Abstract
Background
While specific elements of naturopathic medicine, such as botanical medicines and lifestyle interventions, have supporting evidence, there is limited quantitative data confirming its effectiveness as a comprehensive, whole-person medical approach for patients with metastatic colorectal cancer (CRC).
Objective
This study aims to retrospectively evaluate the integration of naturopathic modalities, including modulated electrohyperthermia (mEHT), into the standard of care for metastatic CRC. We compare survival outcomes between patients at the Integrated Health Clinic (IHC) and a matched control group from the National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) database, a de-identified, publicly available cancer registry in the United States.
Methods
A retrospective chart review was conducted for 131 IHC patients diagnosed with stage IV CRC and treated with mEHT between 2010 and 2021. These patients were matched with 262 controls from the SEER database using propensity score matching. The primary outcome was overall survival, with time zero defined as the first IHC treatment date (with controls assigned the time zero of their matched IHC patient) to account for immortal time bias. Survival analysis was conducted using a Kaplan-Meier curve, log-rank test, and Cox proportional-hazards model.
Results
The overall survival analysis did not achieve a statistically significant difference (HR = .76; 95% CI: .57-1.01) between the IHC (median survival time: 29 month) and SEER groups (median survival time: 18 months). Incorporating time-varying effects, the hazard ratio (HR) for the IHC group compared to the SEER group was .63 (95% CI: .46-.86) for survival <36 months, indicating a lower hazard of early mortality in the IHC group. Moreover, IHC patients who initiated treatment within 90 days of diagnosis had significantly improved survival compared to their matched controls (HR = .45; 95% CI: .28-.70).
Conclusion
This study provides evidence that integrative naturopathic treatment, including mEHT, can significantly improve survival outcomes for CRC patients in the first 36 months post-treatment and when initiated within 90 days of diagnosis.
Keywords
Introduction
Colorectal cancer (CRC) ranks as the third most prevalent cancer and stands as the second most common cause of cancer-related deaths, accounting for approximately 9.3% of all cancer deaths globally. 1 Approximately 33% of patients with CRC will develop metastases either at diagnosis or during follow-up. 2 Although CRC is highly manageable when detected early, the 5-year survival rate for metastatic CRC detected in later stages remains low at 20%. 3
In recent years, CRC has been on the rise among younger adults (under 50) in the United States and Canada.4-6 In a recent JAMA article, authors projected that by 2030, CRC will become the leading cause of death among individuals aged 20 to 49 due to its association with aggressive tumor traits that are less responsive to treatment. 7 These findings underscore the growing significance of CRC as a public health concern, necessitating collaborative efforts within the healthcare community to enhance patient outcomes and survival.
A systemic therapy approach targeting cancer cells throughout the body serves as the mainstay treatment for metastatic CRC. Chemotherapy regimens combining fluoropyrimidines, oxaliplatin, and irinotecan have been widely used as first-line treatments for metastatic CRC, with studies demonstrating improved overall survival and progression-free survival rates.8,9 Immunotherapy has also shown remarkable responses in patients with microsatellite instability-high (MSI-H) or mismatch repair-deficient (dMMR) tumors, leading to durable tumor control and improved survival outcomes. 10
While systemic therapies can have substantial benefits, they may also be associated with side effects that can impact mental well-being and quality of life. Common side effects of chemotherapy and immunotherapies, such as fatigue, nausea, neuropathy, diarrhea, dry mouth, drowsiness and skin rashes can contribute to psychological distress and mental health issues.11-15 Therefore, supportive care measures, including psychological interventions and integrative approaches, may be incorporated into treatment plans to address these aspects, enhance overall well-being, and potentially improve survival rates. Consequently, there exists a critical need to explore adjunctive cancer therapies that can enhance patient outcomes without undue risk.
Integrative approaches combining conventional therapies with complementary naturopathic modalities, have gained popularity among cancer patients seeking to improve quality of life and potentially enhance survival rates.16-18 However, evidence is lacking for its impact on overall survival, highlighting the need for further research.
Integrative oncology modalities range from dietary supplements, herbal remedies, and acupuncture to lifestyle counseling and physical therapies, aiming to support the body’s natural healing processes. One of the less commonly used modalities is hyperthermia, in which body tissue is exposed to high temperatures, typically between 38.5 to 43°C, to induce a number of important physiological changes in the tumour microenvironment, and ultimately aimed at irreversibly damaging cancer cells while minimizing harm to normal tissues. 19 This therapy is often used in conjunction with other cancer treatments, such as radiation therapy and chemotherapy, to enhance their effectiveness. 20
This approach is typically categorized into loco-regional hyperthermia, which targets specific body areas such as tumors, and whole-body hyperthermia, which raises overall body temperature to induce fever-like physiological effects aimed at addressing metastatic cancer. 20 Loco-regional hyperthermia delivers heat through external or internal applicators directly to the tumor site or regions of the body, whereas whole-body hyperthermia is applied systemically to effect cancer cells throughout the body. 21 Although standard loco-regional hyperthermia shows promising effectiveness in combination with other treatments, it lacks precision at the cellular level, potentially impacting both malignant and surrounding healthy tissues.22,23
To address this, modulated electro-hyperthermia (mEHT) has emerged as a unique loco-regional hyperthermia method that uses modulated and selective electromagnetic fields to generate targeted heat within cancer cells, minimizing heating of adjacent normal tissues. 24 This selective heating is based at least partially on the known cellular heterogeneity, aiming to exploit the electrical properties of malignant cells and allow focused treatment of the cancerous area. The disorganized tumour microenvironment leaves sporadic cells surrounded by an extracellular matrix rich in salts and electrolytes which provide passage of the electromagnetic energy to heat the cells. 25 While there is some promising research on the effectiveness of mEHT in cancer populations, the evidence remains limited, primarily focused on glioblastoma, lung and pancreatic cancer.26-31 A search of ClinicalTrials.gov reveals only seven registered randomized controlled trials (RCTs) investigating mEHT, none of which address CRC. Additionally, studies exploring mEHT’s effect on CRC has had mixed outcomes (Supplemental Table 1). While some studies suggest potential benefits, such as improved tumor regression and reduced toxicity,32,33 most findings show no significant improvement in overall survival compared to other therapies.32,34,35 The majority of studies are retrospective,32,33,35,36 with small sample sizes (range: 21-62), single arm,33,36 and varying control treatments, limiting the reliability and generalizability of the results. The heterogeneity in study designs and lack of standardized protocols highlight a significant gap in the evidence for this treatment in this specific cancer type, CRC.
The World Health Organization (WHO) emphasizes the necessity of establishing evidence to verify the safety and effectiveness of naturopathy. 37 Some studies have explored naturopathic modalities like herbal medicine (eg, curcumin, resveratrol), high-dose vitamin C, and mEHT as adjunct therapies for CRC, aiming to enhance conventional treatments and improve patient outcomes.35,36,38-41 While specific elements of naturopathic medicine, such as herbal and nutritional supplements and lifestyle interventions, have supporting evidence, there is a limited availability of quantitative scientific data confirming its effectiveness as a whole person, comprehensive medical approach. In this study, we aim to conduct a retrospective evaluation on the integration of naturopathic modalities to the standard of care, conventional treatment. More specifically, the naturopathic treatment approach included the use of mEHT in the treatment of metastatic CRC. We assessed its effect on overall survival when compared to a matched control group.
Materials and Methods
Study Design
This study employs a retrospective chart review design and was conducted at the Integrated Health Clinic (IHC) in Fort Langley, BC, focused on patients treated between 2010 and 2020. The patients receiving integrative naturopathic treatment were compared to controls sampled from the National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) database, a de-identified, publicly available database of cancer cases in the United States. In this study, we operate under the assumption that the standard of care, including chemotherapy, radiotherapy, and surgery, is consistent between Canada and the USA.
Participants
Patients were eligible for the study if they were diagnosed with CRC at 18 years of age or older and were stage IV, which meant they had metastases to other organs (eg, liver, lung, skeleton) at the time of diagnosis. The IHC patients were required to have received at least six rounds of mEHT treatments between August 1, 2010, and Dec. 31, 2021. Patients were excluded if they were missing chart data for any of the variables used in the analysis (eg, diagnosis date, cancer type/stage, standard of care treatment, and deceased date). In general, in British Columbia, naturopathic treatment is not covered by provincial health care, so patients of the IHC must be able to finance their treatment and travel to the clinic.
Similarly, the SEER database was filtered to records with an age at diagnosis of 18 years or older and a diagnosis of stage IV colorectal cancer with complete data for all variables included in the analysis. For patients with multiple diagnoses (multiple records) in the database meeting the inclusion criteria, the record with the earliest diagnosis date was selected.
In this study, we assume that none of the SEER patients received modulated electrohyperthermia (mEHT) due to its unavailability in the United States. While several FDA-approved hyperthermia devices (https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfPMA/pma.cfm) are used in the U.S. (ie, COOK VH8500 Hyperthermia Treatment System, MEDIFOCUS 100A, PYREXAR BSD 2000, LABTHERMICS Sonotherm 1000), none of these devices provide mEHT specifically. Specific data on the use of other forms of hyperthermia for cancer is lacking; however, it is recognized that this therapy is not widely accessible in the USA.42,43 Regarding integrative naturopathic treatments in general, a study showed that 26.5% of insured SEER patients used complementary and alternative medicine (CAM) services, including chiropractic, naturopathic treatments, message therapy, acupuncture, and other types of CAM. 44 This variation in treatment types and their distinctiveness from the protocols offered by the IHC in this study positions the SEER data as a reasonably comparable control group, representative of the general population of cancer patients, for evaluating the specific contributions of integrative naturopathic treatment.
Intervention
Patients of the IHC were treated using an integrative naturopathic treatment model, which incorporates a personalized cancer care plan tailored to each patient’s needs and circumstances. This treatment model included modalities such as mEHT, intravenous therapies including high dose vitamin C, targeted supplementation, focused immune support, acupuncture, dietary and lifestyle counselling.
mEHT was applied using the EHY-2000 + Oncotherm device that can heat (via radiofrequency) tumors up to 15 cm deep to the target temperature range of 38.5-42.5°C, over 60 minutes with a treatment area of 30 cm in diameter. The EHY-2000 Oncotherm device has been approved as a Health Canada Class III Medical Device.
In addition to integrative naturopathic treatment, all patients received standard conventional care as determined by their oncologist, independent of care received at IHC. Standard care treatment may have included chemotherapy, radiation, and/or surgery. For patients receiving radiotherapy concurrently, mEHT was applied 2-3 times per week during radiotherapy, attempting to apply mEHT within the shortest time frame before or after the radiation. For those receiving chemotherapy (such as FOLFIRI, FOLFOX, FOLFIRINOX, FOLFOXIRI) mEHT was generally applied on days 1, 3 and 5 of a 14-day cycle, and on days 1, 3, 5, 8 and 10 of a 21-day cycle. In the mEHT monotherapy salvage setting, when no further surgical, radiotherapy, or chemotherapy treatments were being used or offered, mEHT treatment was typically administered 2-3 times per week. All treatments were held for a minimum of one week prior to their next scan.
Data Collection
The charts of IHC patients meeting the inclusion criteria were reviewed and their information recorded. Data were obtained through reviewing the patient’s labs, pathology, imaging, consult and treatment notes from medical, surgical and radiation oncologists. The date of death was established by confirming with family members, conducting web searches for obituaries, or identifying the date when eligibility for coverage under the Medical Services Plan (MSP) was terminated. MSP refers to the public health insurance program in British Columbia, Canada, which provides provincially-funded medical care for residents. Data collected and used in the analysis were: date of death, age at diagnosis, sex, year of diagnosis, stage at diagnosis, treatment with surgery (yes/no), treatment with radiation (yes/no), and treatment with chemotherapy (yes/no). A research assistant initially pulled the data, and this information was meticulously re-checked by study principal investigator to ensure the accuracy of data retrieval. Equivalent data were extracted from the SEER database. IHC patients were followed until August 2023 to identify deaths, however, the SEER database only contains data until the end of 2021, so all IHC and SEER patients were censored at loss to follow-up or Dec. 31, 2021 if alive as of that date.
The Research Ethics Board (REB) at the Canadian College of Naturopathic Medicine reviewed and approved the study (study number CCNMREB055.Parmar). Notices were posted at the clinic locations to inform patients of the study and they had the option to opt out of participation. Participation or not had no effect on the patient’s treatment.
Statistical Analysis
Patients in the SEER database meeting the inclusion criteria formed the pool of potential controls. Matched controls were then selected from the pool via nearest neighbour propensity score matching with a ratio of 2 controls to 1 IHC patient and a caliper of .25 in units of logarithmic standard deviation. Logistic regression was used to predict the propensity scores and the variables included in the propensity score model were age at diagnosis, sex, year of diagnosis, treatment with surgery (yes/no), treatment with radiation (yes/no), and treatment with chemotherapy (yes/no). An exact match on sex was required. Propensity score matching attempts to balance the groups with respect to measured variables to remove the influence of measured confounders. This method assumes that there are no unmeasured confounders.
For IHC patients, there was a delay between diagnosis and beginning treatment at the IHC. As such, careful handling of the baseline time was required to avoid immortal time bias. We used the prescription time-distribution matching approach to account for immortal time bias. This method sets time zero equal to the time of first clinic treatment for IHC patients and SEER patients are assigned the same time zero as their matched IHC patient. SEER patients who died before their assigned time zero were excluded. Both groups are followed from time zero until death or the end of follow-up.
Kaplan-Meier curves were used to estimate the survival probability over time per group and the log-rank test was used to test for significant between-group differences. Restricted mean and median survival times were compared between groups. A Cox proportional-hazards (Cox PH) model was used to calculate a hazard ratio comparing the IHC patients to the controls, adjusting for remaining discrepancies between the matched groups in the measured covariates.
To investigate changes in the hazard ratio over the follow-up period, we re-estimated the Cox PH model with a time-varying coefficient for group. Based on the Kaplan-Meier plot and a plot of the Schoenfeld residuals, we selected a cut-off survival time of 36 months and estimated separate coefficients for group for survival times of <36 months and ≥36 months.
We hypothesized that the subgroup of IHC patients who began clinic treatment within 90 days of diagnosis would receive the greatest benefit from the intervention. To test this hypothesis, the analyses were repeated using only the subgroup of IHC patients who began clinic treatment within 90 days of their diagnosis and their matched controls. We also used a Cox PH model to directly compare survival between the group of IHC patients who began clinic treatment within 90 days of diagnosis and the group of IHC patients who did not begin clinic treatment within 90 days of diagnosis.
All analyses were performed using R Statistical Software (v4.1.2; R Core Team). 45 P-values were considered significant at a level of .05. As this study is exploratory, no multiple testing adjustment was applied.
Results
There were 156 adult IHC patients diagnosed with stage IV colorectal cancer and treated with at least 6 cycles of mEHT during the study timeframe. Of these patients, 25 had missing values for at least one key variable and were removed from the analysis, leaving 131 IHC patients in the study sample. In the SEER database, 127 904 patients were diagnosed with stage IV colorectal cancer between 2000 and 2021 at age 18 or older with complete data on all required variables. From this pool of potential controls, 2 matches were selected for each IHC patient, giving a sample of 262 matched controls. After matching and the assignment of time zero, 105 controls died before their assigned time zero and were removed.
Sample Characteristics for the SEER and IHC Groups After Matching.

SMD for diagnosis year calculated as continuous.
Abbreviations: IHC, Integrated Health Clinic; IQR, interquartile range; SD, standard deviation; SEER, National Cancer Institute Surveillance, Epidemiology, and End Results; SMD, standardized mean difference.
IHC patients often had long delays between diagnosis and beginning integrative naturopathic treatment at the clinic, with a median delay of 264 days (IQR: 53-736); 63% of the sample began IHC treatment more than 90 days after diagnosis.
When measuring survival time from diagnosis, the restricted mean survival time (RMST) was 37.3 months [95% Confidence Interval (CI): 29.1, 45.5] for the SEER group and 57.2 months [95% CI: 49.6, 64.7] for the IHC group, while the median survival time was 16 months [95% CI: 13, 20] for SEER and 45 months [95% CI: 39, 57] for IHC (Supplemental Table 2). However, survival time measured from diagnosis suffers from immortal time bias due to the delay between diagnosis and the start of clinic treatment in the IHC group. Using the prescription time-distribution matching approach and measuring time in months from the beginning of clinic treatment, there were no significant differences between the SEER and IHC groups in RMST (SEER: 36.7 [95% CI: 30.0, 43.4]; IHC: 37.9 [95% CI: 31.6, 44.3]) or median survival time (SEER: 18 [95% CI: 15, 24]; IHC: 29 [95% CI: 19, 39]), as shown in Supplemental Table 2. The Kaplain-Meier curve in Figure 1 also shows no significant differences between the two groups (log-rank test P-value = .41). The Cox PH model gave a hazard ratio of .76 (.57, 1.01) comparing the IHC group to the SEER group (Table 2). This indicates a 24% lower hazard of mortality among the IHC patients compared to the SEER patients, but this difference is not statistically significant. Kaplain-Meier curve with time measured from the start of clinic treatment for IHC patients and from the start of clinic treatment of their matched IHC patient for SEER controls. Hazard Ratios From a Cox Proportional-Hazards Model, for the Full Sample and Subgroup Beginning Clinic Treatment Within 90 days of Diagnosis With matched Controls. Survival Time is Measured From the Start of Clinic Treatment for the IHC Group and From the Start of Clinic Treatment of Their matched IHC Patient for the SEER Controls. Full sample: N = 288, number of events = 222; Subgroup: N = 138, number of events = 102. Abbreviations: CI, confidence intervals; HR, Hazar Ratio; IHC, Integrated Health Clinic; Ref, reference level; SEER, National Cancer Institute Surveillance, Epidemiology, and End Results.

Time-Varying Hazard Ratios for Survival: Comparison of IHC and SEER Groups Using the Cox Proportional Hazards Model.
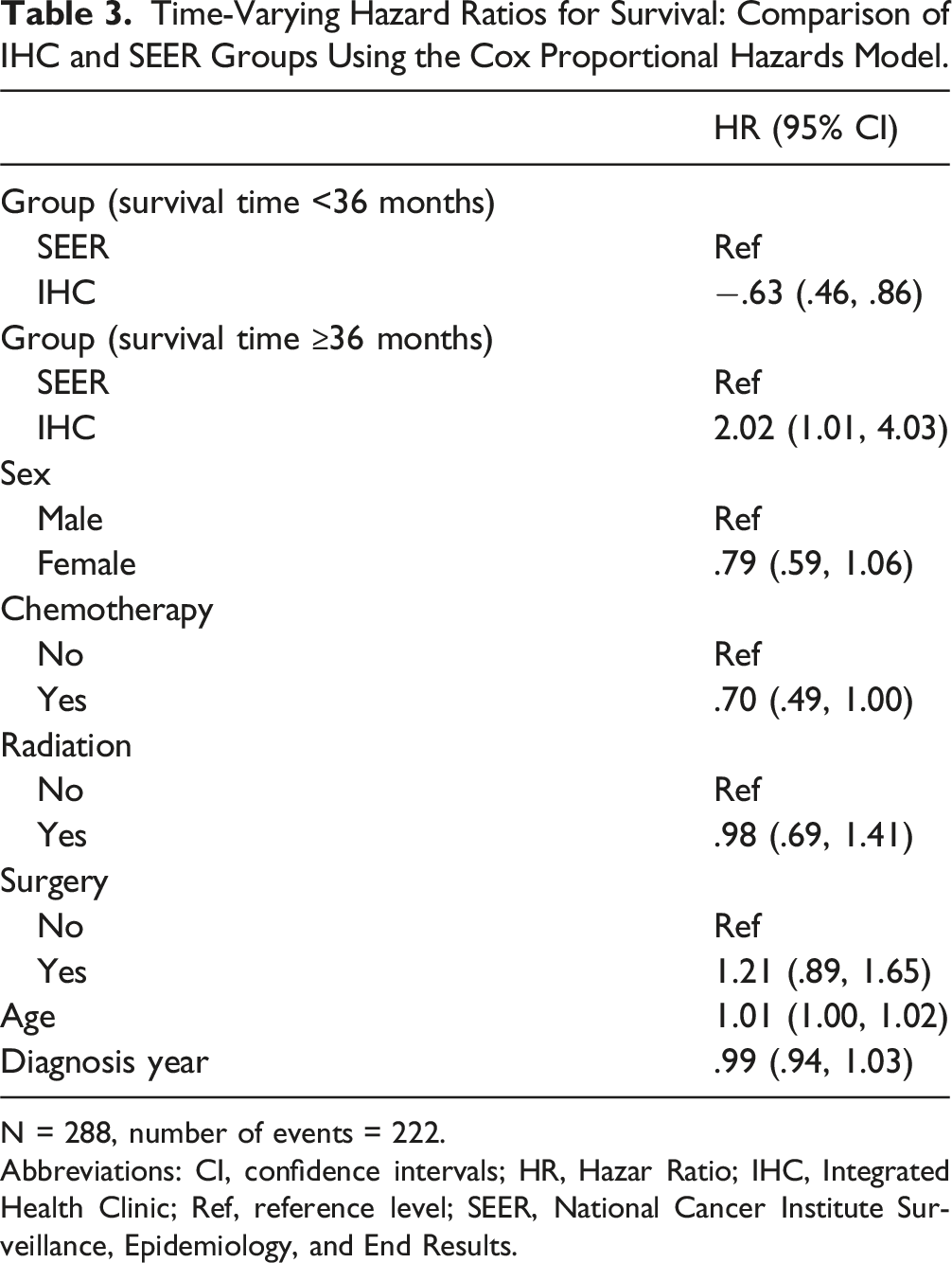
N = 288, number of events = 222.
Abbreviations: CI, confidence intervals; HR, Hazar Ratio; IHC, Integrated Health Clinic; Ref, reference level; SEER, National Cancer Institute Surveillance, Epidemiology, and End Results.
In a subgroup analysis comparing IHC patients beginning clinic treatment within 90 days and their matched controls, the Kaplain-Meier curve shows that the IHC patients have significantly higher survival probabilities (log-rank test P-value = .015; Figure 2). When a Cox PH model is applied to this sample, the hazard ratio is .45 (.28, .70), indicating 55% lower hazard of mortality among IHC patients beginning treatment within 90 days compared to their matched controls, which is statistically significant (Table 2). Kaplain-Meier curve with time measured from the start of clinic treatment for IHC patients and from the start of clinic treatment of their matched IHC patient for SEER controls, for the subset of IHC patients beginning clinic treatment within 90 days of diagnosis and their matched controls.
As the subgroup analysis of 131 patients revealed a survival benefit compared to controls when treatment began soon after diagnosis, we conducted a comparison of survival for IHC patients beginning clinic treatment within 90 days of diagnosis to IHC patients who began clinic treatment after 90 days. The hazard among patients beginning treatment after 90 days was 1.79 (95% CI: [1.09, 2.95]) times greater than among patients beginning treatment within 90 days (Supplemental Table 3).
Discussion
This retrospective study presents evidence that integrative naturopathic treatment can significantly improve survival outcomes for patients with CRC in the first 36 months post-treatment and when it started within 90 days of diagnosis. The median survival time from beginning clinic treatment of 29-months for patients treated at the IHC notably surpasses the 18-month median survival time in the SEER control group. This 11-month increase in median survival, although not statistically significant, is clinically relevant, suggesting that the integrative approach could be a valuable addition to the standard CRC treatment protocol. By introducing a time-varying coefficient into the Cox PH model, we observed a statistically significant lower hazard of mortality for IHC patients within the first 36 months of diagnosis; the hazard after 36 months increased to be greater in the IHC group than the SEER group. This suggests that some early deaths in the IHC group were delayed compared to the SEER group. In addition, the subgroup analysis revealed that patients who commenced the integrative naturopathic treatment model within 90 days of diagnosis had a 55% reduction in the hazard of death compared to the SEER group. This further underscores the potential benefits of early intervention.
We believe that the personalized nature of the integrative naturopathic treatment approach played a significant role in CRC patient outcomes. In our study, each patient’s treatment plan was tailored to their specific clinical context, incorporating a combination of mEHT with other integrative modalities, such as dietary adjustments, lifestyle changes, and mind-body practices. Among the therapies utilized, mEHT was the only consistent intervention across all patients. While this personalized treatment plan limits our ability to study the effects of a single treatment modality, it highlights the adaptability of the integrative model, which seeks to address the complex and unique aspects of each patient’s condition.
The results of our study align with the previous research indicating no significant change in overall survival following mEHT.32,34,35 Although no survival benefit was observed, one study highlighted other potential advantages of mEHT, including improvements in tumor regression and reduced toxicity, suggesting that mEHT may have therapeutic effects beyond overall survival. 32 Similarly, in the current study, we identified a time-dependent survival benefit. Specifically, patients who initiated integrative naturopathic treatment within 90 days of diagnosis exhibited a significant survival advantage compared to their matched controls, with a 55% reduction in the hazard of mortality. In a similar study, no time-dependent survival benefit was observed. 35 The limited sample size (n = 21) for CRC in that study may have reduced the ability to detect a significant effect in this subgroup analysis. 35
While research on the effect of mEHT on CRC is limited, it has been more widely studied in glioblastoma, lung, and pancreatic cancers.26-31 A recent meta-analysis found that adding mEHT to standard chemoradiotherapy significantly improved survival rates in glioblastoma patients, with 1-year survival rates of 42-61% for mEHT-treated patients. 26 Similar to our study, this meta-analysis reported time-dependent survival benefits, with the greatest benefits observed in newly diagnosed cases. 26 While this study focuses on glioblastoma, it suggests that mEHT may enhance treatment outcomes in other cancers, including CRC.
A few preclinical studies have demonstrated mEHT anti-cancer mechanisms in CRC through multiple pathways. mEHT has been shown to upregulate heat shock proteins and enhance damage-associated molecular pattern (DAMP) signaling, which promotes immune recognition of tumor cells. 46 Additionally, mEHT induces p53-mediated apoptosis and cell cycle arrest, which further supports the efficacy of chemotherapeutic agents in CRC cells. 47 Moreover, in vivo, mEHT contributes to both localized and systemic tumor destruction in CRC models, suggesting its potential for robust anti-tumor effects beyond the immediate treatment site. 48
Moreover, our results support the use of integrative oncology including mEHT for CRC patients, but it does not provide evidence on the difference between mEHT and other forms of hyperthermia. Previous research indicates that other forms of hyperthermia, when combined with radiotherapy and chemotherapy, can significantly enhance radiosensitivity and reduce tumor size in patients with metastatic CRC.49,50 Future studies can compare the difference between mEHT and other forms of loco-regional hyperthermia to improve health outcomes of this population.
In addition to mEHT, other integrative approaches have been investigated for their potential benefits in cancer treatment. Recent systematic reviews of complementary and integrative therapies, including acupuncture, yoga, herbal medicine, and mind-body interventions, highlighted their role in alleviating cancer-related symptoms and improving patients’ overall well-being.18,51-53 These findings are consistent with our study, which suggests that incorporating integrative naturopathic treatments can lead to enhanced patient outcomes.
The significant survival benefit identified by the subgroup analysis is supported by previous research that emphasizes the importance of early intervention in cancer treatment and metastatic CRC prognosis. 54 Although not specific to integrative naturopathic treatment, a delay of even four weeks in cancer treatment is associated with increased mortality for surgical, systemic, and radiotherapy treatments across seven types of cancer, including CRC. 54 This suggests the necessity of investigating the effect of treatment delays on survival outcomes for integrative naturopathic treatments. Moreover, patient-reported outcomes, such as quality of life measures, should be utilized in future studies to determine if the quality of life for integrative naturopathic treatment patients improves beyond their survival outcomes.
The discrepancy between the insignificant result in the whole group analysis and the significant improvement in survival in the subgroup analysis results may be explained by several factors. The heterogeneity within the whole group may dilute the effects of the integrative naturopathic treatment. Patients with different treatment histories, metastatic locations or timelines and diverse co-occurring health conditions may obscure the treatment’s efficacy. Alternatively, the discrepancy between the whole group and subgroup results may be due to the treatment only having an effect when applied early. Further research with larger samples is required to identify the effect of integrative naturopathic treatment on CRC patients seeking treatment at all times after diagnosis.
One of the strengths of this retrospective study is the use of a large control group from the SEER database, which enhances the validity of our findings. Moreover, to ensure robust data analysis in our study, we implemented advanced statistical methodologies. We used a technique called propensity score matching to select control patients from a large cancer database, ensuring they were similar to our treatment group on important potential confounders like age, sex, and treatment history. This method helps balance the groups and reduce the effect of measured confounders. We also carefully handled the survival time to avoid immortal time bias. We then compared survival times using established statistical methods to determine if our treatment had a significant impact. Our approach aims to provide reliable and unbiased results, demonstrating the effectiveness of our integrative naturopathic treatment model.
This study supports the effectiveness of an integrative naturopathic treatment model when implemented early following diagnosis. The use of a well-documented control group and advanced statistical methodologies strengthens the validity of these findings, aligning with the principles of evidence-based medicine. Integrative naturopathic treatment model’s emphasis on combining conventional and complementary treatments can enhance therapeutic efficacy and patient well-being when implemented soon after diagnosis, paving the way for more holistic, evidence-based and patient-centered cancer care.
In the context of the Canadian healthcare system, integrative naturopathic treatment presents unique challenges and opportunities. While Canada’s universal healthcare system provides comprehensive coverage for conventional cancer treatments, complementary and integrative therapies, including naturopathic treatments, are often not covered. This can create barriers for patients seeking integrative care, as they must bear the financial burden themselves. However, the potential benefits observed in our study highlight the need for policy changes to support the integration of these therapies into the mainstream healthcare system and health insurance programs.
Future research should focus on prospective, randomized controlled trials to further validate the effectiveness of integrative naturopathic treatment in metastatic CRC, and the necessity of early treatment. Investigating the biological mechanisms underlying the observed survival benefits and evaluating patient-reported outcomes (eg, health-related quality of life) will also be crucial in understanding the full impact of these therapies on patients’ overall wellbeing.
Conducting a cost-effectiveness analysis to determine if public funding by governments leads to cost savings represents a subsequent step in the research on naturopathic treatments. Expanding coverage to include integrative naturopathic treatments could enhance the overall quality of cancer care in Canada. By providing patients with access to a wider range of therapeutic options, the healthcare system can better address the physical, emotional, and psychological needs of cancer patients. The inclusion of mEHT and other integrative treatments could potentially reduce healthcare costs in the long term by improving patient outcomes and reducing the need for more intensive treatments. Therefore, advocating for policy changes and increased research funding is essential to facilitate the adoption of integrative oncology within Canada’s healthcare framework.
Limitations
There are several limitations to consider. The retrospective design and reliance on chart reviews may introduce selection and information biases. We attempted to address these issues by double-checking extracted data and conducting a robust group matching strategy. The personalized treatment plan and variation in the use of integrative treatments between patients prohibits the study of the effects of specific interventions on survival outcomes. Additionally, we were unable to account for the self-selection bias in our IHC cohort due to the requirement that IHC patients must finance their treatment and travel to attend treatment at the clinic; comparable data on socioeconomic status was not available for both groups, therefore, it could not be accounted for in the analysis. The financial ability to access naturopathic treatments may limit the generalizability of our findings to broader populations. While we can be confident that very few SEER controls received any form of hyperthermia, as no devices that provide mEHT are approved for use in the USA and the use of other hyperthermia devices was found to be limited,42,43 it is possible that some of the SEER controls received other naturopathic treatments. 44 If that is the case and the naturopathic treatment did not have a negative effect on the patient’s survival, then our estimates would be conservative estimates of the true effect obtained from comparing a group that received integrative naturopathic treatment to a group that did not receive any naturopathic treatment. Addressing these limitations with a prospective randomized clinical trial would provide more definitive evidence.
Conclusions
The observed survival benefit in the IHC group in the first 36 months post-treatment and among those who began treatment within 90 days of diagnosis, underscores the potential of integrating naturopathic treatments to complement conventional cancer therapies. This approach views the person as a whole, addressing not only the physical but also the emotional, psychological, and social aspects of a patient’s life. Incorporating mEHT and other integrative modalities into standard oncological care could provide a holistic approach to managing metastatic CRC, potentially improving patient outcomes and quality of life. The integration of these treatments into clinical practice requires a collaborative and timely effort among healthcare providers to ensure that patients receive comprehensive and personalized care.
In conclusion, our study contributes to the growing body of evidence supporting the use of integrative naturopathic treatment, including mEHT, in the management of metastatic CRC. If these findings are confirmed through larger, prospective studies, they could lead to significant changes in the management of CRC, offering more hope to patients who are battling this challenging disease.
Supplemental Material
Supplemental Material - Integrative Naturopathic Treatment Model for Colorectal Cancer: A Retrospective Study
Supplemental Material for Integrative Naturopathic Treatment Model for Colorectal Cancer: A Retrospective Study by Sara Izadi-Najafabadi, Lisa McQuarrie, Sarah Denotter, Mark Elderfield, and Gurdev Parmar in Global Advances in Integrative Medicine and Health.
Footnotes
Acknowledgments
We would like to express our heartfelt gratitude to our generous donors and the Oncology Association of Naturopathic Physicians (OncANP) for their invaluable financial support for this study. We also extend our deepest thanks to our patients and their families for their trust and participation in the integrated health clinic. Your contributions have been crucial to the success of this research.
Author Contributions
Conceptualization, G.P., M.E. and S.D.; methodology, G.P., S.I.N., and L.M.; formal analysis, L.M.; writing—original draft preparation, S.I.N.; writing—review and editing, G.P., S.I.N., S.D., L.M.; supervision, G.P.; project administration, M.E., S.D.; funding acquisition, S.I.N., M.E., G.P. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors wish to disclose that one of the authors, Dr. Gurdev Parmar, is a Co-founder and Medical Director of the Integrated Health Clinic from which the data for this study were extracted and analyzed. Although every effort has been made to ensure the integrity and objectivity of the research, the potential for conflicts of interest exists. The findings and conclusions presented in this study reflect the views of the Integrated Health Clinic and its affiliated staff. The donors/funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by donors and the Oncology Association of Naturopathic Physicians (OncANP).
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
