Abstract
Background:
Glioblastoma multiforme (GBM) is an aggressive brain tumor with limited treatment options and poor prognosis. Emerging evidence suggests that integrative oncology approaches may provide survival benefits when combined with conventional treatments. This study examines whether an integrative oncology treatment plan incorporating modulated electro-hyperthermia (mEHT) improves survival in GBM patients.
Methods:
This retrospective cohort study analyzed data from GBM patients treated at the Integrated Health Clinic (IHC) between 2010 and 2024. Survival outcomes were compared between IHC patients receiving adjuvant integrative naturopathic therapies and a matched control group from the National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) database. Kaplan–Meier survival estimates, and Cox proportional hazard models were conducted to assess survival differences. Secondary analyses evaluated the impact of treatment timing (≤120 days vs >120 days post-diagnosis) and age on survival.
Results:
The integrative treatment cohort demonstrated a lower hazard of mortality than the SEER group (HR = .72, 95% CI: .53-1.00, P-value = .05). The treatment benefit was greater among IHC patients who started treatment within 120 days of diagnosis (HR = .52, 95% CI: .33-.83, P-value = .006) and those under age 50 (HR = .51, 95% CI: .31-.85, P-value = .009).
Conclusions:
The findings suggest that an integrative naturopathic approach incorporating mEHT may improve survival outcomes in GBM patients. Patients initiating integrative treatment earlier experienced a greater survival benefit, as did patients under 50 years of age. Further studies, ideally prospective randomized controlled trials, are warranted to validate these findings.
Keywords
Introduction
Glioblastoma multiforme (GBM), or grade IV astrocytoma, is the most common, aggressive, and deadly primary brain cancer in adults. 1 Between 2015 and 2019, in the United States, it accounted for 14% of all central nervous system (CNS) tumors and 50% of all CNS malignant tumors. 2 The median age at diagnosis is 64, and it is 1.6 times more prevalent in men than women. 3 Since 2004, the Stupp protocol has been the standard treatment for newly diagnosed GBM, improving median survival from 12.1 under the previous treatment regime to 14.6 months.4,5 It includes maximal safe resection, radiotherapy, and temozolomide (TMZ) therapy. 4 Despite significant advancements in GBM’s molecular and pathological understanding over the past 2 decades, the standard of care remains unchanged.
GBM is a treatment-resistant malignancy with poor prognosis and limited survival, even with the current standard of care. 6 Given these challenges, there is growing interest in integrative oncology—an approach that combines conventional therapies with evidence-based complementary naturopathic strategies to enhance treatment efficacy, manage symptoms, and improve patient quality of life.7,8 Integrative oncology provides individualized treatment interventions tailored to each patient’s unique needs, utilizing evidence-based complementary therapies that are rigorously evaluated and appropriately integrated into standard of care. 9 Usually as complementary therapies gain stronger evidence, they are incorporated into integrative oncology.9,10
Some of the more promising complementary therapies for GBM include high-dose intravenous vitamin C, which was found to be safe and well-tolerated when combined with radiation and TMZ, potentially improving patient outcomes by enhancing the efficacy of standard treatments11 -14; ketogenic diet, which may impair the growth and survival of cancer cells by restricting glucose intake and has shown potential to enhance progression-free survival and overall survival in GBM patients, as well as increase the effectiveness of certain chemotherapy drugs15,16; acupuncture to alleviate treatment-related side effects improving quality of life; lifestyle counselling to optimize overall health and well-being; and modulated electro-hyperthermia (mEHT), a radiofrequency (RF; 13.56 MHz) capacitive hyperthermia treatment that delivers alternating electric fields to the tumour site.17 -20
mEHT applies a RF carrier-wave with amplitude modulation 21 and creates a strictly impedance-matched resonant circuit, where the patient is a part (an “electric component”) of the RF current loop. The capacitive part of the patient’s impedance is compensated for as much as possible. The frequency could be anywhere in the range of beta-delta dispersion (around 10 MHz) and for practical purposes 13.56 MHz is used, which is the allowed medical standard. The heterogenic complex impedance of the target guides the RF current for selective heating. 22 Energy absorption is concentrated on the tumor-cells and their microenvironment, with selective targeting of the membrane rafts of malignant cells. 23
mEHT employs amplitude-modulated electromagnetic fields to selectively target tumor cells, inducing cellular stress and apoptosis while sparing healthy tissue. 24 Its mechanism of action includes increasing tumor perfusion, reducing hypoxia, and impairing DNA repair pathways, which collectively enhance the efficacy of chemotherapy and radiation therapy. 24 When combined with systemic treatments such as chemotherapy or radiation therapy, mEHT has demonstrated synergistic effects by improving drug delivery to the tumor site, increasing drug uptake, and sensitizing tumor cells to radiation-induced damage. 25 Additionally, mEHT can stimulate immunogenic cell death, potentially amplifying the immune response against cancer.24,26 These combined effects make mEHT a promising adjunctive therapy in oncology.
While integrative oncology is increasingly used in cancer care, evidence on its synergistic effects, potential side effects, and optimal integration models remains limited. Only 11 studies have investigated the effects of mEHT in GBM patients, with varying study designs and sample sizes (See Supplementary Table 1).27 -37 Most studies included mixed high-grade gliomas rather than exclusively GBM, further complicating direct comparisons. Only 3 retrospective studies28,30,36 and 1 prospective study 27 included a control group, limiting the ability to draw definitive conclusions about the efficacy of mEHT. While findings suggest that mEHT is a well-tolerated non-invasive treatment with potential benefits in tumor response, survival, and quality of life, several limitations exist in the current evidence, including small and heterogeneous sample, poor study design, and variability in treatment protocols, all of which introduce potential bias.
Due to the individualized and dynamic nature of integrative oncology, standardizing treatment protocols for research purposes presents a significant challenge. 10 Retrospective observational studies serve as a valuable starting point for identifying patterns, generating hypotheses, and informing the design of future prospective trials that can more rigorously evaluate the effects of integrative oncology approaches on patient outcomes. 10 We recently published a similar retrospective study on colorectal cancer (CRC), which demonstrated promising results. The findings highlighted potential survival benefits associated with an integrative oncology approach including mEHT, 38 supporting the need for further investigation into the application of such a treatment model in other cancer types, including GBM.
This retrospective study evaluates an integrative oncology treatment model for GBM, assessing survival outcomes following the integration of complementary therapies, including mEHT, with conventional care. Specifically, the study seeks to determine whether integrative oncology treatment improves survival in GBM patients compared to standard care, and if the timing of treatment initiation and age modify the treatment effect.
Methods
Study Design
This retrospective chart review study was conducted at the Integrated Health Clinic (IHC) in British Columbia (BC), Canada, focusing on patients treated between 2010 and 2024. Patients who received integrative naturopathic care in conjunction with standard of care (referred to as integrative oncology treatment) were compared with controls derived from the National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) database. The SEER database is a publicly available, de-identified collection of cancer case records from the United States. 39
Ethics Statement
The study received approval from the Research Ethics Board (REB) at the Canadian College of Naturopathic Medicine (CCNMREB055.Parmar). Notices were posted at clinic locations to inform patients of the study, with an option to opt out of participation. Opting out did not affect patients’ treatment plans.
Participants
To be eligible for inclusion, patients in both groups needed to have an initial diagnosis of grade IV GBM at age 18 or older. For IHC patients, an additional requirement was that they had undergone at least 6 sessions of mEHT treatments between August 1, 2010, and August 31, 2024. Patients in both groups were excluded if essential data were missing, such as diagnosis date, cancer type, grade, standard care treatments, or date of death. IHC patients were followed until death, loss to follow-up, or the end of the data collection period on August 31, 2024. Since naturopathic medical care is not covered by British Columbia’s medical services plan, IHC patients had to self-finance their treatment and travel.
The version of the SEER database used for this study was the SEER 17 Nov 2023 submission, which includes diagnoses between 2000 and 2021. Before matching we reduced the SEER database to patients diagnosed between 2008 and 2021 because all IHC patients were diagnosed in 2008 or later. GBM patients were identified from the SEER database as records where the SEER Brain and CNS recode variable contained “1.1.2 Glioblastoma.” In the SEER database, cases diagnosed before 2017 were graded using a different coding system than cases diagnosed after 2017. For this study, for tumors diagnosed in 2017 or earlier, we filtered the grade to “Undifferentiated; anaplastic; Grade IV” and for tumors diagnosed after 2017, we filtered the pathological grade to “4.” Additional details on the SEER database, including the coding of grade, are available in the database documentation. 39 In cases where a single patient had multiple eligible diagnosis records only the earliest record was used. Follow-up in the SEER database ended on December 31, 2021.
It is assumed that SEER patients did not receive mEHT, as the treatment is not available in the U.S. Although several FDA-approved hyperthermia devices are used in the U.S. (eg, COOK VH8500 Hyperthermia Treatment System, MEDIFOCUS 100A, PYREXAR BSD 2000, LABTHERMICS Sonotherm 1000), none provide mEHT specifically. While data on the use of other hyperthermia forms is limited, these therapies are generally not widely accessible in the U.S.40,41 Data on treatment with complementary and alternative medicine were not available in the SEER version we accessed, therefore, we could not exclude patients who received any kind of naturopathic treatments from the SEER group. Previous research has shown that approximately 26.5% of SEER patients with health insurance reportedly accessed complementary and alternative medicine, including chiropractic care, acupuncture, massage therapy, and naturopathic treatments. 42 However, these complementary and alternative medicine treatments differ from the specialized integrative services offered at the IHC, specifically with regard to mEHT. We assume that the standard cancer treatments—Stupp protocol combining surgery, chemotherapy, and radiotherapy4,5—are comparable between Canada and the United States and that the survival of GBM patients receiving standard of care cancer treatments in Canada and the United States are similar. 43 Some of the patients included in the SEER control group will have received some form of integrative naturopathic treatment; assuming this treatment had a positive or no effect on survival, our estimate of the difference in survival between the IHC and SEER control group represents, at worst, a conservative estimate of the difference in survival between a group receiving integrative oncology treatment and a group receiving only standard care.
Intervention
IHC patients received an integrative oncology treatment model (combination of conventional treatments and complementary naturopathic therapies) tailored to individual needs. This model included mEHT, high-dose intravenous vitamin C, targeted supplementation, immune support, acupuncture, and dietary and lifestyle counselling.
The IHC patients in our study received mEHT treatments utilizing the Oncotherm EHY-2000+ device, which delivers modulated, capacitive radiofrequency (13.56 MHz) heating to target tumors with up to a 15 cm depth of penetration, maintaining a temperature range of 38.5°C to 42.5°C throughout the treatment session. The EHY-2000+ device is a Health Canada Class III Medical Device, is produced in the European Union, approved according to the European Medical Device Directive (CE-MDD), and produced in Good Manufacturing Process under ISO13485 standard, certified by TUV Product Service, Munich, Germany.
Patients lay on their side on the Oncotherm EHY-2000+ treatment bed. Their head rested on a memory foam pillow coated with a special metal fabric, which helped carry the treatment signal by acting as part of the electrode system. A second, movable electrode—about 20 cm wide—was placed gently over the area of the head being treated, based on the doctor’s instructions. Patients were given a call bell in case they needed anything during the session and were checked every 10 to 15 minutes to make sure they were comfortable.
The IHC brain protocol used on all GBM patients follows a structured step-up protocol to ensure optimal patient tolerance and safety (Table 1). Applied power and treatment duration were gradually increased over the initial sessions. Power increased in increments of 20 W, starting at 50 W in the first session, followed by 70 W in the second, 90 W in the third, and reaching 100 W by the fourth session. It was then further increased to 120 W in the fifth and stabilized at 140 W from the sixth session onward. Treatment duration followed a similar progression—30 minutes for the first session, 40 minutes for the second, 50 minutes for the third, and 60 minutes for all subsequent treatments. Amplitude modulation is turned on starting with the fourth treatment to further increase the efficiency of applied energy absorption. Modulation applies an amplitude modulated time-fractal signal to the 13.56 MHz carrier wave. This modulation increases the specific absorption rate (SAR) in the extracellular space, enhancing the radiofrequency wave properties for cancer cell selectivity and energy delivery. Treatment areas of 20 cm in diameter were used.
mEHT Treatment Progression: Applied Power, Duration, and Modulation Settings.

Abbreviation: W, Watts (Maximum achieved power).
All IHC patients also received conventional standard-of-care treatments prescribed by their neurosurgeons, medical and radiation oncologists. All patients in the IHC group received mEHT at some stage of their conventional treatment journey—some during radiation, others after radiation, some alongside TMZ, some after completing TMZ, and some following a recurrence of the disease. For patients undergoing radiotherapy with concurrent TMZ, mEHT sessions were administered 2 to 3 times weekly, timed before or after radiation sessions. For patients on the Stupp 5/23 cycles, mEHT was provided 2 to 3 times alongside TMZ, which was taken orally 30-minutes prior to mEHT as an adjunctive chemo-sensitizing treatment (Table 2). In cases where patients were no longer receiving conventional treatments, mEHT monotherapy was typically administered 2 to 3 times per week until a minimum of 1 week prior to the next scan(s). When mEHT was administered as a stand-alone maintenance therapy, in cases where the patient was clinically stable or had no convincing evidence of disease remaining, the most common protocol was to administer mEHT 1 to 2 times per week for a total of 12-treatments until a minimum of 1 week prior to the next scan(s), after which patient response to the protocol is assessed. All patients were prescribed dexamethasone (minimum 2 mg/day) and/or boswellic acid (minimum 2000 mg/day) to manage cerebral edema and risk of seizure. Each treatment was administered under the supervision of the prescribing physician and patients were closely monitored throughout.
Treatment Schedule Using Chemotherapy.

Data Collection
Data from IHC patient charts were meticulously reviewed, including laboratory, imaging, and pathology reports, and treatment notes from medical, radiation, and surgical oncologists. Dates of death were verified through family confirmation, obituary searches, and Medical Services Plan (BC public health insurance) records.
Variables of interest included date of death, age at diagnosis, sex, diagnosis year, grade at diagnosis, and receipt of surgery, radiation, or chemotherapy. A research assistant initially extracted the data, then the data were verified for accuracy by the study’s principal investigator.
Equivalent data for SEER patients were extracted from the database. Survival time was provided in units of months in the SEER database, calculated as the floor of the death date minus the date of diagnosis, divided by the average number of days in a month (365.24/12). The same calculation was applied to survival time for patients in the IHC group to convert from days to months.
Statistical Analysis
From the patients in the SEER database that met the eligibility criteria, matched controls were selected using optimal propensity score matching with a ratio of 6 controls to 1 IHC patient. Age, sex, year of diagnosis, and treatment with surgery, radiation, and chemotherapy were included in the propensity score model and an exact match was required on sex. Propensity score matching controls for confounding at baseline (pre-treatment) by generating a sample of controls with similar distributions of the covariates as the treated group. The quality of the matching is evaluated by the standardized mean difference (SMD) between the 2 groups, and an SMD of less than 0.1 is generally accepted to indicate negligible differences between the matched groups. 44 We must assume that there are no unmeasured confounders.
There was often a delay between diagnosis and the start of IHC treatment, representing a period of “immortal time” included in the survival time when measured from diagnosis for IHC patients that is not present for SEER patients. Survival time measured from diagnosis, therefore, suffers from immortal time bias. To address this, we use the prescription time-matching approach 45 and measure survival time from an index time. For IHC patients, the index time is the start of clinic treatment. For SEER patients, the index time is the start of clinic treatment for their matched IHC patient; the resulting survival time does not include the length of time between diagnosis and the start of clinic treatment of the matched IHC patient. SEER patients who died before their matched IHC patient began clinic treatment had a negative survival time and were excluded. IHC patients were censored if alive as of August 31, 2024, and SEER patients were censored if alive as of December 31, 2023.
The sample was characterized using frequency for categorical variables, mean and standard deviation (SD) for symmetric continuous variables, and median and interquartile range (IQR) for skewed continuous variables. Survival time was visualized using a Kaplan–Meier (KM) plot, illustrating the survival probability over time per group. Descriptive statistics for the outcome included restricted mean and median survival times, calculated by group. The hazard ratio (HR) comparing the IHC group to the SEER group was estimated with a Cox proportional-hazards (Cox PH) model. Age, sex, year of diagnosis, and treatment with surgery, radiation, and chemotherapy were included in the Cox PH model to adjust for any remaining between-group discrepancies in these variables.
In secondary analyses, we explored the effects of age and the timing of clinic treatment using stratification. The age strata were defined as <50 and ≥50 years of age and the treatment timing strata were defined as the initiation of clinic treatment before or after 120 days from diagnosis. SEER controls were assigned to the same strata as their matched IHC patient. The 120-day cutoff was selected to define an early treatment group based on the typical treatment timeline for GBM patients. After diagnosis, patients generally undergo standard-of-care treatment—including craniotomy surgery, radiation with TMZ—which typically spans 90 to 120 days from craniotomy to the completion of 6 weeks of radiation with TMZ. It is typically following this period that patients explore complementary naturopathic treatment options. Hazard ratios were calculated by estimating stratified Cox PH models.
Analyses were performed with R Statistical Software (v4.1.2; R Core Team). 46 A significance level of .05 was applied for P-values and no multiple testing adjustment was applied because this study is purely exploratory.
Results
There were 73 patients diagnosed with grade IV GBM who received at least 6 sessions of mEHT at the IHC between August 1, 2010 and August 31, 2024. Three patients were excluded because they were under age 18 at the time of diagnosis and 3 patients were excluded because they were missing information on standard of care treatments (surgery, radiation, and/or chemotherapy). The remaining 67 patients formed the IHC group.
In the SEER database, there were 60 615 diagnoses of GBM. After restricting to diagnoses between 2008 and 2021, there were 41 948 records remaining. We then excluded diagnoses that were not grade IV (n = 24 920) and records from patients diagnosed before age 18 (n = 175), missing survival time (n = 112), or missing standard of care treatment (n = 9). A further 4 cases were removed during deduplication (ie, the process of removing later records from patients with multiple eligible diagnoses). The remaining 16 278 unique patients formed the pool of eligible controls for matching and 402 controls were matched. Of the matched controls, 159 died during the length of time between diagnosis and the start of clinic treatment of their matched IHC patient and were removed. The final sample included 243 matched controls.
The IHC group had a median age of 52 years and more males (59.7%) than females (40.3%). The diagnoses were spread evenly over the 14-year study time period, with a median year of diagnosis of 2017 (Table 3). IHC patients were likely to receive standard of care treatment in the form of surgery (86.6%), chemotherapy (76.1%), and/or radiation (73.1%). The control group was similar to the IHC group in age (SMD = .045) and sex (.017), with a slight between-group imbalance remaining in the year of diagnosis (SMD = −.108). In the control group, 79.8% of patients were treated with surgery (SMD = .181), 84.8% were treated with radiation (SMD = .288), and 88.1% were treated with chemotherapy (SMD = .315). Achieving ideal balance on the standard of care treatment variables was not possible without discarding patients from the IHC group, which would have made the IHC group less representative of all IHC GBM patients. We included all covariates in the Cox PH models to adjust for the remaining group imbalances. No cases of seizure or other side effects were observed during or immediately following mEHT treatment and other integrative therapies in IHC patient cohort.
Sample Characteristics of SEER and IHC Groups After Matching.

Abbreviation: IHC, integrated health clinic group; IQR, interquartile range; SEER, control group from the surveillance, epidemiology, and end results database; SMD, standardized mean difference.
The median number of days from diagnosis to starting clinic treatment for IHC patients was 124 days (IQR = 41-392). Survival time from diagnosis (in months) was a median of 24 (95% CI: 18-29) months in the IHC group and 15 (95% CI: 13-17) months in the control group. This result is provided for completeness only and should not be used to compare groups due to the issue of immortal time bias discussed previously. Instead, we measured survival time from the index time and found a median of 15 (95% CI: 11-21) months in the IHC group and 11 (95% CI: 10-13) in the SEER group (Table 4). The restricted mean survival time in the IHC and SEER groups was 27.5 (95% CI: 18.9-36.2) and 19.3 (95% CI: 15.7-22.9) months, respectively, and the log-rank test P-value comparing the 2 groups was .049. Figure 1 illustrates the probability of survival over time, measuring time from the index time in months, and shows a higher probability of survival in the IHC group at all times, although the confidence intervals from the 2 groups overlap.
Survival Time for the SEER and IHC Groups.

Abbreviation: IHC, integrated health clinic group; RMST, restricted mean survival time; SEER, control group from the surveillance, epidemiology, and end results database.
The log-rank test evaluates if there is a difference between groups in the probability of death at any time point.
Survival time measured from diagnosis suffers from immortal time bias in the IHC group due to the delay between diagnosis and start of clinic treatment. Survival time from clinic treatment does not suffer from the same bias. In the SEER group, controls were assigned the time from diagnosis to the start of clinic treatment of their matched IHC patient.
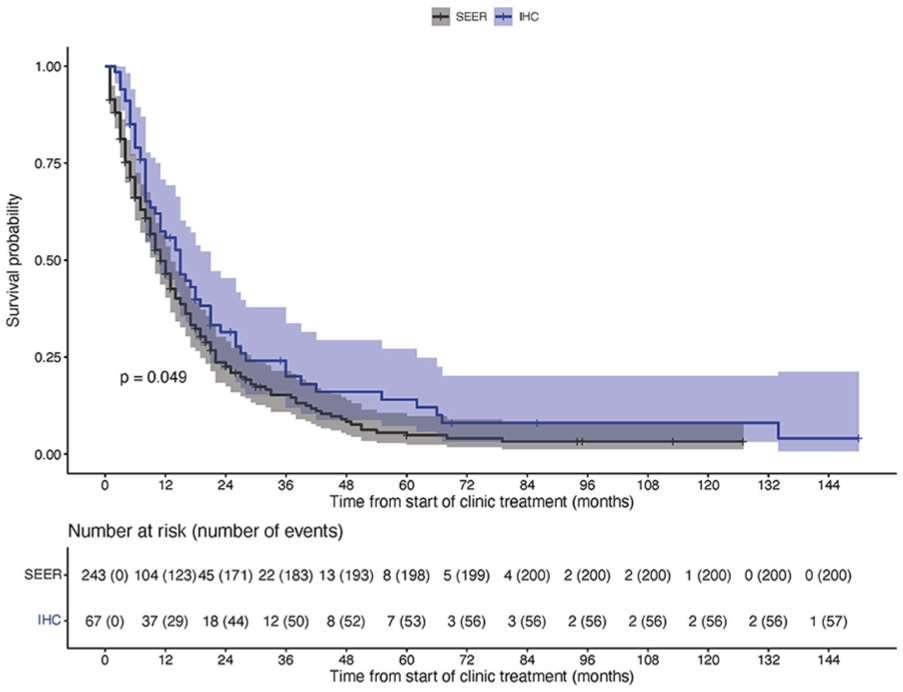
Kaplan–Meier curve with time measured from the start of clinic treatment for IHC patients and from the start of clinic treatment of their matched IHC patient for SEER controls. The blue and grey shaded regions represent the 95% confidence intervals for the IHC and SEER groups, respectively.
Table 5 presents HRs comparing survival in the IHC group to the SEER control group, obtained by estimating a covariate-adjusted Cox PH model (See Supplementary Table 2 for complete model results). In the full sample of IHC and SEER patients, the estimated HR was 0.72 (95% CI: 0.53-1.00, P-value = .05), suggesting a 28% reduction in the hazard of death for IHC patients compared to SEER patients; however, this result was not statistically significant. In the secondary stratified analysis evaluating the timing of treatment, patients starting clinic treatment within 120 days from diagnosis had an HR of .52 relative to their matched SEER controls (95% CI: .33-.83, P-value = .006), indicating a statistically significant 48% reduction in the hazard of death. In contrast, among patients who initiated treatment after 120 days, no meaningful difference in the hazard of death was observed between the 2 groups (HR = .98, 95% CI: .61-1.56, P-value = .92). Patients who were less than age 50 at diagnosis benefitted more from the clinic treatment than patients aged 50 or older (HR relative to SEER controls in <50 strata: .51, 95% CI: .31-.85, P-value = .009; HR relative to SEER controls in ≥50 strata: .96, 95% CI: .61-1.53, P-value = .87).
Hazard Ratios Comparing Survival in the IHC Group to the SEER Control Group, Calculated from a Cox Proportional-Hazards Model.

Abbreviations: HR, hazard ratio; CI, confidence interval; IHC, integrated health clinic group; SEER, control group from the surveillance, epidemiology, and end results database.
Discussion
This retrospective study evaluated the survival outcomes of GBM patients who received integrative oncology treatment including mEHT at the IHC compared to matched controls from the SEER database. Our findings suggest that the integration of mEHT and complementary naturopathic therapies with the standard of care may offer a survival benefit for GBM patients, particularly when treatment is initiated within 120 days of diagnosis. When survival was measured from the initiation of clinic treatment, IHC patients exhibited a median overall survival of 15 months, compared to 11 months in the control group, with an HR of .72, reflecting a 28% reduction in the risk of death. While promising, these results did not achieve statistical significance.
Previous studies have suggested that integrative oncology may enhance treatment efficacy in glioblastoma.11 -16,27 -37 In our study, patients receiving early integrative naturopathic treatment including mEHT experienced a survival benefit, suggesting a potential synergistic effect when combined with standard therapy. These findings are consistent with prior studies, which demonstrated a significant survival advantage in GBM patients receiving mEHT in combination with chemotherapy. 36 Similarly, Fiorentini and colleagues reported improved tumor response, quality of life and survival with mEHT compared to chemotherapy alone, reinforcing the potential role of integrative therapies. 30
mEHT utilizes amplitude-modulated electromagnetic fields to selectively target tumor cells, inducing cellular stress and apoptosis while preserving healthy tissue. 24 When combined with chemotherapy or radiation therapy, mEHT improves drug delivery, increases cellular uptake, and enhances tumor sensitivity to radiation. 25 These synergic effects highlight mEHT’s potential as an adjunctive oncologic therapy.
A well-established and already FDA-approved non-invasive therapy for GBM, tumor treating fields (TTF), 47 shares key mechanistic similarities with mEHT. Both therapies are non-invasive and use electromagnetic fields to selectively target tumor cells inhibiting their growth and inducing apoptosis. They aim to enhance the effects of standard treatments while sparing healthy tissue.18,48 mEHT employs high-frequency electromagnetic fields to selectively target tumor cells, 24 while TTF utilizes low-intensity intermediate-frequency alternating electric fields to disrupt cell division and inhibit tumor growth, leading to apoptosis.49,50 TTF has gained widespread recognition, and when combined with standard chemotherapy, significantly improves progression-free and overall survival.48,51 Given the mechanistic overlap between TTF and mEHT, as well as TTF’s established clinical efficacy, it is reasonable to consider mEHT as a promising therapeutic approach. A recent meta-analysis comparing mEHT and TTF in GBM found similar survival outcomes, with 1-year survival rates of 60.63% for mEHT and 63.56% for TTF. 18 This suggests that mEHT could offer benefits comparable to TTF and should be further explored as a viable adjunctive therapy in GBM management.
Notably, patients who began integrative treatments within 120 days of diagnosis experienced a statistically significant 48% reduction in the hazard of death (HR = .52) compared to controls. In contrast, those who initiated treatment after 120 days did not show a meaningful survival benefit (HR = .98). These results align with recent advancements in GBM treatment, where novel therapies such as mEHT have shown promise in improving survival18,19,30,32 and underscores the potential importance of early integration of complementary and naturopathic therapies into standard of care in enhancing patient outcomes. 37
Early intervention is essential for the effectiveness of all treatment modalities for GBM. Early intervention, including diagnosis and surgery within specific timeframes, significantly improves survival outcomes, with patients diagnosed within 2 weeks of symptom onset and who underwent surgery within 3 weeks living longer than those who received treatment at later stages. 52 While the benefits of early intervention in conventional therapies are well-established, research on integrative oncology treatments is limited and often low in quality. A recent meta-analysis demonstrated that combining mEHT with standard chemoradiotherapy significantly improved survival in glioblastoma patients, with 1-year survival rates ranging from 42% to 61%. 18 Aligning with our findings, the meta-analysis highlighted a time-dependent survival benefit, with the most substantial gains seen in newly diagnosed patients. 18
Further stratification of our patient cohort by age revealed that patients diagnosed before age 50 experienced a significantly greater survival benefit from integrative oncology treatment compared to those aged 50 or older. This finding suggests that younger GBM patients may be more responsive to complementary and integrative therapies, potentially due to differences in tumor biology, immune response, or treatment tolerability. Prior studies have similarly noted that age is a key prognostic factor in GBM, with younger patients exhibiting better overall survival and a more favorable response to novel therapeutic approaches.53 -55 Similarly, 2 studies on mEHT reported age-related differences to treatment response in GBM patients.33,36 However, Fiorentini and colleagues did not find a statistically significant age-dependant response to mEHT treatment in GBM patients; the authors did not state the size of each age strata in their results and did not present an effect size for the between-strata differences in survival, so low power may explain the statistically insignificant result. 30 These findings highlight the importance of personalized and age-adapted treatment strategies in GBM, as younger patients may derive greater benefit from integrative approaches.
Beyond survival benefits, multiple studies have emphasized the safety and tolerability of mEHT.28,33 -36 Our findings align with these studies, as patients tolerated integrative therapies well, with no unexpected adverse effects. This is particularly important for recurrent GBM patients who may have limited treatment options due to toxicity concerns.
Our results underscore the value of integrative oncology as a holistic approach to cancer care that extends beyond conventional treatments. Integrative oncology aims to combine conventional treatments with evidence-based complementary therapies to improve patient outcomes and quality of life. In our study, the integrative approach included a minimum of 6 sessions of mEHT within a comprehensive naturopathic oncology treatment approach. While our findings suggest a potential survival benefit associated with the early initiation of integrative therapies, the retrospective nature of our study and its limitations prevent us from drawing definitive conclusions about causality.
One key consideration in integrative oncology is the disparity in access to such treatments. Studies have shown that uninsured GBM patients or those with lower socioeconomic status often have reduced survival rates and are less likely to receive standard treatments,56 -58 let alone complementary and naturopathic therapies. Even when financial factors were controlled for by providing care free of charge, disparities in access to integrative oncology treatments are still observed, influenced by factors such as ethno-nationality and immigration status, gender, and age. 59 This disparity highlights the need for broader discussions on integrating evidence-based complementary and naturopathic therapies into standard oncology care to ensure equitable access.
Limitations
Our study is subject to several limitations, including its retrospective design, potential selection biases, and the inherent challenges of standardizing integrative oncology protocols. Patients pursuing integrative treatments often need to finance their care and travel to specialized clinics, and comparable socioeconomic data were not available for both groups in our study. Some patients used mEHT throughout their disease course receiving more treatments, while others used it for a specific time during treatment, and not throughout their care. These differences in dosing and timing may have also had an impact on individual disease progression and thus survival outcomes. Furthermore, while integrative therapies such as mEHT are not widely available in certain regions, it is possible that some control patients received other complementary treatments, which could lead to conservative estimates of the true effect of integrative approaches.
In addition, although survival differences were observed, a causal relationship cannot be established without a randomized controlled trial. The retrospective design limits our ability to fully control for confounding factors, and residual bias cannot be excluded despite the matched design and multivariate survival analysis. Although we attempted to balance the IHC and SEER cohorts, only a limited number of matching variables were available (age, sex, year of diagnosis, and receipt of surgery, radiation, or chemotherapy). Other clinically meaningful factors—such as tumor molecular profile, extent of surgical resection, performance status, comorbidities, or socioeconomic determinants of health—were not available. Thus, residual confounding cannot be excluded, and our analyses should be interpreted as exploratory and hypothesis-generating rather than definitive. Importantly, quality-of-life outcomes—central to evaluating the value of integrative approaches—were not systematically collected in this study and could not be assessed.
Future Direction
Despite all the limitations, the observed association between early initiation of integrative therapies and improved survival outcomes warrants further investigation. Our study serves as a preliminary step toward understanding the role of integrative oncology in GBM treatment. Given the individualized nature of integrative therapies, designing structured randomized controlled trials poses challenges. Future research should focus on prospective studies that not only evaluate survival outcomes but also assess patient-centered metrics such as quality of life and functional status over time. This comprehensive approach will provide a more holistic understanding of the potential benefits and limitations of integrative oncology in GBM care.
Conclusions
Our findings suggest that incorporating integrative naturopathic therapies with standard care may offer survival benefits for GBM patients, particularly when initiated early in the treatment course. These results contribute to the growing body of literature exploring holistic approaches to cancer care and underscore the potential of integrative oncology in improving outcomes for patients with aggressive malignancies like GBM.
Supplemental Material
sj-docx-1-ict-10.1177_15347354251378069 – Supplemental material for The Effect of Integrative Naturopathic Oncology Including Modulated Electrohyperthermia on Survival Outcome among Glioblastoma Multiforme Patients: A Retrospective Study
Supplemental material, sj-docx-1-ict-10.1177_15347354251378069 for The Effect of Integrative Naturopathic Oncology Including Modulated Electrohyperthermia on Survival Outcome among Glioblastoma Multiforme Patients: A Retrospective Study by Sara Izadi-Najafabadi, Lisa McQuarrie and Gurdev Parmar in Integrative Cancer Therapies
Supplemental Material
sj-docx-2-ict-10.1177_15347354251378069 – Supplemental material for The Effect of Integrative Naturopathic Oncology Including Modulated Electrohyperthermia on Survival Outcome among Glioblastoma Multiforme Patients: A Retrospective Study
Supplemental material, sj-docx-2-ict-10.1177_15347354251378069 for The Effect of Integrative Naturopathic Oncology Including Modulated Electrohyperthermia on Survival Outcome among Glioblastoma Multiforme Patients: A Retrospective Study by Sara Izadi-Najafabadi, Lisa McQuarrie and Gurdev Parmar in Integrative Cancer Therapies
Footnotes
Acknowledgements
We would like to express our heartfelt gratitude to our generous donors and the Oncology Association of Naturopathic Physicians (OncANP) for their invaluable financial support for this study. We also extend our deepest thanks to our patients and their families for their trust and participation in the integrated health clinic. Your contributions have been crucial to the success of this research. We would also like to thank Mark Elderfield, and Drs. Sarah Denotter, and Adeleke Fowokan for their support with study administration and for providing valuable feedback throughout the process. Your assistance and insights have been deeply appreciated.
Ethical Considerations
The study received approval from the Research Ethics Board (REB) at the Canadian College of Naturopathic Medicine (CCNMREB055.Parmar).
Consent to Participate
Due to the retrospective design of this study and the secondary use of data, individual informed consent was not obtained. Instead, informational notices were posted at clinic locations, allowing patients the opportunity to opt out. Declining participation had no impact on their treatment plans.
Consent for Publication
Not applicable.
Authors Contribution
Conceptualization, GP; Methodology, GP, SI-N, and LM; Formal analysis, LM; Writing—original draft preparation, SI-N; Writing—review and editing, GP, SI-N, and LM; Supervision, GP; Project administration, GP; Funding acquisition, GP. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by donors and the Oncology Association of Naturopathic Physicians (OncANP).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors wish to disclose that one of the authors. Gurdev Parmar, is a Co-founder and Medical Director of the Integrated Health Clinic from which the data for this study were extracted and analyzed. Although every effort has been made to ensure the integrity and objectivity of the research, the potential for conflicts of interest exists. The findings and conclusions presented in this study reflect the views of the Integrated Health Clinic and its affiliated staff. The donors/funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Data Availability Statement
The IHC patients data are not publicly available to protect patient privacy and confidentiality. However, de-identified data may be made available upon reasonable request to the corresponding author, subject to institutional and ethical approvals.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
