Abstract
Study Design
Descriptive study (/review containing supporting case studies).
Objective
The temporomandibular joint has some distinctive features that were not taken into account during the initial development of temporomandibular joint prostheses, such as laterotrusive movements that are necessary for the proper grinding of food as well as synchronous and congruent movements made in conjunction with the healthy, contralateral joint. The aim of this article is to describe the development of a novel type of TMJ prosthesis that optimizes temporomandibular joint replacement.
Methods
The development was initiated by using contemporary technologies like computer-aided design customization, additive manufacturing, and surface treatments. Biocompatibility, proper fixation, and wear resistance, being prerequisites for the longevity of prostheses, were investigated next. Individual variables (condylar path, condylar axis angle, Bennet shift) were introduced as the main basis for physiological movements with the restoration of all functions. Early post-operative results ranging from 1 month to 4.5 years (11 patients, average follow-up period was 23.3 months) were assessed.
Results
The experience with 16 individualized total joint replacements in 11 patients is presented. The 3-year follow-up results of two-patient detailed studies are discussed, which show evidence of the promising restoration of all mandibular movements, when preoperatively present.
Conclusions
By incorporating various innovative and novel features (scaffold for re-attachment of lateral pterygoid, patient-specific functional parametrization, saddle-like design for retention, …) into a novel TMJ prosthesis concept, a major advance in function-reconstructive temporomandibular joint replacement was achieved. Early in vivo results (1 year after surgery) showed promising outcomes, involving both high increases in mandibular movements and decreases in pain scores.
Keywords
Objective
The temporomandibular joint (TMJ) has multiple supportive functions (breathing, chewing, supporting the upper airway, sucking, swallowing, making facial expressions, vocalizing, and sustaining correct pressure in the middle ear) which are all derived from protrusion, retrusion, and lateralization of the mandible and opening of the mouth. Indications for prosthetic replacement include TMJ ankylosis and end-stage joint disease resulting from trauma, infection, degenerative arthrosis, cancer, developmental/inherited craniofacial anomalies affecting the mandible and TMJ, failed/failing TMJR devices or failed prior invasive surgery.1‐4 The decision to replace the affected joint is based on the severity of the reduced quality of life, mainly related to mandibular function, food intake and pain. While stock prostheses may reduce pain and aid mouth opening, they do not naturally function in alignment with the healthy, contralateral joint because they have not been adapted based on the patient’s anatomy nor do they allow for proper grinding movements. This is because the lateral pterygoid muscle was sacrificed during condylectomy and not re-attached. Optimal biological integration and acceptable wear of alloplastic components are prerequisites for any TMJ prosthesis. Moreover, for optimal success, the TMJ prosthesis should be made of biocompatible materials, should be able to withstand the loads delivered over the full range of function of the joint, must be stable in situ and the surgery to implant the prosthesis must be performed for the proper indications, and it must be performed aseptically. 5 Regardless of whether the TMJ is reconstructed with alloplastic, allogeneic, or autogenous material, it should improve mandibular function and form, reduce suffering and disability, contain excessive treatment and cost and prevent morbidity. 6
According to a review performed by De Meurechy et al. 7 no extensive research has been conducted (over the last 20 years) to improve TMJ prostheses regarding both materials and functionality.
To optimize and improve these existing TMJ stock prosthesis concepts, all physiological movements that are required for the abovementioned functions of a normal TMJ should be restored on both the replaced and contralateral (healthy or replaced) sides.
The objective of this article is to discuss the development of such an improved TMJ prosthesis, called the TMJ Parametro (Figure 1a and b, Vid. 1) (CADskills BV, Ghent, Belgium). Renders of a TMJ Parametro with pterygoid muscle reattachment. (a) Lateral view of a TMJ Parametro total joint. (b) Frontal view of a bilateral TMJ Parametro total joint. (c) Frontal view of the Ti6Al4V mandibular component with the HadSat® coating. (d) The fossa component consisting of highly cross-linked polyethylene and a grade 23 Ti6Al4V extra-low interstitial. (e) The bone-implant interface, which shows the 3D-printed lattice structure used to induce osseointegration as a secondary fixation method. (f) Lateral pterygoid muscle reattachment using bone chips and the corresponding enthesis.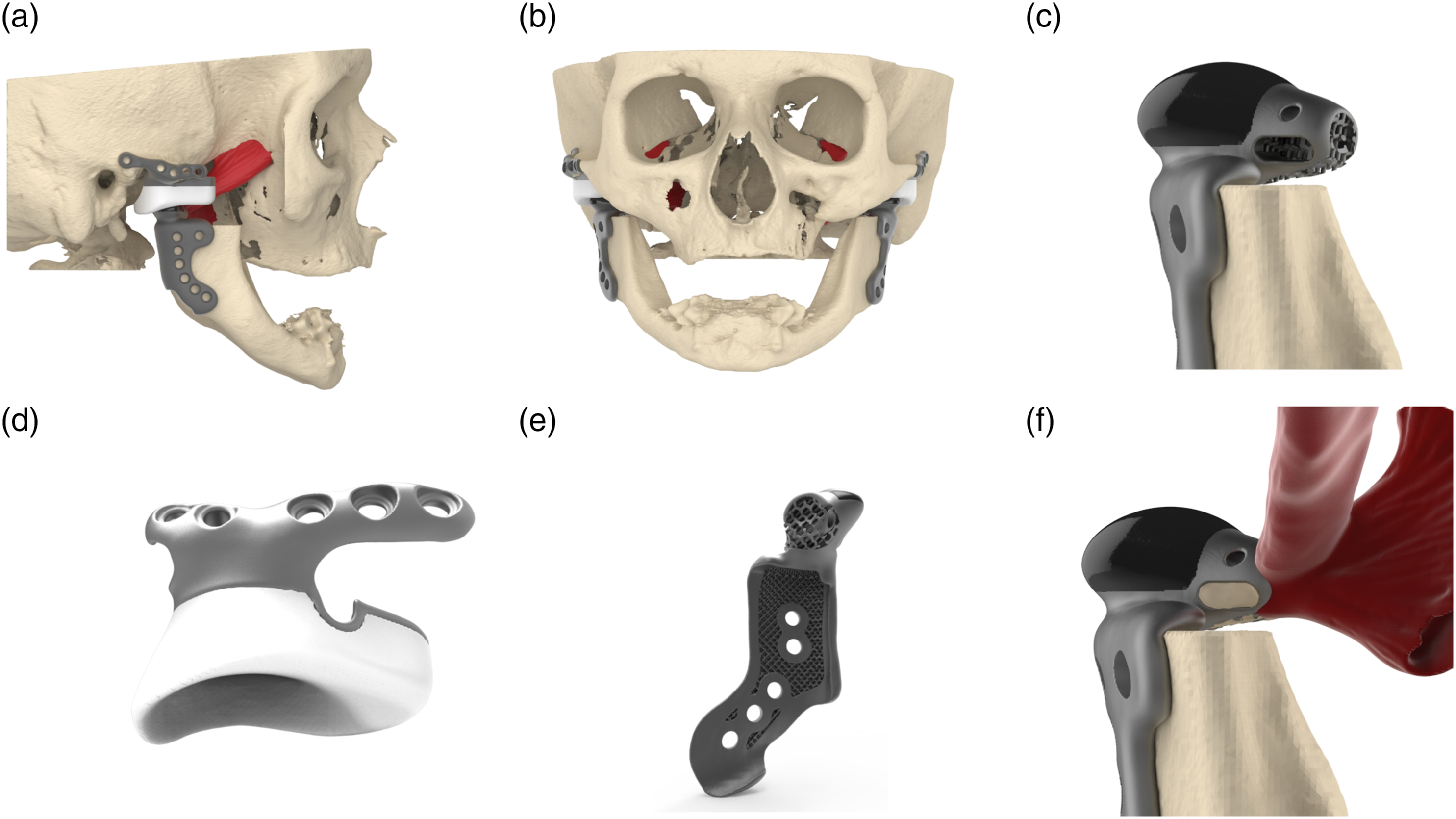
Methods
All the procedures in studies involving human participants were performed in accordance with the ethical standards of the institutional and/or national research committee (Centraal Studieloket, UZ Brussel, Code of approval: EC-2022-075) and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The authors certify that they have obtained all appropriate patient consent forms. On the form, the patients have given their consent for their images and other clinical information to be reported in this journal. This study encloses a descriptive technical review/report, a summary of the early results and two case studies.
Implant Design
Metallic component
The mandibular component and the skull base segment of the fossa component are additively manufactured using a Ti6Al4V ELI. The condyle is resurfaced using a diamond-like carbon (DLC) coating (Figure 1c and Figure 2), which is applied using the nondisclosed HadSat® protocol with a Vickers hardness (HV0.05) of 3500 ± 500 and a friction coefficient of .1. The HadSat® coating is a nontoxic, carbon-based coating that meets the Food and Drug Administration guidelines. The biocompatibility of this coating was tested under the International Standard ISO 10993-1 by the North American Science Associates. The test results are summarized in Table 1. Microscopic views (Magnification 500x) of the condylar surfaces of two different TMJ Parametro implants using scanning electron microscopy. (a) A condyle that was coated with HadSat® showing few irregularities. (b) An uncoated, polished condyle showing multiple grooves. Overview of Biocompatibility Tests Performed on the HadSat® Coating by the North American Science Associates.
Polymeric component
The articulating part of the fossa component, which is in contact with the condyle, is made of γ-irradiated tocopherol-enriched highly cross-linked UHMWPE (HXLPE) (Figure 1d). This HXLPE component is hot pressed onto the scaffold of a Ti6Al4V component, which in turn is fitted onto the glenoid fossa. Processing parameters as temperature, time, and pressure settings are confidential.
Surface Finishing
A sandblasted, large-grit, and acid-etched (SLA) surface at the bony interface of the mandibular component and the skull base of the fossa component of the TMJ Parametro is achieved by both micro-shot peening with alumina grit (∅ = 550 μm) and etching using 2 wt% oxalic acid at 85°C for 10 min. This enhanced surface roughness allows for bone ingrowth which reduces the stress on the screw-bone interface quite rapidly, allowing a reduction in the number screws required for primary stability from seven to five 7. (Figure 1e)
Functionality
Kinematics of the prosthetic joint
When comparing the load on the contralateral side of a mandible that has undergone total TMJ replacement with the load on the condyle of a healthy mandible, the load increase is approximately 15% when using a stock prosthesis.8‐13 Increases in mechanical loads have been shown to stimulate cartilage production and articular disc damage 14 which can negatively affect the patient (e.g., pain) and should thus be avoided. 15
In order to prevent disease development in the unaffected joint, an attempt was made to prevent an increase in load in the untreated joint. This was done by allowing the replaced side to move synchronously with the other joint during both rotational movements with the other joint and vice versa, as well as during translative movements (e.g., when performing movements of opening and closing, both the healthy and replaced condyle should move without causing interferences in each other’s joint space). In comparison with the Groningen TMJ prosthesis, where a more bulky fossa component with a low rotational point is used, 16 the articular surface of the TMJ is placed more cranial, to allow for a more natural movement. The same study, concerning the Groningen TMJ prosthesis, did indicate that a more accurate planning and prediction was feasible thanks to the patient-specific fit.
Furthermore, mastication involves laterotrusion, which is only possible with intact lateral pterygoid muscle function since occasional recruitment of the medial pterygoid muscle and minimal support by the masseter muscle cannot be predicted. Reattachment of the lateral pterygoid muscle is one of the main (innovative) features of the discussed joint. 17 In order to realize this reattachment, a scaffold in the condylar neck area (optionally, with a tunnel for temporary fixation with bioresorbable sutures) was provided, to form a bony union with the enthesis that has been carefully chiseled from the pterygoid fovea prior to condylar resection (Figure 1c–f). Therefore, both the condylar axis angle, the Bennett shift and free excursion at the anteromedial joint space should be integrated into the design of the prosthesis.
Prosthetic joint design
The occurrence of material wear is unavoidable, due to constant friction during mastication and other jaw movements. Also, to keep the center of rotation as high as possible, so as to mimic the original TMJ position, the HXLPE has a central thickness of only 2 mm. Despite the possible occurrence of a more uneven wear pattern caused by the more natural movements of the TMJ Parametro artificial condyle when compared to regular stock implants, the replacement of the fossa component may be required. This exchange could be facilitated by applying a tongue-and-groove fixation between the HXLPE and titanium parts as to minimize the invasiveness of the revision surgery. However, the use of such a fixation would also increase the surface area available for bacterial colonization by pumping actions, potentially resulting in the formation of a biofilm and in turn an acute infection.
As previously discussed, a rigid fixation is achieved by compressing the HXLPE onto a thin titanium scaffold (Figure 1d). Tests performed in sheep (de Meurechy et al, 2022, to be published) have demonstrated that this type of fixation is protective against infection, while at the same time counteracting undue deformation over time. In order to allow for replacement of the fossa component (typically after 20 years or more) no residual scaffold was provided at the interface between the fossa component and the skull base, which is likely thin at the middle cranial fossa and easily out-fractured, as to prevent excessive force during replacement.
Because the design of the joint is specific to the patient, care should be taken when extending the fixation plate of the fossa component anteriorly (not surpassing the mid-tubercular level to protect the frontal branch of the facial nerve) and posteriorly (making use of the non-pneumatized part of the temporal squama).
The size of the anterior extension of the fossa component depends on whether the coronoid process was resected or maintained. With the origin and insertion of the temporalis muscle intact, the anterior shift of the condyle is limited and anterior dislocation of the condyle is not anticipated. The more limited space and less anterior shift are likely to shorten the extension of the condylar path of the fossa component. The reverse situation applies when the coronoid process is resected.
If no undercuts are present or when they can be eliminated, a saddle-like design can be used to fit over the resection stump. This physically prevents any potential downward, medial and lateral movements. In doing so, a minimal set of screws are sufficient to counteract upward movements, which by themselves are minimized by the action of the masseter and medial pterygoid muscles.
Finally, the design of an improved joint prosthesis should also consider psychosocial functions. Asymmetries in the lower face, which can lead to a compromised self-image, can be addressed by using design software that has mirroring tools (e.g., Geomagic Freeform Plus, 3DSystems, Rock Hill, SC, USA) to achieve correct aesthetic outcomes postoperatively. By correcting side differences in the gonial angle and mandibular border using the mirrored side as a reference and whilst taking into consideration the quantity and quality of overlying soft tissue, the TMJ prosthesis act as a facial contouring implant as well (Figure 3). Thus alleviating stigmata of pathological deformations. A more prominent jaw angle can be used in the mandibular component to avoid or compensate for asymmetries.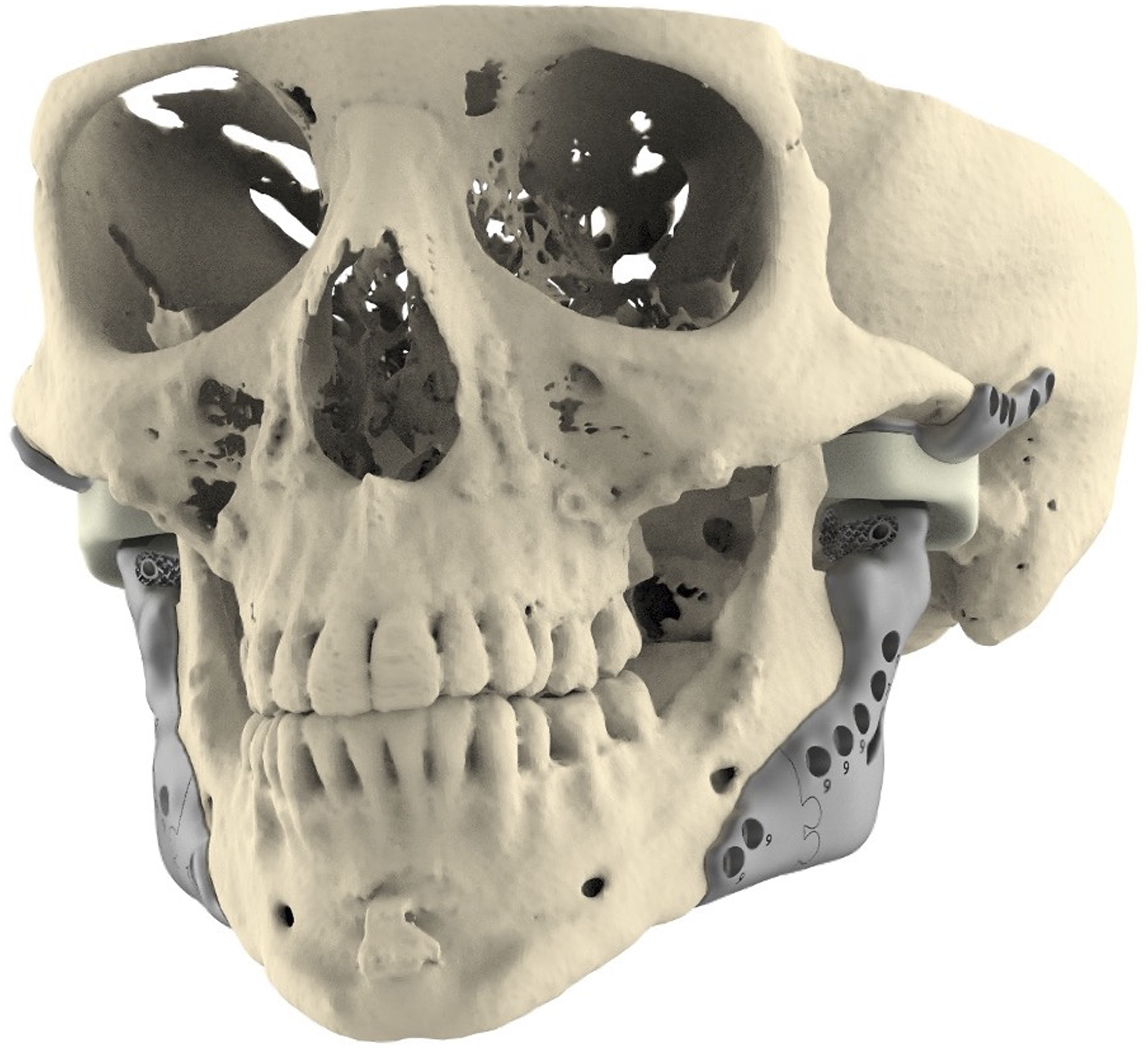
Patients and Methodology
After thoroughly evaluating the proposed implants in sheep experiments,18,19 eleven patients (2 men, 9 women; mean age at surgery of 49 years, 1 months) received all together 16 customized total TMJ Parametro prostheses. The surgery was performed by one surgeon in the same hospital. Follow-up ranged between 1 month and 4.5 years. Four patients suffered from end-stage degenerative arthrosis/arthritis due to disc pathology. Three had conservatively treated subcondylar fractures with subsequent degenerative joint disease. One patient had osteomyelitis in the ascending ramus after a ballistic trauma. One showed bilateral condylar resorption after orthognathic surgery. There was one female adolescent with unilateral craniofacial microsomia and one with TMJ ankylosis as a result of radiotherapy in childhood for a rhabdomyosarcoma. The indications for surgery varied between severe pain, refractory to conservative treatment and/or tissue sparing surgery, and severe trismus with severe dietary restrictions. Results were recorded in the electronic medical files, using Helkimo’s index (Helkimo, 1974) and a patient-reported outcome measure questionnaire. The criteria and indications for these TMJ replacements are as described by Sidebottom 20 and as mentioned in CADskills BV’s TMJ manual.
Results
The main aim of the paper is to present technical evolutionary steps, not to analyze clinical end-results. However, in order to demonstrate the clinical behavior of the novel prosthesis, early results of this first small group of patients are described here for completeness.
Group Results
Because the heterogeneity of indications, descriptive statistics about pain relief, increased mandibular movements, and dietary improvements are not representative for individual changes in wellbeing. The ankylosis and hemifacial microsomia caused no pain, whereas a maximal mouth opening of 28 mm was present in the patient with bilateral condylar resorption, who scored 10 in the Visual Analogue Scale (VAS, 0–10) before joint replacement. Therefore, the following results should be interpreted with caution. Two cases are described in detail to complement the group results.
One patient was excluded from the descriptive statistics because she twice received joint replacements within a year interval, once on the right-hand side and once on the left-hand side, leading to a disrupted follow-up. The total number of patients that were included in the descriptive analysis was 11, including one patient with a major component of neuropathic facial pain, whose pain score remained 8.
Important to remark is that the reattachment of the lateral pterygoid muscle was not always achievable, nor favorable. In cases with too much osteogenic capacity (young, ankylotic joint) or in absence of the lateral pterygoid muscle altogether (hemifacial microsomia, Pruzansky type III), no reconstruction of the muscle enthesis was attempted. In 25% of the discussed joint replacements, an enthesis reconstruction could not be performed, otherwise, the lateral pterygoid reattachment was carried out as described in the work of Prof Mommaerts. 17
Post-operative maximal mouth opening increased from 25.9 (SD 4.3) mm to 32.5 (SD 1.3) mm. The preoperative average pain score of 8.1 (SD 1.2) dropped to 1.4 (SD 1.3), whilst the mean preoperative diet score of 1.7 (1= liquid, 2 = soft, 3 = solid; SD 0.4) increased to 2.8 (SD 0.3). The average follow-up period was 23.3 months.
Case Studies
To illustrate the functionality of the TMJ Parametro, unilateral and bilateral replacement cases are discussed.
Case study #1: unilateral total joint replacement
In the early 1990s, a male patient was treated using intermaxillary fixation for 11 months (according to the patient, unverified) following a facial trauma. Since that time, the patient has experienced progressive worsening of joint function and increasing pain. This persistent pain became unbearable in 2017, forcing the patient to sleep upright. The majority of the pain was located on the right side, both at rest and while medicated. While speaking, the patient had to push the right ascending ramus into protrusion using his index finger. In 2018, a maximal mouth opening of 40 mm was measured, and laterotrusive motions of 10 mm and 5 mm to the left and right, respectively, were observed. Both at rest and during movement, capsulitis arthralgia was noticeable, which limited the patient’s diet to only liquid and very soft foods. A visual analogue scale (VAS) pain score of 10/10 was obtained, which led to an overall Wilkes Stage 5 classification
21
and a clinical dysfunction degree (Helkimo Index) of III.
22
CT scans showed bilateral, degenerative changes of both TMJs, narrowing of both joint spaces, and bilateral formation of osteophytes with flattening of the condyles (Figure 4). Since the clinical symptomatology was worse on the right side, the surgeons opted for a unilateral (right) joint replacement. A 3D model of the temporomandibular joint on the right side of the first patient, showing formation of osteophytes and flattening of the condyle (red arrow).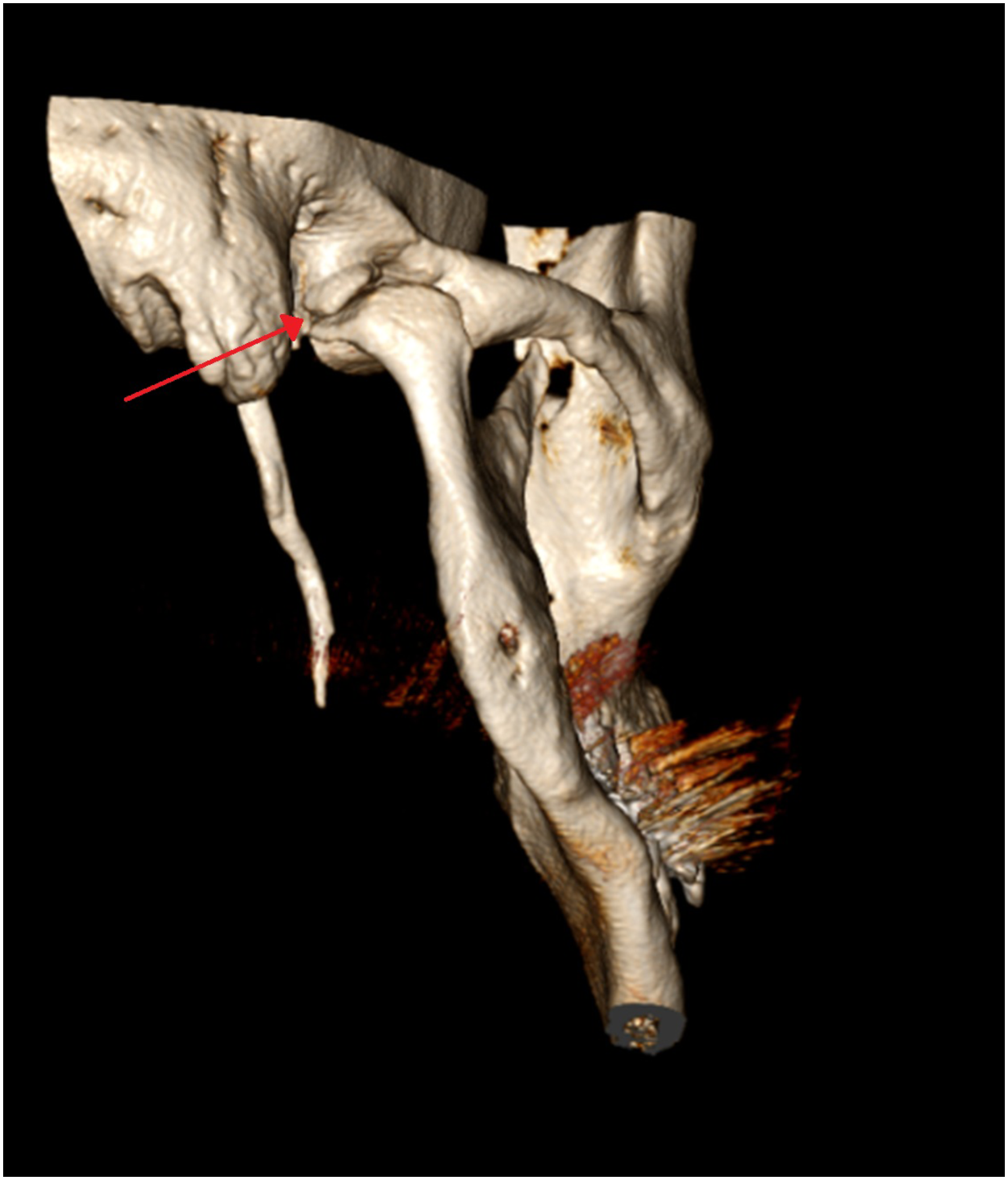
In 2018, at the age of 55 years, he received a TMJ Parametro prosthesis on the right side. The lateral pterygoid tendon was fixed to the scaffold in the condylar neck of the mandibular component.
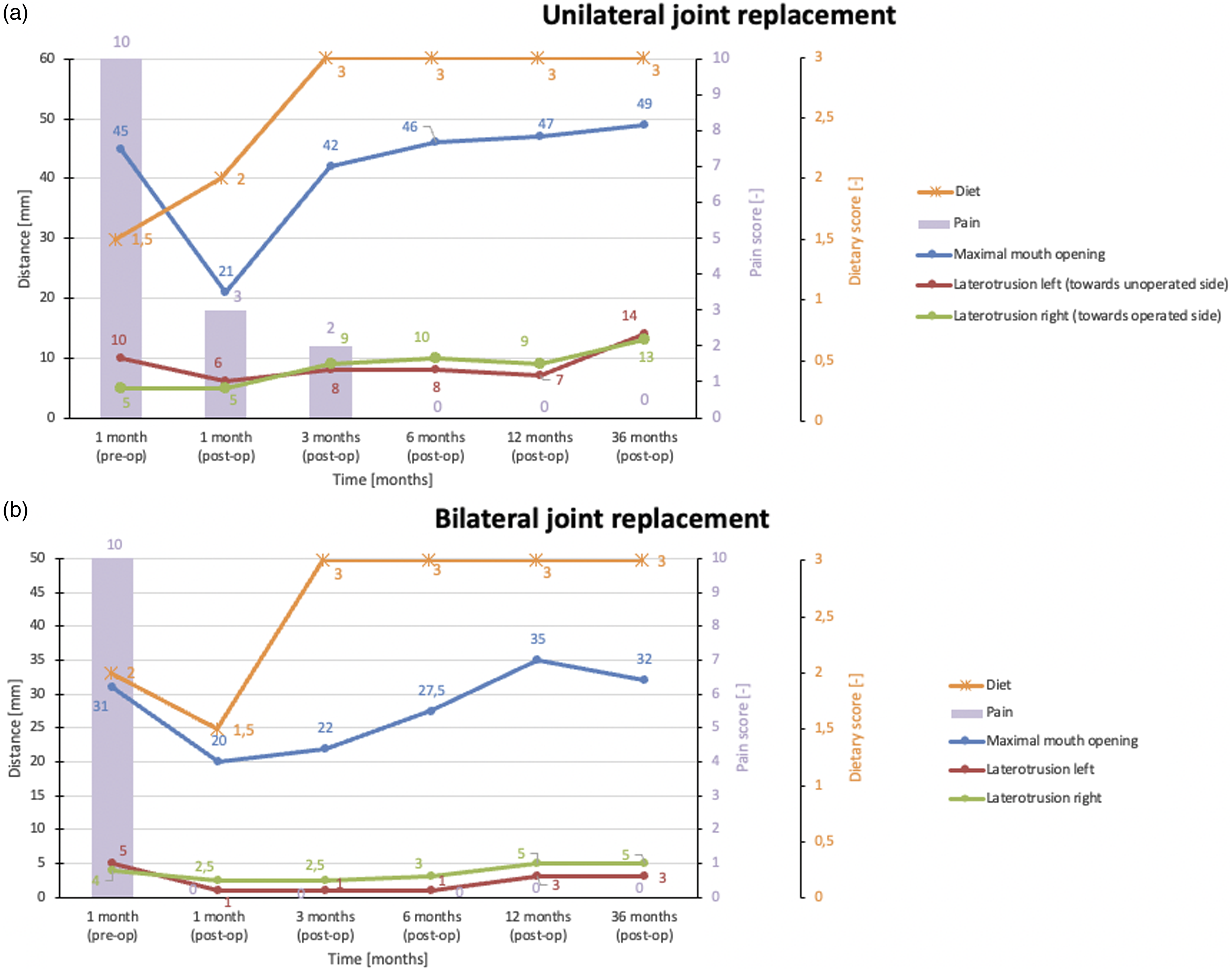
The postoperative maximal mouth opening progressed from 21 mm (1 month postoperatively) to 49 mm (3 years postoperatively) (Figure 5a), while the laterotrusive motion to the left (towards the unoperated side) increased from 6 mm to 14 mm during the same time period (Figure 5b). Meanwhile, the laterotrusive motion towards the operated side increased from 5 mm to 13 mm. The results from the follow-up of his maximal mandibular movements during this 3-year period are shown in Figure 6a. His VAS pain scores (on a scale of 10) decreased from 10 (preoperatively) to 3 (1 month postoperatively), 2 (3 months postoperatively), and 0 during his next three check-ups (6 months, 1 year and 3 years postoperatively) (Figure 6a). After 3 months, the patient was able to eat solid food again (Figure 6a). Clinical visualization of the patient’s maximal mouth opening (a) and lateral movement towards the unoperated side. (b) after unilateral temporomandibular joint replacement. Follow-up results of the patients who received either unilateral (a) or bilateral (b) joint replacement. Maximal movements (left y-axis), pain scores (middle y-axis), and dietary scores (right y-axis) are shown. The pain scores range from 0 (no pain at all) to 10 (unbearable pain). The dietary scores are as follows: 1: liquid; 1.5: liquid/soft; 2: soft; and 3: solid.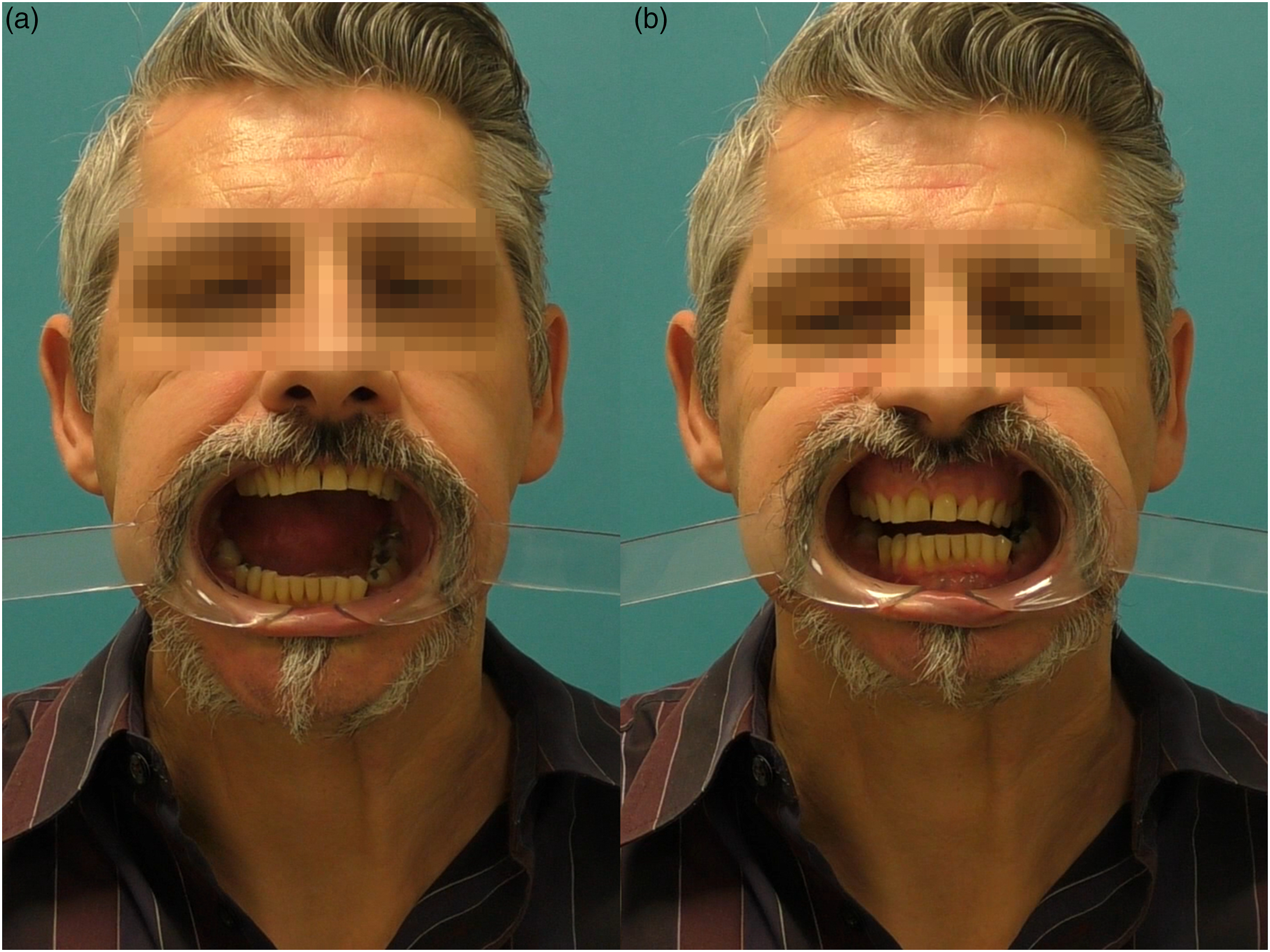
Case study #2: bilateral total joint replacement
A 77-year-old female patient underwent conservative treatment for bilateral arthrogenic TMJ pain that had persisted since 1986. In 2007, a CT scan showed an extensive degenerative process in both joints. In 2011, a CT scan showed extreme narrowing of the joint spaces and a dysmorphic appearance of the condyles, including osteophytic and resorptive processes. In 2017, she visited multiple hospitals with pain in both TMJs which, at rest, radiated temporally and worsened during movement. Her maximal mouth opening was restricted to 25 mm. VAS pain scores of 8/10 (right) and 6/10 (left) were obtained, which led to a VAS dietary score of 4 (where 0 is a liquid diet and 10 is a normal diet) and an overall Wilkes Stage 5 classification 22 with variable pain at rest and crepitations and pain during movement.
In 2019, she underwent bilateral total joint replacement with a customized TMJ Parametro prosthesis at the Universitair Ziekenhuis Brussel. Both left and right lateral pterygoid tendons were reinserted into the scaffold in the condylar neck of the corresponding mandibular components
17
(Figure 7). Intra-operative pictures of case 2. (a) Condensed bone chips in the scaffold in the condylar neck for lateral pterygoid muscle attachment (red arrow). (b) The implanted fossa component.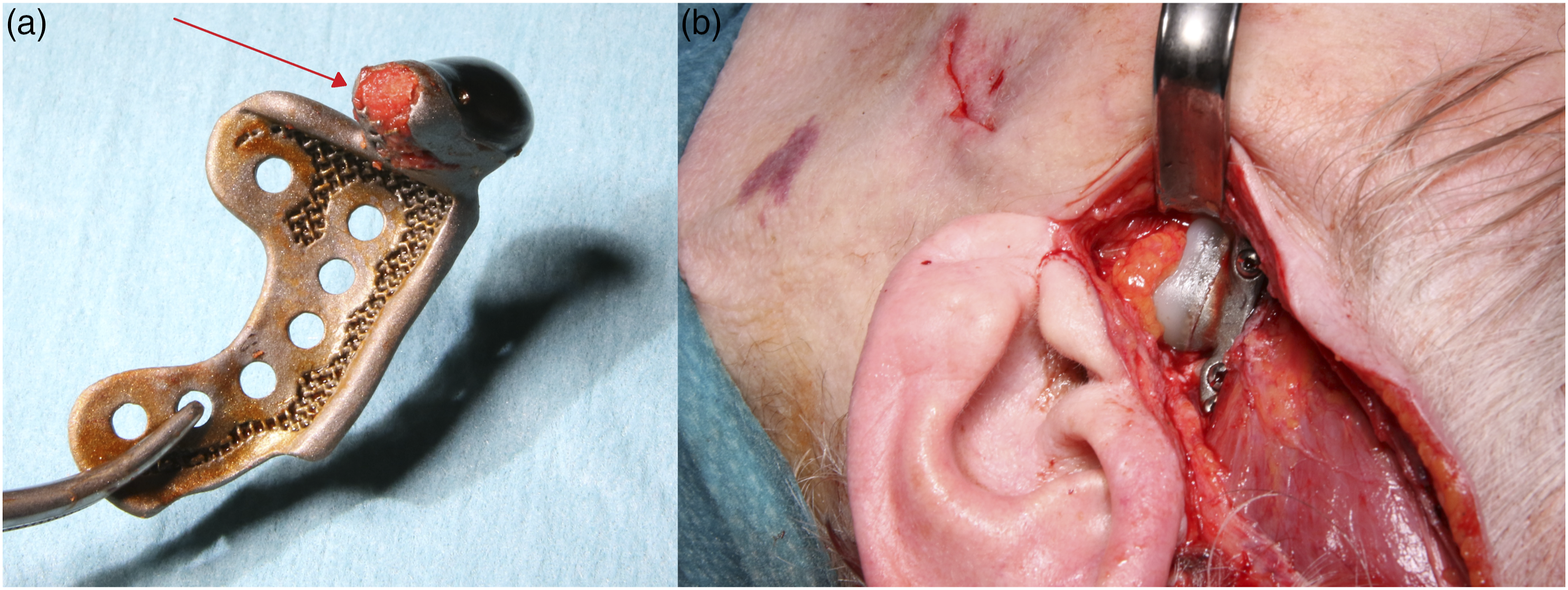
Her postoperative maximal mouth opening progressed from 15 mm (1 month postoperatively) to 32 mm (3 years postoperatively), while her laterotrusive motion to the right increased from 1.5 mm to 5 mm during the same period. Meanwhile, the opposing laterotrusive motion increased from 1.5 mm to 3 mm. The results from her current follow-up of her maximal mandibular movements during this 3-year period are shown in Figure 6b. At the 1-month postoperative check-up, her pain had already completely disappeared (VAS pain score of 0, Figure 6b), and after the 6-month mark, she was finally able to eat solid food again (Figure 6b).
Discussion
Abovementioned post-operative results, which mimic healthy biomechanical movements of the mandible, were achieved by extensive research and careful selection of the most suited biomaterials and features, which are being discussed here. The main limitation of this study is the currently small sample group and short follow-up. Moreover, the electromyographic results of lateral pterygoid muscle activity could not be monitored. It would be interesting to correlate such findings with various lateral pterygoid muscle enthesis reconstructions.
Metals
A meta-analysis of implant-related metal sensitivity showed that 10% of the general population is allergic to at least one or more alloy components (usually nickel) found in orthopedic implants. In patients with a functioning prosthesis, this number increased to 23%, while for patients with a failing prosthesis, it was as high as 63%. 23 A more recent study reported that nickel, chromium, and cobalt induce allergic skin reactions in 20%, 4%, and 7%, respectively, of the general population in Europe and in 14%, 4%, and 9%, respectively, of the population in the United States. 24 The prevalence of metal sensitivity appears to be rising and is most pronounced in nickel-containing implants. 25 In contrast, only occasional sensitivity has been reported for titanium. 23 In a recent review, only two studies presented strong evidence of sensitization to commercially pure titanium. 26 In contrast to chromium-cobalt particles, titanium-aluminum-vanadium-containing particles of a similar size to those found in the surrounding tissues of failed prostheses in humans showed little toxicity in an in vitro study using rat macrophages, even at high concentrations. 27
These findings result in Ti6Al4V being the preferred titanium alloy in small load-bearing implant applications. Important to remark is that the use of grade 23 Ti6Al4V extra-low interstitial (ELI) is preferred for long-term implants, such as in joint applications. Because of the reduced oxygen, nitrogen, and iron content, this grade shows enhanced biocompatibility compared to industrial grade 5 Ti6Al4V. Grade 23 is also most frequently used as a starting powder during the additive manufacturing of titanium implants.
Besides Ti’s excellent strength and manufacturability, it also boasts a high corrosion resistance. This is thanks to the presence of a thin (1.5 – 10 nm in thickness) but stable oxide film on the surface which minimizes the release of metal ions from the bulk.28,29 This layer is mainly composed of amorphous TiO2 with small amounts of suboxides TiO and Ti2O3 near the metal/oxide interface, and depending on the alloying elements, traces of Al2O3, V2O3 or V2O5, …30,31 The nearly-stoichiometric structure of TiO2 with few ionic defects/vacancies makes this compound an excellent barrier for ionic migration from the bulk metal to the environment. 32 As a result and in contrast to other bioinert implant materials, Ti alloy implants are not encapsulated by fibrous tissue. Even in particulate form, tissue activation remains weak because of this protective layer. 33
However, the presence of other metal oxides in the passive film on the Ti6Al4V alloy does raise some concerns. Although Al2O3 has never been associated with toxicity or allergy after orthopedic biomaterial degradation, 34 vanadium oxide can cause allergic reactions, 35 as well as toxicity at low concentrations and with continuous exposure. 36 Moreover, the presence of alloying metal ions (Al, V) having a different valence than the host metal (Ti) can alter the ionic transport across the oxide layer. Whereas the stable Al2O3 decreases the anion vacancies thereby enhancing the barrier function of TiO2, vanadium oxide dissolves from the passive film creating vacancies that enable ionic transport and therefore increase metal ion release. 32
Despite the limited Ti ion release from Ti based implants, it can still be a problem for certain percentage of the patient population. Prospective skin patch testing of orthodontic patients who wear titanium- and nickel-containing appliances demonstrated a nickel allergy prevalence of 14% and a titanium allergy prevalence of 4%. 37 It is thus imperative to subject potential candidates for TMJ replacement to skin patch testing for titanium hypersensitivity. However, a standardized patch test is not yet available. An important aspect to testing is sensitization. Should allergy or sensitization susceptibility be tested? Specific immunoglobulin E (IgE) antibodies are produced after prior exposure to a substance that consequently becomes an allergen. Hence, should patch testing be repeated after 3 months to ensure that the original test has not sensitized the candidate to that substance? Is epicutaneous sensitization possible, or is intradermal testing mandatory? T-lymphocytes are constantly observed surrounding titanium debris in tissues. Titania microparticles can act as adjuvants to drive antigenic T helper 2 cell differentiation and the IgE response. Should titania microparticles be injected intradermally to rule out hypersensitivity?
A second remark that has to be made with concern to the use of titanium alloys is the material’s poor abrasion/wear resistance. While compression forces are on average 66 N/cm2 in a TMJ,12,38 shear forces do play a greater role. Even more so by restoring the lateral pterygoid muscle (LPM) function, as all movements, including protrusion and lateralization, remain simultaneously present. Despite the low forces generated, low friction and a hard coating are advised for the condylar head to prevent wear of the opposing fossa component, which usually consists of softer polyethylene.
The latter can be achieved by using a diamond-like carbon coating (DLC). 39 DLC is an amorphous carbon composed of a mixture of sp3 and sp2 carbon bonds with various levels of hydrogen. Coatings of materials within the DLC family can be fabricated based on hydrogen content, the addition of metallic and nonmetallic doping elements, the presence of interlayers, and the choice of bonding and deposition methods. These parameters can be controlled for the engineering of a broad range of thin (1–5 μm) coatings with a hardness of 8–80 GPa or higher. Diamond is the hardest known material to date, with 70–150 GPa Vickers hardness. The coefficient of friction, surface finish, and application temperature can also be manipulated. After the application of the coating, a polishing process can be used to increase the tribological properties of the prosthesis. 40
Delayed delamination from its substrate because of corrosion poses a serious issue for implant stability. Delamination occurs because of the dissolution of the silicon-adhesion-promoting interlayer and has been observed in noncemented hip prostheses. 41 Consequently, excessive wear of the polyethylene counterpart occurs. Interfacial and interlayer properties should, therefore, be carefully monitored.
HXLPE
Medical-grade, ultra-high molecular weight polyethylene (UHMWPE) wear debris (ie, small particles generated from articular surfaces in joint prostheses) often triggers an inflammatory response. 42 The infiltration of monocytes and the activation of fibroblasts and histiocytes into the pseudosynovial membrane lead to the production of chemokines, cytokines, and osteoclastogenic factors. Monocytes and macrophages differentiate into osteoclasts, which are responsible for osteolysis and loosening of the implant. The formation of submicron-size particles (<1.0 mm) leads to a higher proinflammatory cytokines production compared to particles that are larger than 1 mm, 42 which induce giant cell formation.
In order to limit the amount of UHMWPE wear, cross-linking can be achieved using ionizing irradiation. This leads to the production of free radicals that can recombine and form the cross-links. 43 While highly cross-linked UHMWPE (HXLPE) exhibits decreased volumetric wear, 44 the immune reaction to these HXLPE particles is higher than to conventional UHMWPE particles. Nevertheless, as there is a significant decrease in total particle volume, less inflammation and foreign body reaction occurs when using HXLPE, making it for instance preferrable to conventional polyethylene for hip prostheses. 45 HXLPE bearings exhibit a reduced incidence of aseptic loosening and osteolysis.
As previously discussed, the HXLPE-component has also been treated with tocopherol. Vitamin E, which acts as an antioxidant will prevent oxidation during compression molding, radiation cross-linking (due to γ-irradiation), and shelf storage. Furthermore, it will also protect the HXLPE from oxidation after implantation, and implantation as free radicals are generated in vivo by both cyclic loading and the reactions of lipids absorbed from the synovial fluid.43,46 As a result, HXLPE blended with vitamin E exhibits good resistance to fatigue wear. 47 However, important to remark is that, clinically, the addition of tocopherol has not been proven to be an asset, even when reduced total femoral head penetration was observed at a 3-year follow-up. 48
Tissue Integration
Another important aspect of an implant (endoprosthesis) besides biocompatibility is tissue integration. Osseous integration is the apparent direct attachment of bone to a biocompatible material without intervening tissue. A recent study 49 found that there is a direct relationship between the roughness of the titanium surface and the stimulation of bone formation, with pores measuring 600 μm (macro roughness) show greater bone ingrowth compared to a smaller (100–300 μm) pore diameter.50-53 Secondly, sandblasted, large-grit, and acid-etched (SLA) surfaces (micro roughness) show increased osseointegration compared to smooth surfaces. 50 It is believed that these mechanical and chemical abrasions induce the adsorption of fibronectin and other proteins that, in turn, trigger osteoblasts to form focal adhesions via an integrin-mediated mechanism.54,55 Removing surface contaminants while imparting wettability is equally useful and may trigger hard tissue formation as well.56-58 Further, plasma activation induces the initial adhesion of proteins and bone marrow cells. Unfortunately, steam sterilization after plasma activation completely removes this increase in wettability.
In comparison to osseointegration, soft tissue integration is less precisely defined. It is rather described as “a strong soft tissue-implant seal … with a thin capsule containing few inflammatory cells and fibroblasts … and collagen fiber orientation preferably oblique to the implant surface or randomly oriented”. 59 A surface roughness Ra value between .5 and 1 μm has been shown to induce soft tissue adhesion. Smoother surfaces, with the exception of acid-polished and anodized titanium (Ra = .2 μm), prevent adhesion. Micro-arc oxidation (also known as plasma electrolytic oxidation) significantly increases the percentage of soft tissue adhesion. 60 Similarly, a fibroblast growth factor-2/apatite composite coating applied by immersion (for 48 h) induced significantly less inflammation and yielded promising skin-screw interfaces. 61 Both processes have a low cost-effectiveness. 59
Heterotopic Ossification
A last point of discussion that has to be touched upon in light of reattachment of the LPM’s enthesis, is the occurrence of heterotopic ossification (HE). 62 HE is defined as “a heterogeneous disorder characterized by pathologic endochondral ossification with hematopoietic bone marrow in soft tissues, such as subcutaneous tissue, skeletal muscle, or fibrous tissue adjacent to joints”. 63 About 10% of HE cases result in limitations in range of motion. Once it develops, surgical removal is the only effective treatment, followed by local irradiation, which in turn may induce malignancy, and/or nonsteroidal anti-inflammatory agents to prevent recurrence. 64 A strong relationship between trauma (e.g., arthroplasty) and the involvement of multiple organ systems 63 seems to exclude the influence of the type of material or its surface characteristics.
An important question is whether the pores of the titanium scaffold must be filled with particulate bone, calcium phosphate, stem cells, or growth factors to enhance bone formation and guarantee bony union with the reattached enthesis. In a sheep model of TMJ replacement, postoperative function suggested that filling the scaffold with autologous bone chips was sufficient. 18 The addition of calcium phosphate may hinder reattachment, even if more bone will be formed within the pores, 65 and the addition of bone marrow-derived mesenchymal stem cells has not been clinically proven to enhance bony fusion.66,67
Even though an increase in movement capabilities can be seen by using this method, longstanding limitations of lateral movements cannot be undone by lateral pterygoid reattachment. Disuse atrophy of the lateral pterygoid muscle does not appear to be reversed by exercise. Supplementation with branched-chain amino acids and anabolic steroids was not investigated in that respect.
Conclusion
A careful analysis of the requirements for a successful TMJ replacement has led to the development of a new type of individualized, artificial joint that mimics both normal joint anatomy and function. Even though various features contribute greatly to optimal functionality and biocompatibility, the final outcome of the replacement will not only depend on these added features but also on the underlying disease and its duration, as well as on compliance with postoperative physiotherapy.
Even though a larger sample size (potentially with division between indications) is needed to have sufficient evidence on the added values of this prosthesis, the case series still supports further investigations on the use of the prosthesis.
Early clinical results are promising. Results in a sheep experiment and a small study series indicate that further clinical use is justified. Further long-term follow-up in a larger sample is planned for.
Supplemental Material
Footnotes
Acknowledgments
A.B. acknowledges the receipt of a starting grant from Internal Funds KU Leuven (STG/17/024).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Ir. Stijn Huys is the R&D officer at CADskills, which produces the patient-specific implants that were presented. Ing. Ruben Van de Sande is the production manager and Prof. Maurice Mommaerts is the innovation manager and co-owner of CADskills.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Baekeland scheme of the Flanders Agency for Innovation and Entrepreneurship (VLAIO), HBC.2017.0575.
Helsinki statement
The principles stated in the Declaration of Helsinki and its later amendments were adapted.
Informed consent
The authors certify that they have obtained all appropriate patient consent forms. On the form, the patients have given their consent for their images and other clinical information to be reported in this journal.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
