Abstract
Objective
Poor sleep and depression are common problems during pregnancy, but there has been little investigation into the association between them. This prospective study aims to examine the relationship between sleep quality and depression during pregnancy.
Methods
Pregnant women under 14 weeks’ gestation attending routine outpatient antenatal care in Singapore’s largest maternity hospital were recruited between 2012 and 2014. Women with multiple pregnancies and deemed at high risk of miscarriage were excluded. Six hundred and forty participants completed the Pittsburgh Sleep Quality Index (PSQI) and Edinburgh Postnatal Depression Scale (EPDS) at the three trimesters during pregnancy.
Results
Mean PSQI score was highest in the third visit, suggesting poorer quality sleep in the late third trimester compared to other trimesters. 15.6% of participants at each time point fulfilled the criteria for antenatal depression according to the EPDS cut-off score > 14. PSQI scores were significantly correlated with EPDS scores, and also prospectively predicted EPDS scores in all three trimesters.
Conclusion
Sleep quality in Singaporean pregnant women was poorest in the third trimester, and was associated with the development of depressive symptoms. With more than 1 in 10 women having antenatal depression, interventions targeting sleep quality might be particularly beneficial.
Introduction
Sleep problems and depressive symptoms are commonly experienced by pregnant women. A US National Sleep Foundation survey found that more than 79% of women report sleep changes during pregnancy compared to other times. 1 Common sleep disorders pregnant women face include sleep restriction, poor sleep quality and efficiency, sleep-disordered breathing and parasomnias.2–6 These symptoms are thought to be associated with pregnancy-related physical discomfort4,7 and fluctuations in hormone levels. 8 Women are also vulnerable to mood disorders during pregnancy, with an estimated worldwide prevalence of antenatal depression ranging between 10% and 25%.9–10 Locally, the prevalence ranges from 12.2% in the general pregnant population 11 up to 18% for individuals having high-risk pregnancies. 12 Such mood fluctuations are believed to be multifactorial in origin, with changes in hormonal levels, socio-cultural and psychosocial factors associated with pregnancy having been implicated aetiologically.
Considerable research has demonstrated a close bi-directional relationship between derangements in sleep and mood disorders in the general population. Sleep deprivation has a significant effect on human functioning, 13 including loss of concentration, irritability and lethargy, 14 which are symptoms analogous to those experienced by depressed individuals. Sleep disturbance is also a core symptom in mood disorders, occurring in more than 80% of individuals diagnosed with depression. 15 Another study showed that poorer sleep quality, as measured by the Pittsburgh Sleep Quality Index (PSQI), was associated with a lower ambulatory positive effect. 16 This finding was significant even after adjusting for the diagnosis of depression and self-reported anxiety. They concluded that impaired sleep quality could explain reduced pleasurable experiences in daily activities, possibly a critical factor in the pathophysiology of depression. 17
There is also extensive evidence illustrating how sleep disturbances can predict the onset and recurrence of depression in susceptible individuals.17–20 Previous research on community samples of young adults and elderly individuals showed that sleep disturbances predicted subsequent depression across 2 to 3 years.17,21 Individuals with persistent complaints of interrupted sleep and insomnia were also found to have an elevated risk of developing depressive episodes within a year. 22
In pregnancy, there is a paucity of studies looking into the relationship between sleep problems and depression, despite existing literature depicting considerable rates of both insomnia and depression amongst women who are pregnant. One of the reasons might be due to the mistaken presumption by many that sleep disturbances are common and expected during pregnancy and post birth. Indeed, conventional measures used to screen for mood disorders during pregnancy and after delivery, such as the Edinburgh Postnatal Depression Scale (EPDS), also exclude sleep disturbances as a core symptom, instead qualifying insomnia as a consequence of low mood.
There is increasing evidence that poor sleep in early pregnancy can predict subsequent depression in both ante- and postnatal phases.23–26 In addition, pregnancy-related physical symptoms have also been found to predict depressive symptoms in the third trimester, both directly and indirectly, via the mediation of reduced sleep quality. 24 Such changes in maternal sleep quality and mood state can result in adverse pregnancy outcomes including intra-uterine growth restriction, preterm labour27,28 and prolonged labour. 29
Objectives of the Study
Our primary aim is to ascertain the prevalence of sleep disturbances and depressive symptoms in Singaporean pregnant women, and examine prospectively their link. We also aim to identify demographic and clinical risk factors for antenatal depression in Singapore.
The relationship between insomnia and depression in pregnancy is poorly established, especially in an Asian setting. To the best of our knowledge, this is the first study examining a sizable Southeast Asian sample. This study will provide valuable evidence for future studies examining the impact of insomnia in pregnant women and exploring possible treatment options. Findings from this study will also have public health implications in terms of service provision and healthcare planning.
Methods
This study was nested in a prospective cohort perinatal study on adverse pregnancy outcomes, entitled ‘Improving the Adverse Outcomes of Pregnancy – Clinical and Translational Research Program in Perinatal Medicine’. The sample was derived from KK Women’s and Children’s Hospital Singapore (KKH), a tertiary maternity hospital. KKH handles approximately 12,000 deliveries annually, accounting for 25–30% of all national births.
The study was granted ethics approval by the SingHealth Centralized Institutional Review Board (CIRB). Written informed consent was collected from all participants upon recruitment.
Participants
Pregnant women attending antenatal care in KKH at less than 14 weeks’ gestation were screened for eligibility between August 2012 and April 2014. Women with uncomplicated singleton pregnancies were invited to participate in this study. Women deemed to have higher risk pregnancies, such as multiple pregnancies and chronic medical conditions (e.g. systemic lupus erythematosus or renal disease), were excluded from this study. Women with a history of pregnancies complicated by aneuploidies and cervical incompetence were also excluded.
Study Procedures
Women who met study criteria and recruited into the study were followed up at outpatient clinics during the antenatal periods between 11–14 weeks’, 18–22 weeks’ and 28–32 weeks’ gestation. These time points correspond to the first, second and third trimesters of gestation, respectively. Detailed interviews were conducted by trained staff who are well-versed in the primary languages spoken locally. Vital demographic data consisting of age, marital status, accommodation type, monthly income and educational levels was obtained. Participants were also asked to complete self-rated questionnaires during each visit. The questionnaires were made available in English, Chinese and Malay.
Measures
Assessment of depressive symptoms
Depressive symptoms were assessed using the EPDS, a ten-item self-rated questionnaire that is widely used to screen for postnatal depression. It has also been validated for use in pregnant populations. 30 Each question has a score from zero to three with a maximum total score of 30. A cut-off score of greater than 14 is used to identify probable cases of antenatal depression. 31 Previous validity studies have shown a specificity of 98% and sensitivity of 57% for this score in identifying probable antenatal depression. 32
Assessment of sleep quality
The Pittsburgh Sleep Quality Index is a 19-item self-report measure that measures seven subscale domains of sleep. These areas comprise sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbance, use of sleeping medication and daytime dysfunction over the previous month. It consists of both open-ended and multiple-choice questions. Each subscale has a possible score of zero to three, with a maximum global score of 21. A cut-off of more than five is used to identify probable cases of poor sleep quality. 33 The PSQI and its psychometric properties have been validated in pregnant women with good internal consistency as well as convergent and divergent reliability properties.26,34
Statistical Analysis
Statistical analysis was done using the Statistical Package for Social Sciences Version 21.0 (IBM SPSS v21.0 for Windows XP).
All assumptions of parametric tests were met.
The explanatory variables were examined using descriptive statistics. PSQI scores were grouped into good sleep quality (PSQI total score ≤ five) and poor sleep quality (PSQI total score > five). Categorical data were examined using chi-squared analysis. Continuous data were examined using independent t-tests and repeated measures ANOVA. Pearson’s correlations explored prospective relationships between EPDS scores and PSQI scores. Univariate logistic regression analysis was used to determine the impact of each socio-demographic or clinical variable associated with the presence of depression in pregnancy. Multivariate logistic regression analysis was then performed to adjust for the effect of confounders on presence of depressive symptoms at any time point.
Statistical significance was set at two-tailed p-value < 0.05.
Results
Subjects
Six hundred and forty participants consented to participating in the study while attending their regular outpatient appointments at the clinic, and completed the questionnaires for all three visits. Eligible participants who did not complete the study were observed to suffer from higher levels of depressive symptoms based on the mean EPDS scores (8.3 vs 7.1, p = 0.003). There were no other statistically significant demographic differences between responders and non-responders.
Demographic and Clinical Characteristics of Participants
The mean age was 30.4 ± 4.7 years (range = 18–43). Over half the participants were Chinese (54.2%, n = 347), with the others being Malay (24.1%, n = 154), Indian (11.5%, n = 154) and others (10.2%, n = 74). This reflects Singapore’s multi-racial distribution. Most were married (92.5%, n = 592). 69.7% had at least tertiary education (n = 446), and 93.0% of the women (n = 595) stayed in public housing. In comparison with the national statistics, 32.4% of Singaporeans have at least tertiary education, and 81.9% of them stay in public housing. 35
The numbers of women with probable depression in each of the three trimesters were comparable. However, mean EPDS scores were higher in the first trimester than in the second trimester (7.6 vs 7.0, p < 0.001) and third trimester (7.6 vs 7.2, p = 0.038).
A majority of women (n = 480, 75%) experienced poor sleep at least during one time point. Mean PSQI scores were highest in the third visit, followed by the first and second visits. More than half of the participants experienced poor sleep quality in the first (n = 325, 50.8%) and third (n = 375, 58.6%) trimesters. Women reportedly slept longest hours in the first trimester with a mean duration of 7.0 ± 1.5 h (range = 2–12). The time spent asleep gradually decreased with the subsequent trimesters. More than 40% (n = 268) of the women recruited had persistently poor sleep, experiencing poor sleep in two or more consecutive trimesters.
Demographic characteristics of the sample.

Comparison of Mean Pittsburgh Sleep Quality Index Scores Between Depressed and Non-Depressed Women
Comparison of mean PSQI scores between depressed and non-depressed women.

There was homogeneity of variances, according to Levene’s test for equality of variances, for first (F = 2.8; p = 0.9) and third (F = 0.4; p = 0.6) visits. Independent t-tests showed that depressed participants consistently scored higher on the PSQI than non-depressed participants, with mean differences of 3.1 ± 0.4 (p < 0.001) in the first trimester and 2.8 ± 0.4 (p < 0.001) in the third trimester. The assumption of homogeneity of variances was violated for results in the second trimester (F = 6.2; p = 0.01), with a t-test of −6.2 (p < 0.001). Similarly, independent t-tests showed that depressed participants scored higher on the PSQI than non-depressed participants in the second trimester, with mean differences of 3.5 ± 0.6 (p < 0.001).
Prospective Relationship Between Sleep Quality and Depression
Correlations between Pittsburgh Sleep Quality Index (PSQI) and Edinburgh Postnatal Depression Scale (EPDS, n = 640) at three time points (T1 to T3) in pregnancy.

aCorrelation is significant at the 0.001 level (2-tailed).
PSQI and EPDS scores were stable across trimesters. Earlier time point PSQI scores had a correlation between later time point PSQI scores. EPDS scores earlier in gestation had a stronger correlation to later EPDS scores (p = 0.001). These findings suggest that women who complained of poor sleep quality and depressive symptomatology earlier in the pregnancy were more likely to experience persistence of symptoms up till the third trimester.
EPDS scores in the first trimester significantly predicted PSQI global scores in the second and third trimesters. Similarly, EPDS in the second trimester predicted PSQI scores in the third trimester (p = 0.001).
PSQI scores prospectively predicted EPDS scores significantly across all time points. PSQI global scores in the first trimester predicted EPDS scores in the second and third trimesters. PSQI scores in the second trimester were significantly more predictive of EPDS in the third trimester.
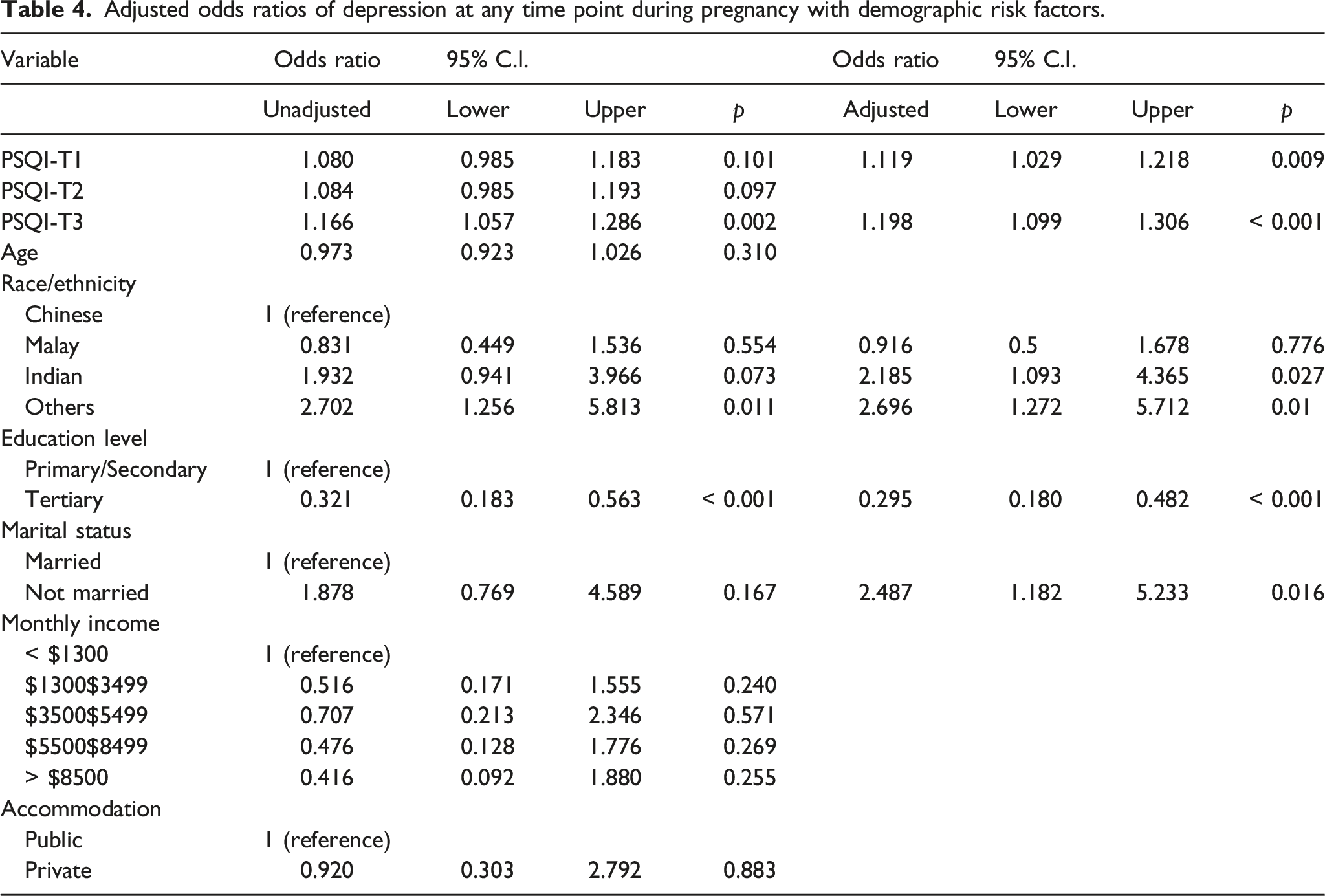
Predictors of Depression at any Time Point
Multivariate logistic regression analysis, using all PSQI scores and demographic data as variables, was used to identify factors associated with depression at any time point. This model accounts for 24.9% of variance (Nagelkerke) and shows adequate goodness of fit with the observed distribution, with Hosmer and Lemeshow χ2(df = 8) = 6.7, p = 0.6.
Adjusted odds ratios of depression at any time point during pregnancy with demographic risk factors.
Discussion
Our study found the prevalence of antenatal depression and sleep disturbances to be 15.6% and 75%, respectively. This is consistent with worldwide and local figures.6,11,12,36,37
Results also indicate that first-trimester mood and sleep disturbances tend to persist throughout pregnancy. This supports our proposed hypotheses and is in line with findings of previous research,22,25,38,39 suggesting that common complaints of poor sleep and mood changes early in pregnancy can predict depressive mood in later pregnancy.
In a comparable study,
25
the strength of correlation between the predictive nature of poor sleep on depression during pregnancy was higher (r = 0.42–0.64), compared with (r = 0.30–0.36) in our current study. However, these two study populations differed in ethnicity and demographic variables, which may be reflective of inherent differences in genetic and cultural factors. Another Singaporean birth cohort study found that pregnant women who reported poor sleep quality (PSQI > five) were statistically more likely to have probable antenatal depression (EPDS
Our findings also support a weak but significant predictive correlation between initial depressive symptomatology and later poor sleep quality in pregnancy, consistent with past studies examining the predictive relationship between mood disorders and sleep disturbances in healthy individuals.22,38 These findings were not replicated in previous studies involving pregnant women, 26 and require further clarification.
Nonetheless, our findings are in line with existing literature that sleep problems earlier in pregnancy may contribute to the development of depressive symptomatology later in pregnancy. Given that transient depressive symptoms can progress to clinical depression in both antenatal and postnatal periods, the implications of our findings are significant and warrant further research into evaluating the impact of such experiences on pregnancy and postnatal care.
Our findings also suggest that women of certain ethnic groups such as Indian and other races are at a higher risk of developing depressive symptoms. On the contrary, women with higher educational qualifications are protected from antenatal depression. Such findings are not surprising and are consistent with existing local data obtained from the Singapore Mental Health Study, a nationwide mental health epidemiological study conducted in from 2009 to 2010. 40 Our study also found that women who were unmarried during pregnancy were at a higher risk of having probable antenatal depression. This could be explained by reduced emotional support, a known risk factor for antenatal depression. 11
Strengths and Limitations of This Study
This is a large prospective cohort study exploring the longitudinal relationship between sleep quality and depressive symptomatology from the first to third trimesters of pregnancy. This study is also the first of its kind to examine the bi-directional relationship between sleep quality and pregnancy in an Asian population. The broad inclusion criterion for study participants also enhances the applicability and generalizability of the findings to routine clinical practice.
However, there are several limitations in this study. The findings of this study were based on self-report with majority of recruited subjects having tertiary education and higher socio-economic status as reflected by income, compared to the average Singaporean. 35 More unbiased measures of depression and sleep quality such as the Structured Clinical Interview for DSM disorders (SCID) for the diagnosis of depression and sleep polysomnography or actigraphy could have provided more objective data, and can be utilized for future studies. A minor limitation to the generalizability of our study was the difference in levels of depressive symptomatology between study subjects and non-participants, although a one-point difference in the severity of depressive symptoms might not be clinically meaningful. In retrospect, the study population could have been further profiled to include other critical demographic markers of life conditions, such as weight and BMI, smoking and alcohol use. As a result of such limitations in study design, we were unable to ascertain presence of pre-existing psychiatric conditions as well as psychosocial and lifestyle factors previously found to be associated with the development of antenatal depression. 11
The findings from this study can potentially influence clinical practice and service provision. Obstetricians and other healthcare workers should be alert to the relationship between sleep deprivation and depression in pregnancy. Health promotion strategies can be developed especially targeted at expectant women and encouraging them to prioritize healthy sleep habits during pregnancy despite a hectic lifestyle. As antenatal and postnatal depression are closely related, further research should be done to evaluate the impact of poor sleep in early gestations on birth outcomes and postpartum depressive symptoms, both directly and indirectly via antenatal sleep quality and depressive symptoms. In addition, divergent outcomes of study participants with stable or improving PSQI despite worsening EPDS could be explored to potentially yield meaningful and actionable insights.
Conclusion
Sleep quality in Singaporean pregnant women was poorest in the third trimester, and was associated with the development of depressive symptoms, highlighting the need for prompt assessment and treatment of at-risk individuals early in pregnancy.
Footnotes
Acknowledgements
We thank the contributions of study participants, NORA study group and all clinical staff involved. We would also like to thank Joshua GOOLEY, Neuroscience and Behavioral Disorders Program, Duke-NUS Graduate Medical School for his input.
Author Contributions
HC and KHT conceptualized and designed the project. NSR supervised the recruitment, data collection and managed the data. SHP drafted the manuscript, supervised by TEC and HC; CC edited the manuscript for journal; and KHT and HC obtained funding and supervised the study. All authors read and approved the final version of the manuscript.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study, Neonatal & Obstetric Risk Assessment (NORA) Improving the adverse outcomes of pregnancy: a clinical and translational research programme in perinatal medicine, was supported by National Medical Research Council (NMRC/PPG/KKH/2010 Project 1 NORA) and the SingHealth Foundation (SHF/FG510S/2011). The funding agencies did not play any role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Ethical Approval
Ethical approval to report these cases was obtained from CIRB Ref 2010/214/D.
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article
Availability of data
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.
