Abstract
Keywords
Introduction
Facial fractures are a common consequence of trauma, affecting individuals across various age groups and demographics.1-3 These fractures can have significant physical, functional, and psychological consequences for the affected individuals. 3 As such, primary treatment goals should focus on enhancing quality of life and helping these patients regain a sense of normalcy by restoring form and function. 4 To achieve this, it becomes crucial for the surgeon to both thoroughly understand the trauma and implement the most appropriate surgical management that will lead to optimal results and recovery. 5
One of the significant breakthroughs in craniofacial surgical management has been the widespread use of 3D imaging and virtual surgical planning (VSP).6-10 It has become a reliable tool for elective craniofacial surgery in both adult and pediatric patients. VSP enables surgeons to simulate and plan procedures, optimizing the placement of implants and the restoration of facial harmony prior to exposing the fractures themselves. Adoption of VSP has been able to improve efficiency, accuracy, and predictability in these procedures through operative simulation and production of guides and custom 3D plates that reproduce the expected gaps and demonstrate the 3-dimensional movements encountered intraoperatively.6-10
In facial trauma, traditional management has relied on 2-dimensional (2D) photos, computed tomography (CT) scans, and dental impressions to guide fracture repair. A typical workflow for craniofacial trauma reduction begins with obtaining imaging, with CT scans being the gold standard, along with dental impressions if occlusal involvement is present. Next, surgical exposure is performed to visualize all fractures and assess necessary interventions. The reduction is then carried out sequentially, starting with the most stable fragments and working toward the most unstable areas. This method involves referencing the preoperative CT scan and intraoperative findings, while balancing reduction of bone fragments, maintaining good exposure, and bending plates to fit the contours of the fracture before drilling.1,4 Relying on these traditional methods, which are already complex, becomes even more challenging in a surgical field characterized by limited visualization, bleeding, bony comminution, loss of 3D architecture, and obscured landmarks. It also requires the efforts of skilled surgeons and assistants to contour plates while maintaining the spatial orientation of comminuted fragments, a time-consuming process that can lead to inaccurate and suboptimal results.
Technology, specifically VSP and 3D printed operative deliverables, has been described in isolated regions of the craniofacial skeleton, specifically for orbital and zygomatic fractures.6,8,9,11-17 However, while we recognize these initiatives have shown benefits in isolated areas, there are other areas of significant challenges for accurate reduction and fixation, such as nasoorbitoethmoid (NOE), complex comminuted occlusal fractures involving both maxilla and mandible, and particularly when multiple areas of the facial skeleton are involved, for example, NOE together with a zygomaticomaxillary complex (ZMC) fracture. Traditional methods for reducing these complex fractures, even under correct operative sequencing, carry the risk of improperly reduced fragments propagating errors throughout the entire sequence. 18 While technology offers potential solutions, its adoption has been limited due to high costs, time constraints, and challenges in visualization and plate contouring in vivo due to obstructing soft tissue.
In this study, we introduce a comprehensive protocol- virtual surgical planning with intraoperative deliverables (VSPD)- integrating established key components as well as additional steps to enhance surgical precision and outcomes. Our approach incorporates VSP web sessions for identifying and reducing fractures, generation of operative deliverables that include a reduced/perfected skull model, intraoral splints, and intraoperative plate bending on the 3-dimensional printed template using standard plating sets. Additionally, we employ plate etching to denote fracture lines, which facilitates plate registration in vivo. We present our experience using the VSPD protocol, utilizing 3 representative cases of our larger patient series to demonstrate the technique. In a subsequent manuscript, results from the use of this protocol in varying regions of the craniofacial skeleton will be analyzed. We aim to demonstrate that utilizing technology can enhance the efficiency and accuracy of facial fracture management for complex trauma.
Methods
We performed a retrospective analysis of all patients who underwent craniomaxillofacial surgery at our institution between January 1, 2020, and January 1, 2025. The IRB approved this study under the ID: STUDY00005292. Inclusion criteria were patients 18 years of age or older who underwent craniomaxillofacial surgery for traumatic injury at our institution between the specified dates. Patients were excluded if they were evaluated but did not receive an operation by a craniofacial surgeon within our institution, if they were pediatric patients under the age of 18, or otherwise considered a vulnerable population (confidential inmates, pregnant patients, etc.). Patient demographics, medical history, primary injuries, surgical treatments, complications, and reoperation information were collected. To describe our virtual surgical planning with intraoperative deliverables protocol, we chose 3 representative cases from the larger cohort that demonstrate different craniofacial regions, levels of complexity, and the unique advantages of using this technique.
Treatment Protocol
The steps of the complete VSPD treatment protocol are represented in Figure 1. Upon admission, CT craniofacial imaging was performed utilizing 1 mm cuts and 3D reconstructed DICOM (Digital Imaging and Communications in Medicine) images. Images were sent to 3D Systems® (USA) and Stryker® (USA) for segmentation and preparation of the webinar. Web-based planning sessions were held with the surgeon and engineer (typically within 48 hours). All fractures were identified, and the anatomy reviewed. Once fracture segmentation was completed, a 2-part process was used to achieve the “best form.” This involved a virtual attempt to reduce the fracture fragments. If the fractures were unilateral, then the use of mirror imaging and blending with the ipsilateral skeleton helped to assess proper reduction. A combined approach was used to achieve the best results in reduction, known as a reduction/perfection model. In bilateral trauma cases, sized cohort images were superimposed as a model for proper fracture reduction. Validity of virtual reduction was checked by assessing occlusion, symmetry, and overall appearance of the craniofacial skeleton, so that appropriate virtual perfected morphology was achieved. Optimal occlusion was achieved by identifying the best combination of bone reduction, intercuspation, and, if possible, centric relation.
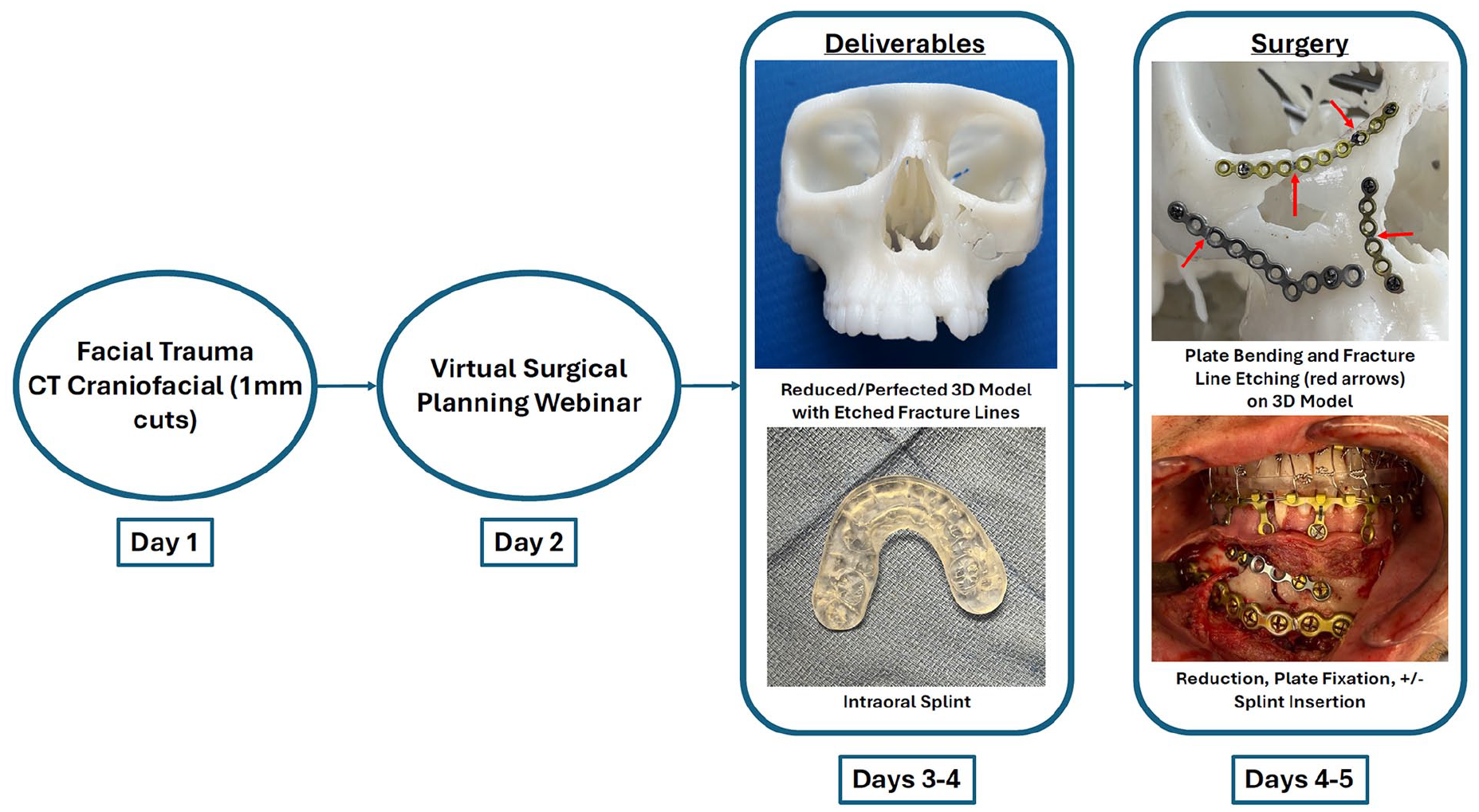
Workflow diagram illustrating VSPD protocol.
Three-dimensional models were generated based on the reduced/perfected model obtained during the VSP session. After surgeon approval of the final plan and manufacturability checks, the models were printed with marked fracture lines. Occlusal splints were also generated, if indicated. Expedited turnaround time for delivery of the virtually planned reduction/perfection sterilizable models and splints was as little as 48 hours after the VSP webinar.
During the surgery, there was an exposure table and a back table, which held the plates and splints. Standard surgical incisions were used, dictated by the fracture patterns involved and the amount of exposure needed to ensure adequate reduction and fixation; existing lacerations or previous scars were used when possible. For cases involving LeFort or mandibular fractures, arch bars were placed, standard Erich or hybrid mandibulomaxillary fixation (MMF), and patients were put in rigid occlusion using occlusal splints to ensure the patient was in proper occlusion. (Figure 2) Once adequate exposure was achieved, the fractures were reduced, working from most stable to most unstable. The surgeon then moved to the back table to prepare the plates. The 3D printed models, as mentioned above, were sterilized and available on the field prior to the start of the case, as well as any occlusal splints. Stryker® (USA) craniomaxillofacial standard plating sets were utilized. On the back table, the appropriate plate to span a given defect was selected, cut, and shaped to the appropriate length and contour as measured against the 3D printed perfected model. The contoured plates were secured to the model to achieve exact conformation. A 1.1 mm fissure bur was used to etch the fracture lines onto the plate to ensure correct placement in vivo, especially in areas of spanning fractures or comminuted thin maxillary injuries. When possible, obvious and stable bony landmarks were utilized to have an additional reference point for proper plate registration when transferring from the model to in vivo, for example, using the very edge of the orbital rim. Pictures were taken of the plates on the models to be used as a reference for placement in vivo. The plates were secured with corresponding mono-cortical screws, or bi-cortical in the case of mandible fractures. After plating, soft tissue was resuspended and incisions closed per standard practice. We assessed the reduction by obtaining craniofacial CT scans (1 mm cuts) 2 days postoperatively. The patients were followed up in the clinic weekly for the first 2 to 3 weeks postoperatively, then at the 1-, 3-, and 6-month marks.
Reduced virtual planning images illustrating occlusal splint based on CT data (left), Intraoperative photo demonstrating use of 3D printed splint from VSP (right).
Results
We have used this VSPD protocol on a large patient series since its inception 5 years ago. Three representative patient cases were analyzed, showcasing the diverse range of fractures that can be effectively treated with this protocol. Results demonstrated that reduction/perfection models marked with fracture lines and occlusal splints were useful to transfer the virtual plan surgically. Our approach was successfully applied in a regionally comprehensive manner throughout the craniofacial skeleton to include frontal, orbital, zygomatic, mandible bones, and occlusion. No revisions were required for any of the cases. No postoperative complications presented.
Clinical Presentation—Case 1
A 32-year-old male presented after a firework blast trauma with a comminuted left ZMC, left LeFort II, left NOE fractures, and left orbital floor and medial wall fractures. (Figure 3). During virtual surgical planning, fracture fragments were reduced with mirror imaging and blended with the ipsilateral side, thus creating a reduction/perfection image. Operative deliverables delivered within 72 hours of the planning session included a perfected skull model with marked fracture lines. The surgical approach was completed with lynch and gingival buccal sulcus incisions, along with the patient’s periorbital and lower eyelid traumatic lacerations. Standard upperface and midface plates were bent and secured to the perfected model to achieve exact adaptation. A 1.1 mm fissure bur was then used to etch the plates at each fracture site, creating real-time custom 3D plates with fracture location registration to guide correct placement in vivo. The fractures were reduced and fixated in the periorbital area and medial and lateral buttresses of the Le Fort II and zygomatic fractures. Total operative time was 4 hours and 17 minutes.
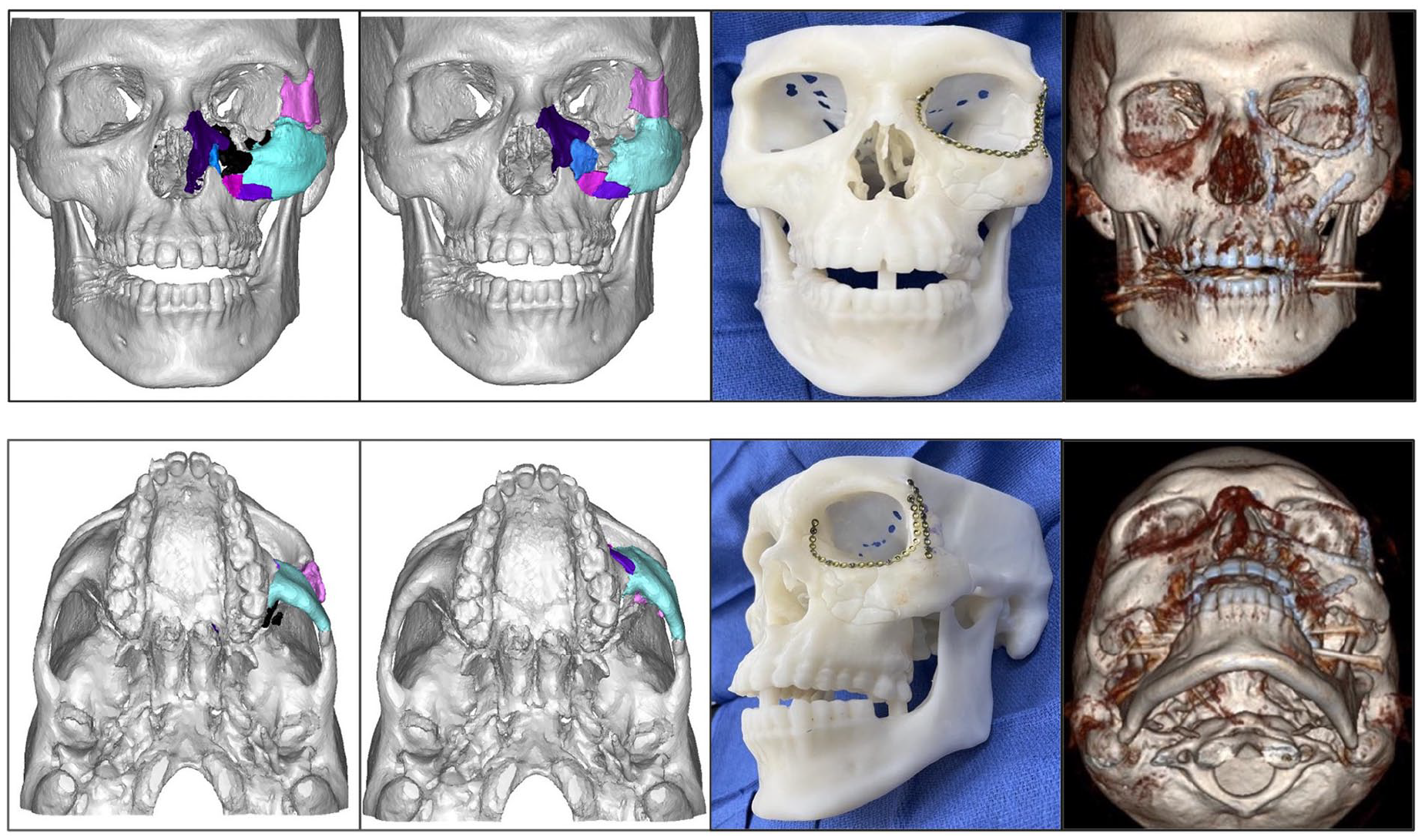
From left to right: VSP (unreduced and reduced), operative deliverable showing prebent plate and fracture etching, and postoperative 3D scan.
The post-operative scan revealed good anatomic reduction and adherence to the VSP plan. Orbital reconstruction was delayed due to initial post-traumatic and surgical swelling at the time of initial reduction/fixation. The patient underwent definitive orbital floor and medial wall reconstruction with a Medpor 3D Titan implant 2 weeks postoperatively. There were no postoperative complications.
Clinical Presentation—Case 2
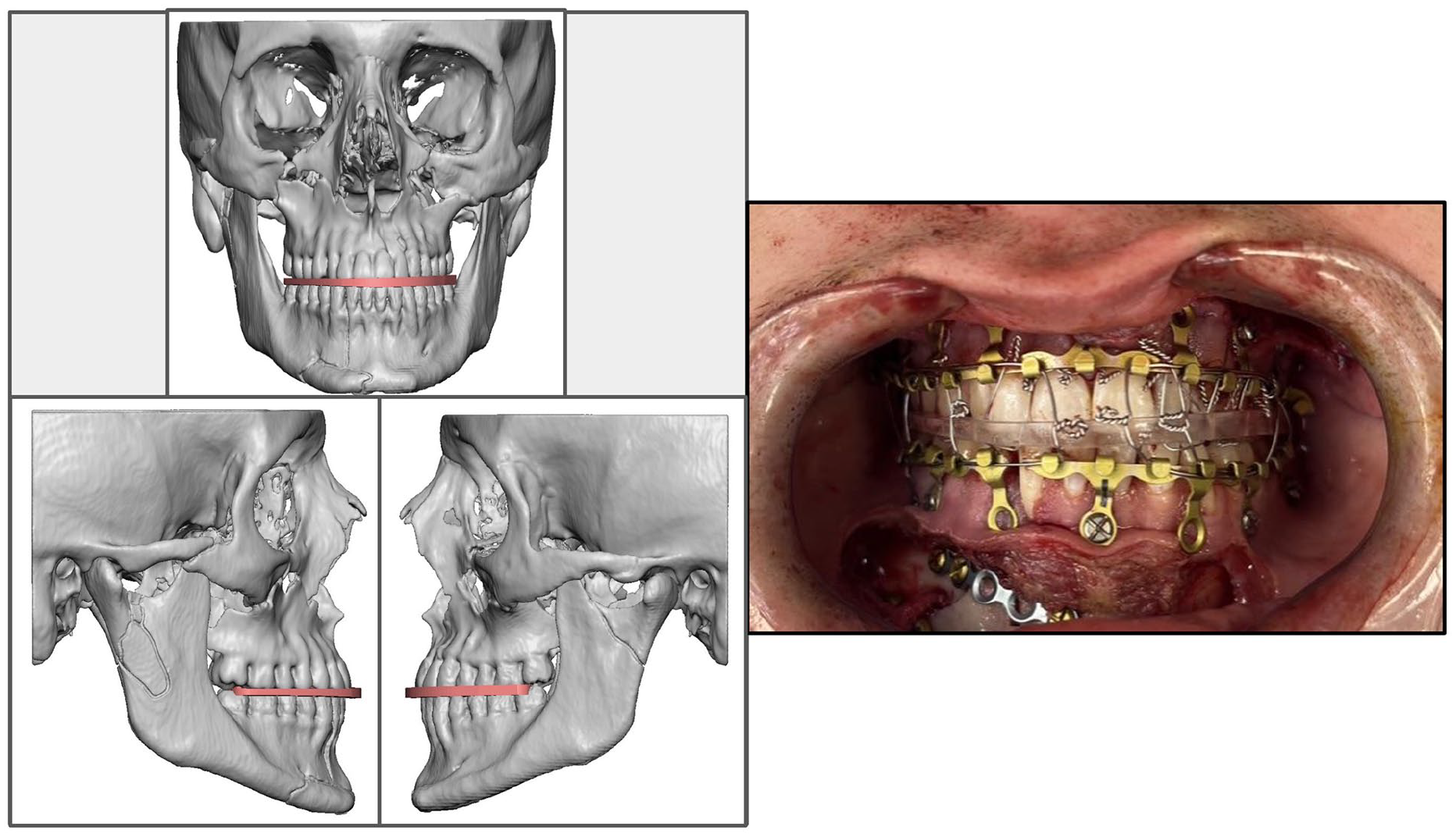
A 41-year-old male presented after a motor vehicle collision with a comminuted left subcondylar mandibular fracture (Figure 4). VSP was carried out 48 hours after the initial CT scan. Proper reduction was assessed using his virtual occlusion and checking the condyle position in the fossa. There was premature contact on the affected side, with mandibular deviation to the left upon opening. The splint was designed based on maximum intercuspation, condylar seating, and bone reduction, enabling ideal occlusion. Operative deliverables were delivered within 72 hours, including a perfected mandible model with marked fracture lines and an occlusal splint. During surgery, the occlusal splint was inserted before MMF. A retromandibular transparotid approach was utilized to expose the subcondylar fractures. The mandible strut plate was bent and secured to the 3D reduction model, and fracture lines were etched with the 1.1 mm fissure bur. A devascularized butterfly bone segment from the subcondylar fracture was fixated as a bone graft to the pre-bent plate. To achieve this, the 3D model was burred out to accommodate the piece of bone before in vivo application, ensuring the bone fragment would fit stably between the distal and proximal fracture segments. Total operative time was 2 hours and 36 minutes.
From left to right: VSP (unreduced and reduced images), operative deliverable showing prebent plate housing devascularized butterfly bone segment, postoperative 3D scans.
The post-operative scan revealed good anatomic reduction of the comminuted fracture and a mandibular height that was equivalent to the uninjured contralateral side. Adherence to the VSP plan was maintained. Rigid MMF was held for 2 weeks, followed by early motion with light elastics. At 2 months follow-up, the patient had normal occlusion and maximal interincisal opening (MIO). There were no postoperative complications or malocclusion.
Clinical Presentation—Case 3
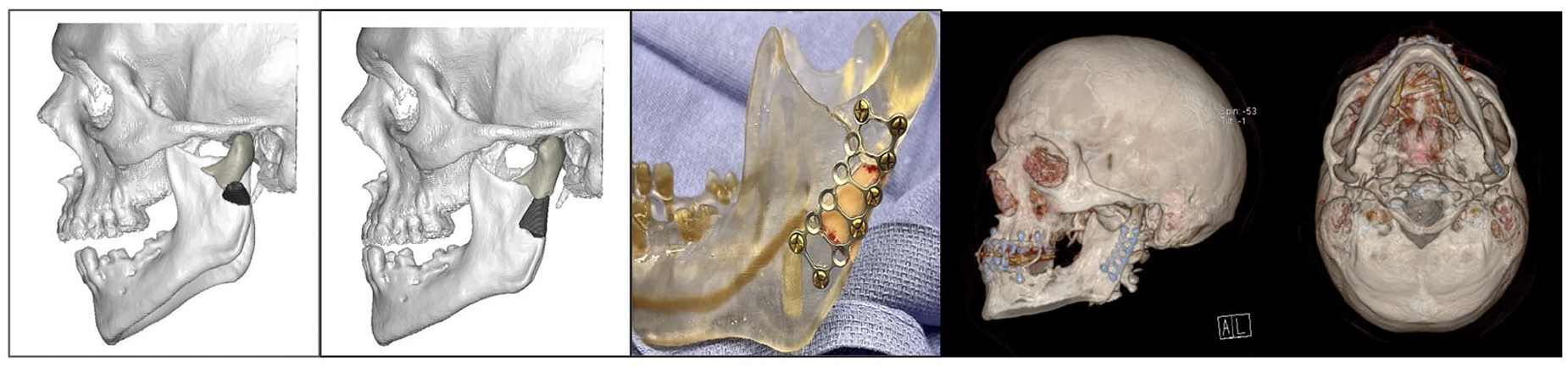
A 22-year-old male presented after an all-terrain vehicle (ATV) rollover with bilateral ZMC, LeFort I, II, and III, NOE, mandible, subcondylar, and orbital floor fractures, as well as a palatal fracture and right parasymphyseal mandible fracture (Figure 5). A CT scan (1 mm cuts) was taken the day of admission; however, surgical intervention was delayed due to the patient’s unstable condition, including acute respiratory distress syndrome, GI bleed leading to hemodynamic instability, and C. difficile infection. During VSP, fracture fragments were identified and reduced with superimposition of a normative skull due to bilateral injury. For the occlusal fractures and bite relationship, reduction of fractures to their ideal anatomical position was performed, and the subsequent best bite was achieved that optimally maintained the reduction of fractures. Operative deliverables included: a perfected skull model with marked fracture lines and an occlusal splint. Both the reduced/perfected skull model and occlusal splint were delivered to the hospital within 48 hours of the webinar.
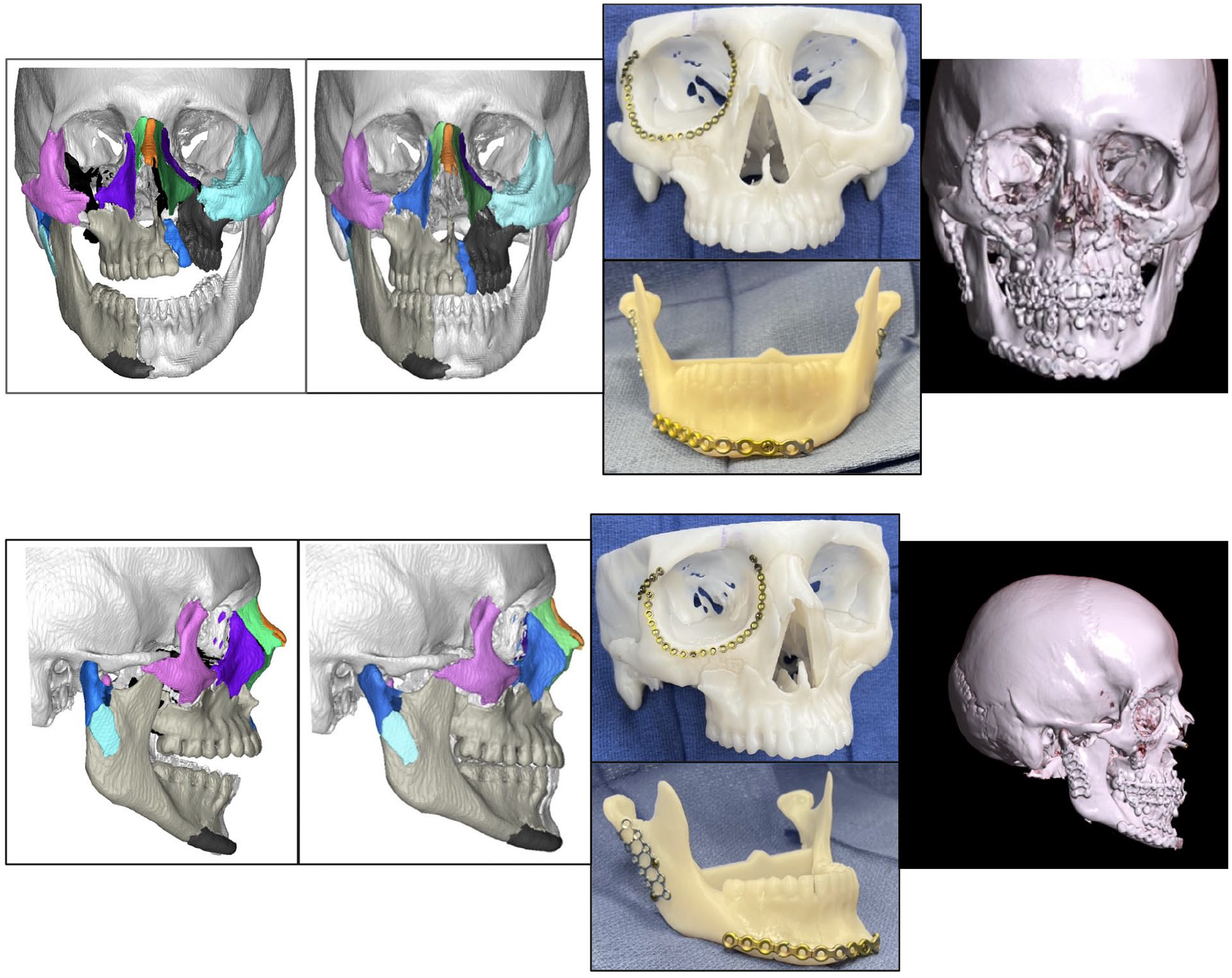
From left to right: virtual surgical planning (unreduced and reduced), operative deliverable showing prebent plates, and postoperative 3D scan.
Surgical exposure included multiple approaches: lynch incisions, transconjunctival lower lid, upper lid, existing lacerations, and intraoral. The occlusal splint was inserted before obtaining MMF. Standard midface and mandible plates were bent and secured to the perfected model to achieve exact conformation. A 1.1 mm fissure bur was then used to etch the plates at each fracture site, thus creating real-time custom 3D plates with fracture location registration to guide correct placement in vivo. The mandibular condyle fractures were approached through a preauricular incision. Impacted condylar heads were reduced and fixated with a lag screw and discopexies were performed using Mitek anchors. Appropriate reduction and fixation were achieved by pre-drilling the lag screw on the perfected skull model and referencing condylar height data from the VSP session. Total operative time was 9 hours and 42 minutes.
The patient experienced no complications and went on to heal well. Postoperative CT scans revealed high fidelity to the preoperative plan with respect to facial bony morphology and occlusion. At the 8-month follow-up visit, the patient reported no significant complications, including vision changes, trismus, malocclusion, or significant facial asymmetry or contour deformities.
Discussion
Traditional approaches to managing facial fractures have long relied on 2-dimensional photographs, CT scan data, and dental impressions paired with handmade splints. Since the 1980s, CT imaging has been used to reconstruct the 3-dimensional facial skeleton, while computer-based cephalometric analysis has enabled surgeons to evaluate skeletal fracture patterns and obtain linear and angular measurements to guide surgical repair. 19 Lab-based fabrication of occlusal splints, typically derived from dental impressions and stone models, has also played a key role in directing bone movement to achieve fracture reduction based on a normalized bite.20,21 These conventional techniques have been particularly effective in elective craniofacial procedures and in treating simple, non-displaced, non-comminuted fractures.
However, as facial trauma becomes more complex, traditional methods face significant limitations. Surgeons are often challenged by limited visualization, loss of anatomical projections, unstable and comminuted fragments, and the difficulty of identifying reference points for hardware fixation—all while aiming to minimize visible scarring with stealth incisions. Furthermore, assessing a patient’s premorbid occlusion in such scenarios can be especially problematic.
Technological advancements in facial fracture management have greatly enhanced the precision, safety, and esthetic outcomes of surgery. Although the integration of technology is not a new concept—endoscopic techniques for orbital trauma emerged in the 1970s, and intraoperative navigation for zygomaticomaxillary complex (ZMC) reconstruction began in the 1990s22,23—recent innovations such as VSP have revolutionized the field. While traditional planning remains more cost-effective in some cases, the increasing adoption of VSP is driven by its ability to improve surgical efficiency and predictive accuracy.10,19 For instance, Schwartz reported an average reduction of 60 operative minutes per orthognathic case when using VSP, without compromising, and often improving surgical precision. 21 This has had a transformative impact on elective craniofacial surgeries, particularly orthognathic procedures. Yet, this level of precision has not been fully translated into craniomaxillofacial trauma care.
In the trauma setting, additional technologies such as intraoperative imaging and navigation systems have been employed to improve outcomes. Tools like the C-ARM and O-ARM provide real-time X-ray or CBCT imaging to assess fracture alignment, although their availability is limited in many trauma centers. 24 Intraoperative CT scanning has also proven valuable in offering real-time visual confirmation of fracture reduction, particularly in complex cases. 25
Navigation systems—comprising a localizer, tracking probe, and CT data—function similarly to a GPS, allowing for precise localization of anatomical landmarks in 3D. Systems like Stryker® (USA) and StealthStation® (USA) use infrared optical tracking to avoid interference from surrounding metal and enhance accuracy. By establishing the spatial relationship between the surgical instrument and the patient’s anatomy, these systems help correlate real-time positioning with preoperative CT data through a registration process, ultimately improving surgical outcomes. 26
However, despite their advantages, these technologies have notable limitations. Intraoperative imaging exposes patients to additional radiation, and navigation systems, while helpful for orientation, cannot compensate for tissue shifts during surgery or guide physical bone manipulation. They also require a high level of surgical skill and judgment to interpret and act upon the information provided. Crucially, unlike our VSPD protocol, these tools do not offer a tangible 3-dimensional guide to assist with the reduction of complex, comminuted fractures, highlighting a significant gap in current trauma care techniques.
Virtual Planning for Facial Trauma
Virtual surgical planning6-10 has been a powerful tool in both adult and pediatric craniofacial surgery since its adoption in the mid-2000s and has continued to develop more widely used applications. It has been able to improve cephalometric analysis, operative simulation, and splint modeling. This modality has greatly enhanced the correction of facial irregularities, especially when considering the added complexities of dentofacial asymmetry, otherwise improperly visualized in 2D surgical plans. VSP has also been able to improve both intraoperative efficiency and accuracy through the production of templates that reproduce the expected gaps and demonstrate the 3-dimensional rotation encountered when performing the procedure, as well as, in some cases, provide patient-specific custom implants and cutting guides.
However, the utility of VSP has been limited thus far to mostly planned orthognathic procedures or secondary reconstruction. VSP has been difficult to implement on a large scale in acute trauma patients due to multiple challenges encountered.
The webinar process already involves a multi-step workflow and efficient collaboration between surgeons and engineers for preoperative planning and surgical simulation. However, when applied to complex trauma cases, added difficulties arise. First, the software was initially designed to simulate elective osteotomies, not to segment and reduce facial fractures. 27 The process of electively osteotomized bone segmentation is simpler than the identification and reduction of irregularly shaped fracture fragments. Elucidating all fractures on a comminuted virtual skeleton requires detailed discernment from the engineer as well as input from the surgeon, who has referenced physical exam and CT interpretation. Engineers also face a steeper learning curve when adapting the software to accommodate complex injuries, as a higher level of precision and customization is required. All of these added challenges can ultimately affect timeliness when both finalizing the plan and generating the operative deliverables. These are important challenges to consider in acute presentations.
Traditionally, operative deliverables (3D “perfected” models, patient-specific implants, splints) are generated via third-party vendors after online web seminars. As described in the treatment protocol, 3D models are created from DICOM files, printed at an external facility, and delivered to the surgeon. Delivery times vary depending on whether the surgery is elective or trauma-related. In elective surgery, deliverables arrive generally within 10 to 14 days from injury, if the CT scans are sent immediately after acquisition. 28 Recently, the ability to perform expedited VSP and manufacturing for trauma cases to create operative deliverables has opened the door for using this technology in the acute setting; deliverables can arrive in as little as 72 hours from initial presentation.
Currently, there has been an increased interest in in-house/point-of-care printing. Point-of-care (POC) manufacturing allows for anatomic models to be generated directly at the “place of care” using in-house Computer-Aided-Design (CAD)/Computer-Aided-Manufacturing (CAM) technology. This approach is becoming more feasible as the cost of 3D printers is decreasing and the learning curve to use the software is more manageable. While there may potentially be higher upfront costs depending on the equipment purchased, POC printing has been able to produce models in as little as 1 hour. However, the problem of Food and Drug Administration (FDA) approval remains for in-house segmentation and printing. 28 To date, there is no FDA-approved solution to sterilizing point-of-care printed products as would be received from an approved third-party vendor.
A major concern with the use of technology in facial trauma management has been timeliness. Historically, its adoption in trauma care has been limited by the prolonged manufacturing, shipping, and turnaround times associated with the use of physically printed modalities. In an acute trauma situation, the time needed to adequately design and transport deliverables may compromise an optimal outcome for the patient by delaying treatment. However, now that major plate manufacturers such as Stryker® (USA), KLS-Martin® (USA), and DuPuy-Synthes® (USA) have relocated printers domestically and streamlined the process of CT model generation, fracture reduction, and 3D printing so that materials can be expedited within the week, it is reasonable to plan for a same-admission surgery utilizing these tools. With the addition of the point-of-care approach as well, a potential future solution, models and splints can be printed without transportation delay. Thus, same-admission VSP-assisted craniofacial trauma reconstruction may soon become the standard of care for complex patients, especially those with pan-face or complicated occlusal injuries.
Operative Deliverables
Sharaf et al11,12 described the use of the EPPOCRATIS protocol, which stands for Expedited Preoperative Point of Care Reduction of Fractures to Normalized Anatomy and 3-dimensional Printing to Improve Surgical Outcomes. In brief, preoperative CT images (1 mm cuts) were used for VSP. A 3-dimensional model was printed and used to bend fixation hardware preoperatively, which was then sterilized. In ZMC fractures, Abdul Lateef Hassan and Abbood Mohammed 6 presented VSP and pre-bending fixation hardware, showing good fidelity to the uninjured orbital volume. Fawzy et al 16 also discussed the use of a 3D printed mirror template based on the uninjured orbit to create custom pre-operatively bent titanium mesh constructs and prepare bone grafts in severe multi-wall orbital trauma. In their study, they were able to significantly reduce the widening of the fractured orbit and reapproximate the orbital measurements and volume of the contralateral side, thus achieving symmetry and accuracy of fixation. Other studies report the use of additional types of technology to assist in fracture repair surgery, including intraoperative CT-guided navigation and prefabricated custom patient-specific implants.7,15,19-21,29
While these previous studies have shown promising results in terms of surgical accuracy and increased efficiency by describing the use of pre-bent plates for facial fracture repair, limitations remain.6,8,9,11-14 Preoperatively bending plates to fit an ideal skeleton provides a helpful guide. However, these plates still require transportation and sterilization, which risks altering the ideal shape and accuracy. Moreover, these pre-bent operative deliverables may not provide a precise, real-time guide for in vivo fixation when a surgeon is working in a comminuted 3-dimensional space. To be properly placed, the plate must localize the fracture site in the context of the surrounding fracture lines and angles. Otherwise, the fixation could be somewhat arbitrary. Intraoperatively bending and fixing plates to the 3D template and plate etching with the fracture lines, allows for accurate 3D plate structure with references to in vivo orientation with respect to fractures.
Mandibular and occlusal fractures can also create more of a challenge regarding virtual planning of operative deliverables. Identifying premorbid occlusion is challenging when we don’t know if centric relation was equal to centric occlusion. Therefore, making isolated dental splints to help reduce occlusal fractures is inherently difficult. Traditional dental impressions, model surgery, followed by occlusal splint creation, create a bite irrespective of the surrounding bony anatomy and fractures. When placed intraoperatively, the dental lab splint may not induce near-bony reduction in the surrounding fractures, and if anything, may cause malreduction. In occlusal type fractures, the virtual surgical planning consultation and involvement of the software engineering team become vital to achieve accuracy of fracture reduction along with the patient’s ideal bite relationship. Yoon et al 30 reported a small case series highlighting the use of virtual 3D-generated acrylic splints to enhance the accuracy and effectiveness of open reduction and internal fixation procedures for midface and mandibular fractures. Similarly, Shakya et al 10 conducted a non-randomized controlled trial comparing conventional clinical judgment with preoperative VSP and 3D model-based reduction for mandibular condyle fractures. Their method, which closely resembles the approach used at our institution, involved planning the necessary disimpaction and rotational movements for plate or screw fixation. The study demonstrated a significant reduction in operative time and improved accuracy in the VSP group, as confirmed by postoperative CT measurements. At our institution, we have also found that generating intermediate and final occlusal splints from CT-based craniofacial data, without the need for dental impressions or cast models, has been especially beneficial. Splints created at the same time as bony reduction solve this issue of optimizing both dental and bone alignment.
Managing craniofacial trauma, particularly panfacial fractures, is an intricate and technically demanding process due to the variability in fracture injury patterns and operative sequencing protocols. The most effective order for fracture reduction and fixation has been widely discussed in the literature, whether from top-down, bottom-up, inside-out, or outside-in, as each method can have distinctive advantages depending on the injury and the surgeon’s familiarity with the anatomy.31,32 For example, Dr. Joseph Gruss popularized the top-down outside-in approach to establish facial width and projection at the frontal bar and progress downwards, while in contrast, Dr. Paul Manson’s bottom-up, inside-out approach establishes occlusion and posterior facial height first at the mandible and builds upward toward the midface.31,32 The critical reference points used to guide the rest of the reduction differ between these 2 approaches. What remains common, however, is the emphasis on beginning with stable, reliable bony landmarks and working from “known to unknown” regions. This is essential because if fragments are improperly reduced early in the sequence, these errors can propagate throughout the procedure, leading to significant functional and aesthetic complications, such as malocclusion, facial widening, enophthalmos, etc. For example, failing to recognize and reduce a hemi-NOE fracture can cause lateral displacement of the ZMC, resulting in an orbit that is too large and a face too wide. 33 Given the complexity of visualizing and reducing multiple fracture points, which can often not be seen simultaneously and risks displacing 1 site when manipulating another, operative sequencing without clear planning risks performing open internal fixation without proper reduction. This may result in the unnecessary use of bone grafts or poor outcomes that require secondary surgeries. Therefore, this is where the integration of technology and operative deliverables becomes invaluable. The ability to use a reduced and perfected model intraoperatively allows real-time plate bending to the targeted form. This helps verify reductions, ensure anatomic relationships are restored, and eliminates much of the intraoperative guesswork with complex panfacial fractures, thus enhancing precision, improving outcomes, and ensuring better restoration of facial form and function.
Intraoperative Observations
Operative efficiency is crucial when managing complex facial fractures, which involve multiple technical steps such as surgical exposure, fracture reduction, plate bending and registration, and splint adaptation. While it may seem that incorporating advanced technology would lengthen surgical time, it often enables parallel workflow efficiencies. Whenever possible, surgical exposure and backtable utilization, such as plate bending and splint refinement, can occur simultaneously. This parallel processing helps avoid additional operative time and can even lead to time savings. However, in settings with more limited staffing, these tasks are performed in series. Of note, our patient in Case 3 required particularly efficient surgical execution due to his critical medical condition. In such high-stakes scenarios, optimized workflows and intraoperative precision are essential to minimize anesthesia time and overall physiological stress.
In addition to time savings, this approach also offers cost advantages. Although there may be concerns with increased costs with advanced technologies, additional expenses depend on each hospital’s contract, and in most cases, there is no added cost to the patient due to bundled billing. A study by Stryker (USA) found that using a hybrid MMF system saved $4588 per case by reducing OR time by 74 minutes. 34 Our approach similarly streamlines the surgical process. Furthermore, the lack of implants eliminates implant-related costs, and by removing intraoperative guesswork, we simultaneously enhance accuracy and reduce the risk of complications, thereby minimizing potential costs associated with secondary revisions, as shown by our case series with no reoperations required.
A key benefit of this approach is increased intraoperative confidence in hardware fit and orientation, which minimizes the need for iterative adjustments and enhances accuracy in fracture reduction and fixation. The ability to fabricate and fix real-time custom plates eliminates the need for rebending plates and streamlines proper plate placement.
Several valuable insights have emerged through this series of cases. For instance, when repairing multiple fractures, we found that a single, hand-bent 3D plate registering across multiple fractures is more accurate than using several smaller plates. A single large plate helps define the final anatomical form, whereas multiple smaller plates attempt to unify irregular bone fragments without a cohesive structural guide.
Additionally, we discovered that intraoral splints derived from CT scans require additional acrylic thickness for proper fabrication. As a result, a contouring burr is now routinely used to thin and refine the splint intraoperatively, creating a lower profile fit and reducing the risk of patient irritation or postoperative irregularities.
Limitations
Our study has several limitations. It is subject to selection bias, as we have chosen to include cases that we believe best illustrate our protocol and the range of fractures it can address. Additionally, our assessment of improved efficiency and accuracy is based on subjective observations, as we did not directly compare procedure duration or outcome metrics with traditional protocol. Perceived improvements were based on visual alignment between postoperative CT scans and preoperative plans, as well as the surgeons’ subjective impression that the reduced need for intraoperative adjustments streamlined the workflow. We plan to address these issues in the subsequent manuscript, where there will be a detailed quantitative and qualitative analysis in our large patient series.
Future Directions
This study depicts in detail just 3 cases of a larger patient series to review a single institution’s experience with VSP and intra-operative plate bending and etching on a 3-D printed template. We believe that the use of 3D printed models, occlusal splints, pre-bended plates, and plates etched with the fracture lines enhances the ability to achieve proper reduction and fixation efficiently while creating a better understanding of the patient’s condition. This comprehensive approach to craniofacial trauma treatment can be utilized for fracture repair ranging from limited regional to panfacial across the craniofacial skeleton. The additional steps and costs incurred using a technologically enhanced method are offset by the surgical accuracy and potential avoidance of operative secondary post-traumatic deformities. While this paper presents a proof of concept, a second manuscript will provide a subanalysis of this technique as applied to specific craniofacial anatomic regions.
Conclusion
While the use of this VSPD protocol involves certain limitations, namely the additional time for preoperative planning and generation of deliverables as well as the potential increased costs, we believe these factors are offset by meaningful benefits. Even in time-sensitive cases such as mandibular fractures, where intervention is ideally within 72 hours, this approach did not negatively impact outcomes and likely contributed to improved surgical precision. This time and financial investment are further justified by the improved operative workflow, increased anatomic accuracy, and potential decreased need for secondary procedures.
Footnotes
Acknowledgements
We would like to sincerely thank the engineers we collaborated with for the considerable amount of time and effort they put into helping plan and create all models for the patients we treated.
Data Availability Statement
Data files stored in a password-protected cloud source.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Author Raymond Harshbarger is a member of the Editorial Board of FACE and did not take part in the peer review or decision-making process for this submission.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
The Institutional Review Board at the University of Texas at Austin determined that this protocol (IRB ID: STUDY 00005292) met the criteria for exemption from IRB review on April 3, 2024.
Consent to Participate
Consent to participate in research was obtained when the data was originally collected.
Consent for Publication
Retrospective analysis with de-identified data. All patients signed a photographic consent for teaching and publication.
Informed Consent
Informed consent was obtained for all participants when the data was originally collected.
