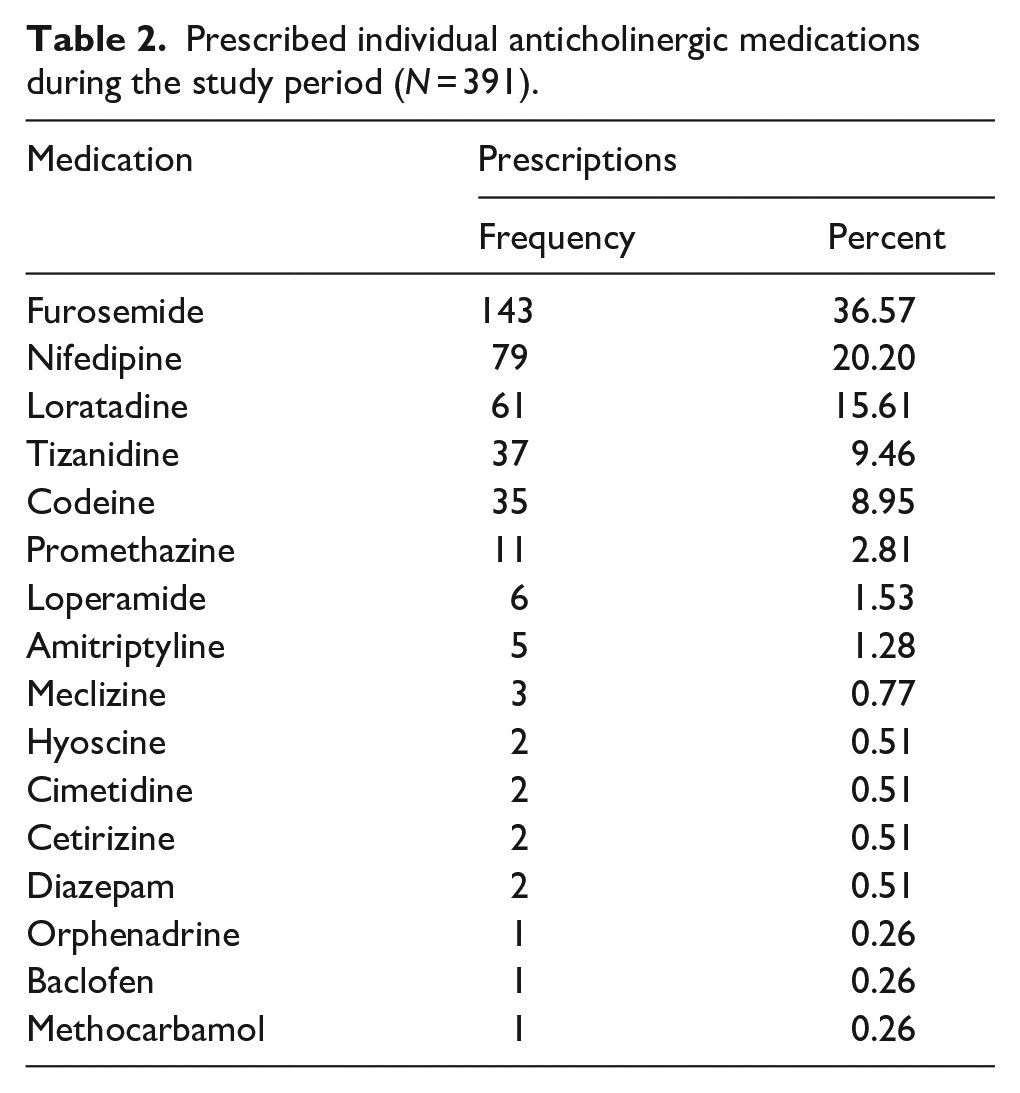
Table 2 shows the pattern of individual anticholinergic medication prescribed during the study period. Furosemide (36.57%, n = 143/391), nifedipine (20.20%, n = 79/391), and loratadine (15.61%, n = 61/256) were the most commonly prescribed medications with anticholinergic effects (Table 2).
Prescribed individual anticholinergic medications during the study period (N = 391).
The analysis of the predictors of exposure to high-risk ACB medications
The univariable analysis indicates that disease state was significantly associated with high-risk ACB medication prescription. A significantly higher proportion (57.4%) of those who had acute diseases were prescribed high-risk ACB medications (p = 0.001), as shown in Table 3.
Distribution of study population according to ACB score categories.

ACB: anticholinergic cognitive burden.
Chi-sqaure test is significant at p < 0.05.
The multivariable logistic regression analysis after adjusting for confounders (number of medications per prescription, sex, and age) showed that patients who had acute diseases were 2.5 times (adjusted odds ratio (AOR) = 2.49, 95% confidence interval (CI): 1.40–4.45) more likely to be exposed to high-risk ACB medications than those with chronic diseases (Table 4).
Multivariable analysis for the predictors of older adults’ exposure to high-risk ACB score medications.

ACB: anticholinergic cognitive burden; CI: confidence interval.
Significant at p < 0.05.
Discussion
To the best of our knowledge, this is the first study to investigate the anticholinergic medication burden in older adults in northern Nigeria. According to the study findings, a considerable proportion of the study population was exposed to polypharmacy and high-risk ACB medications. Furosemide, nifedipine, and loratadine were the most commonly prescribed anticholinergic medications, although exposure to high-risk ACB medications was significantly associated with the treatment of acute disorders diseases.
The current study indicated a high frequency of polypharmacy. The value from the present study is comparable to the rate (54.5%) recorded in a study conducted in southwest Nigeria. 27 It is worthy to note that polypharmacy can have serious consequences for patients and the healthcare system. For patients, this may entail ADRs, falls, and hospitalization, as well as poor quality of life and increased nonadherence to prescriptions, increased healthcare costs, and mortality. 34 On the contrary, the effects of polypharmacy on the healthcare system include decreased healthcare provider productivity and quality of care, and an increase in medication errors. 35 As a result, there is an urgent need for interventions such as pharmacists’ educating physicians on rational prescription, pharmacist-led medication reviews, and cautious deprescribing. These techniques will aid in the prevention of ADR, and improve medication adherence and disease processes in older adults. Conversely, the prevalence rate noted in our study is remarkably greater than the rates (23.9%, 29.5%, and 35.7%) recorded in other studies conducted in southwest Nigeria.19,28,36 Differences in physicians’ prescribing habits and disease burden may explain the observed discrepancies. Furthermore, varied prevalence rates of polypharmacy among older adults have been observed in different countries. A much lower prevalence of 13.5% was found in Brazil, 31 while 42.7% was recorded in Ethiopia. 29 However, far higher prevalence rates of 80.5% and 92.0% were noted in Saudi Arabia and Australia, respectively.30,32 The disparities in results could be attributed to differences in patient type, disease burden, and prescribing culture of the physicians across countries.
In our study, there was a high prevalence of exposure to high-risk ACB medications. This finding highlights the need for the use of low-risk ACB medications in older adults in the study setting. The ACB tool can be used to select appropriate medications with low ACB scores for older adults or facilitate deprescribing to limit exposure to needless anticholinergic medications. Reducing unnecessary anticholinergic medications in older adults can help to prevent or lessen cognitive decline, 37 falls, 38 and other functional impairments in this high-risk population. A previous study in southwest Nigeria found a higher exposure rate of 40.8% (60/147) to high-risk ACB medications, 19 compared with 26.6% (188/256) in our study. Furthermore, higher prevalence rates of 36.4% (20/55) and 48.4% (30/62) were reported in Malaysia and the United States, respectively.39,40 These disparities could be attributed to differences in physicians’ prescribing culture, anticholinergic burden assessment methodologies, and cutoffs.
In our study, cardiovascular drugs and antihistamines ranked highest as the most frequently prescribed anticholinergic medication classes. In contrast, antidepressants and opioid receptor agonists were the most frequently used anticholinergic medication classes in older adults in a previous study. 41 Furthermore, anxiolytics and antidepressants were reported in Australia, 12 while anxiolytics/hypnotics and antidepressants were reported in Ireland. 24 The analysis of the individual anticholinergic medications revealed furosemide and nifedipine as the most frequently prescribed. These findings were expected due to the high frequency of hypertension and pedal edema in our study population. Of interest here is that mild anticholinergic effects of furosemide and nifedipine are typically ignored by physicians, necessitating vigilance when they are required to be administered concurrently with other anticholinergic medications to avoid harmful effects due to their cumulative effects. Conversely, zopiclone, temazepam, and tamsulosin were reported as the most commonly prescribed individual anticholinergic medications in Finland, 42 while estazolam and terazosin were found in China, 26 and codeine and tramadol were reported in Ireland. 24 The observed discrepancies could be explained by differences in disease patterns, prescribing culture, and drug formularies across countries.
The analysis of the study data for potential independent predictors of exposure to high-risk ACB medications revealed treatment of acute disease conditions as a significant predictor. The combined effects of antihistamines, such as loratadine in the treatment of some acute respiratory disorders, as well as other anticholinergic medication(s), could explain this finding. However, the observed high prescription of antihistamines, particularly loratadine, is not surprising due to the high prevalence of seasonal allergies in the research location caused by the dusty environment that lasts several months each year. It is worth noting that this significant independent predictor of exposure to high-risk ACB medications is modifiable. As a result, tailored interventions focused on minimizing unnecessary anticholinergic medications used in the treatment of acute disorders in older adults may result in improved cognitive and functional outcomes. Evidence demonstrates that prioritizing high-risk groups and focusing on medications that can be safely substituted or terminated could make medication reviews more successful.43–46 A previous study has shown that a pharmacist-led multidisciplinary medication review can reduce the dispensing of unnecessary anticholinergic medications to older adults, resulting in a considerable reduction in the anticholinergic burden score. 6 Another study revealed that providing information to general practitioners about patients’ drug burden index (DBI) scores resulted in lower DBI scores in older adults. 47 The recommendation of pharmacists to replace high ACB score medications with low ACB score medications could also be a useful technique for limiting older adults’ exposure to high-risk ACB medications.
The implication of the findings of the present study is the critical need for the involvement of pharmacists in the multidisciplinary care team for older adults to help reduce unnecessary anticholinergic medications, ADEs, and improve their overall health outcomes.
Study limitations
The main limitation of the study was a retrospective study design that was unable to gather information on other medications that patients were taking which were not documented in their medical records kept in the study hospital, and the inability to determine changes in patients’ exposure to anticholinergic medications over time. Also, the study analysis was dependent on the quality of the medical records and the amount of detail they contained. The consequences of exposure to ACB medications were not analyzed due to a lack of such data in the patients’ medical records. Future studies that would assess the relationships between potential adverse effects, physical function outcomes and ACB scores are recommended. Second, although a priori sample size was not estimated, the study included all patients that met the inclusion criteria. Third, this was a single-center study, and only included outpatients, so the findings cannot be generalized to other hospitals, and departments/units. Further studies that would include a larger sample size of outpatients and hospitalized patients are warranted. Also, benchmarking the study institution to others through future comparative retrospective studies may enhance the quality of the study.
Conclusion
The present study revealed a high prevalence of polypharmacy and exposure to high ACB medications in older adults. Our findings demonstrate that older adults’ exposure to high ACB medications was significantly associated with the treatment of acute diseases, likely driven by high loratadine prescriptions in conjunction with other anticholinergic medications required for the treatment of other diseases. These findings highlight the need for a multidisciplinary care team that includes a clinical pharmacist. A clinical pharmacist in the team can provide targeted interventions such as physicians’ education and training on rational prescribing, pharmacist-led medication reviews, and deprescribing of unnecessary anticholinergic medications to improve the quality of care for older adults.
Footnotes
Appendix 1

| Anticholinergics | |
|---|---|
| Alprazolam | Homatropine |
| Amantadine | Hydroxyzine |
| Amitriptyline | Hyoscyamine |
| Atropine | Imipramine |
| Baclofen | Loperamide |
| Benztropine | Loratadine |
| Brompheniramine | Loxapine |
| Cetirizine | Meclizine |
| Chlorpheniramine | Methocarbamol |
| Chlorpromazine | Methscopolamine |
| Cimetidine | Nifedipine |
| Clemastine | Nortriptyline |
| Clomipramine | Olanzapine |
| Clozapine | Orphenadrine |
| Codeine | Oxybutynin |
| Colchicine | Paroxetine |
| Cyproheptadine | Prochlorperazine |
| Desipramine | Promethazine |
| Dexchlorpheniramine | Propantheline |
| Diazepam | Protriptyline |
| Digoxin | Pseudoephedrine Hcl/Triprolidine Hcl |
| Dimenhydrinate | Ranitidine |
| Diphenhydramine | Scopolamine |
| Diphenoxylate | Thioridazine |
| Disopyramide | Thiothixene |
| Doxylamine | Tizanidine |
| Fesoterodine | Trifluoperazine |
| Fluphenazine | Trihexyphenidyl |
| Furosemide | Triprolidine |
Author contribution(s)
Availability of data and materials
The dataset generated and/or analyzed during the current study is available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethics approval (SSH/GEN/641) for the study was obtained from the Ethics and Research Committee of the study hospital before the commencement of data collection. Due to the retrospective nature of the study, the need for informed consent for inclusion in and/or collection/use of data for the present study was waived by the ethics committee. The confidentiality of patients’ information was maintained throughout the study duration by using anonymous codes.
Informed consent
Not applicable
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
