Abstract
In some individuals, tinnitus can be modulated by specific maneuvers of the temporomandibular joint, head and neck, eyes, and limbs. Neuroplasticity seems to play a central role in this capacity for modulation, suggesting that abnormal interactions between the sensory modalities, sensorimotor systems, and neurocognitive and neuroemotional networks may contribute to the development of somatosensory tinnitus. Current evidence supports a link between somatic disorders and higher modulation of tinnitus, especially in patients with a normal hearing threshold. Patients with tinnitus who have somatic disorders seems to have a higher chance of modulating their tinnitus with somatic maneuvers; consistent improvements in tinnitus symptoms have been observed in patients with temporomandibular joint disease following targeted therapy for temporomandibular disorders. Somatosensory tinnitus is often overlooked by otolaryngologists and not fully investigated during the diagnostic process. Somatic disorders, when identified and treated, can be a valid therapeutic target for tinnitus; however, somatic screening of subjects for somatosensory tinnitus is imperative for correct selection of patients who would benefit from a multidisciplinary somatic approach.
Keywords
Introduction
Tinnitus is defined as the perception of sound without an accompanying external auditory stimulus. This condition affects millions of people. A recent meta-analysis and systematic review by McCormack et al. 1 covering 40 manuscripts and 39 different studies revealed that the prevalence of tinnitus ranges from 5.1% to 42.7%. Tinnitus is commonly observed in individuals above the age of 60 years and affects 8% to 20% of the elderly population; however, chronic tinnitus can occur at any age. 2 Approximately 1% to 2% of the populations in Western industrialized countries reportedly experience incessant tinnitus, 3 sometimes with severe consequences on quality of life. 4
Subjective tinnitus, often perceived as a nonspecific buzzing, tonal sound, hissing, humming, ringing, or roaring, can be triggered by a variety of causes. In most cases, no single factor such as chronic progressive hearing loss is considered sufficient to elicit tinnitus; instead, symptoms develop when more than one factor act synergistically.5–11 An increased prevalence of tinnitus is associated with hearing loss, which can be diagnosed in up to 90% of patients with tinnitus, and with ototoxic drug use, infections, psychological stress, and a range of medical conditions that can affect hearing function.3,5,12–16 Such patients are considered to have “otic tinnitus.” 17
Tinnitus can also be evoked or modulated in some individuals by inputs from the somatosensory, somatomotor, and visual–motor systems following interactions with the auditory system.6,18–21 This has led to the term “somatosensory modulation of tinnitus.” 18 These interactions can modulate the psychoacoustic attributes of tinnitus, such as loudness and pitch, in a temporary manner. Such modulations may occur immediately following stimuli such as forceful muscle contractions of the temporomandibular joint (TMJ), head and neck, and limbs;20,22,23 vertical or horizontal eye movements;24–26 pressure application on myofascial trigger points; 27 cutaneous stimulation of the hands or fingertips; 28 electrical stimulation of the median nerve and hand; 29 finger movements 30 ; head rotation; 31 orofacial movements; 32 transcranial direct current stimulation; 33 and intracochlear electrical stimulation. 34 Somatosensory modulation of tinnitus may be elicited with or without concomitant somatic disorders. 18 When tinnitus appears to be preceded or strictly linked to an underlying somatic disorder and therefore related to problems of the musculoskeletal system rather than of the ear, the term “somatic tinnitus” or “somatosensory tinnitus” has been proposed. 20
The association between tinnitus and the anatomical relationship of both the somatosensory and auditory pathways has been extensively studied.3,11,35-40 Research on animal models indicates that the integration of auditory and somatosensory afferents occurs as early in the auditory pathway as in the cochlear nucleus (CN), at the site of convergence of the projections from the auditory nerve and trigeminal and dorsal column ganglia and brain stem nuclei. 3 Development of ipsilateral tinnitus in response to somatic injury supports the hypothesis of a possible somatosensory component in the origin of tinnitus.18,22 The anatomical and physiological characteristics of somatosensory tinnitus are elaborated in more detail in a later section of this review.
Anatomical and physiological basis of somatosensory tinnitus
The possibility of somatic modulation of the loudness and pitch of tinnitus has prompted a search for neural connections between the auditory and somatosensory systems.36–38 The pathophysiologic background of somatosensory tinnitus is still partly unclear; however, theories have been proposed based on anatomical and physiological evidence.
Experimental studies have demonstrated the presence of auditory connections to the dorsal column and trigeminal systems, where the dorsal root and trigeminal ganglia cells send axonal projections that terminate in the CN. 3 These projections, along with those from the brain stem somatosensory nuclei, terminate primarily in the granule cell domain of the CN that surrounds the ventral CN (VCN) and extends into the second layer of the dorsal CN (DCN). 36 Some researchers have documented excitation of the VCN neurons in the absence of sound upon stimulation of the trigeminal ganglion, 39 and others have documented both excitation and inhibition in DCN neurons. 40 The localization and response characteristics of these units upon stimulation of the trigeminal ganglion are consistent with those of the fusiform or giant cells 41 in the DCN and bushy or stellate cells in the VCN. 39
Somatosensory stimulation can affect both sound-driven and spontaneous firing rates following cessation of the stimulation, which might be a result of long-term potentiation or depression. 42 Preceding an acoustic stimulus by electrical stimulation of somatosensory pathways can alter the spike timing of the sound-evoked response and synchrony of firing between neurons in the DCN, an additional proposed correlate of tinnitus. 43 Most importantly, a trigeminal stimulus preceding an acoustic stimulus can modulate the firing rates as well as the temporal response patterns to the sound.17,44
According to the leading animal model theory proposed by Shore et al., 36 cochlear damage has been hypothesized to trigger somatosensory tinnitus by inducing aberrant neural changes in central auditory structures following deafferentation due to an increase in the spontaneous firing rates in the DCN and VCN neurons 45 and an upregulation of excitatory nonauditory projections. 46 Previous studies have revealed that fusiform cells show increased spontaneous firing rates in the DCN47–49 after deafening, which might be a result of upregulation of the glutamatergic somatosensory innervation of both granule and magnocellular cells in the CN 50 or changes in glycine receptors, unmasking the fusiform cell excitability. 51 Increased spontaneous firing rates in these groups of neurons can be caused by increased synchrony of firing between neurons, 52 which is generally confined to a restricted region of cochlear damage and maximal at frequencies above the traumatizing frequency. 53
Animal experiments have revealed that at 1 and 2 weeks after unilateral cochlear ablation, the number of vesicular glutamate transporter 2 (VGLUT2+) terminals receiving somatosensory inputs increases while the number of vesicular glutamate transporter 1 (VGLUT1+) terminals decreases, indicating an enhanced somatosensory influence on the CN after decreased auditory nerve innervation.3,50 This alteration in the balance of inputs from auditory and somatosensory structures could affect bimodal integration and strengthen the somatosensory inputs. 36 These changes cause the DCN neurons to become more responsive to somatosensory stimulation following cochlear damage, 48 with decreases in the latencies and thresholds to somatosensory stimulation and enhancement of bimodal integration. These results are in agreement with those of studies in humans, in whom the tinnitus frequency is correlated with the edge frequency of the audiogram, the frequency of the greatest threshold shift, or the frequency range of the hearing loss.3,54
Although not directly relevant to tinnitus, tractography (a technique used to visually represent neural tracts) has provided evidence of connections between the primary auditory cortex (A1) and secondary somatosensory cortex (SII) that could represent an alternative route to the ascending auditory system highlighted in animal models. 55 Using functional magnetic resonance imaging, Beauchamp and Ro 55 examined the neural substrates of acquired auditory-tactile synesthesia in a patient with a lacunar infarct to the right VLN of the thalamus. The imaging examination demonstrated a double dissociation in the patient’s secondary somatosensory cortex with increased responses to auditory stimulation and decreased responses to somatosensory stimulation. These findings suggest that stroke-induced plasticity can result in abnormal connections between sensory modalities that are normally separate and that synesthesia can be caused by inappropriate connections between nearby cortical territories.
In summary, it appears that neuroplastic changes initiated by damage to either the somatosensory or auditory input pathways to the DCN may result in compensatory shifts of excitation and inhibition. These changes are reflected in the upregulation of glutamatergic inputs from somatosensory pathways after deafening and increased sensitivity of DCN neurons to somatosensory stimuli. Thus, somatic tinnitus could be a result of increased spontaneous firing rates of specific sets of neurons that are excited by somatosensory inputs.36–55
Clinical features of somatosensory modulation of tinnitus
The perception and intensity of tinnitus can be somatically modulated in a subpopulation of individuals. As previously discussed, somatosensory modulation of tinnitus originates from the complex somatosensory–auditory interactions arising from musculoskeletal anatomic regions such as the TMJ, craniocervical junction, cervical vertebrae, and neck and shoulder muscles.56–61
Comparison of previous studies on somatic modulation of tinnitus. The average prevalence of modulation was 69%. The main somatic regions resulting in tinnitus modulation were the temporomandibular joint and head and neck region, followed by the eyes and limbs.
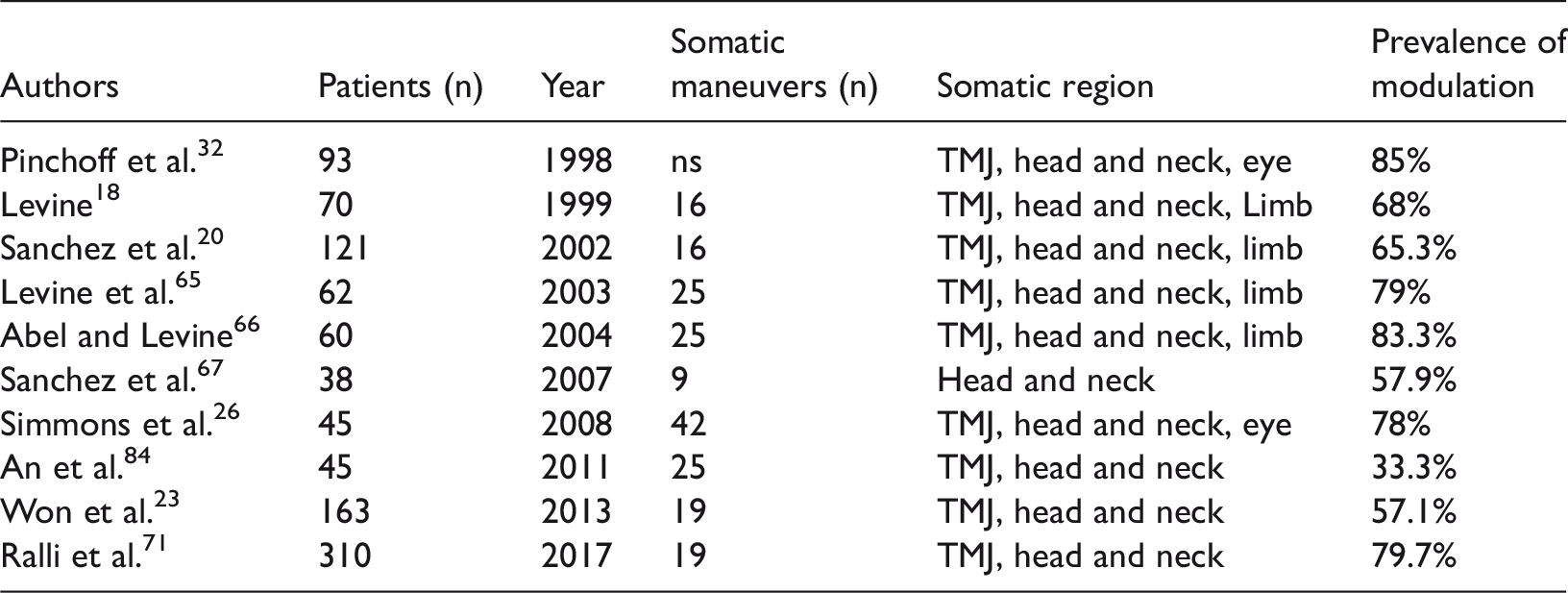
TMJ, temporomandibular joint
Large variations have been reported in modulating the loudness and pitch of tinnitus. Individual diurnal fluctuations in loudness perception have also been described. Some patients experience louder tinnitus upon awakening, while others experience the absence of tinnitus upon awakening with a return of tinnitus during the day; this might be related to somatic factors such as stretching of the neck muscles when the head passively falls forward while sleeping in a sitting position. 58
Because tinnitus is a heterogeneous condition, subtyping of different forms of tinnitus has been proposed for appropriate clinical diagnosis and management. 62 Ward et al. 63 recently investigated the characteristics of somatic tinnitus within a large UK cohort. The authors aimed to determine the prevalence of somatosensory tinnitus and whether it was linked to a specific hearing loss profile, etiology, or other characteristics using a model prediction method. The authors found that the prevalence of a self-reported ability to modulate tinnitus was 16% and that somatic tinnitus was significantly predicted by age, pulsatility, loudness, and TMJ complaints. Among individuals with somatic tinnitus, a high proportion had pulsatile tinnitus, were younger than 40 years, reported variation in the loudness of their tinnitus, and reported TMJ disorders, thus profiling somatic tinnitus as a distinct subtype of general tinnitus. 63
Temporomandibular joint, head and neck, and eye movements that modulate tinnitus
A large percentage of individuals with tinnitus report tinnitus modulation by TMJ and head and neck maneuvers, while very few report modulation by manipulation of the extremities. This suggests that the auditory pathway inputs from the cranial nerves and upper cervical region of the spinal cord are more important in modulating tinnitus than the inputs from the caudal spinal cord. 22
The percentage of positive tinnitus modulation sorted by somatic region based on several recent studies is shown in Figure 1. The TMJ is the most frequent tinnitus-modulating region. Rubinstein
68
reported modulation of tinnitus in one-third of subjects who performed jaw movements or applied pressure on the TMJ. In 1999, Levin
18
conducted a systematic study of 70 consecutive patients and reported that 68% of the patients could somatically modulate their tinnitus with head or neck maneuvers. Furthermore, when TMJ maneuvers were added in another study by Levine et al.,
65
80% of the patients could somatically modulate their tinnitus. This finding was confirmed by Sanchez et al.,
20
who obtained similar results (65%). In an analysis of 163 patients at National University in Seoul, Won et al.
23
reported that all jaw muscle maneuvers, especially clenching of the teeth and opening of the jaw with or without pressure, increased tinnitus loudness.
Percentage of patients with positive tinnitus modulation sorted by somatic region based on the review of the literature listed in Table 1. An average of 69.4% of patients with tinnitus showed some degree of modulation, while 30.6% reported no modulation. The region with the highest degree of modulation was the temporomandibular joint, followed by the head and neck region, limb, and eye. TMJ, temporomandibular joint.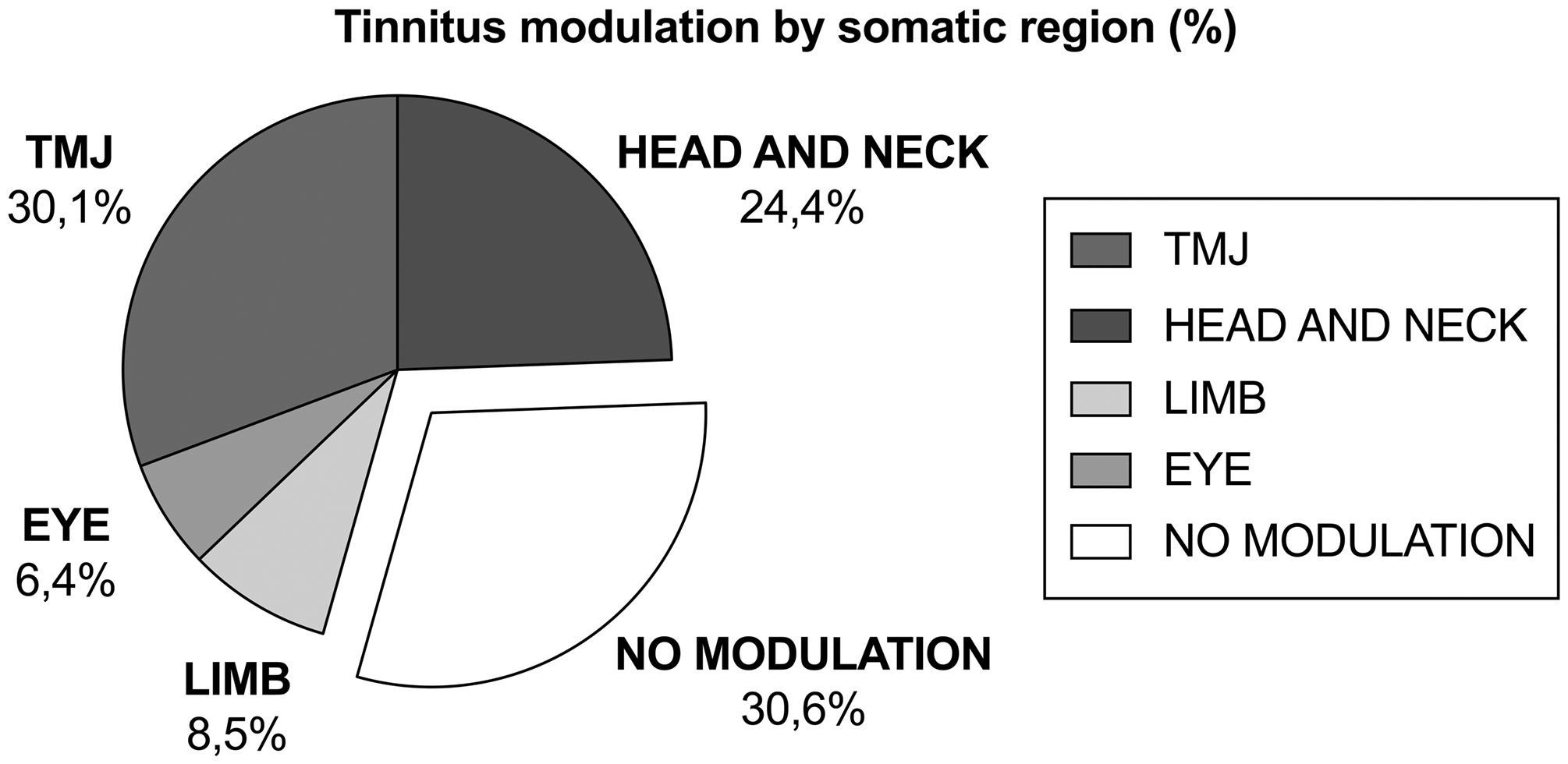
The head and neck is the second most frequent tinnitus-modulating region. Kapoula et al. 69 reported that 14 of 23 patients (61%) examined in their clinic could modulate their tinnitus with jaw movements, 10 (43%) with head movements, 9 (39%) with muscle pressure, 3 (13%) with eye movements, and 2 (9%) with a global muscular effort. In one study, application of head and neck maneuvers revealed that 41% of patients could only increase their tinnitus loudness, 17% could only decrease their tinnitus loudness, and 10% could either increase or decrease their tinnitus loudness depending upon the maneuver. 22
One of the first cases of modulation of tinnitus by shifting the eye gaze from a straight to side gaze in the right and left directions as a complication of acoustic neuroma surgery was reported by Whittaker 24 in 1982. Since then, many similar cases have been reported by other authors.25,70 While recruiting patients for their imaging study on somatosensory tinnitus, Simmons et al. 26 found that 87 of the 113 responding patients who had undergone acoustic neuroma surgery had developed gaze-evoked or gaze-modulated tinnitus. Furthermore, in 95% of the patients, tumor removal resulted in complete hearing loss in the affected ear, indicating a higher prevalence of gaze-induced tinnitus than previously thought. In another study, most patients (77%) experienced tinnitus in the ear or the side of the head on which the tumor was removed, and no patient experienced tinnitus exclusively in the ear or side of the head opposite the side of tumor removal. 19 The lateral gaze resulted in increased loudness of tinnitus in approximately 99% of patients with acoustic neuroma, and most patients also reported an increase in the tinnitus pitch with eye movements (approximately 89%). 19 Furthermore, the loudness of gaze-invoked tinnitus could be modulated by jaw movements in 22% of patients with acoustic neuroma in another study. 21 Interestingly, Simmons et al. 26 also found that preexisting tinnitus was a major risk factor associated with the development of gaze-invoked tinnitus in patients who had undergone acoustic neuroma resection.
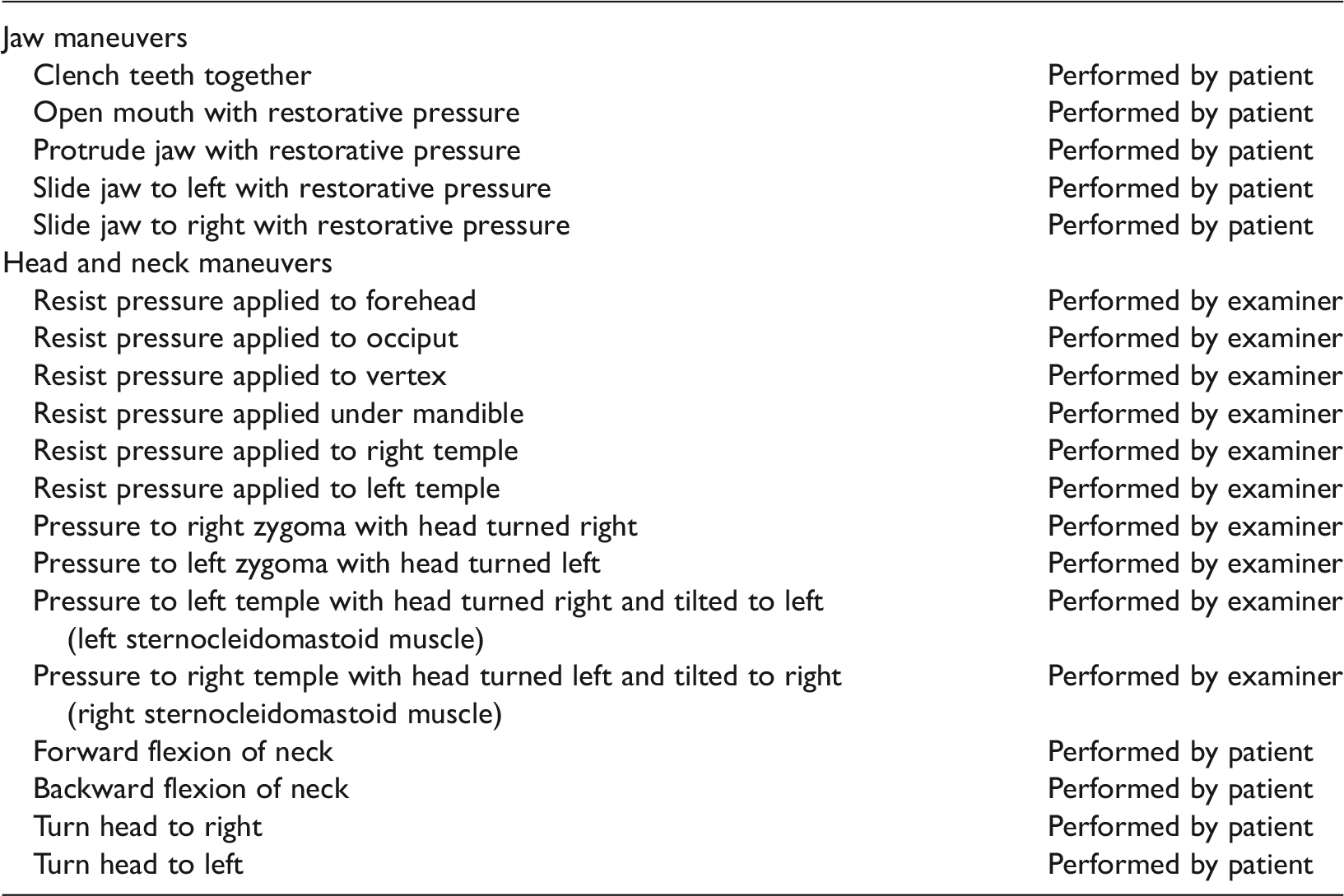
Maneuvers used for somatic testing in a previous study from our group (Ralli et al., 2017).

Characteristics of tinnitus modulation in patients with somatic tinnitus described in a recent paper from our group (Ralli et al., 2017), sorted by target somatic region (temporomandibular joint vs. head and neck). Temporomandibular joint maneuvers significantly increased tinnitus loudness, while head and neck maneuvers increased loudness in 58.3% of patients and decreased it in 41.7% of patients. TMJ, temporomandibular joint.
Association between tinnitus and somatic disorders
The association of tinnitus with signs and symptoms of TMJ disorders has been reported by many authors.20,64,72–78 Epidemiological studies have documented that patients with concurrent tinnitus and TMJ problems are younger and more often female and have better hearing function than patients with tinnitus but without TMJ symptoms. 73
Improvements in tinnitus symptoms upon treatment of TMJ disorders have also been described.73,79,80 Tullberg and Ernberg 81 reported that 2 years after treatment of TMJ disorders, 43% of patients in the treatment group had decreased tinnitus compared with only 12% of patients in the control group. In a cross-sectional analysis of data from the Study of Health in Pomerania (SHIP 0), TMJ disorders were found to be the strongest predictor of tinnitus apart from headache. De Felicio et al. 82 observed an improvement tinnitus symptoms in patients with TMJ disorders after using bite splints for a period of 8 weeks, and Wright and Bifano 80 reported improvements in tinnitus symptoms in patients who had undergone cognitive therapy, use of bite splints, and home exercises for the treatment of TMJ disorders. Vielsmeier et al. 61 investigated the possibility of identifying the presence of comorbid TMJ complaints with a subtype of tinnitus and found that patients with concurrent tinnitus and TMJ complaints (22% of 1204 patients analyzed) were significantly younger, had a lower onset age, and were more likely to be female. Furthermore, patients with TMJ complaints could modulate or mask tinnitus more frequently by somatic maneuvers and by music or sound stimulation. 61
In a cross-sectional study, Bernhardt et al. 72 investigated whether the symptoms associated with TMJ disorders could act as a risk factor for the development of tinnitus in a set of 3134 patients. Among 191 patients with pain upon palpation of the TMJ, 24 (12%) were diagnosed with tinnitus after 5 years with a risk ratio of 2.4 (95% CI, 1.6–3.7) after adjustment for sex, age, education level, and frequent headache. Buergers et al. 60 reported that the incidence of tinnitus was more than 8-fold higher in patients with than without TMJ disorders (36.6% vs. 4.4%, respectively). They also demonstrated that stomatognathic therapy including intraocclusal stabilization and physiotherapy (passive muscle stretching and massaging of the affected masticatory elevator muscles, thermotherapy with moist heat, traction of the TMJs, and coordination exercises) improved tinnitus symptoms in 11 (44%) of 25 participants. More recently, in a retrospective cohort study of patients with tinnitus, Lee et al. 77 observed a 3.22-fold higher risk of developing tinnitus within the 3-year follow-up period (95% CI = 2.67–3.89). Manfredini et al. 83 found many correlations between tinnitus and TMJ disorders. They reported a tinnitus prevalence of approximately 30% in patients with TMJ disorders.
Comparison of results of previous studies on tinnitus changes following temporomandibular joint (TMJ) therapy. Sixteen studies published from 1964 to 2016 were found in the literature. The average rate of TMJ disorders among patients with tinnitus was 44%; after TMJ disorder treatment, 69% of patients reported improvement or resolution of their tinnitus, while 32% reported no changes.

TMJ, temporomandibular joint
Future clinical and research perspectives
The increased prevalence of bruxism, TMJ dysfunction, and neck pain associated with increased modulation of tinnitus by somatic maneuvers reported by many authors highlights the role of somatosensory afferents, depicting somatosensory tinnitus. While these findings are interesting, many questions remain regarding the clinical approach to patients with somatosensory tinnitus.
How close is the association between the capability to modulate tinnitus following somatic maneuvers and the presence of a somatic disorder? Evidence of this association could help to identify underlying somatic disorders in patients with tinnitus with modulation capabilities. Apart from the presence of underlying somatic disorders, are there specific individual demographic or tinnitus-related factors, such as age, sex, or type of tinnitus, that increase the chance of tinnitus modulation? Identification of a somatic tinnitus profile could help to facilitate patient selection during somatic testing. What is the relationship between tinnitus modulation following specific maneuvers and the efficacy of somatic treatment focused on the regions evoking modulation? The changes in tinnitus pitch and loudness that occur after somatic maneuvers are mainly transitory; however, some patients report that such changes last for longer periods of time after manipulation. Could this represent a basis for long-lasting effects of manual therapies?
Current evidence supports a link, mainly for TMJ disorders, between the presence of a somatic disorder and higher modulation of tinnitus, especially in patients with a normal hearing threshold. However, the presence of such a correlation is still debated.20,61,79,84 The severity of tinnitus is reportedly related to the severity of TMJ pain; 74 patients with tinnitus who have been diagnosed with a TMJ disorder have a higher chance of modulating their tinnitus with somatic maneuvers in the TMJ region61,73; furthermore, the prevalence of tinnitus appears to be higher in patients with than without TMJ disorders.64,73–78,85 In a study from our group described in a previous section of this review, 71 we focused on the correlation between a positive history of a somatic disorder and positive tinnitus modulation, paying special attention to whether such a correlation was present within the same somatic region. We found a significant association within the same patients between a positive history and positive tinnitus modulation in the same region, mainly for the TMJ in patients with unilateral tinnitus (76.2%) and for the TMJ and head and neck region in patients with bilateral tinnitus (56.0%) (p < 0.001). 71 This confirms the presence of the correlation and supports the findings of other authors. Such a connection could help identify, among patients with tinnitus, those with underlying head and neck dysfunctions that could play a role in their tinnitus and who could benefit from further multidisciplinary investigation and physical therapy. In these cases, it is important to seek the cooperation of other specialists such as dentists, gnathologists, osteopaths, orthopedic specialists, and physiotherapists for a second-level evaluation of a possible disorder affecting the musculoskeletal system and begin focused treatment.5,71
Identification of specific individual demographic or tinnitus-related factors that increase the chance of tinnitus modulation can be helpful in the management of patients with somatosensory tinnitus. In a previously discussed study, Ward et al. 63 reported that among individuals able to modulate tinnitus, a higher proportion had pulsatile tinnitus and were under the age of 40 years. Pinchoff et al. 32 found a strong male preponderance among people able to modulate tinnitus. This was not consistent with the findings reported by Won et al., 23 who found that female sex was more closely associated with modulation. In a study from our group, 71 we did not find strong statistical significance of specific variables. However, some factors seemed to be more prevalent in patients with positive modulation: a high-pitched tinnitus sound was present 2.4 times more frequently than other tinnitus sounds, and male patients had a 1.4- to 2.0-times greater chance of tinnitus modulation than female patients; no differences were found in tinnitus onset or age. The differences found among these studies make it difficult to hypothesize the presence of common demographic or tinnitus-related factors in patients who are able to modulate their tinnitus, a characteristic that seems more closely related to somatic components such as the presence of musculoskeletal disorders than to specific demographic profiles.
Several therapeutic approaches have been proposed to treat somatosensory tinnitus, such as stabilization splints for TMJ disorders, exercises that include repetition of movements that individually modulate tinnitus, 29 relaxation of muscle tension in the head and neck, 86 cervical manipulation, 87 deactivation of myofascial trigger points by injection of local anesthetics such as lidocaine, 88 laser therapy, pressure release, transcutaneous electrical nerve stimulation, and magnet therapy. 89 In our opinion, although some of these treatments may have a positive effect on tinnitus, their effect depends on correct identification of the underlying somatic disorder, when present. In our experience, the most efficacious treatments are stabilization splints when a TMJ disorder is present and osteopathy sessions with cervical manipulation when a head and neck muscle disorder is present. 71
Conclusion
Somatosensory tinnitus is certainly a widespread condition, and further animal studies are required to better understand its pathophysiologic basis. Human studies are necessary to investigate the clinical correlates and determine to what extent a correct diagnosis and treatment of a possible underlying somatic disorder could contribute to the management of this subtype of tinnitus. Somatic disorders have been shown to play a central role in a large portion of patients with tinnitus and, when correctly identified and treated, can represent a valid therapeutic option. However, accurate screening of patients for somatosensory modulation of tinnitus is imperative to correctly select patients who would benefit from a multidisciplinary somatic approach.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
