Abstract
Introduction
Bevacizumab is a monoclonal antibody, which, in association with combination chemotherapy regimens, has been shown to be active in metastatic colorectal cancer. Other biologic agents active in the same setting are cetuximab and panitumumab, both of which are monoclonal antibodies directed against the antiepidermal growth factor receptor. The objective of this study was to compare treatment costs of first-line regimens for metastatic colorectal cancer in Italy.
Methods
A set of first-line regimens was considered, according to the Italian Association of Medical Oncology and the European Society for Medical Oncology guidelines. A targeted review of the literature was undertaken to identify clinical study references for treatment regimens. The total cost of a regimen was calculated in the perspective of the Italian healthcare system summing up drugs, administration, and adverse event costs, based on year 2016 prices and tariffs.
Results
Bevacizumab 7.5 mg + capecitabine was the least expensive regimen, with a total cost of €16,754 per patient. When we consider regimens based on FOLFOX, bevacizumab 5 mg + FOLFOX4 was the least expensive (€32,709 per patient), compared to panitumumab + FOLFOX4 (€42,815), cetuximab + FOLFOX4 (€42,725), and cetuximab + FOLFOX (€37,995). If we consider combination regimens based on FOLFIRI, the association of FOLFIRI and bevacizumab was less expensive than regimens that included cetuximab (€28,389 for bevacizumab 5 mg + FOLFIRI and €35,310 for cetuximab + FOLFIRI).
Conclusions
From the perspective of the Italian health care system, bevacizumab appears to be a convenient option among the first-line regimens for metastatic colorectal cancer. Further study, based on real-world evidence, would be necessary to confirm this result.
Introduction
Colorectal cancer (CRC) is the third most common tumor and the fourth most frequent cause of death worldwide (1). It develops through abnormal proliferation of the intestinal mucosa cells as a result of interaction between environmental and genetic factors. The main risk factors are those related to the lifestyle, such as unhealthy diet, obesity, tobacco and alcohol use, and to physical inactivity, ageing, inflammatory bowel diseases, and inherited genetic disorders (2). The signs and symptoms of CRC may include blood in the stool, a change in bowel movements, weight loss, and fatigue (2).
The disease is characterized by chromosomal instability that causes changes of oncogenes and tumor suppressor genes (APC, TP53, KRAS, BRAF, PTEN, SRC, TGF-b, SMAD 2, and 4), as well as thymosin b-4, which participate in the neoplastic transformation. All RAS mutations are present in about 55% of colorectal carcinomas, according the Italian Association of Medical Oncology (AIOM) (3). The same society recommends the analysis of all RAS genes in patients with metastatic CRC (mCRC) (3).
In 2012, 694,000 CRC deaths were estimated worldwide (4). In 2015, the Italian Association of Cancer Registries estimated 20,609 CRC deaths (11,850 men; 8,758 women), and 60,916 new cases of CRC yearly, with a rate of 99 new cases per 100,000 in men and 81 per 100,000 in women (4, 5).
Up to 20% of patients present with mCRC (6), of which a minority are eligible for resection. In unresectable patients, the major aim of the treatment is to stop tumor progression and to prolong the overall survival (OS), while controlling for symptoms and maintaining quality-of-life.
Several new drugs and combination regimens have been recently introduced in clinical practice for the treatment of unresectable mCRC, determining a substantial increase in life expectancy of advanced CRC patients. However, the availability of several biological agents associated with several multidrug chemotherapy schedules has complicated decision-making processes, making it difficult to choose the best regimen. Several cytotoxic agents, such as 5-fluorouracil (5-FU), capecitabine, oxaliplatin, and irinotecan are currently available for the treatment of advanced CRC. Although they can be used as single agents, they are mainly incorporated into combination regimens, such as folinic acid/5-FU/oxaliplatin (FOLFOX), folinic acid/5-FU/irinotecan (FOLFIRI), and capecitabine/oxaliplatin (XELOX). Biologic drugs directed against vascular endothelial growth factor (VEGF; i.e., bevacizumab) and drugs based on epidermal growth factor receptor (EGFR; i.e., cetuximab and panitumumab) have been approved in combination with chemotherapy and have demonstrated their efficacy in improving the outcomes for advanced CRC patients.
Bevacizumab is a monoclonal antibody that recognizes and binds VEGF and is registered for the treatment of mCRC patients regardless of all RAS mutation status. When incorporated into oxaliplatin and/or irinotecan-based regimens, bevacizumab induced statistically significant improvement in OS and progression-free survival (7, 8).
The choice of a biologic treatment in mCRC patients is dictated, in the first instance, by the clinical judgment of what is most suitable for the specific condition of the patient. However, the high prices of chemotherapeutics and biologics risk a challenge to the sustainability of the system. As a consequence, the oncologists must today add the cost dimension to the efficacy and safety considerations of their decision-making process. Few economic analyses are available in the literature that have assessed the economics of biologic treatments in mCRC. Some of these discussed the applicability of a foreign cost-effectiveness analysis (USA-based) to the Italian setting (9–11). Another study analyzed the cost impact of a sequence of two biologics (bevacizumab followed by cetuximab in second-line treatment) in the perspective of the Veneto region (12). The objective of this study was to help inform this decision-making process in Italy by estimating and comparing treatment costs of a range of first-line regimens that include anti-VEGF and anti-EGFR biologics in mCRC patients.
Methods
The study population was defined by patients affected by unresectable mCRC with any genotypic profile, according to AIOM guidelines (3).
We considered first-line regimens that incorporated biologic drugs in association with the most common combinations of chemotherapeutics, such as FOLFOX, FOLFIRI, and XELOX, according to the AIOM and the European Society of Medical Oncology (ESMO) guidelines (13), and the Summary of Product characteristics (SPCs).
Multidrug regimens containing capecitabine and irinotecan (CAPIRI) was also included in this analysis, even though this combination regimen should be used with specific attention to side effects (3). Capecitabine plus bevacizumab was also taken into consideration as it represents a possible option for patients with contraindications to the doublet chemotherapy.
For each drug regimen included in the analysis, a clinical study reference was identified (Tab. I) (7, 8, 14–22). In the case of multiple references, we considered the most recent publication or the phase (III vs. II) of the clinical trial, with the exception of studies with direct comparison (head-to-head) between the biologics (23, 24), which were always included.
Chemotherapy regimens included in the cost analysis
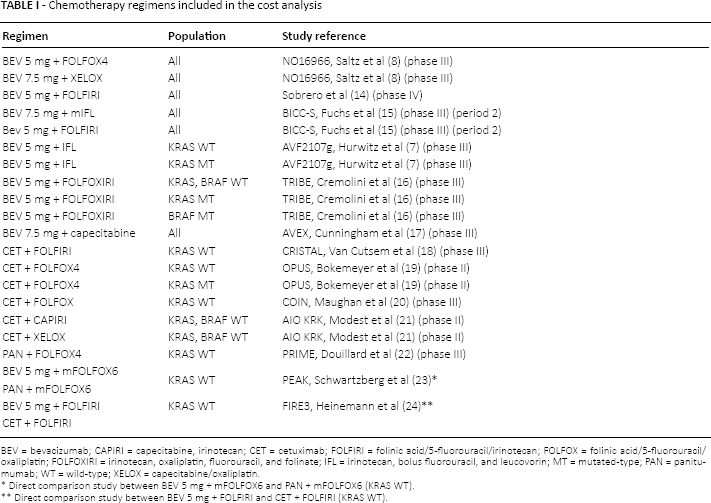
BEV = bevacizumab; CAPIRI = capecitabine, irinotecan; CET = cetuximab; FOLFIRI = folinic acid/5-fluorouracil/irinotecan; FOLFOX = folinic acid/5-fluorouracil/oxaliplatin; FOLFOXIRI = irinotecan, oxaliplatin, fluorouracil, and folinate; IFL = irinotecan, bolus fluorouracil, and leucovorin; MT = mutated-type; PAN = panitumumab; WT = wild-type; XELOX = capecitabine/oxaliplatin.
Direct comparison study between BEV 5 mg + mFOLFOX6 and PAN + mFOLFOX6 (KRAS WT).
Direct comparison study between BEV 5 mg + FOLFIRI and CET + FOLFIRI (KRAS WT).
The analysis was conducted using an analytical model developed in MS Excel (Microsoft). We defined the study outcome as the total average cost per patient treated. This was estimated in the perspective of the Italian health care system. Data concerning the duration of treatment exposure were available in a limited number of cases. Therefore, we estimated the average treatment cost assuming fixed treatment duration for all regimens. This duration was assumed equal to 6.1 months, based on the opinion of a panel of clinical experts reported in a previously published economic evaluation (25).
The total cost per patient was calculated as the sum of the costs of drugs, administration, and the management of adverse events. The costs of that drugs we considered were reported from publicly available sources (Tab. II) (26) and do not take into consideration confidential discounts negotiated with AIFA (Agenzia Italiana del Farmaco).
Unit costs for drugs and administration
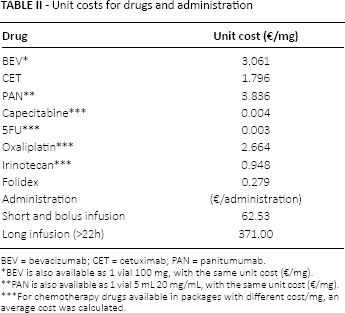
BEV = bevacizumab; CET = cetuximab; PAN = panitumumab.
BEV is also available as 1 vial 100 mg, with the same unit cost (€/mg).
PAN is also available as 1 vial 5 mL 20 mg/mL, with the same unit cost (€/mg).
For chemotherapy drugs available in packages with different cost/mg, an average cost was calculated.
For chemotherapy, the generic drug prices were considered (27). An average price was calculated for those products that have a different cost per mg among the various packages available. The average dose was estimated considering an individual of 70 kg and 169 cm, yielding to a body surface area of 1.81 m² (28). For each regimen, the administration costs were calculated according to the schedule reported in the corresponding reference study. We accounted for a single cost for multiple co-administrations when the different chemotherapies had a corresponding cycle length. The unit cost of an intravenous administration for short and bolus infusion of €62.53 was derived from a previous economic study, inflated to December 2015 Euros (29). The unit cost of administration for long infusions (>22 hours) was estimated in €371.00 based on the corresponding national DRG tariff (DRG 410) (30). An initial one-off cost was calculated to account for the treatment and the administration costs with regimens requiring an initial escalation or charge dose.
The cost evaluation of adverse events was based on in-patient and day hospital DRG tariffs (30), and outpatient care tariffs (31). The incidence of adverse events was derived from the clinical reference studies. Only grade 3 and 4 adverse events were included, regardless of their incidence.
Results
The total costs of treatment for each first-line regimen, including bevacizumab and other biologic drugs, are summarized in Table III.
Total average cost per patient and cost breakdown for first-line mCRC treatment regimens given a fixed treatment duration of 6.1 months
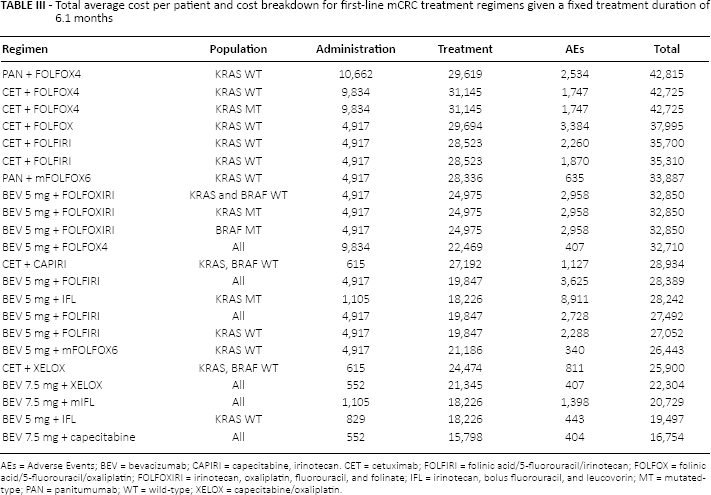
AEs = Adverse Events; BEV = bevacizumab; CAPIRI = capecitabine, irinotecan. CET = cetuximab; FOLFIRI = folinic acid/5-fluorouracil/irinotecan; FOLFOX = folinic acid/5-fluorouracil/oxaliplatin; FOLFOXIRI = irinotecan, oxaliplatin, fluorouracil, and folinate; IFL = irinotecan, bolus fluorouracil, and leucovorin; MT = mutated-type; PAN = panitumumab; WT = wild-type; XELOX = capecitabine/oxaliplatin.
Panitumumab + FOLFOX4 regimen was the most expensive regimen, with a total cost of €42,815 (of which €29,619 was due for the acquisition of the drug) per patient treated. When we compare the regimens incorporating a biologic with FOLFOX, bevacizumab 5 mg + FOLFOX4 was less expensive (€32,710; of which €22,469 was the acquisition cost) compared to panitumumab + FOLFOX4 (€42,815), cetuximab + FOLFOX4 (€42,725; acquisition cost: €31,145), and cetuximab + FOLFOX (€37,995; acquisition cost: €29,694).
The comparison between the regimens based on the combination of biologics + FOLFIRI showed that bevacizumab was less expensive than cetuximab. The total cost was estimated in €28,389 (of which €19,847 was the acquisition cost) for bevacizumab 5 mg + FOLFIRI and in €35,310 (acquisition cost: €28,523) for cetuximab + FOLFIRI. Considering the head-to-head study by Heinemann and colleagues (24), the costs for the same regimens were estimated at €27,052 (of which €19,847 was the acquisition cost) and €35,700 (acquisition cost: €28,523) for bevacizumab 5 mg + FOLFIRI and cetuximab + FOLFIRI, respectively.
Cetuximab + CAPIRI had a total cost of €28,934 (of which €27,192 was the acquisition cost), and cetuximab + XELOX of €25,900 (acquisition cost: €24,474). Bevacizumab 7.5 mg + capecitabine was the less expensive among all the considered regimens with a total cost of €16,754 (acquisition cost: €15,798).
Discussion
In this study, a range of first-line drug regimens for mCRC based on bevacizumab, cetuximab, and panitumumab were compared in terms of the total cost of treatment evaluated in the perspective of the Italian health care system. Bevacizumab 7.5 mg + capecitabine resulted the less expensive regimen with a total cost estimated at €16,754.49 per patient for a conventionally assumed treatment duration of 6.1 months. On the other hand, panitumumab + FOLFOX4 resulted the most expensive (€42,815.05). Bevacizumab, in association with FOLFOX and FOLFIRI, was less costly than cetuximab and panitumumab. These results were in line with the ancillary substudy concerning the cost-analysis of a phase III trial (CALGB/SWOG 80405) in which patients with all RAS WT mCRC received FOLFIRI or mFOLFOX6 (medical doctor/patient choice at enrolment) and were randomized to either cetuximab or bevacizumab: in fact, according to USA 2014 costs, the analysis showed that the cost for chemo + bevacizumab was $66,075; whereas it was $105,339 for chemo + cetuximab (32). The drug regimens that included capecitabine (i.e., cetuximab + CAPIRI, cetuximab + XELOX, bevacizumab 7.5 mg + XELOX, bevacizumab 7.5 mg + capecitabine) had a total cost lower than the other drug regimens. These results may be a consequence of lower adverse events and/or administration costs – the latter being linked to the oral administration of capecitabine, and they were in line with a previous study in which the costs of the regimens with capecitabine were lower than those with 5-FU infusion in first-line CRC treatments (33). The study was conducted considering the use of capecitabine and 5-FU as monotherapy or in combination with other chemotherapy agents. The costs were estimated at €6,841 per patient treated with 5-FU-monotherapy versus €2,056 with capecitabine only, and €12,620 with 5-FU in combination versus €9,745 with capecitabine in combination.
Some limitations can be ascribed to our study; the most relevant is the indirect estimate of the treatment costs. The drug regimens considered in this analysis were derived from the literature, and the corresponding costs were not estimated in a real clinical setting. Moreover, the patient populations were heterogeneous. The unit costs of health care resources were derived from the literature and from the DRG and outpatient tariffs, as a proxy of real clinical practice costs, which were not available. Another consideration should be taken on the variability of the DRG tariff among Italian regions. For instance, DRG 410, which was used to estimate the administration cost of long infusions, has a national tariff of €371, while its value could be as low as €150 in Sicily. Therefore, national tariffs could be seen as an average among all regions, but the degree of local variability, affecting the actual computation of costs, is high.
Another limit was the fact that the duration of the treatment was not available in all reference studies. For this reason, we decided to calculate the costs assuming a fixed treatment duration of 6.1 months for all regimens. This approach was already used in a previous economic evaluation, and was validated by clinical experts (25). A possible alternative would have been to consider, for each regimen, progression-free survival as a proxy of the time on treatment. It is well known that in oncology, progression-free survival provides a poor approximation of the duration of the treatment, because patients could stop the treatment before progression or prolong it despite progression, then the cost calculation based on progression-free survival would have introduced an additional imbalance in the study. For these reasons, we preferred to assume a conventional fixed duration of treatment, in order to compare the costs of the regimens on the same basis. A similar treatment duration (about 6.0 months) was considered appropriate in other published studies (34–36).
An important consideration is linked to the unit costs of the drugs used in the analysis, which were based on publicly available information and which did not correspond to the real prices actually paid by the Italian health care system. These real prices are, in fact, not publicly available as the actual discount negotiated is kept confidential.
In conclusion, from the cost perspective of the Italian health care system, bevacizumab appears to be a convenient option because the schemes containing this drug were generally less expensive than those based on other biologic first-line treatments for mCRC. Further studies, based on real-world evidence, are necessary to confirm these results.
Footnotes
Financial support: The study was supported by Roche SpA.
Conflict of interest: SI, CD, SL received compensation or salary from SIHS SLR related to the development of this project. RB receive consulting fees from SIHS related to this project. The authors have no other conflicts of interest to disclose not related to this project.
