Abstract
Background
Patients with pulmonary
Objective
To evaluate the physical, immunological, and psychiatric effects of Jiinshihoto in patients with pulmonary MAC who were ineligible for standard antimicrobial treatment.
Methods
This single-center, open-label, prospective study was conducted from March 2018 to January 2020. Patients who were not eligible for standard treatment for pulmonary MAC disease were enrolled. Jiinshihoto (3.0 g) was administered three times per day for 12 months. The primary endpoints were 3-month changes in chronic obstructive pulmonary disease assessment test (CAT) scores, body weight, and natural killer (NK) cell activity. Secondary endpoints were 3-month changes in self-rating depression scale (SDS) scores. This study was registered in the UMIN-Clinical Trials Registry (UMIN000033590).
Results
In total, 24 patients were enrolled. The mean age was 68 years, and 74% were female; the mean body mass index was 19.3 kg/m2. Of these, 23 patients completed the 3-month follow-up. For the primary endpoints at 3 months, no significant changes were observed in CAT scores (from 11.2 to 11.0,
Conclusion
Jiinshihoto did not improve respiratory symptoms, weight, or NK cell activity in patients with pulmonary MAC ineligible for standard treatment but may be beneficial for depressive symptoms.
Introduction
Nontuberculous mycobacteria (NTM) are increasingly recognized as important opportunistic pathogens worldwide.
1
In Japan, the estimated incidence of pulmonary nontuberculous mycobacteria infection was 14.7 (per 100 000 population),
2
with pulmonary
Kampo formulae have shown potential as effective treatments for NTM, with some formulae demonstrating desirable treatment outcomes.4,5 For example, Hochuekkito, a Kampo formula, has been found to suppress the increasing number of bacteria discharged in sputum, prevent chest deterioration observed through imaging, and improve appetite. 4 Hochuekkito also increases the population of natural killer (NK) cells 6 ; however, there is limited evidence supporting Kampo therapy in patients who cannot receive standard treatment.
Jiinshihoto is a Kampo formula traditionally used for patients with chronic respiratory symptoms such as cough, sputum, and general weakness, particularly in women. 7 It has been used to treat chronic inflammatory airway diseases and contains multiple herbal components with effects on both respiratory and psychological symptoms. 8 Based on these properties, we hypothesized that Jiinshihoto might be beneficial for patients with pulmonary MAC disease who are not candidates for standard antimicrobial therapy. Therefore, this study aimed to evaluate the efficacy of Jiinshihoto in improving physical, psychological, and immunological outcomes in this patient population.
Materials and Methods
Study Design
This single-center, open-label, prospective study was conducted at the University of Fukui Hospital from March 2018 to January 2020. A target sample size of 30 patients was determined considering the number of patients with MAC disease who visit our facility.
This study was conducted in accordance with the Declaration of Helsinki. The protocol and informed consent documents were approved by the Ethics Committee of the Faculty of Medical Sciences of the University of Fukui (Registration No: Fukui 20170186, approval date: 21/02/2018) and were registered in the UMIN-Clinical Trials Registry (UMIN000033590). Written informed consent was obtained from all study participants.
Study Population
This study enrolled patients with pulmonary MAC disease who were considered ineligible for long-term standard antimicrobial therapy or had declined such therapy after informed discussion. The inclusion criteria were as follows: (i) patients who were diagnosed with pulmonary MAC according to the diagnostic criteria of the American Thoracic Society (ATS) and the Infectious Diseases Society of America (IDSA) 3 ; (ii) patients aged between 20 and 80 years; (iii) patients who did not use rifampicin, ethambutol, azithromycin, clarithromycin, streptomycin, kanamycin, rifabutin, sitafloxacin, or levofloxacin. The use of these drugs for a duration of less than 1 week for purposes other than treating pulmonary MAC disease was allowed.
Patients were considered ineligible for standard antimicrobial therapy for one or more of the following predefined reasons: (i) mild non-cavitary nodular bronchiectasis without radiological or clinical progression, for which careful observation was deemed appropriate; (ii) previous intolerance to standard antimicrobial therapy due to adverse events that precluded continuation of long-term treatment, as determined based on documented adverse reactions during previous treatment; (iii) refusal to receive standard therapy after being informed of potential adverse effects, uncertain long-term benefits, or the possibility of disease recurrence; or (iv) advanced age or comorbidities judged by the treating physician to potentially heighten the risk of treatment-related adverse events.
The exclusion criteria were as follows: (i) patients receiving standard antimicrobial therapy for pulmonary MAC disease; (ii) patients currently undergoing treatment for pulmonary tuberculosis; (iii) patients with hypokalemia; (iv) patients with heart failure; (v) patients with drug allergies; (vi) pregnant or breastfeeding women; and (vii) patients who had participated in another clinical trial within the past 4 months.
All patients diagnosed with pulmonary MAC disease, with the exception of those with mild disease who were managed with observation alone (as described above), were initially offered standard antimicrobial therapy in accordance with current clinical guidelines. 9 Only patients who met the aforementioned criteria for ineligibility or who declined standard therapy after adequate explanation were enrolled in this study.
Study Procedure
Eligible patients were enrolled at our respiratory medicine outpatient clinic and received baseline assessments including clinical evaluations, laboratory tests, psychological assessments, and chest computed tomography (CT) imaging before initiating treatment. Jiinshihoto extract granules (3.0 g) were administered three times daily for 12 months.
Follow-up assessments were conducted at 3 months (primary endpoint assessment) and 12 months (final assessment). At each visit, patients underwent clinical examination and safety monitoring including blood pressure and electrolyte levels to detect potential pseudoaldosteronism.
Patients were continuously monitored for disease progression throughout the study period. Disease progression requiring standard antimicrobial therapy was defined as follows: (i) new bronchiectasis, bronchiolitis, or nodules appearing in a previously unaffected lung lobe; (ii) appearance of new infiltrative or cavitary shadows in the same or another lung lobe; or (iii) appearance of fresh, blood-stained sputum. Patients meeting these criteria were withdrawn from the study and received standard antimicrobial therapy. In cases where the study was terminated due to disease progression, the data obtained at termination were carried forward and regarded as the 12-month data.
Adherence to medication was assessed based on treatment persistence. It was defined as completion of the planned 12-month Jiinshihoto treatment period. Patients who discontinued treatment as a consequence of disease progression, adverse events, or withdrawal of consent were classified as non-completers.
Endpoints
The primary endpoints were 3-month changes in chronic obstructive pulmonary disease assessment test (CAT) scores, body weight, and NK cell activity. The secondary endpoints were the changes at 3 and 12 months in self-rating depression scale (SDS) scores, State-Trait Anxiety Inventory (STAI) scores, chest CT findings, as well as the safety of Jiinshihoto. Various cytokines and chemokines were examined as exploratory endpoints.
Assessment of the Respiratory Symptoms
The patients completed the CAT questionnaire before treatment, 3 months after treatment, and at the end of the study. The CAT is a short, validated questionnaire to be completed by patients and is used to assess the impact of chronic obstructive pulmonary disease on health status. It comprises eight items (cough, phlegm, chest tightness, breathlessness going up hills/stairs, activity limitations at home, confidence, sleep, and energy) that are formatted using a 6-point differential scale.10-13 As the items correspond to all chronic respiratory diseases, CAT can be used for chronic obstructive pulmonary disease as well as a wide range of respiratory diseases and was applied in our study to evaluate the results. Previous studies have shown a strong correlation between CAT and the St. George’s Respiratory Questionnaire 14 ; CAT was selected based on its simpler format, which was suitable for this study.
Psychological Tests
SDS and STAI scores were measured before treatment, 3 months after treatment, and at the end of the study. The SDS score represents the self-rated depression of a patient, derived from answers to a 20-item questionnaire that was developed through a factor analytic study of depression and depressive symptoms. SDS scores were categorized according to the degree of depression as follows: 20-39, normal; 40-47, mild depression; 48-55, moderate depression; and ≥56, severe depression.15,16
The STAI consists of two scales: state anxiety (anxiety at the time of measurement) and trait anxiety (anxiety as a personality trait). Both scales include 20 items rated on a 4-point scale. High anxiety thresholds were ≥42 points for state anxiety and ≥44 points (males) or ≥45 points (females) for trait anxiety.17,18
Assessments of Inflammation, NK Cell Activity, Cytokines, and Chemokines
Erythrocyte sedimentation rate, white blood cell count, and C-reactive protein levels were measured to assess inflammation. Lymphocyte count and albumin, total cholesterol, and cholinesterase levels were measured to assess the nutritional status. Biological defense mechanisms can be evaluated by measuring the cytotoxic activity of NK cells. NK cell activity was determined using the 51Cr release method, which calculates activity values by adding effector cells (NK cells) to target cells (K-562) marked with 51Cr, culturing them, and measuring 51Cr released due to target cell damage. 19 The standard value is 18-40%.
Serum cytokine CC chemokine ligand 17/thymus and activation-regulated chemokine, interferon-α, interferon-γ, interleukin (IL)-2, IL-4, IL-5, IL-6, IL-12/IL-23 p40, IL-13, IL-17/IL-17A, IL-18/IL-1F4, and tumor necrosis factor-α were assayed using MILLIPLEX® MAP Human Cytokine/Chemokine/Growth Factor Panel A 48 Plex Premixed Magnetic Bead Panel-Immunology Multiplex Assay (MERCK Millipore Corporation, Billerica, MA, USA). Levels of granulocyte-macrophage colony-stimulating factor were measured using an enzyme-linked immunosorbent assay (Quantikine; R&D Systems, Minneapolis, MN, USA).
Assessment of the CT Images
Chest CT findings were evaluated by two respiratory physicians using a scoring system for pulmonary MAC disease developed by Song et al. 20 In case of discrepancies, final decisions were reached by consensus.
Safety Evaluation
Safety was evaluated by monitoring for side effects throughout the study period. Because Jiinshihoto contains glycyrrhiza, which can cause pseudoaldosteronism, patients were monitored for increased blood pressure and sodium levels, fluid retention, edema, hypokalemia, and associated myopathy. The severity of adverse events was graded based on the Common Terminology Criteria for Adverse Events, version 5.0, developed by the U.S. National Cancer Institute. 21
Statistical Analysis
Continuous variables are presented as mean and standard deviation, and categorical variables as frequency and percentage. The analysis population included patients who received at least one dose of Jiinshihoto and had complete primary endpoint data at 3 months.
Changes in primary and secondary endpoints from baseline to 3 months and 12 months were analyzed using paired
Results
In total, 24 patients were enrolled (Figure 1). One patient discontinued after 2 weeks of treatment owing to worsening liver dysfunction, and two patients withdrew consent at 3 months (one declined to continue taking the study medication, and one failed to attend the scheduled outpatient visits). Four additional patients were discontinued between 3 and 10 months owing to symptom progression. Among the enrolled patients, the reasons for ineligibility for standard antimicrobial therapy were as follows: mild nodular bronchiectasis in 12 patients (50.0%), previous evidence of drug intolerance due to adverse events in one patient (4.2%), refusal of standard therapy owing to concerns regarding adverse effects in six patients (25.0%), and advanced age or comorbidities, such as liver dysfunction, in five patients (20.8%). Disease progression was indicated by radiological progression on chest CT in three patients and the development of hemoptysis in one patient, for all four of whom, standard antimicrobial therapy was subsequently initiated. Flow diagram of patient enrollment and analysis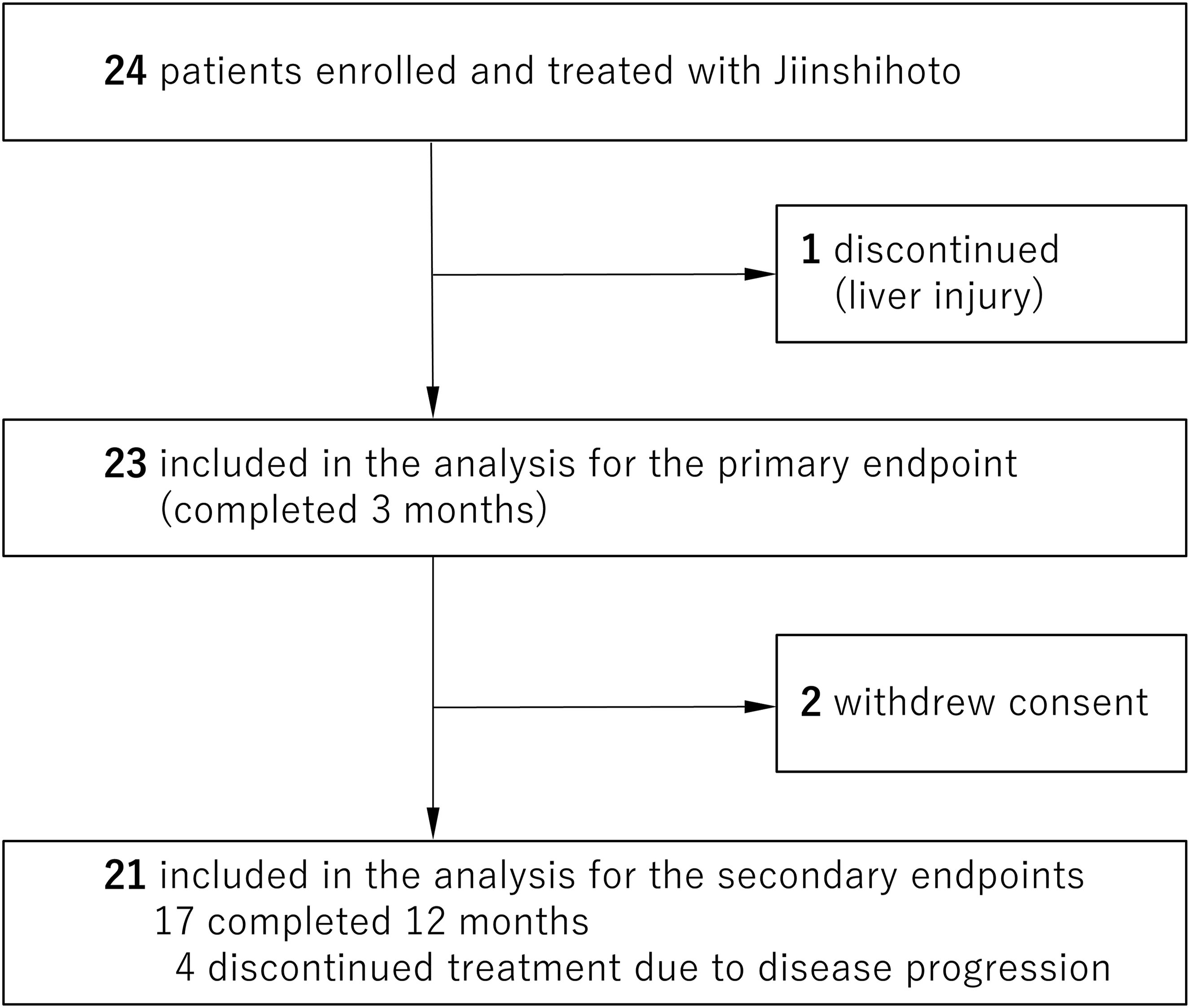
Patient Characteristics
Baseline Characteristics of Patients

Values are presented as mean (standard deviation) or n (%) unless otherwise specified.
Abbreviations: BMI, body mass index; CAT, chronic obstructive pulmonary disease assessment test; CCL17, C-C motif chemokine ligand 17; CRP, C-reactive protein; CT, computed tomography; ESR, erythrocyte sedimentation rate; FAS, full analysis set; IL, interleukin; LDH, lactate dehydrogenase; NK, natural killer; SD, standard deviation; SDS, self-rating depression scale; STAI, state-trait anxiety inventory; TARC, thymus and activation-regulated chemokine; TNF, tumor necrosis factor; WBC, white blood cell count.
Primary Endpoints
At 3 months, there were no significant changes in any of the three primary endpoints compared with baseline. The mean change in CAT score was −0.3 (95% confidence interval [CI], −3.4 to 2.9; Changes in primary endpoints from baseline to 3 months. Changes in chronic obstructive pulmonary disease assessment test (CAT) scores (A), body weight (B), and natural killer (NK) cell activity (C) from baseline to 3 months. Data are presented as individual patient values (gray dots) connected by lines, with box plots indicating the 25th and 75th percentiles, median (horizontal line), and mean (cross). 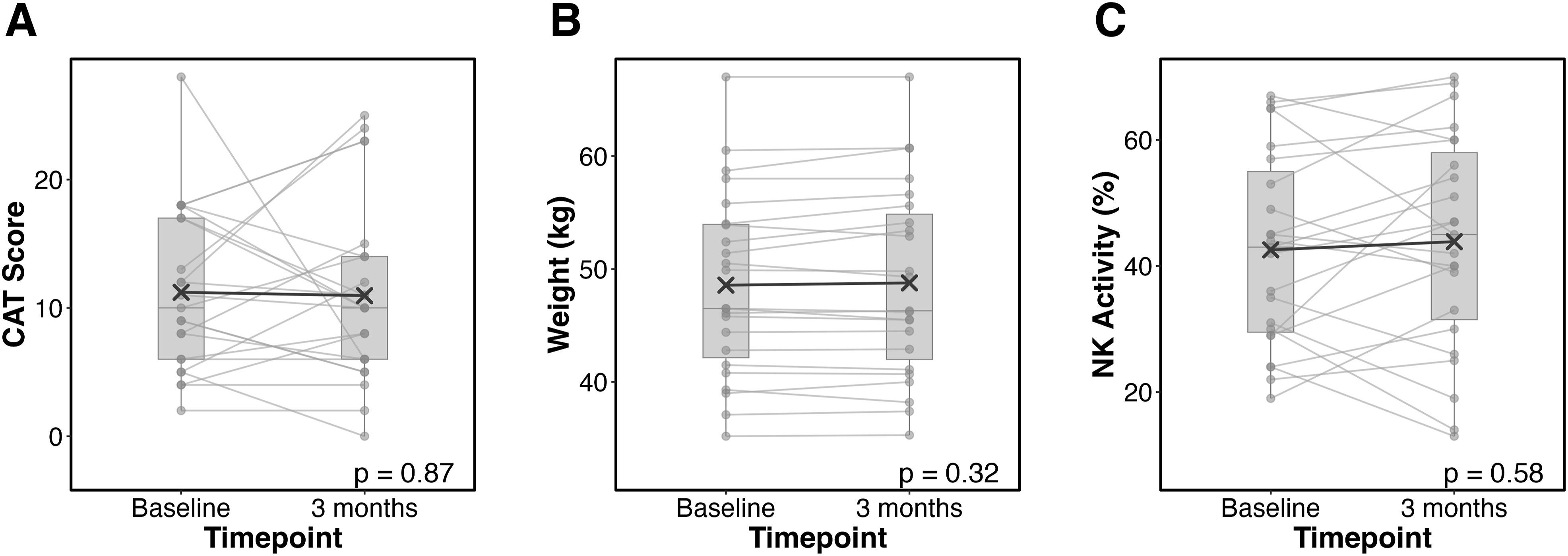
Secondary Endpoints
Psychological assessments showed a significant improvement in SDS score, with a mean change of −3.9 points (95% CI: −7.3 to −0.4,
Exploratory Analysis
During the study period, we detected no significant changes with respect to the CAT score, body weight, or NK cell activity. From baseline to 12 months, the mean change in the CAT score was +0.7 points (p for trend = 0.65), body weight declined by 0.5 kg (p for trend = 0.28), and NK cell activity increased by 2.4% (p for trend = 0.45).
Imaging and Inflammatory Outcomes
Imaging and inflammatory marker scores worsened over time. CT scores increased by + 6.8 points at 3 months and +7.4 points at 12 months ( Longitudinal changes in psychological, imaging, and inflammatory parameters during Jiinshihoto treatment (A) chronic obstructive pulmonary disease assessment test (CAT) scores, (B) body weight, (C) natural killer (NK) cell activity, (D) self-rating depression scale (SDS) score, (E) state-trait anxiety inventory (STAI) trait score, (F) STAI state score, (G) computed tomography (CT) score, and (H) erythrocyte sedimentation rate (ESR) were assessed at baseline, 3 months (3M), and 12 months (12M). Data are presented as mean ± standard deviation, with the number of patients shown below each timepoint. 
Adherence to Medication
On the basis of their completion of the full 12-month treatment period, 17 of the 24 enrolled patients (70.8%) completed the prescribed Jiinshihoto regimen. None of the patients discontinued treatment as a consequence of adverse drug reactions. The remaining seven patients failed to complete the 12-month treatment period owing to either a refusal to continue the prescribed treatment or a progression of the disease that required the initiation of standard antimicrobial therapy.
Safety Evaluation
None of the patients treated with Jiinshihoto experienced any adverse events including pseudohypoaldosteronism, myopathy, anorexia, gastric discomfort, nausea, or diarrhea. One patient with mild liver injury prior to treatment discontinued the study because it worsened during the study period. Four patients with disease progression discontinued the study and received standard treatment.
Discussion
In this prospective study evaluating the effects of Jiinshihoto in patients with pulmonary MAC disease who were not receiving standard antimicrobial therapy, we detected no significant improvements in the pre-specified primary endpoints at 3 months, namely, the CAT score, body weight, and NK cell activity. These findings indicate that Jiinshihoto does not produce a measurable short-term effect on respiratory symptoms, nutritional status, or innate immune function in this patient population.
Previous studies4,5 of Kampo formulations in pulmonary MAC disease have reported radiological stabilization and increased NK cell indices with Hochuekkito, and case reports have noted weight gain with Ninjinyoeito. In contrast, in our cohort, none of the primary endpoints showed significant improvement. Several factors may explain these negative findings: the natural disease progression in this patient population, which may have offset any potential therapeutic benefits, and limited potential for further NK cell activation because most patients had baseline levels within the normal range. Additionally, the relatively short 3-month observation period may be insufficient to detect meaningful changes.
Despite no improvement in the primary endpoints at the pre-specified 3-month evaluation, a significant and sustained improvement in depressive symptoms, as assessed by the SDS score, was observed at both 3 and 12 months. This psychological benefit was observed despite a significant worsening of CT scores over the same period. Thus, the effects on depressive symptoms represent secondary outcomes rather than efficacy against pulmonary MAC disease.
Psychological distress, including depression and anxiety, is highly prevalent among patients with pulmonary MAC disease and has been reported to persist independently of objective disease severity or radiological progression.22,23 From this perspective, the dissociation between worsening imaging findings and improvement in depressive symptoms observed in the present study is clinically plausible and indicates that Jiinshihoto may have beneficial effects on mental well-being that are independent of disease activity.
Our study population comprised typical patients with MAC disease, predominantly elderly women with low body weight and concurrent psychological symptoms including mild depression and elevated anxiety. These characteristics align with established epidemiological patterns of pulmonary MAC disease.22,23 In our study, 3 months after Jiinshihoto administration, the SDS score had decreased to the non-depressive range. While this change may not be considered a major effect, it has the potential to contribute to reduced psychological burden and improved quality of life in this population.
Several components of Jiinshihoto, such as
The enrolled patients in the current study had mild nodular bronchiectasis or had declined standard antimicrobial therapy owing to concerns about drug tolerance or potential adverse effects. While the ATS/ERS/ESCMID/IDSA clinical practice guidelines recommend antimicrobial treatment over observation for patients meeting NTM disease diagnostic criteria, 9 many patients with pulmonary MAC disease cannot tolerate or are unwilling to undergo standard therapy. For these patients, supportive treatments including antitussives, expectorants, and Kampo formulations represent therapeutic options. Although our findings in this study fail to provide evidence for the use of Jiinshihoto as a substitute for standard antimicrobial therapy, this herbal medicine may have utility as a supportive treatment that could contribute to improve psychological well-being rather than as a therapy targeting respiratory symptoms, immune function, or disease progression.
This study has some limitations. First, the small sample size limited statistical power to detect clinically meaningful differences and precluded multivariable adjustment for potential confounding factors. Second, as this was a single-center study, our institutional practices and patient management approaches may have influenced outcomes, limiting the generalizability of findings to other settings. Third, the open-label design resulted in potential bias in outcome assessments. Fourth, we did not systematically control for or analyze the impact of concurrent supportive therapies, lifestyle modifications, or other interventions that patients may have received during the study period, which could have confounded our results. In addition, given that we included only those patients who were not receiving standard antimicrobial therapy at enrollment, we could not evaluate the potential additive or synergistic effects of Jiinshihoto when combined with standard treatment. Consequently, we could not extrapolate our findings to patients undergoing standard antimicrobial therapy, and thus further randomized controlled trials comparing standard therapy alone with standard therapy plus Jiinshihoto are warranted. Finally, the short follow-up period limits conclusions about long-term psychological and physical efficacy, and longer observation might have revealed more durable effects.
Conclusion
Though Jiinshihoto did not improve clinical symptoms, body weight, or NK cell activity in patients with pulmonary MAC disease who were not eligible for standard antimicrobial therapy, a sustained amelioration of depressive symptoms was observed in these patients upon Jiinshihoto treatment, suggesting a potential role of Jiinshihoto in supporting psychological well-being in this patient population.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
