Abstract
Background
Allergic rhinitis (AR) is a common inflammatory disease of the upper respiratory tract mainly triggered by allergens such as dust mites, pollen, spores, and viral or bacterial infections. AR is primarily associated with symptoms such as nasal itching, sneezing, rhinorrhea, nasal congestion, and watery, itchy, or red eyes. AR significantly affects an individual’s quality of life. Probiotics have been proven effective in the clinical management of AR through immunomodulation. However, studies on the use of Alkalihalobacillus clausii to alleviate the symptoms of AR have rarely been reported.
Objective
This study aimed to explore the clinical efficacy, safety, and possible underlying mechanism of Alkalihalobacillus clausii 088AE in alleviating the associated symptoms of acute AR in patients.
Methods
A prospective, interventional, randomized, double-blinded, placebo-controlled, monocentric clinical study was conducted on patients with acute AR (N = 40) randomized into two groups, test (N = 20) and placebo (N = 20). Patients in the test arm received a probiotic strain, A. clausii 088AE, whereas patients in the placebo arm received Maltodextrin. The primary endpoints (efficacy) were total 4 nasal symptoms scores (T4NSS), total 2 ocular symptoms scores (T2OSS), cough scores, and immunological parameters (T-helper 1 (Th1), Th2, Th17, and T-regulatory (Treg) cells, Interleukin (IL)-4, IL10, IL17, IL22, interferon-gamma (IFN-γ), tumor necrosis factor-alpha (TNF-α) and -beta (TNF-β), and forkhead box P3 (FOXP3)) evaluated from baseline to end of treatment (EOT). The secondary endpoints (safety) assessed were vital physical signs, hematology, and biochemical parameters, along with an assessment of adverse or serious adverse events (AEs and SAEs).
Results
A. clausii 088AE supplementation significantly reduced the T4NSS (rhinorrhoea, nasal stuffiness, nasal itching, and sneezing; P < 0.001), T2OSS (itching and watery eyes; P < 0.001), and cough scores (P < 0.01) by the EOT compared to baseline. The placebo group reported a significant increase in all the above symptom scores at the EOT from their baseline values (P < 0.001). The intergroup analysis between A. clausii 088AE and placebo indicated a significant change in T4NSS, T2OSS, and cough score (P < 0.001). Further, the immunological parameters were improved (non-significant, P-value ≥ 0.05) with the probiotic supplementation. No adverse events (AEs) or serious adverse events (SAEs) leading to termination of study participation were reported with the use of A. clausii 088AE in the study. No clinically significant vital signs and physical examinations were reported as AEs or SAEs by the investigator.
Conclusion
A. clausii 088AE supplementation improved the clinical symptoms in patients with AR. At the administered oral dose, A. clausii 088AE was found to be safe and tolerable in adult subjects with acute allergic rhinitis. A. clausii 088AE can be recommended to support the clinical pathophysiology of AR-related symptoms of the host. Besides, clinical studies with a larger population, multiple centres, and prolonged intervention periods are necessary to validate the significance of this study further.
Introduction
Allergic rhinitis (AR) (also known as Hay fever) is an inflammation of the nasal airways triggered by exposure to allergens that stimulate immunoglobulin E (IgE)-mediated inflammatory responses in the nasal mucosa.1,2 AR is generally characterized by symptoms including rhinorrhea, nasal congestion, itching, sneezing, and in some cases, rhinoconjunctivitis, such as watery, itchy, or red eyes. 3 AR can be intermittent or persistent based on the course of the symptoms, while mild, moderate, or severe based on the grade of clinical manifestations. 4 Allergic rhinitis affects more than 400 million people globally, with incidence rates ranging from 10% to 30% among adults and over 40% among children. 5 It can potentially lead to physical, psychological, and social consequences, affecting the overall quality of life of patients. Further, AR affects sleep and work performance and may cause depression and anxiety.3,5 Thus, it is imperative to explore effective treatment strategies to avoid the prevalence and alleviate symptoms of AR.
The best approach to managing AR is avoiding corresponding allergens, such as dust mites, animal dander, molds, and pollen.4,5 However, this approach is rarely achievable and may require a complete change in lifestyle. Unfortunately, complete resolution of AR symptoms is typically very difficult to achieve. 2 Consequently, several effective allergy medications are used to treat AR, including antihistamines, decongestants, and intranasal corticosteroids. Despite their effectiveness in managing AR in the short term, many associated symptoms, including nasal congestion, remain unaffected. 6 Besides, the use of these drugs may cause several side effects, such as dry mouth, drowsiness, dizziness, and safety concerns. Further, these drugs do not modify the immune background which causes allergic inflammation and may pose safety concerns with prolonged treatments.4,7 All these issues surface the need for alternative and safe treatment for managing AR symptoms.
One such treatment that has drawn the favorable attention of researchers to modulate the immune responses in AR patients is the use of probiotics. Probiotics are viable microorganisms that benefit the health of the host by improving the intestinal microbial balance. 7 Probiotics can stimulate the immune system at all mucosal surfaces, primarily preventing atopic diseases. Probiotic intake is thought to promote healing of the enteric mucosa by reducing gut permeability, enhancing local intestinal immune responses (particularly Immunoglobulin-A (IgA) synthesis), and restoring a balanced gut microbiota.4,7,8 Interestingly, the gut microbiota can be linked with the respiratory tract (“gut-lung axis”). The alterations in the composition, biodiversity, or activity and function of gut microbiota, known as dysbiosis, can affect the immunity and microbiota of the lung and vice versa. 4 Reflecting on the fact that changes in the gut microbiota are one of the most important indicators of allergic diseases, 3 probiotics possess excellent potential in treating AR. Further, it has been reported that the intestinal microflora plays an important role in regulation of T-helper 1/T-helper 2 (Th1/Th2) balance, the possible key mechanism involved in allergic diseases. 9 Probiotics are known to suppress the Th2-related cytokines, Interleukin (IL)-4, IL-5, and IL-13, and stimulate immunity by promoting T-helper 1 (Th1) and T-regulatory 1 (Treg-1) cells.8,9 However, the results of many studies differ, and the role of imbalance in Th1/Th2 responses in the development of allergic diseases is still incomprehensible. Therefore, the precise mechanism of probiotics in treating allergic rhinitis remains ambiguous. 9 Notably, Th2 polarization characterized by high levels of IL-4 and low levels of interferon-gamma (IFN-γ) have been reported in allergic children, indicating that probiotics have important therapeutic advances in treating allergies, specifically allergic rhinitis. 8
Evidently, the most common probiotics studied for allergic rhinitis are the Lactobacillus and Bifidobacterium groups. A few studies found a protective effect of probiotics on AR10,11 and several others reported no association between them.12,13 Lactobacillus plantarum strains inhibited the allergic cytokine 14 and increased the percentage of Th1 cells significantly in adults with seasonal allergic symptoms. 15 Lin et al 16 reported Lacticaseibacillus paracasei GM-080 to improve allergic rhinitis symptoms in children. Furthermore, Bifidobacterium lactis NCC2818, 6 Lactobacillus paracasei ST11, 17 and Lactobacillus gasseri A5 18 reduced IL-5, IL-13, and tumor necrosis factor (TNF)-α generation from peripheral blood mononuclear cells (PBMCs) of AR patients, but did not alter serum total IgE, serum allergen-specific IgE, or IL-10 production.
On the other hand, Alkalihalobacillus clausii (formerly known as Bacillus clausii) was found to modulate cytokine patterns at the nasal level in allergic children with recurrent respiratory infections. 8 Particularly, A. clausii restored physiological Th1 polarization and induced T-regulatory cell response, as documented by increased levels of IL-10 and tumor growth factor (TGF)-β after treatment. 8 Further, A. clausii has been reported as a safe and frequently prescribed probiotic that modulates immune response and prevents gastroenteric and respiratory infections in children. 19 It is currently available as an over-the-counter (OTC) medical product and used as a complementary treatment. 20 However, very few studies investigated the effect of A. clausii on respiratory infections and established the therapeutic efficacy against ailments related to allergic rhinitis.8,21,22 Be that as it may, these studies have either evaluated the symptom scores or monitored the immunological markers and not considered both these parameters to give a broader spectrum of the possible mechanism of A. clausii in the treatment of AR.
Thus, considering the lack of in-vivo data on A. clausii 088AE in treating AR, the present study comprises a prospective, randomized, double-blind, placebo-controlled, monocentric, interventional trial evaluating the effect of Alkalihalobacillus clausii 088AE on participants suffering from acute AR. The total 4 nasal symptom score (T4NSS), total 2 ocular symptom score (T2OSS), cough scores, and immunological markers were evaluated as primary endpoints, whereas physical, hematological, and biochemical parameters as secondary endpoints. To the best of our knowledge, this is the first study to determine the efficacy of Alkalihalobacillus clausii 088AE against AR by evaluating both the relevant symptom scores covering a wide range of symptoms of AR and the corresponding immunological parameters.
Materials and Methods
Investigational Product
The investigation product comprised probiotic Alkalihalobacillus clausii 088AE (containing 2 billion CFU of probiotic/g). The placebo product consisted of Maltodextrin. Both the probiotic and placebo products with required compliant quality standard were provided Advanced Enzyme Technologies Ltd, Thane, India. The packaging, labeling, and physical appearance of both these products were identical, except for their unique batch numbers for distinguishing the products.
Informed and Ethical Consent
The trial was registered under the “Clinical Trial Registry of India” as per regulations with the given registration number CTRI/2020/09/027657 dated September 08, 2020 [https://ctri.nic.in/Clinicaltrials/pmaindet2.php?EncHid=NDcwNzg=&Enc=&userName=CTRI/2020/09/027657], prior to the enrolment of any patients for the clinical study. The study protocol, all its amendments, and the patient information sheet were reviewed and approved by the institutional ethics committee (Vagus Institutional Ethics Committee; EC approval reference number: ECR/1181/Inst/KA, 2019). The trial was conducted as per the “Declaration of Helsinki 2013” (64th World Medical Association General Assembly, Fortaleza, Brazil, October 2013), 23 ethical principle based on the recent revision and ICH harmonized guidelines integrated addendum to ICH E6 (R1): Guidelines for Good Clinical Practice ICH E6 (R2), New Drugs and Clinical Trial Rules (2019) 24 and following the “National Ethical Guidelines for Biomedical and Health Research involving Human Subjects” issued by the Indian Council of Medical Research (2017) 25 and all other applicable laws and regulations of the country. The written informed consent was obtained from all the participants enrolled in the study. The CONSORT checklist of information of all the details and events in the current clinical trial are presented in Table S1 (supplementaty file).
Subject Selection for the Study
A prospective, randomized, double-blind, placebo-controlled, monocentric, interventional trial was conducted on 40 participants (aged 18 to 65 years) suffering from acute allergic rhinitis (AR). The site of the study was Vagus Super Speciality Hospital, malleshwaram, Bangalore, 560003, India. The trial involved a total of 5 scheduled visits to the clinical site by the participants. These participants were selected based on the following defined inclusion and exclusion criteria.
Inclusion Criteria
The male and female subjects enrolled in the study were included based on the following criteria: (i) Subjects willing and able to provide written informed consent before any investigation-related activities, and adhering to all applicable guidelines; (ii) Subjects of either sex, aged between 18 to 65 years (both inclusive), with AR; (iii) At the time of the enrolment the subjects had an investigator assessed: (a) Total Four Nasal Symptom Score (T4NSS)* of 4 to 10 (both inclusive) with minimum nasal stiffness score of 2 [*T4NSS included assessment of (1) rhinorrhea, (2) nasal stuffiness, (3) nasal itching, and (4) sneezing using a 4-point scale for each (0 = no symptoms, 1 = mild, 2 = moderate, and 3 = severe)]; (b) Total Two Ocular Symptom Score (T2OSS)* of 1 to 5 (both inclusive) [*T2OSS included assessment of (1) eye itching and (2) watery eyes or tearing using a 4-point scale for each (0 = no symptoms, 1 = mild, 2 = moderate, 3 = severe)]; and (c) Cough score of 0 to 1 in a 4-point scale based on its severity.
Exclusion Criteria
The enrolled subjects were excluded from the study, who were (i) receiving antihistamines with a history of hypersensitivity to antihistamines or any of the excipients of study formulations, (ii) suffering from nasal structural abnormalities (including deviated nasal septum, nasal polyps, etc.), (iii) using nasal sprays or with nasal, ocular, sinus surgery in past 6 months, and (iv) having a history of disorders of nasolacrimal drainage; rhinitis medicamentosa (defined as a condition characterized by nasal congestion without rhinorrhoea or sneezing that is triggered using topical nasal decongestants for more than 4 to 6 days). Further, subjects were excluded from the study who (i) received immunotherapy for at least 6 months prior to enrolment in this study, (ii) received intranasal/systemic corticosteroids in the past 4 weeks or cromolyn sodium in the last 2 weeks, or were having any antibiotic therapy, and (iii) would require concomitant chronic or intermittent use of oral, intravenous, intramuscular, topical, or inhaled corticosteroids.
Subjects with significant disease(s) or disorder(s) other than seasonal allergic rhinitis were also excluded from the study that, in the opinion of the investigator, may (a) put the subject at risk because of participation in the study, (b) interfere with the study evaluations, and/or, (c) cause concern regarding subject’s ability to participate in the study; Additionally, subjects with a history of substance abuse were removed from the trial. Women who were pregnant or breastfeeding or of child-bearing potential not using medically acceptable methods of contraception or with a positive serum pregnancy test at screening were also excluded from the study. Nonetheless, subjects–(i) unable or unwilling to comply with the study procedures, (ii) participated in another investigational study within the last 3 months prior to entry into this study, (iii) likely to travel outside the study area for a substantial portion of the study period, (iv) had self-reported chronic diseases like diabetes, kidney diseases, cardiovascular disease, and (v) who were on treatment with any kind of central nervous system (CNS) disease (self-reported) were not considered for the study.
Study Design, Randomization, and Supplementation
Forty patients, including male and female, were randomized in a 1:1 ratio to either the test arm (N = 20) or the control arm (N = 20) using the SAS random number generation method. The test arm received 1 sachet of 1 g of A. clausii 088AE (2 billion CFU/g) powder, and the control arm received a placebo containing maltodextrin (1 g per sachet) to consume orally. Either of the treatments was received by the patients twice a day orally (morning and evening) for 4 weeks. The investigation product and placebo were packed in a similar packaging material with the appropriate batch numbers placed on it. The subjects were asked to return the unused investigation products and placebo by the end of the trial. The subject randomization, treatment allocation, and study protocol are represented in Figure 1. The investigator enrolled the participants and assigned the intervention based on the randomization list. Supportive medications and treatments, if needed, were recommended and provided to the patients by the concerned physician/investigator. The participants, the investigators, and the study team were blinded to the treatment allocation. The study protocol was strictly followed, and no changes or amendments were made to the approved study protocol once the trial commenced. In addition, no interim analysis was performed during the study period. Study flow chart for the clinical study on evaluating the efficacy and safety of Alkalihalobacillus clausii 088AE in alleviating the symptoms of acute allergic rhinitis.
Endpoints: Efficacy and Safety of Variables
Primary and secondary endpoints evaluated different efficacy and safety variables associated with the use of A. clausii 088AE for treating AR. The total interventional period was 4 weeks and this analysed the efficacy of the investigational product, whereas the final safety follow-up on 42nd day assessed the safety and tolerability in terms of AEs and SAEs of the investigational product. The primary - T4NSS [*T4NSS included assessment of (1) rhinorrhea, (2) nasal stuffiness, (3) nasal itching, and (4) sneezing using a 4-point scale for each (0 = no symptoms, 1 = mild, 2 = moderate, and 3 = severe)], T2OSS [*T2OSS included assessment of (1) eye itching and (2) watery eyes or tearing using a 4-point scale for each (0 = no symptoms, 1 = mild, 2 = moderate, 3 = severe)], cough scores [0 to 1 in a 4-point scale based on its severity], immunological markers, and secondary–physical, hematological, and biochemical parameters were investigated. The endpoint results of the test arm were compared with the corresponding results of the placebo (control) arm.
Primary Endpoints
Primary endpoints evaluated in this study for efficacy assessment were changes in the symptom scores–Total Four Nasal Symptom Score (T4NSS), Total Two Ocular Symptom Score (T2OSS), and cough scores in patients from clinical visits 1 to 5 with respect to baseline. Further, the study also evaluated the effect of probiotic supplements on immunological parameters, including T helper (Th) cells–Th1, Th2, and Th17, as well as T-regulatory (Treg) cells (Determined by Flow cytometer, BD FACS - Fluorescence-activated cell sorting). Correspondingly, the changes in cytokines such as interleukin (IL)–IL4, IL10, IL17, and IL22, and interferon-gamma (IFN-γ), tumor necrosis factor-alpha (TNF-α) and -beta (TNF-β), and Forkhead box P3 (FOXP3) [Determined by ELISA Method on an ELISA plate reader (Spectramax I3X plate reader, Molecular Devices). The details of analytical kits are provided in Table S2 of Supplemental file] were monitored from visits 1 to 5 with respect to baseline values in all subjects.
Secondary Endpoints
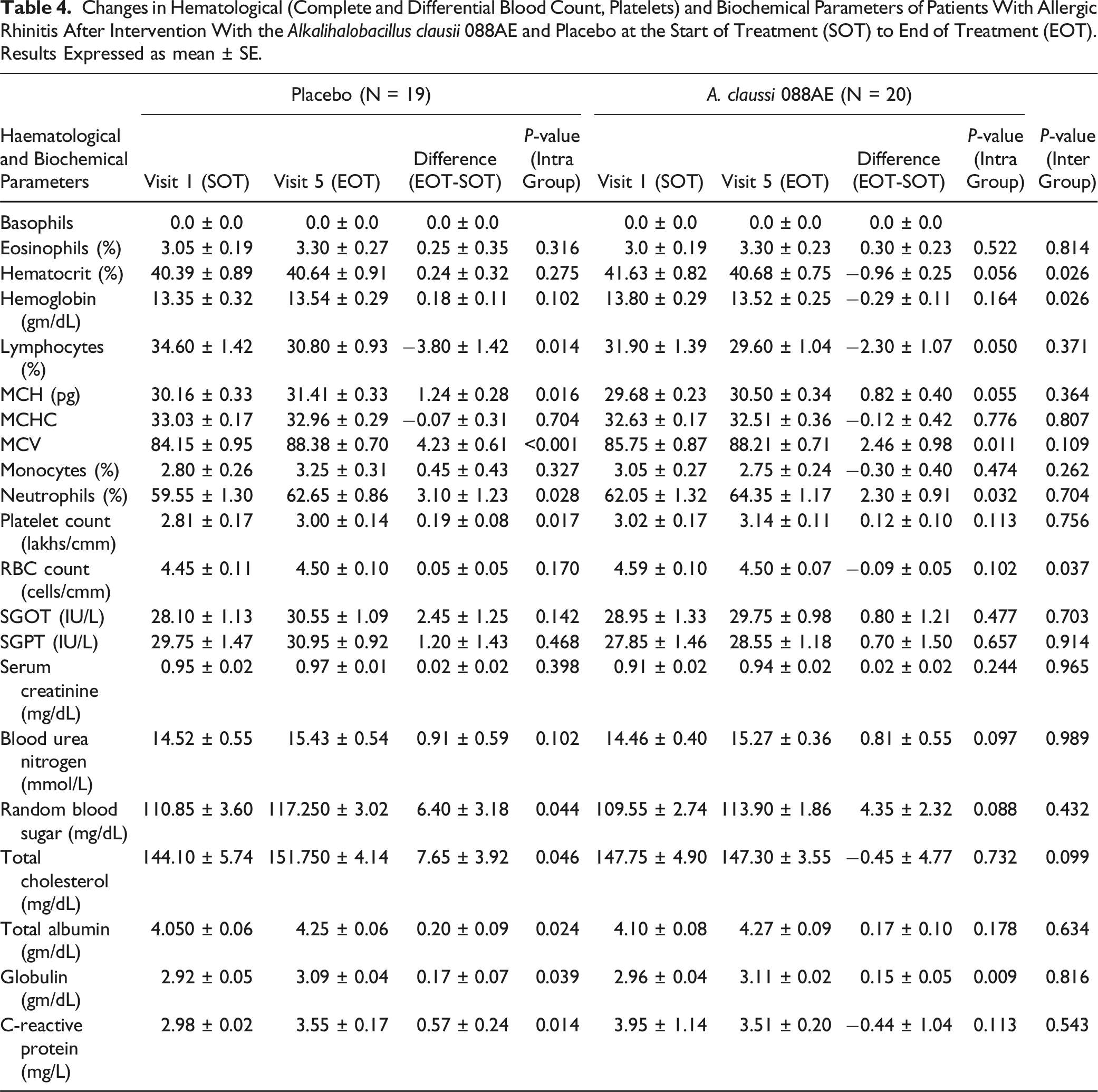
Secondary endpoints evaluated in this study for safety assessment were changes in physical parameters, including body temperature, systolic blood pressure, diastolic blood pressure, pulse rate, and respiration rate at each visit. Additionally, changes in hematological parameters–complete and differential blood count and platelets, biochemical parameters– Serum Glutamic-Oxaloacetic Transaminase (SGOT), Serum Glutamic Pyruvic Transaminase (SGPT), serum creatinine, blood urea nitrogen (BUN), random blood sugar (RBS), total cholesterol, total albumin, and globulin, and C-reactive protein (CRP). The hematological and biochemical tests were conducted on visits 1, 3, and 5 for all 40 subjects. Further, the rate of incidence of adverse events (AE) or serious adverse events (SAEs) was also monitored during the study.
Sample Power and Statistical Analysis
The study recruited a total of 48 (40 + 08) subjects (incorporating 20% of patients to compensate dropouts and/or withdrawals). The distribution of subjects was based on computer generated randomization in 1:1 ratio. Each treatment arm had at least 20 subjects. Data was analyzed with a 5% significance level (confidence interval 95%) and maintained a minimum power of 80% for study using SAS software, version 9.1. All the data sets were analyzed using Microsoft Excel 2016, and the findings of all the experiments are presented as mean ± standard error. The statistical analysis followed the statistical analysis plan (SAP) and was performed using R CRAN software, version 4.0.3. Multiple unpaired t-tests were performed for all the parameters studied to compare the difference between control and test arms at the start of treatment (SOT) and end of treatment (EOT). The P-value ≤0.05 was considered statistically significant unless signified.
Results
Demographic Characteristics of Subjects in the Placebo and Alkalihalobacillus clausii 088 AE Arms That Participated in the 4-Week Intervention Study.
Assessment of AR Symptoms
The efficacy of the A. clausii supplementation on AR symptom alleviation was evaluated by monitoring the changes in T4NSS, T2OSS, and cough scores in patients from clinical visits 1 to 5 with respect to baseline. The T4NSS included assessment of rhinorrhea, nasal stuffiness, nasal itching, and sneezing using a 4-point scale for each (0 = no symptoms, 1 = mild, 2 = moderate, and 3 = severe). The T2OSS included an assessment of eye itching and watery eyes or tearing using a 4-point scale for each (0 = no symptoms, 1 = mild, 2 = moderate, 3 = severe). The cough scores were also evaluated as 0 to 1 on a 4-point scale based on its severity.
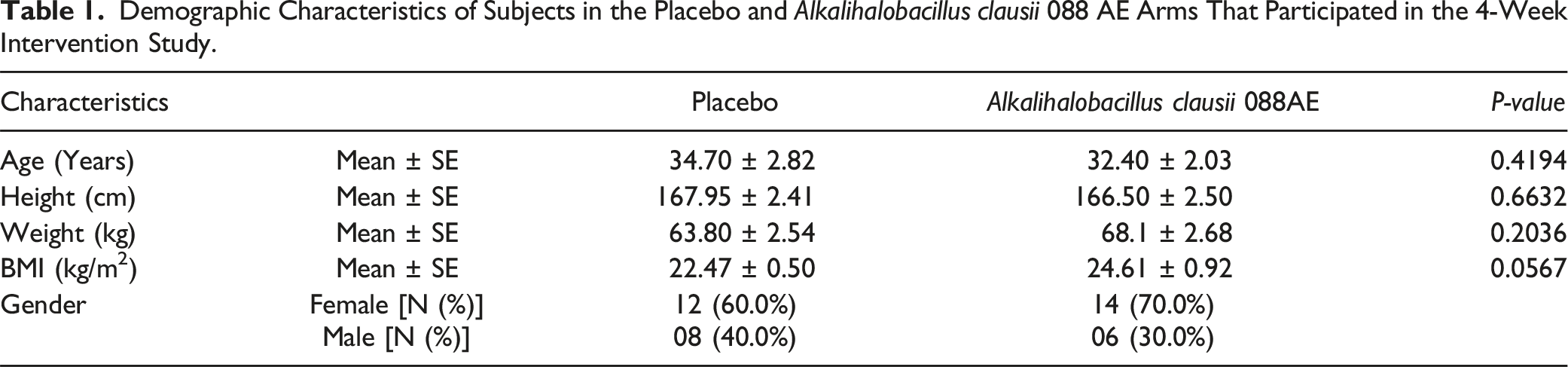
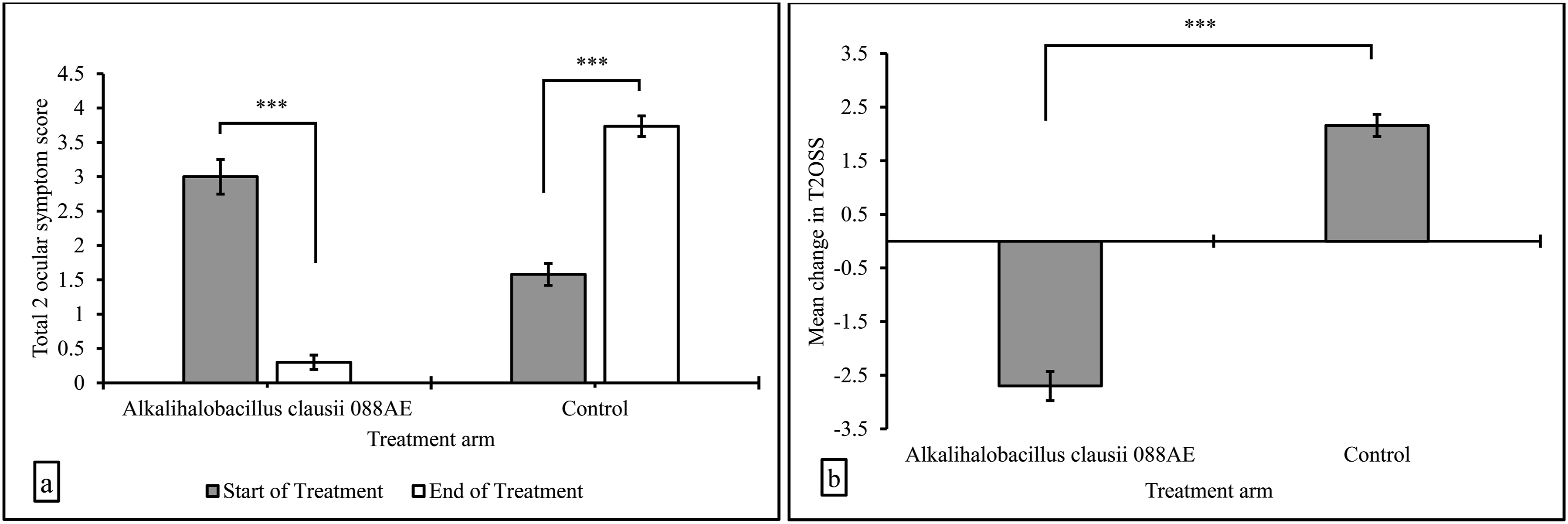
Treatment of AR with A. clausii 088AE showed a significant decrease in the T4NSS with respect to its baseline value (Figure 2a). The average or mean T4NSS value in the test group was 7.80 ± 0.19 at the start of the treatment, which decreased significantly to 1.75 ± 0.42 by the end of the treatment (P < 0.01). However, the placebo group showed a significant increase (P < 0.01) in the average T4NSS value from 5.70 ± 0.22 to 8.05 ± 0.25. The intergroup analysis displayed a statistically significant variation in the change (EOT−SOT) in T4NSS score between A. clausii 088AE and placebo, and this were −6.05 ± 0.54 and 2.35 ± 0.24, respectively (Figure 2b). The difference in mean change in T4NSS was statistically significant (P < 0.01). Similarly, A. clausii 088AE group showed a significantly decreased T2OSS with respect to its corresponding baseline levels (Figure 3a). The mean T2OSS value in the test group was 3.0 ± 0.25 at SOT, which decreased significantly to 0.30 ± 0.11 by EOT (P < 0.01). On the contrary, the T2OSS values in the placebo group showed a significant increase from 1.55 ± 0.16 to 3.75 ± 0.15 (P < 0.01) by EOT. The intergroup difference of T2OSS between A. clausii 088AE and placebo was −2.75 ± 0.26 and 2.20 ± 0.21 and this was statistically significant (P < 0.01) (Figure 3b). Further, the changes in the cough score were evaluated for both the A. clausii and placebo groups. The average cough score decreased significantly by EOT with the A. clausii supplementation (Figure 4). The average cough score in the A. clausii group was 0.4 ± 0.11 at the start of the treatment, which reduced to zero by the end of the treatment, definitely suggesting a significant change in the cough scores with probiotic treatment (P < 0.01) (Figure 4a). Notably, the placebo group showed a significantly higher cough score of 0.95 ± 0.05 by the end of the treatment (P < 0.01). The intergroup analysis revealed a statistically significant variation in the change (EOT−SOT) in cough scores between A. clausii 088AE and placebo, which were −0.40 ± 0.11 and 0.85 ± 0.08 (Figure 4b), respectively (P < 0.01). Changes in the symptoms of allergic rhinitis as evaluated by (a) T4NSS, and (b) mean change in T4NSS after intervention with the Alkalihalobacillus clausii 088AE and placebo (Results expressed as mean ± SE, ***P-value < 0.01). Changes in the symptoms of allergic rhinitis as evaluated by (a) T2OSS, and (b) mean change in T2OSS after intervention with the Alkalihalobacillus clausii 088AE and placebo (Results expressed as mean ± SE, ***P-value < 0.01). Changes in the symptoms of allergic rhinitis as evaluated by (a) cough score, and (b) mean change in cough score after intervention with the Alkalihalobacillus clausii 088AE and placebo (Results expressed as mean ± SE, ***P-value < 0.01).
Assessment of Immunological Parameters
Changes in Immunological Parameters of Patients With Allergic Rhinitis After Intervention With the Alkalihalobacillus clausii 088AE and Placebo at the Start of Treatment (SOT) to End of Treatment (EOT). Results Expressed as mean ± SE.

Correspondingly, the study also evaluated the changes in cytokines: IL-4, IL-10, IL-17, and IL-22, and IFN-γ, TNF-α, TNF-β, and FOXP3 from visits 1 to 5 with respect to baseline values in all subjects. Similar to previous observations, no significant changes were observed in any of these parameters from baseline values. However, both the A. clausii 088AE and placebo groups showed an increase in IL-17 and IL-22 levels from SOT to EOT. Precisely, the A. clausii group showed an increase in IL-17 levels from 60.80 ± 21.16 pg/mL to 74.10 ± 30.03 pg/mL, and from 75.60 ± 23.83 pg/mL to 79.10 ± 20.77 pg/mL, in A. clausii 088AE and place group, respectively. Whereas, for the IL-22, the A. clausii 088AE group showed an increase from 53.80 ± 10.66 pg/mL to 62.50 ± 5.84 pg/mL, and placebo caused an increase from 54.40 ± 8.34 pg/mL to 61.10 ± 7.52 pg/mL, respectively. Additionally, although insignificant, the IL-10 values increased in the placebo group but decreased slightly in the test group, whereas the IL-4 values showed a marginal increase in both groups. Notably, the IFN-γ level decreased in the placebo group from 60.20 ± 10.12 pg/mL to 45.10 ± 4.94 pg/mL, but even though it slightly increased from 44.20 ± 3.51 pg/mL to 45.80 ± 6.09 pg/mL in the A. clausii 088AE group by the end of the treatments. Surprisingly, the FOXP3 values decreased and were identical in both the groups at the start and end of the treatments. Further, TNF-α values increased in both groups by the EOT. This change was higher in the A. clausii 088AE group than the placebo group, resulting in increased TNF-α values from 76.50 ± 6.22 pg/mL to 85.00 ± 7.87 pg/mL. On the other hand, the TNF-β value showed a slight increase in the test group, whereas it substantially reduced in the placebo group from 53.50 ± 4.49 pg/mL to 38.20 ± 4.25 pg/mL.
Effect on Th1/Th2 Balance
Allergic disorders are associated with a shift in the balance of Th1/Th2 ratio towards the activation of Th2. 26 Thus, to explain the effect of probiotics on AR symptoms, the ratio Th1/Th2 was evaluated. Accordingly, a favorable shift in the Th1/T2 balance towards Th1 was observed with the A. clausii 088AE supplementation. The Th1/Th2 ratio in the A. clausii group was 0.94 ± 0.23 at baseline, which slightly decreased to 0.87 ± 0.13 compared with the ratio in the placebo group, which decreased from the baseline value of 1.41 ± 0.33 to 1.00 ± 0.26. The reduction in the Th1/Th2 ratio brings clarity to the balance of cytokines, which is beneficial for alleviating the symptoms of AR, suggesting that the A. clausii 088AE supplementation is more effective than the placebo treatment.
Safety Assessment: Effect on Physical, Hematological, and Biochemical Parameters
The effect of test and placebo supplementation on the secondary endpoints was evaluated to assess the safety of the products. The safety of the investigational product, A. clausii 088AE, was assessed for tolerance, adverse events, serious adverse events, and systemic biomarkers such as physical, haematological, and biochemical parameters.
The investigational product, A. clausii 088AE, was well tolerated by all the participants included in the study. During the entire clinical study, the patients reported no serious adverse event, nor did the physician recognize any. Among the 39 patients who completed the 4-week interventional study, the most common adverse events were gastric (1 in 19 patients; 4.5%), and nausea (1 in 19 patients; 4.5%) in placebo group, while none in A. clausii 088AE group. The compliance was higher (≥95%), indicating the safety of investigational products for oral administration. Further, no (zero) reports related to deaths or serious adverse events leading to the termination of the clinical study were documented.
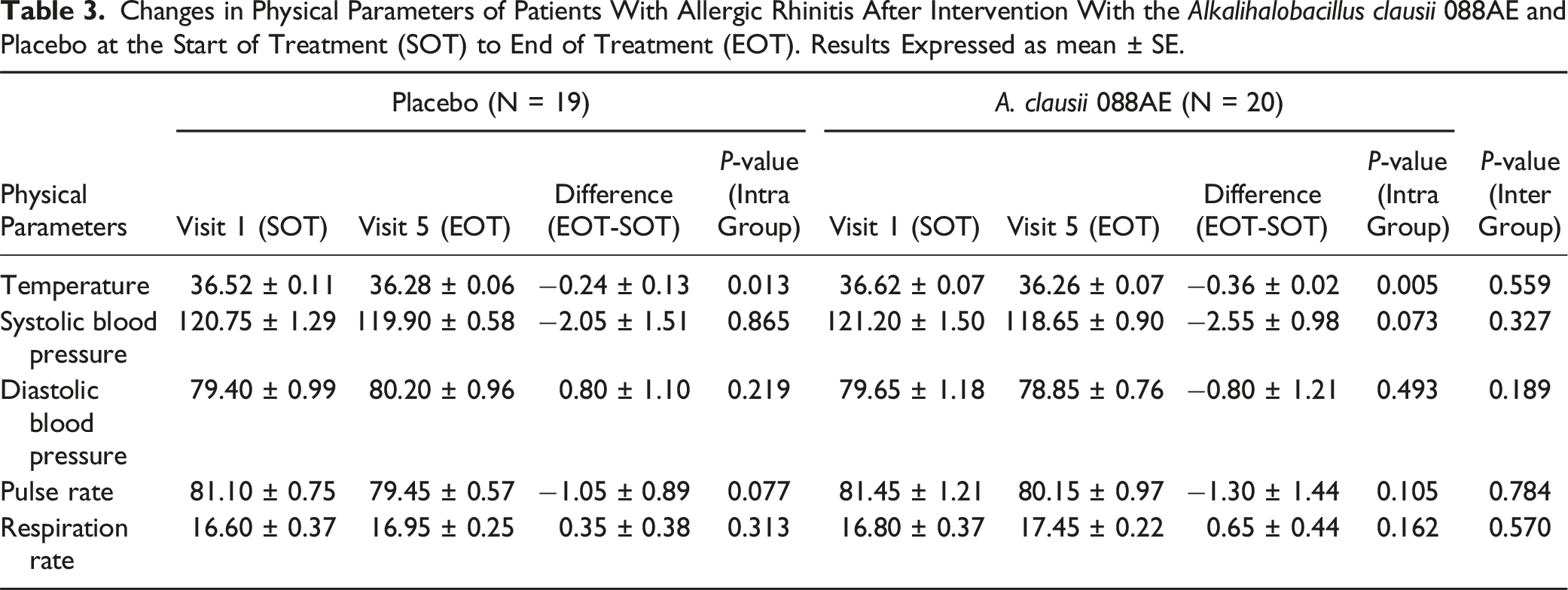
Changes in Physical Parameters of Patients With Allergic Rhinitis After Intervention With the Alkalihalobacillus clausii 088AE and Placebo at the Start of Treatment (SOT) to End of Treatment (EOT). Results Expressed as mean ± SE.
Changes in Hematological (Complete and Differential Blood Count, Platelets) and Biochemical Parameters of Patients With Allergic Rhinitis After Intervention With the Alkalihalobacillus clausii 088AE and Placebo at the Start of Treatment (SOT) to End of Treatment (EOT). Results Expressed as mean ± SE.
Discussion
The current study represents a prospective, randomized, double-blind, placebo-controlled, monocentric, interventional trial evaluating the therapeutic impact of A. clausii 088AE on allergic rhinitis in human subjects. Specifically, the effect of probiotic A. clausii 088AE was evaluated on adults with acute allergic rhinitis using a set of primary (symptom scores and immunological parameters) and secondary (physical, hematological, and biochemical parameters) endpoints. To the best of authors knowledge, this study is the first to evaluate the T4NSS, T2OSS, and cough score, incorporating all the essential symptoms that need to be monitored along with the corresponding changes in the immunological markers for treating and managing the symptoms of AR with A. clausii 088AE.
In the present study, the Alkalihalobacillus clausii 088AE showed a prominent efficacy in alleviating the symptoms of acute allergic rhinitis (AR). Quantitatively, this decrease in the AR symptoms was evaluated using T4NSS, T2OSS, and cough scores with respect to their baseline values. T4NSS assessment includes a score on the basis of the severity of parameters- rhinorrhea, nasal stiffness, nasal itching, and sneezing. The test arm showed a statistically significant decrease in the mean T4NSS value (P < 0.01) by the end of the treatment from the baseline value, indicating the positive impact of A. clausii on the AR symptoms. However, the placebo group showed a significant increase (P < 0.01) in the mean T4NSS value. A statistically significant change (EOT-SOT) in T4NSS amongst A. clausii 088AE and placebo was seen after the intervention period, suggesting a superior efficacy of A. clausii treatment over the placebo supplementation for resolution of symptoms of allergic rhinitis. Further, the placebo treatment failed to deliver any positive effect on the T4NSS symptoms, which might have resulted in the increased severity of the symptoms over time.
Previous studies have reported a significant decrease in nasal symptom scores with the probiotics treatment. Specifically, Ciprandi et al 8 reported a significant reduction in the nasal total symptom scores (P = 0.049) in children treated with B. clausii spores for 3 weeks at a dosage of 4 billion spores daily. The clinical studies on B. clausii treatment for AR are limited, many studies have reported a decrease in AR symptom scores with other probiotic supplementations following the same mechanism of improving the immunomodulatory effect.6,26–28
In addition to T4NSS, this study involved evaluating the T2OSS and cough score values to further understand the role of probiotics in alleviating the AR symptoms, which was rarely reported by previous studies for treating AR with B. clausii.8,22 On this point, our study has some distinct advantages over previous studies. The present study clearly exhibits a significant decrease in the T2OSS and cough score, which complements the results observed for TNSS. The inter group analysis of T2OSS and cough scores indicated a statistically considerable reduction in the symptoms, revealing the efficacy of A. clausii 088AE in resolution of symptoms associated with ocular and cough in allergic rhinitis patients. In a meta-analysis by Güvenç et al., 29 a combined symptom score of 21 studies suggested a significant decrease in the ocular symptoms with other probiotic supplementation as compared to the placebo treatment. Specifically, Nagata et al., 15 Costa et al., 30 and Ishida et al. 31 reported a significant improvement in the ocular symptom scores with probiotic supplementation than that with the placebo treatment. These results are in agreement with the observations in the present study for ocular symptom scores. Interestingly, the cough scores diminished completely after 4 weeks of probiotic treatment; however, these scores increased substantially in the placebo group by the end of the treatment. Notably, all three symptom scores increased significantly with respect to the baseline values by the end of the treatment in the placebo group. This suggests that the placebo supplementation had little to no effect on the alleviation of AR symptoms during the study period, which might have led to an increase in symptom severity with time and increased symptom scores by the end of the treatment compared to the corresponding baseline values.
Evidently, many studies have attributed the improvement in clinical symptoms of AR to the changes in the immunological profile.8,21,26,28 Accordingly, changes in the immunological markers were evaluated to identify the role of A. clausii supplementation on AR symptom alleviation. Probiotics are known to modulate the immune response in the host by identifying specific patterns in microbes, target molecules with nod- and toll-like receptors, and, as a consequence, may reduce allergic responses. 30 Probiotics can also have functions in down-regulating the type-2 immunological response, declining the production of pro-allergic cytokine and allergen-specific IgE, and upregulation of allergen-specific IgA and IgG production in the body. 32 Mechanisms of probiotics have been known mostly as suppression of Th2-related cytokines and generation of regulatory T cells (Tregs) and Th1-related cytokines, as studied in mouse model. 9
Interestingly, in the present study, no significant changes were observed in Th1, Th2, Th17, and Treg cells in the test or placebo groups. Similarly, no significant changes were observed in cytokines: IL-4, IL-10, IL-17, and IL-22, and IFN-γ, TNF-α, TNF-β, and FOXP3 from visits 1 to 5 with respect to the baseline values in both groups. A similar observation was reported by Yan et al 33 in a meta-analysis of the beneficial effects of probiotics on AR by collecting 30 randomized controlled trials. The results revealed that the consumption of probiotics improved the scores of Rhinitis Quality of Life and Rhinitis Total Symptom but not the immunological parameters.
Besides, the A. clausii 088AE treatment in the present study resulted in a marginal change in the %Th1 cells, whereas the placebo treatment decreased the %Th1 cells. Further, probiotics could help induce the maturation of dendritic cells to restore the balance between type-1 T-helper cells and type-2 T-helper cells. This ratio is attained by either the production of IL-12 and IFN-γ or by suppression of the response of Th2 cells by downregulating the IL-4 levels and upregulating the IgA, IgE, and IgG1. In addition, the stimulation of IL-10 34 and tumor growth factor-β 8 could suppress the response of Th2 cells and enhance the activity of the Treg cells. 28 Moreover, allergic disorders are generally associated with the shift in Th1/Th2 cytokine balance towards Th2. 26 Th1 immune responses are believed to produce anti-infection effects in the respiratory tract by recruiting inflammatory cells to the site of infection through the coordinated expression of leukocytes and vascular adhesion molecules. On the other hand, the Th2 immune response causes deleterious changes in the upper airways, counteracting the Th1 immune response and resulting in long-term airway adverse remodeling. Thus, the balance between Th1 and Th2 (or the Th1/Th2 balance) determines mucosal immune homeostasis. 26
The results revealed maintaining the balance of Th1/Th2 towards Th1 with the probiotic treatment, whereas the ratio decreased with the placebo treatment. These results are in agreement with those observed with B. clausii (Enterogermina) 8 and other probiotics15,26 on AR. Specifically, it has been demonstrated that B. clausii stimulates Th1 and Treg immunity, promoting IL-12, IFN-γ, IL-10, and transforming growth factor-beta (TGF-ß) synthesis, and down-regulates Th2 response, inhibiting IL-4 production, in allergic children with respiratory infections. 8 Lin et al 16 reported a non-significant decrease in TNSS and IgE levels but an increase in IFN-γ levels with the consumption of Lacticaseibacillus paracasei GM-080 with 3 months of consumption of 2 billion CFU/day. Meng et al 26 reported diminished levels of IL-4 and IL-13 and a marked increase in the IFN-γ levels after 8 weeks of treatment with Broncho-Vaxom (BV), a mixture of 8 probiotics, indicating a favorable shift in the Th1/Th2 balance. Thus, the present study, through its findings, may serve as strong clinical evidence in support of the view that A. clausii 088AE produces a favorable shift of the Th1/Th2 balance towards the Th1 response by inducing the production of the Th1-type response (IFN-γ) in the nasal mucosa. Hence, it is highly plausible that restoring and maintaining the normal Th1/Th2 cytokine balance is 1 of the mechanisms underlying the efficacy of A. clausii 088AE against AR.
Moreover, the safety risk assessment unveiled that A. clausii 088AE was well tolerated by all the studied patients administered with the probiotic. The safety of A. clausii 088AE was examined by evaluating the vital physical, hematological, and biochemical parameters of all subjects within the study duration. As expected, the values of these parameters at SOT did not vary significantly from the values obtained at EOT in both arms (P ≥ 0.05). These clinically relevant and important findings prominently indicate the safety of the probiotic strain A. clausii 088AE upon oral consumption. In addition, at the specified dose, no serious adverse events were reported by the participants or the physician. Nonetheless, the most common adverse events reported were gastric (1 patient), and nausea (1 patient). The higher compliance (≥95%) indicated the tolerability and safety of investigational products (A. clausii 088AE) for oral administration and therapeutic use. B. clausii was found safe and effective at the studied dose for four weeks and 6 billion CFU/day for 3 weeks in children with allergic rhinitis.8,21 The safety and efficacy of the B. clausii probiotic with 2 other probiotic strains at a dosage of 10 billion CFU/day for 14 days has been proven and documented for resolving post-COVID fatigue. 35 Precisely, A. clausii 088AE, at a dosage of 6 billion CFU/day for 7 intervention days, was found to be safe and effective in treating antibiotic-associated diarrhea. 20
The current clinical study illustrates the therapeutic safety and efficacy of Alkalihalobacillus clausii 088AE in resolution of symptoms of allergic rhinitis. Overall, the oral supplementation of this probiotic could significantly mitigate the nasal, ocular and cough related symptoms in patients with allergic rhinitis. The observed data signifies that the oral administration of A. clausii 088AE could confer health benefits to patients with symptoms of allergic rhinitis.
Conclusions
The findings from the present study agree with the notion emerging on the use of probiotics in treating allergic rhinitis. Probiotics, specifically A. clausii 088AE, surely exert a beneficial effect in alleviating AR symptoms and exhibits the potential to bring immunological changes in patients with allergic diseases. The supplementation of A. clausii 088AE at the studied dose was well tolerated by the patients and found to be safe for consumption. The probiotic treatment significantly alleviated the symptoms of allergic rhinitis and partially showed a positive response in modulating the immunological parameters. This study highlighted the possible mechanism of action of A. clausii 088AE against AR via immunomodulation. However, the use of this probiotic in AR needs to be investigated further for varied study conditions and parameters to gain a broader insight into the effectiveness of A. clausii in the treatment of AR. Accordingly, prospective, double-blind studies involving larger-scale interventional studies at multiple centres, and prolonged follow-up periods are necessary to validate these findings.
Supplemental Material
Supplemental Material - A Prospective, Interventional, Randomized, Double-Blinded, Placebo-Controlled, Monocentric Clinical Study to Evaluate the Efficacy and Safety of Alkalihalobacillus clausii 088AE in Resolution of Acute Allergic Rhinitis Symptoms
Supplemental Material for A Prospective, Interventional, Randomized, Double-Blinded, Placebo-Controlled, Monocentric Clinical Study to Evaluate the Efficacy and Safety of Alkalihalobacillus clausii 088AE in Resolution of Acute Allergic Rhinitis Symptoms by Abhijit Rathi, PhD, Rajeshree A Khaire, M Chem, PhD, and VL Rathi, MS in Global Advances in Integrative Medicine and Health.
Footnotes
Acknowledgments
The authors would like to thank Advanced Enzymes Technologies Ltd, Thane for providing the probiotic supplement required for the clinical study. The authors would also like to thank Dr. Abhijeet B. Muley for assistance in manuscript writing.
Author’s Contributions
Conceptualization, methodology: AR and VLR; Data curation, formal analysis: AR; Visualization: AR; Writing – original draft and data interpretation: AR and RAK; Writing – review & editing: AR, and VLR.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All the named authors except V. L Rathi are paid employees of Advanced Enzymes Technologies Limited, Thane, India that has a corporate affiliation with Specialty Enzymes and Probiotics, USA. V. L. Rathi is a chairperson of Advanced Enzymes Technologies Limited. Specialty Enzymes and Probiotics, USA had no role in study design and actual conduct of the study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Clinical Trial Registration
The clinical trial registry of India CTRI/2020/09/027657 [Registered on: 08.09.2020].
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
