Abstract
Background:
Cardiac rehabilitation using mobile health is increasingly being described as effective in improving exercise habits and physical function. However, the effectiveness of combined mobile health and hospital-based cardiac rehabilitation in older patients with cardiovascular disease is unknown.
Objective:
To investigate the effect of a wearable watch device combined with hospital-based cardiac rehabilitation in older patients with cardiovascular disease compared with those with usual care.
Methods:
A randomized, controlled trial was conducted in patients with cardiovascular disease aged ⩾60 years who underwent hospital-based cardiac rehabilitation. The patients were randomized 1:1 in those with a wearable watch (Fitbit Versa 3) or usual care. During out-of-hospital cardiac rehabilitation, patients with a wearable watch were advised to check their heart rate with a wearable watch and to target their physical activity at the heart rate determined by cardiopulmonary exercise testing. The primary endpoint was a change in physical activity obtained from the International Physical Activity Questionnaire (metabolic equivalents [METs]-minute/week) between the 2 groups during 12 weeks.
Results:
Among 39 patients (median age = 73 years, 82% men), the change in physical activity was significantly greater in patients with a wearable watch than in those with usual care (median: 2073 [1155-4400] vs 148 [−1261-899] METs-minute/week; analysis of covariance; P < .001). In patients with a wearable watch, 6-minute walk distance, the peak oxygen uptake, peak METs, and anaerobic threshold were significantly improved, whereas no significant increase was observed in patients with usual care from baseline to 12 weeks.
Conclusions:
In older patients with cardiovascular disease, a wearable watch device combined with hospital-based cardiac rehabilitation increases physical activity and improves exercise capacity outside of hospital.
Introduction
Cardiac rehabilitation is a recommended comprehensive program for patients with cardiovascular disease.1,2 Cardiac rehabilitation, with a focus on exercise training, contributes to improved exercise capacity and reduced symptoms, such as dyspnea and fatigue on exertion, and is associated with an improved long-term prognosis.3,4 Despite the demonstrated benefits of cardiac rehabilitation, participation rates in cardiac rehabilitation are low.5,6 This low participation rate is attributed to geography and physical or psychological barriers to frequent hospital visits for hospital-based cardiac rehabilitation. 7 Participation in cardiac rehabilitation is especially low in older people. 8 Furthermore, this situation has been exacerbated by the coronavirus disease 2019 pandemic. 9 Hospital-based cardiac rehabilitation comprises a short session during the week, and the duration of the intervention is short term (once a week, each session consists of 40-60 minutes, and until 150 days, rehabilitation is covered by the Japanese insurance system). To address these medical and social issues, implementing continuous out-of-hospital cardiac rehabilitation, such as outside of hospital or home-based, is important. However, out-of-hospital cardiac rehabilitation often results in decreased motivation in cardiac rehabilitation, an inadequate physical workload, and safety concerns owing to the lack of direct supervision. 10
Cardiac rehabilitation using mobile health is increasingly being described as effective in improving exercise habits and physical function.11,12 In the past, problems such as a lack of technology and low availability of mobile health for older people have limited the introduction of cardiac rehabilitation using mobile health. In recent years, the use of smartphones and Internet availability in older people has dramatically increased, and there is a need to verify whether cardiac rehabilitation using mobile health can be implemented safely and effectively. 13 To date, there have been few reports on the effectiveness of mobile health cardiac rehabilitation in older people. Therefore, this study aimed to investigate whether a combination of a wearable watch device and hospital-based cardiac rehabilitation in older patients contributes to increased daily physical activity and improved exercise capacity outside of hospital.
Methods
Study Design
This was a single-center, randomized, controlled trial. Patients were recruited at Kindai University Nara Hospital between April 2023 and October 2024. The patients were randomized by concealed allocation to 1 of 2 groups (wearable watch use or usual care) before cardiopulmonary exercise testing. A Fitbit Versa 3 (Fitbit Inc., San Francisco, CA, USA) wearable watch was used, and it was provided to the patients by the hospital. The Fitbit series is widely used worldwide as a wearable watch device to observe physical activity. 14 Both groups underwent cardiac rehabilitation under the supervision of a physical therapist. In addition, all patients were advised to perform their own aerobic and muscle resistance training outside of the hospital setting. Cardiac rehabilitation was performed for 12 weeks, and follow-up evaluations were conducted. This study complied with the Declaration of Helsinki. Written informed consent was obtained from all participants. In this study, the analysis protocol was approved by the ethics committee of Kindai University Nara Hospital (approval number: 690).
Study Population
The subjects were patients aged 60 years or older with cardiovascular disease and they received outpatient hospital-based cardiac rehabilitation. Patients with cardiovascular disease were defined as those with a diagnosis of angina pectoris, myocardial infarction, or chronic heart failure. The exclusion criteria were as follows: patients who were unable to walk independently; those who had severe atrioventricular block; those who had a pacemaker, implantable cardioverter defibrillator, or cardiac resynchronization therapy implanted; those who were undergoing hemodialysis; those who had an active malignancy; those who had decompensated heart failure; those who had severe valvular disease; those who had undergone a heart transplant; and those who had dementia that made handling the wearable watch difficult.
Intervention
The patients underwent cardiopulmonary exercise testing at baseline and center-based cardiac rehabilitation with a target heart rate at the anaerobic threshold. In accordance with the guidelines of European Association of Preventive Cardiology, all patients were instructed to perform out-of-hospital cardiac rehabilitation consisting of moderate physical activity for at least 30 minutes each day, 5 days each week, or 15 minutes of vigorous physical activity for at least 15 minutes each day, 5 days each week, or a combination of both for at least 10 minutes. 1 We requested that the patients with a wearable watch wear it continuously for their daily activities. When exercising outside the hospital, they were instructed to check their heart rate with the wearable watch and engage in moderate to vigorous physical activity as close to the target heart rate as possible. The patients were informed that their daily step count would be recorded via the wearable watch. The daily step count could be checked by the patients themselves by operating the watch device, and when they visited the hospital, the step count information was shared between the cardiologist and the patients. We also instructed the patients to engage in moderate physical activity with a target of 11 to 13 on the Borg assessment scale. 15
Outcome
The primary outcome was the difference in an increase in self-reported physical activity (metabolic equivalents [METs]-minute/week) derived from the International Physical Activity Questionnaire (IPAQ) between the 2 groups over 12 weeks. The major secondary outcomes were an increase in the IPAQ score within each group and the difference in an increase in peak oxygen uptake (VO2) between the 2 groups. Other secondary outcomes included the 6-minute walk distance, grip strength, calf circumference, Short Physical Performance Battery score, bioelectrical impedance analysis, other cardiopulmonary exercise testing parameters, Patient Health Questionnaire-9 (PHQ-9) score, blood test results, and the left ventricular ejection fraction. Correlations were examined for the mean step count and the change in peak VO2 over 12 weeks in patients with a wearable watch.
Data Collection
The body mass index and medical history were collected at baseline. Blood tests, echocardiography, IPAQ scores, 6-minute walk distance, grip strength (GRIP-D; Takei Scientific Instruments Co., Ltd., Tokyo, Japan), calf circumference, Short Physical Performance Battery score, bioelectrical impedance analysis (InBody S10; InBody Co., Ltd., Tokyo, Japan), cardiopulmonary exercise test, and PHQ-9 scores were measured at baseline and 12 weeks later. Questionnaire was collected at baseline and at the 12-week follow-up to determine how motivated the patients were to exercise on their own. The patients selected 1 of 4 items from the questionnaire (not at all motivated, slightly, some, or a lot) in response to the question “Are you motivated to exercise on your own?” The patients were considered to be self-motivated to exercise if they chose “some” or “a lot” of motivation.
Cardiopulmonary Exercise Test
The cardiopulmonary exercise test was performed with a cycle ergometer (Strength Ergo 8; Fukada Denshi, Tokyo, Japan) using a ramp stress test. Heart rate, blood pressure, a 12-lead electrocardiogram (FCP-8800; Fukada Denshi), and respiratory gas analysis (Cpex-1; Fukada Denshi) were recorded. Testing was considered maximal if the respiratory gas exchange ratio was >1.1 or heart rate was >85% of the maximum predicted heart rate. Peak VO2 was defined as the oxygen uptake in the last 30 seconds of testing. The anaerobic threshold was determined using ventilatory gas exchange analysis, and was identified by the simultaneous occurrence of the following: a sustained increase in a respiratory exchange ratio >1.0, a sharp inflection point in the VCO2 versus VO2 relationship (V-slope method), a divergence in the ventilatory equivalents for oxygen and carbon dioxide (VE/VO2 increases while VE/VCO2 plateaus or decreases), and an increase in end-tidal oxygen concentrations. Daily step counts during the 12 weeks of the study were obtained in patients with a wearable watch.
International Physical Activity Questionnaire
Physical activity was assessed using the IPAQ at baseline and after 12 weeks. The IPAQ score was calculated by classifying physical activity into 3 levels (walking, moderate physical activity, and vigorous physical activity) over the last 7 days of the questionnaire and the total time (minutes) of each activity in a week. 16 The following formulas were used for assessment: physical activity related to walking = 3.3 METs × minutes of walking per day × days walking per week; physical activity related to moderate physical activity = 4.0 METs × minutes of moderate physical activity per day × days performing moderate physical activity per week; and physical activity related to vigorous physical activity = 8.0 METs × minutes of vigorous physical activity per day × days of vigorous physical activity per week. The total of these 3 scores was calculated as total physical activity (METs-minute/week).
Sample Size
On the basis of a previous study of mobile health in patients with cardiovascular disease, the difference in total MET-minute/week after the intervention between mobile health users and non-users was assumed to be 900 METs-minute/week. 17 The standard deviation was 1000 METs-minute/week. The Mann-Whitney U test was used to detect statistically significant differences between groups, with a significance level of 5% to obtain 95% power, resulting in 19 patients in each group. A total of 42 enrollments were planned, taking into consideration that 10% of patients would be lost to follow-up.
Statistical Analyses
Continuous variables are presented as the median and interquartile range, and categorical variables as the number and percentage. Wilcoxon’s signed rank test was used to analyze treatment effects within groups. The Mann-Whitney U test was used to compare treatments between groups. The primary endpoint, which was a comparison of the change in total physical activity between the 2 groups, was assessed by analysis of covariance (ANCOVA) with baseline total physical activity as an adjustment factor. Correlations between step counts per day and the change in peak VO2 between baseline and follow-up in patients with a wearable watch were assessed by Pearson’s rank correlation coefficient. Statistical significance was set at P < .05. JMP Pro 18.1.0 software (SAS Institute Inc., Cary, NC, USA) was used for all statistical analyses.
Results
Patients
Among the 54 patients who were recruited, 10 declined to participate and 5 withdrew during the course of the study. Therefore, the final analysis included 39 patients (Figure 1). No clinical events, such as cardiovascular events, heart failure hospitalization, or death, were observed during the 12-week observation period. Among the patients (median age = 73 years, interquartile range = 68-78 years, 82% men), there were no significant differences in baseline characteristics between the 2 groups, except for male sex and statin use (Table 1). The majority of patients had comorbid hypertension and coronary artery disease. Most patients had a left ventricular ejection fraction >50%.

Study flowchart.
Baseline characteristics.

Abbreviations: ACEi, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; BNP, B-type natriuretic peptide; HbA1C, glycated hemoglobin; HDL, high-density lipoprotein; LDL, low-density lipoprotein; LVEF, left ventricular ejection fraction; SGLT2, sodium-glucose cotransporter 2 inhibitor.
Data are presented as the median (25th-75th percentiles) or number (%).
Primary Outcome
The change in total physical activity was significantly greater in patients with a wearable watch than in those with usual care (median: 2073 [1155-4400] vs 148 [−1261-899] METs-minute/week; ANCOVA P < .001; Figure 2a).
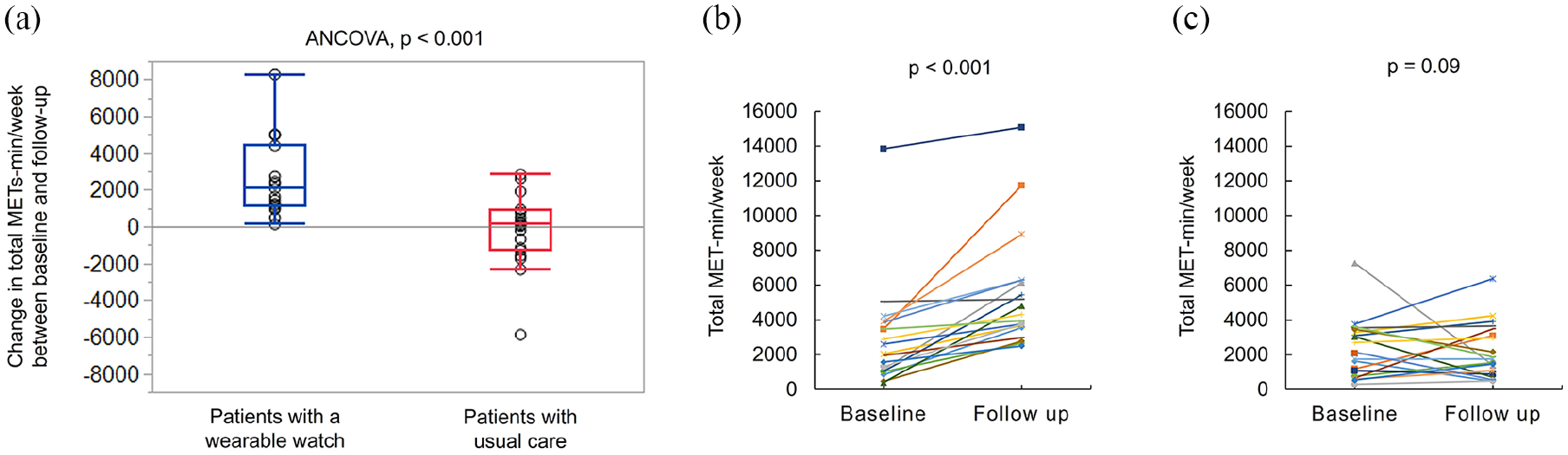
Change in physical activity (METs-minute/week) between baseline and 12 weeks of follow-up. (a) Comparison of the change (between baseline and follow-up) in total METs-minute/week between the 2 groups of patients. Patients with a wearable watch (blue) showed a significantly greater change in total METs-minute/week than patients with usual care (red). (b) Baseline and follow-up total METs-minute/week in individual patients with a wearable watch. (c) Baseline and follow-up total METs-minute/week in individual patients with usual care.
Comparison Between Baseline and Follow-Up Within the Groups
Patients with a wearable watch showed a significant increase in total physical activity at the follow-up compared with baseline (2034 [1040-3852] vs 4305 [3560-6285] METs-minute/week; P < .001; Figure 2b). In contrast, patients with usual care showed no significant increase in total physical activity between baseline and follow-up (2424 [836-3411] vs 1813 [930-3388] METs-minute/week; P = .09; Figure 2c).
A comparison of physical parameters within the groups between baseline and follow-up is shown in Table 2. In patients with a wearable watch, 6-minute walk distance (P = .02), peak VO2 (P = .02), peak METs (P = .01), and the anaerobic threshold (P < .001) were significantly increased at follow-up compared with baseline. In contrast, there was no significant increase in these variables in patients with usual care. Grip strength, calf circumference, Short Physical Performance Battery score, bioelectrical impedance analysis, and PHQ-9 score were not significantly different within the groups between baseline and follow-up.
Physical parameters at baseline and follow-up.

Abbreviations: 6MWD, 6-minute walk distance; AT, anaerobic threshold; HR, heart rate; METs, metabolic equivalents; PHQ-9, Patient Health Questionnaire-9; RER, respiratory exchange ratio; SPPB, Short Physical Performance Battery; VE/VCO2, ventilatory equivalent for carbon dioxide; VO2, oxygen uptake.
Data are presented as the median (25th-75th percentiles) or number (%).
A comparison of laboratory parameters and the left ventricular ejection fraction between baseline and follow-up within the groups is shown in Supplemental Table 1. There were significant alterations in serum creatinine, glycated hemoglobin, and high-density lipoprotein cholesterol concentrations at follow-up compared with baseline in patients with a wearable watch. There were no significant differences in laboratory parameters and left ventricular ejection fraction between baseline and follow-up in patients with usual care.
Comparison of the Change From Baseline to Follow-Up Between the 2 Groups
A comparison of the change from baseline to follow-up between the 2 groups is shown in Table 3. The change in peak VO2 was not significantly different between the 2 groups. The change in the anaerobic threshold was significantly improved in patients with a wearable watch compared with those with usual care (P < .01). There were no significant differences in the 6-minute walk distance, grip strength, calf circumference, SPPB, bioelectrical impedance analysis, or PHQ-9 scores between the 2 groups.
Change in physical parameters between baseline and follow-up.

Abbreviations: 6MWD, 6-minute walk distance; AT, anaerobic threshold; HR, heart rate; METs, metabolic equivalents; PHQ-9, Patient Health Questionnaire-9; RER, respiratory exchange ratio; SPPB, Short Physical Performance Battery; VE/VCO2, ventilatory equivalent for carbon dioxide; VO2, oxygen uptake.
Data are presented as the median (25th-75th percentiles) or number (%).
Correlation Between the Mean Step Count and Peak VO2
In patients with a wearable watch, the correlation between the mean step count per day and the change in peak VO2 from baseline to follow-up was examined (Figure 3). Pearson’s rank correlation analysis showed a significant positive correlation between these variables (r = .51, P = .02).

Relationship between the mean step count and change in peak VO2. Pearson correlation analysis shows a positive correlation between the mean step count and change in peak VO2.
Changes in Motivation to Exercise
At baseline and the 12-week follow-up, a questionnaire was administered to determine the degree of the patients’ motivation to exercise on their own (Figure 4). The proportion of patients with a wearable watch who were motivated to exercise was 73.7% (14/19 patients) at baseline and 89.5% (17/19 patients) at follow-up (P = .21). The proportion of patients with usual care who were motivated to exercise was 80.0% (16/20 patients) at baseline and 70.0% (14/20 patients) at follow-up (P = .47). There was no significant difference in the rate of patients who were motivated to exercise at follow-up between those with a wearable watch and those with usual care (P = .13).

Changes in motivation to exercise.
Discussion
To the best of our knowledge, this study is the first to examine the effect of integrating the use of a wearable watch in older patients with cardiovascular disease in an established cardiac rehabilitation program. Patients with a wearable watch had significantly greater physical activity as assessed by the IPAQ than those with usual care. In patients with a wearable watch, 6-minute walk distance, peak VO2, peak METs, and the anaerobic threshold were significantly improved between baseline and follow-up. A significant correlation was found between the mean step count per day and a change in peak VO2 in patients with a wearable watch. None of the patients had adverse cardiovascular events during the 12 weeks of the study period. Older patients are less likely to benefit from cardiac rehabilitation improvements in exercise capacity than young patients. 18 Potential mechanisms of this finding include the fact that older patients are less likely than young patients to exercise at higher intensities and are less likely to benefit from exercise programs because of multiple comorbidities.19,20 However, opportunities to provide cardiac rehabilitation to older patients with cardiovascular disease are increasing in clinical practice. 21 Our study showed that using the combination of a wearable watch outside of the hospital setting and a hospital-based cardiac rehabilitation program contributed to an increase in physical activity and improved exercise capacity in older patients.
Assistive devices used in cardiac rehabilitation include a variety of interventions, such as computer-assisted, exercise video games, tablets, smart phones, and wearable devices. In cardiac rehabilitation, many studies have reported interventions using mobile phones or wearable watches. 13 The use of mobile phones or wearable watches have the following benefits: increased accessibility for patients who are unable to participate in traditional cardiac rehabilitation; easier access to information resources on cardiac rehabilitation treatment planning; a potentially improved commitment to cardiac rehabilitation treatment planning; more flexible scheduling of cardiac rehabilitation; and the ability of patients to perform cardiac rehabilitation on their own. 13
A meta-analysis examined whether interventions with older adults using a wearable watch help promote physical activity. 22 The wearable watch was shown to be an effective intervention for increasing steps and physical activity, and decreasing the sedentary time. This meta-analysis included 69 randomized controlled trials, of which only 1 trial investigated the effectiveness of a wearable watch as part of cardiac rehabilitation. 23 Therefore, the effectiveness of using a wearable watch in cardiac rehabilitation for older patients with cardiovascular disease has not been adequately evaluated.
Despite the reported positive effects of cardiac rehabilitation on exercise capacity and the long-term prognosis, the percentage of outpatient cardiac rehabilitation introduced in Japan is extremely low at 7%. 6 Older patients are particularly underrepresented in cardiac rehabilitation. 8 Cardiac rehabilitation is primarily provided as a hospital-based protocol, and is affected by socioeconomic factors including transportation to hospitals. 24 Cardiac rehabilitation of older patients with cardiovascular disease through the use of a wearable watch can overcome geographic barriers, logistical barriers (eg, transportation, scheduling, and center capacity), and other related barriers. Furthermore, in our study, we observed a significant improvement in daily physical activity and exercise capacity in patients with a wearable watch. This finding suggests that hospital-based cardiac rehabilitation combined with mobile health outside of the hospital can be safely and effectively implemented.
The potential mechanisms by which a wearable watch improves physical activity and exercise capacity outside of the hospital are unclear. However, mobile health interventions for cardiac rehabilitation were shown to improve adherence in a meta-analysis, suggesting that mobile health contributes to behavioral change in patients. 25 Improving the effectiveness of mobile health is expected to depend more on the design of the engagement strategy than on the characteristics of the technology. 25 Personal encouragement, competitiveness and cooperation, and support through effective feedback related to human behavior are considered effective. In our study, we assessed the motivation to exercise at baseline and follow-up using a questionnaire. We found no significant difference in the proportion of patients who were motivated to exercise at follow-up between those with a wearable watch and those with usual care. However, the proportion of patients who were motivated to exercise was decreased from baseline to follow-up in patients with usual care, but this proportion was increased in patients with a wearable watch. These findings suggested that the intervention using a wearable watch had a partial effect on behavioral change. In our study, patients with usual care also received education and coaching by physical therapists in hospital-based cardiac rehabilitation, and there was no incentive or competitive system for exercise. This approach may not have had a strong effect on the change in motivation to exercise between the 2 groups. Our study was conducted in a small number of cases. The association between therapeutic intervention with a wearable watch and improved motivation to exercise in older patients needs to be examined in a larger number of cases.
In our study, men accounted for 82% of the patients, which is likely owing to the large proportion of patients with coronary artery disease who were included. Most previous studies on cardiac rehabilitation using mobile devices included approximately 80% of male patients, which is a background similar to our study. 26 In healthy adults, a study showed sex differences in the effects of exercise training on exercise capacity, and increased peakVO2 was lower in women, regardless of the body mass index. 27 However, a meta-analysis of healthy adults reported no difference in improved exercise capacity with aerobic exercise between men and women. 28 Whether sex differences affect the effectiveness of a wearable watch differently is unknown and requires further examination.
Although improving exercise capacity is important for older patients with cardiovascular disease, increasing physical activity in daily life is equally important. The reason for this finding is that increased physical activity helps to prevent or delay the development of diabetes, heart disease, and chronic disease, leading to an overall effect on secondary prevention of cardiovascular disease. 29 The guideline of European Association of Preventive Cardiology also recommends increased physical activity in out-of-hospital cardiac rehabilitation. 1 Mobile health is currently being used as an intervention to increase physical activity because of its affordability and convenience, and has established itself as an important strategy that can change users’ lifestyles. Therefore, a strength of our study is that we showed that a wearable watch integrated with hospital-based cardiac rehabilitation increased daily physical activity in older patients with cardiovascular disease. Additionally, the use of heart rate as an indicator provided safe and effective cardiac rehabilitation.
This study has some limitations, such as the small sample size at a single center. A further analysis with a larger number of cases at multiple centers is warranted. Additionally, we used self-reporting to assess changes in physical activity, which may have been insufficient for objective assessment of a wearable watch. In contrast, a meta-analysis reported a strong correlation between self-reporting and objective measures of physical activity. 12 We found a significant positive correlation between self-reported changes in physical activity and the step count. Furthermore, objective data on exercise capacity significantly improved in patients with a wearable watch, suggesting a clinical benefit of a wearable watch. Another limitation is that our study examined the effects of a wearable watch during 12 weeks but did not examine the long-term benefits. Further investigation of whether the use of wearable watch leads to the maintenance of a long-term increase in physical activity should be considered.
Conclusions
In older patients with cardiovascular disease, heart rate monitoring by a wearable watch combined with hospital-based cardiac rehabilitation contributes to an increase in physical activity and exercise capacity outside of the hospital. Active use of mobile health is recommended for older people.
Supplemental Material
sj-docx-1-rpo-10.1177_27536351251343538 – Supplemental material for Integration of Wearables Into a Cardiac Rehabilitation Program and Its Impact on Physical Activity and Exercise Capacity in Older Patients With Cardiovascular Disease
Supplemental material, sj-docx-1-rpo-10.1177_27536351251343538 for Integration of Wearables Into a Cardiac Rehabilitation Program and Its Impact on Physical Activity and Exercise Capacity in Older Patients With Cardiovascular Disease by Eijiro Yagi, Koichiro Matsumura, Yuki Uchigashima, Jun Shiroyama, Mitsuki Hase, Tomoya Nanba, Nobuhiro Yamada, Yohei Funauchi, Masafumi Ueno, Kiyonori Togi, Manabu Shirotani and Gaku Nakazawa in Advances in Rehabilitation Science and Practice
Footnotes
Correction (September 2025):
In table 2, the value for “Whole skeletal muscle mass” has been corrected from “2.7” to “20.7”, and the value for “VE/VCO2 slope” has been corrected from “4.1” to “40.1”.
Ethical Considerations
This study complied with the Declaration of Helsinki. In this study, the analysis protocol was approved by the ethics committee of Kindai University Nara Hospital (approval number: 690).
Consent to Participate
Written informed consent was obtained from all participants
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author, KM, upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
