Abstract
Background
To improve the implementation of clinical trial interventions, there is a need to facilitate communication between key stakeholders and research teams. Community Advisory Boards (CAB) bring together a range of stakeholders not historically included in the research process to inform and work collaboratively with research teams.
Objective
To describe our procedures and processes for (1) integration of a CAB into a pragmatic clinical trial of a telehealth-delivered group mindfulness program for persons with chronic low back pain (cLBP) within primary care, and (2) for the rapid uptake and implementation of CAB recommendations.
Methods
The CAB we convened includes persons with cLBP who have undergone the mindfulness intervention, health care system leadership, advocacy groups, and mindfulness experts. The CAB members underwent a two hour initial training that introduced the research process and the CAB’s role as research partners. The CAB met monthly for 1 hour. We used the Lighting Report method to summarize meetings and share feedback with the research team.
Results
The recommendations of the CAB during the first year they met were divided into recruitment, informed consent, and survey recommendations. The study website also was overhauled based on recommendations, including a more engaging first page with rotating images of nature and testimonials. The language on the website was edited to be more concise and participant-friendly. The CAB recommended talking points to discuss with participants during screening or informed consent about the benefits of participating in research.
Conclusion
We established a CAB that represented diverse perspectives, organizations, and experience with cLBP and mindfulness. The differing perspectives of the CAB resulted in recommendations that the research team itself would not have decided on their own. The Lightning Reports were also an effective way to efficiently communicate the CAB recommendations to the research team.
Introduction
To improve the implementation of clinical trial interventions, there is a need to facilitate communication between key stakeholders and research teams. Community Advisory Boards (CAB) bring together a range of stakeholders not historically included in the research process to inform and work collaboratively with research teams. The importance of CABs to clinical research is being increasingly recognized since the input of a diverse set of voices informs the approach, recruitment, and dissemination of the findings, ultimately resulting in research most relevant to the people it is intending to reach. 1
Mindfulness-Based Stress Reduction (MBSR) is a mind and body therapy that is now included in the evidence-based guidelines of the American College of Physicians for initial treatment of chronic low back pain (cLBP). MBSR is also a Best Practice recommendation of the U.S. Department of Health and Human Services.2,3 Despite MBSR’s effectiveness at decreasing pain and improving function it remains underutilized and has not been embedded into routine clinical care. Research into the barriers and facilitators of MBSR implementation has focused on patient level variables. 4 Researcher’s hypothesized that additional likely barriers to MBSR’s integration include lack of procedural guidelines to deliver a group program in the clinic, unfamiliarity with MBSR, uncertain reimbursement, and importantly, lack of stakeholder involvement in MBSR and mindfulness research and dissemination.
Recognizing the importance of stakeholder engagement to our pragmatic clinical trial of a telehealth delivered mindfulness group program for persons with cLBP, the investigators in the OPTIMUM (Optimizing Pain Treatment In Medical settings Using Mindfulness) pragmatic clinical trial (PCT) convened a CAB. The CAB helped inform the trial which was at the unique intersection of mind and body medicine, chronic low back pain, and diverse health systems in the United States (that included a safety net health system in Boston, Massachusetts; community health centers in the Piedmont region of North Carolina, and large academic medical centers in Pittsburgh, PA and Chapel Hill, NC). Rapid qualitative analysis methods were critical to the integration of CAB recommendations in a time-sensitive manner. Our goal is to describe our procedures and processes for (1) integration of the CAB into OPTIMUM, and (2) for the rapid uptake and implementation of CAB recommendations.
Methods
OPTIMUM is a PCT of MBSR for cLBP delivered via telemedicine, clinicaltrials. gov NCT04129450. 5 OPTIMUM is being conducted in 3 health care systems: the largest safety net hospital in New England, a federally qualified health centers in North Carolina and academic health centers in Chapel Hill North Carolina and Pittsburgh, Pennsylvania. These sites serve large numbers of underserved people as well as a diverse patient population. OPTIMUM’s primary goal is to determine the impact of this intervention in real-life clinical settings. Four-hundred-fifty persons with cLBP ≥18 years are randomized to (1) OPTIMUM (n = 225) + Primary Care Provider (PCP) Usual Care; or (2) PCP Usual Care (n = 225). Our primary hypothesis is that patients in OPTIMUM will have significantly improved pain intensity and interference at completion of the program and 6-month (primary endpoint) and 12-month later, as compared to PCP Usual Care.
Composition of Community Advisory Board (CAB).

CAB Recruitment Procedures
Potential CAB members were recommended through personal connections by research team members from each of the 3 clinical sites. CAB nominees were selected with attention to diversity of role (lived experience with cLBP, working in primary care, and/or teaching mindfulness), age, gender, and race/ethnicity. Research team members invited each CAB member via email solicitation, and each potential member was provided with a “Frequently Asked Questions” document about participating. Ten of 11 potential CAB members accepted the research team’s invitation to participate, with 1 person declining due to a family emergency. CAB members were recruited over the course of several weeks, about 6 weeks before the first CAB meeting was scheduled. CAB members who agreed to participate in the study were compensated as contractors from the health care system that administered the OPTIMUM study and a W9 completed.
Training
We used the “Connecting Community to Research: A Toolkit” as our guide to creating a meaningful relationship with the CAB members. 6 The toolkit is a step-by-step guide to train the CAB by introducing the research process and developing an understanding of the CAB’s contribution as partners in our research study. The CAB participated in a two-hour training.
Monthly Meetings
The decision for monthly meetings (vs quarterly or yearly) was to ensure that the CAB would provide real-time relevant feedback on current issues the OPTIMUM study was facing. 7 The topics of the meetings were initially determined apriori (recruitment procedures and materials, screening telephone scripts), but then as the meetings progressed included immediate study needs (such as reviewing a new measure). The meetings last for 1 hour and are facilitated by one of the co-investigators (IR).
We utilize the Lighting Report method to summarize meetings and share feedback with the research team. 8 With this method real-time synthesis of the CAB’s discussion occurs under classifications of “what works” (Plus), “what needs change” (Delta), “insights, ideas or recommendations” (Insights), and finally, action items. The synthesis is discussed towards the end of the meeting with the CAB to increase the accuracy and validity of the points discussed. Two research team members (ER, RR, or JB) take notes in the Lightning Report format, and these notes are synthesized into a final 1-page report that is disseminated to the full multi-site research team. We then brought these points to the study investigators in one-two weeks at the regularly scheduled team meetings (all investigators, coordinators, and staff).
The Lightning Report recommendations were then presented to the research team, who discussed the recommendations and how to implement them. The responses to and implementation of the CAB recommendations were recorded. As many recommendations involved changing the appearance and wording of flyers (for example) or the wording of the screening introduction, a research member or members were assigned the task of implementing the CAB recommendations. Once it was implemented this was reported back to the CAB. The report includes how the recommendation was adopted or if not, why not. Figure 1 reviews the workflow of the CAB meeting. The parent OPTIMUM study was approved by the University of Pittsburgh single Institutional Review Board on 09/10/2019 (STUDY20110378). Community Advisory Board work flow.
Results
Implemented Community Advisory Board Recommendations.

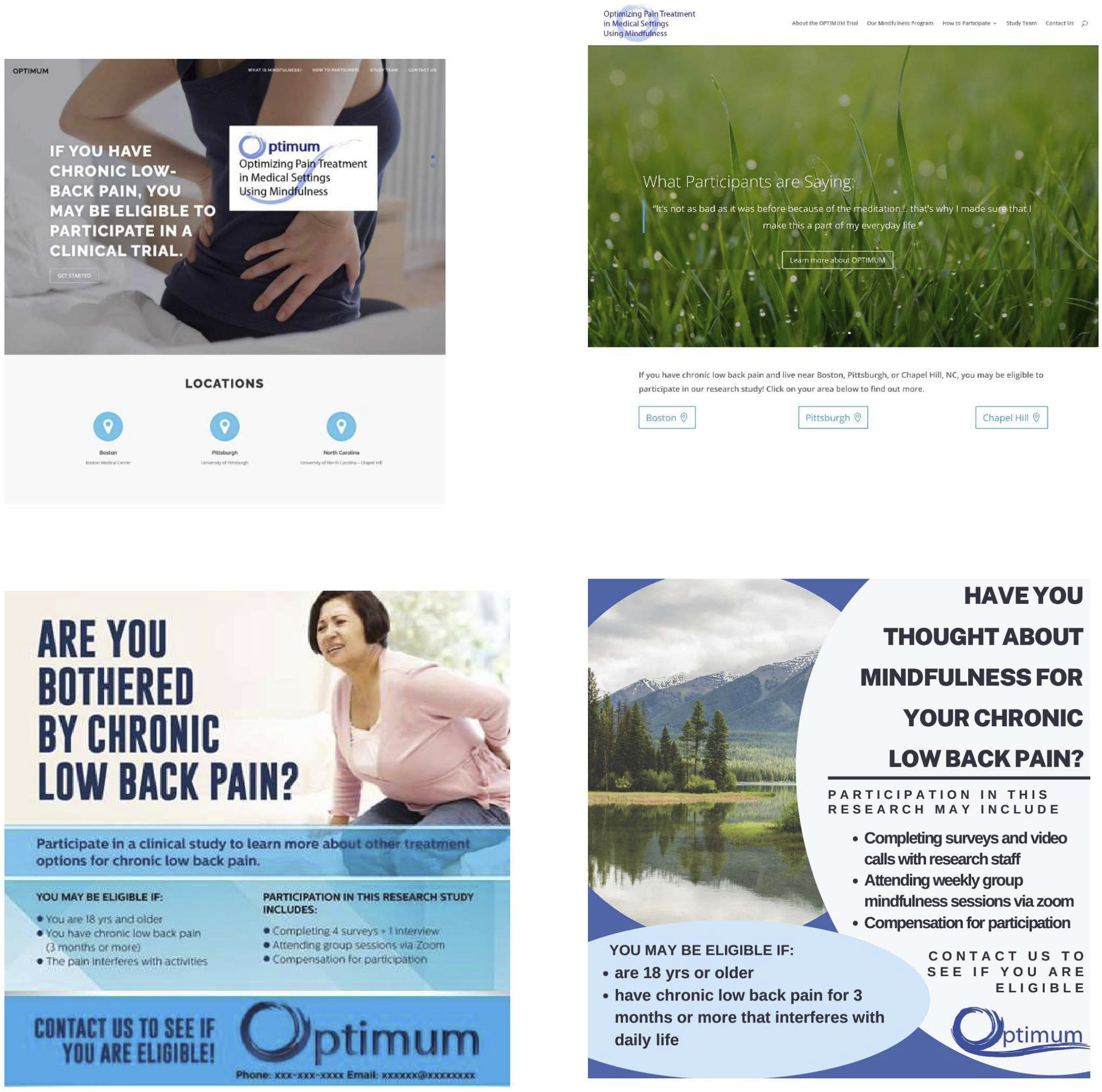
Top left webpage with static image. Top right shows changes recommended by Community Advisory Board that included nature scenes that scroll and include quotes from participants. Bottom left shows flyer before and bottom right after Community Advisory Board recommendations to include a nature scene and highlight mindfulness.
The study website also was overhauled based on recommendations, including a more engaging first page with rotating images of nature and testimonials. The language on the website was edited to be more concise and participant-friendly as the CAB feedback was that it contained too many scientific terms.
We also reviewed an additional survey on chronic overlapping pain and the CAB gave recommendations to clarify the language of the interview guide and remove a figure.
The CAB recommended talking points to discuss with participants during screening or informed consent about the benefits of participating in research. These talking points were for research assistants to use when they interacted with potential participants during the screening process or informed consent. The points included the benefits of participating in research (research findings may help others who are dealing with chronic low back pain, may help discover useful ways to treat and manage chronic low back pain, may reduce the number of people needing to use opioids to manage chronic pain, may help us understand how to implement mindfulness in the clinical setting), of participating in the OPTIMUM study (gives participants a “natural” approach to manage their chronic pain), and the evidence-based benefits of mindfulness (studies have shown that mindfulness can decrease the experience of chronic pain). There were additional talking points on the purpose of data collection, as participants did not understand why surveys were repeated over time (“we ask you the same questions each month to track any changes in pain you may have”).
The CAB also reviewed in an iterative fashion the animated video that was created for recruitment purposes. All aspects of the video were modified because of the feedback. This included changing the animated people to include diversity in age, size, race, ethnicity, and clothing. A mock-up of the animated video was reviewed slide by slide and what was pictured, text, the actual animation, and the narrative were all changed based on the iterative feedback of the CAB. The final narrative was edited based on the CAB’s feedback. Due to feedback from clinical sites, a teaser version (30 second) version of the video was also developed in collaboration with the CAB, to be used on social media. The CAB edited the text and provided feedback on visuals for the teaser version of the animated video. CAB attendance is on average 9 out of 11 persons at each monthly session.
Discussion
We established a CAB that represented diverse perspectives, organizations, and experience with cLBP and mindfulness. The CAB underwent training to review the goals of clinical trial research as well as the goals and expectations for the CAB. The CAB met monthly so that it became an integral part of the study team and provided input in a timely manner. We also established processes for rapid dissemination and implementation of the CAB recommendations which included utilizing the Lightning Report method to summarize key recommendations. These findings were usually brought to the team within 2-4 weeks for discussion and implementation.
Community engagement was essential to promoting health equity in our clinical trial. This is consistent with the first of 8 principles of stakeholder engagement described by Goodman et al of a focus on community perspectives and determinants of health. 9 The differing perspectives of the CAB resulted in recommendations that the research team itself would not have decided on their own. This included adding confidentiality language to scripts and recruitment letters, broadening the depictions of people or animated characters in our recruitment materials, or exchanging or adding images of nature scenes which the CAB believed was more reflective of mindfulness. We also changed the language in instrument instructions to be more understandable for a non-research audience. Our approach is consistent with the Patient-Centered Outcomes Research (PCORI) published set of principles that guide equity and inclusion in research. Its four principles of Inclusion, Equitable Partnerships, Trust and Trustworthiness, Accountability and Actionability are a framework in which we could purposely hold our research procedures accountable. 10
There are several features of our process that were likely related to the success of the CAB. This included the initial training, monthly meetings, regular communication with feedback on recommended changes, compensation, and experienced CAB facilitation. The Lightning Reports were also an effective way to efficiently communicate the CAB recommendations to the research team. Our work is in line with others who have found that valuing and leveraging CAB members’ expertise and utilizing a shared decision-making approach were key components of a successful CAB. We demonstrated this by rapidly implementing CAB recommendations and discussing recommendations and coming to consensus on decisions with the entire team, and then reporting back to the CAB.7,11,12 We also maintained engagement of the CAB by continued education in research procedures and ethics.
Additional elements of continued CAB engagement were a commitment from the research leadership and staff to put in the time and effort required to run the CAB, representation and diversity of the CAB, and well-defined agendas and questions for CAB meetings.
Limitations
The CAB was convened more than 2 years after the grant was written and after the study began. This was in part due to delays at the start of the study due to the pandemic. Nevertheless, the CAB did not weigh in on issues like study design, clinical sites, inclusion/exclusion criteria, and key outcome endpoints. Ideally, a CAB is engaged much earlier to inform research priorities and contribute to the design of research methods, among other factors.
The selection of CAB members was a subjective process, and therefore subject to forms of bias. Future projects that build CABs might include a more extensive recruitment and interview process with pre-specified selection criteria to select CAB members. This could help to mitigate bias and ensure that the CAB is made up of diverse perspectives.
Future directions for CABs need to include measurement of the downstream impact of the CAB on the research study design, outcomes, and dissemination. Stakeholder engagement funding needs to happen while grants are being written to have a truly participatory process.7,13
Conclusion
The Community Advisory Board in the OPTIMUM trial represented diverse perspectives, organizations, and experiences with cLBP and mindfulness. The differing perspectives of the CAB resulted in recommendations that the research team itself would not have decided on their own. The Lightning Reports were also an effective way to efficiently communicate the CAB recommendations to the research team.
Footnotes
Acknowledgements
The content is solely the responsibility of the authors and does not necessarily represent the official views of the NCCIH or the NIH or its HEAL initiative.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported within the National Institutes of Health (NIH) Pragmatic Trials Collaboratory through the NIH HEAL Initiative under award number UH3AT010621 administered by the National Center for Complementary and Integrative Health (NCCIH). This work also received logistical and technical support from the PRISM Resource Coordinating Center under award number U24AT010961 from the NIH through the NIH HEAL Initiative.
