Abstract
Rationale:
Respiratory muscle dysfunction is a common sequela in long coronavirus disease (COVID). Inspiratory muscle training (IMT) can improve inspiratory muscle strength, endurance, functional capacity, and dyspnea. However, evidence supporting its effectiveness through telerehabilitation remains limited.
Objective:
To evaluate the effects of a supervised, home-based telerehabilitation IMT program on inspiratory muscle performance, dyspnea, and functional capacity in individuals with long COVID.
Methods:
This prospective quasi-randomized controlled study enrolled patients with long COVID, assigned to either an IMT group (12 weeks of supervised telerehabilitation) or a control group (usual care). Baseline and after 12-week assessments included dyspnea (baseline dyspnea index/transitional dyspnea index [TDI]), pulmonary function, sustained maximal inspiratory pressure, inspiratory muscle endurance test, and functional capacity (1-min sit-to-stand test).
Results:
A total of 53 individuals were enrolled. Baseline inspiratory muscle performance was comparable between the groups (p > 0.05). After 12 weeks, the IMT group showed significant improvements compared with controls in maximal inspiratory pressure (+31.6 cmH2O [95% confidence interval {CI}: 25.0–38.2] vs. +6.7 cmH2O [−6.1–19.5]; p < 0.01) and endurance time (+52.8 sec [17.3–88.2] vs. −9.5 sec [−90.3–70.1], p = 0.04). Dyspnea decreased significantly with clinically relevant improvements in all TDI domains (p < 0.001). No between-group differences were observed in lung function or functional capacity.
Conclusion:
A supervised, telerehabilitation IMT program is a safe, feasible, and low-cost intervention that improves inspiratory muscle strength, endurance, and dyspnea in patients with long COVID following severe infection. These findings support the use of supervised telehealth approaches to enhance respiratory muscle performance.
Introduction
The coronavirus disease 19 (COVID-19) pandemic posed an unprecedented challenge to health care systems worldwide, leading to long-term sequelae that affected the functional status and quality of life in individuals recovered from COVID-19. 1 These persistent effects are collectively referred to as long coronavirus disease (COVID), characterized by symptoms such as dyspnea, chronic fatigue, exercise intolerance, psychological distress, and cognitive dysfunction lasting >12 weeks after infection.2,3 Up to 90% of hospitalized patients experience long COVID, and ∼33% report limitations in daily activities.3,4 Patients who required high-flow oxygen therapy or prolonged invasive mechanical ventilation appear to be the most severely affected, requiring pulmonary rehabilitation.1,5 The pandemic introduced significant changes in health care delivery, including long-distance support such as telerehabilitation programs and web-based interventions.6–8
Inspiratory muscle training (IMT) has been shown to improve maximal inspiratory pressure (MIP), enhance exercise tolerance, and reduce dyspnea in individuals with chronic respiratory disease. 9 More recently, studies in patients recovering from COVID-19 and participating in outpatient clinical rehabilitation programs have reported that IMT can increase exercise tolerance, reduce dyspnea, 10 and improve functional capacity as well as inspiratory muscle strength and endurance.6,11,12 These benefits are especially relevant for individuals with long COVID, who often present inspiratory muscle dysfunction and exertional dyspnea. 13 However, few studies have evaluated real-time supervised IMT programs delivered via telerehabilitation in this population.14,15 The lockdown restrictions limited access to outpatient clinical rehabilitation; therefore, telerehabilitation emerged as a feasible, cost-effective, and safe alternative. 12 This approach requires minimal infrastructure and has been shown to promote adherence to home-based interventions.15,16 IMT has been delivered through telerehabilitation platforms; however, most available programs lack real-time supervision, which compromises patients’ adherence and exercise quality. 17 Thus, the present study addresses the gap in evidence regarding the feasibility and effects of synchronously supervised IMT through telerehabilitation for patients with long COVID. While previous studies have evaluated home-based or asynchronous IMT programs, evidence remains scarce regarding synchronous supervised IMT delivered through telerehabilitation.18,19 We hypothesized that a supervised telerehabilitation IMT program would enhance inspiratory muscle strength and endurance, reduce dyspnea, and improve pulmonary function and functional capacity in individuals with long COVID. This study aimed to compare the effects of a supervised IMT telerehabilitation program on inspiratory muscle performance and to investigate the relationships among improvements in inspiratory function, functional capacity, and reductions in dyspnea in long COVID patients.
Methods
Study design and participants’ recruitment
This was a quasi-experimental, controlled, open-label, and single-center study. Participants were allocated to the IMT or control group using convenience-based, nonrandomized stratification, considering practical aspects such as availability for remote supervision and scheduling while maintaining a balance in key baseline characteristics. This method facilitated logistics but does not constitute true randomization. The study was conducted between June 2020 and July 2022 with individuals admitted to the intensive care unit requiring ventilatory support (either invasive mechanical ventilation for >48 h or high-concentration oxygen therapy at a rate of >3 L/min) and who had a confirmed COVID-19 diagnosis by polymerase chain reaction (PCR). Additionally, individuals may present with dyspnea symptoms after 30 days of hospital discharge, as assessed by the baseline dyspnea index/transitional dyspnea index (BDI/TDI). The exclusion criteria included pregnancy and the presence of preexisting respiratory or neuromuscular disease. The study protocol was approved by the ethics committee of the hospital (protocol number: 34115720.5.0000.0068), and all patients provided written informed consent. Safety distancing and personal protective measures were implemented during the lockdown period to ensure the safety of patients and health care personnel during all assessments.
Baseline and postintervention assessments were performed during two hospital visits. To mitigate detection bias, all assessments were conducted by respiratory physiotherapists who remained blinded to group allocation and were not involved in the telerehabilitation IMT training. During the first visit, comprehensive medical history, physical examination, smoking history, symptoms of dyspnea, and lung function testing were assessed. During the second visit, MIP and endurance muscle strength and functional capacity were assessed. After inclusion, participants were consecutively allocated to either the telerehabilitation IMT group or the control group. The intervention lasted 12 weeks
Intervention
Supervised telerehabilitation IMT group
The IMT group underwent a 12-week, 5 times a week, supervised training program. At the end of the 12-week period, all participants returned for a comprehensive postintervention evaluation. A telerehabilitation communication software program (Discord, Discord Inc., San Francisco, USA) was installed on the patient’s smartphone or laptop. Discord is a free text, video, and voice communication mobile application (app), and participants received a linear load device (Powerbreathe, Medic Plus model, UK) with instructions for use. During each video call session, the appropriate breathing technique was reinforced. The proper breathing technique included fully exhaling to near residual volume (RV), followed by a maximal and sustained inspiratory maneuver for as long as possible. The training protocol consisted of six sets of 1-min bouts of inspiratory efforts performed against a prescribed load, beginning at 40% and progressing up to 70% of each participant’s MIP. Training intensity was increased every 2 weeks according to the patient’s perceived difficulty. Inspiratory resistance was increased in 7-cmH2O increments until reaching 70% of the individual’s MIP. Patients manually adjusted the resistance on the training device, and the supervising respiratory physiotherapist verified the correct load before each supervised telerehabilitation session.
During each 1-min bout, participants performed as many inspirations as possible, followed by 1-min rest. For the first 4 weeks, IMT was performed daily under supervision; thereafter, supervision was conducted once a week. During the supervised session, the respiratory physiotherapist ensured correct load adjustment, confirmed appropriate posture, and checked the interface positioning to prevent air leakage. Throughout the intervention, patients recorded the number of breaths in each set and their perception of dyspnea using the modified Borg scale. To isolate the effect of the intervention, no additional rehabilitation strategies or educational guidance provided to the control group were offered to the IMT group. Adherence was defined as the percentage of completed sessions and was documented daily during the intensive supervised phase (weeks 1–4) and verified through log review during weekly supervision in the maintenance phase (weeks 5–12).
Control group
The control group received usual care, which included appropriate pharmacotherapy and a structured, in-person educational session on physical activity delivered by a physiotherapist during the baseline assessment. The session provided standardized guidance consistent with World Health Organization (WHO) recommendations for adults, 20 emphasizing the importance of avoiding inactivity and gradually increasing low-impact activities (such as walking or stair climbing) according to each patient’s functional status and daily routine. No formal rehabilitation program or supervised exercise was prescribed for this group.
Outcomes
Dyspnea symptoms
Dyspnea was assessed using the BDI. The questionnaire has three domains: functional impairment, magnitude of task, and magnitude of effort, each of which is scored from 0 (severe dyspnea) to 4 (no dyspnea). The total score ranges from 0 to 12. 21 The TDI assesses changes from the baseline score, ranging from −3 (major deterioration) to +3 (major improvement) per domain, yielding a total TDI score from −9 to +9. An increase of 1 point or more is considered clinically relevant for the patient.22,23
Lung function
Lung function was assessed using spirometry following the guidelines of the European Respiratory Society and the American Thoracic Society. 24 The variable forced expiratory volume in the first second (FEV1) and forced vital capacity (FVC) were expressed in absolute (L) and percent predicted values. Reference values were calculated based on predictive equations validated for the Brazilian population. 25
Sustained maximal inspiratory pressure
Inspiratory muscle strength and endurance were assessed using the Powerbreathe KH2 Medical® (HaB Ltd., Southam, UK) connected to Breathe-Link® software. For accurate measurements, patients were seated comfortably and instructed to use a nose clip and mouthpiece to prevent air leaks during the sustained maximal inspiratory pressure (SMIP) maneuvers. Individuals were asked to exhale completely near RV, followed by a maximal inspiratory maneuver near total lung capacity (TLC). Standardized verbal encouragement was provided throughout the maneuver to ensure maximal effort. 26 Each patient performed a minimum of three and a maximum of five trials, with 1-min rest intervals between attempts. The following outcome measures were recorded and analyzed: peak pressure (PP), MIP, area under the curve of sustained inspiration (AUC), and inspiration time (IT). The SMIP curve with the highest combination of peak inspiratory pressure (PIP) and sustained MIP was selected for analysis. 27
Endurance inspiratory muscle test
A 7-min inspiratory muscle endurance test (EIMT) was performed using the Powerbreathe KH2 Medical device (HaB Ltd., Southam, UK) connected to Breathe-Link software. The device was set at a constant inspiratory load equivalent to 60% of each participant’s MIP. To assess submaximal inspiratory performance under continuous, time-controlled conditions. The system incorporates an electronic vacuum sensor (0–300 cmH2O) and a pressure-sensitive valve synchronized with the software, which provides real-time graphical feedback for pressure (cmH2O), power (watts), flow (L/s), volume (L), and energy expenditure (Joules). A trained respiratory physiotherapist conducted all assessments. Patients were seated comfortably, and they received verbal and visual instructions to ensure the proper technique. Patients were instructed to seal their lips tightly around the mouthpiece, and a nose clip was used to prevent air leakage. The test protocol required patients to exhale near RV before executing a deep inspiratory maneuver near TLC. The endurance test was terminated under the following conditions: patient completion of the full 7-min duration, failure to open the inspiratory resistance valve for three consecutive attempts, and voluntary termination due to exhaustion despite verbal encouragement. Real-time visual feedback from the software program helped regulate breathing cadence, minimizing the risk of hyperventilation and its associated adverse effects. 28 Dyspnea, heart rate (HR), and oxygen saturation were assessed before and after the test.29–31
Functional capacity
Sit-to-stand-test
Patients were asked to stand up and sit down as quickly as possible without using arm support during the test. The test pace was determined by verbal encouragement. Peripheral oxygen saturation (SpO2), HR, blood pressure, dyspnea (Borg scale), and lower limb fatigue were measured before and after the test. 32
Statistical analysis
The sample size was determined using G*Power (version 3.1.9.7; Heinrich-Heine-Universität Düsseldorf, Düsseldorf, Germany) for a two-arm parallel trial (two-sided α = 0.05; power = 80%; 1:1 allocation) to detect the minimal clinically important difference (MCID) in an MIP of 18 cmH2O, as reported by Del Corral et al. 12 Assuming a pooled standard deviation (SD) of ∼19 cmH2O (based on the SD of MIP change reported by Del Corral et al. 12 ), the required sample size is 18 participants per group (36 total). Allowing for 15% attrition, the recruitment target was set to 21 participants per group (42 total).
All analyses were performed using R (version 4.2.3; R Foundation for Statistical Computing, Vienna, Austria). Continuous variables with a normal distribution were summarized as mean ± standard deviation; non-normally distributed variables were summarized as medians with interquartile ranges. Categorical variables were summarized as counts and percentages. Normality was assessed using the Shapiro–Wilk test. Baseline comparisons between the IMT and control groups were conducted using independent-samples t-tests for normally distributed variables, Mann–Whitney U tests for non-normal variables, and Fisher’s exact test for categorical variables, as appropriate. Significant baseline imbalances were identified in several key covariates, including orotracheal intubation, length of hospital stay, FVC (absolute and percent predicted), and FEV1 (absolute and percent predicted). To mitigate selection bias, a propensity score (PS) was calculated using logistic regression that included all variables differing between the groups at baseline. Dyspnea, assessed using the ordinal TDI, was analyzed using ordinal logistic regression, incorporating group (IMT vs. control) and PS as predictors. The intervention effect was expressed as an odds ratio (OR) with 95% confidence interval (CI), representing the adjusted likelihood of greater improvement in dyspnea in the IMT group.
The effects of the intervention on continuous outcomes (e.g., MIP, peak power, and AUC) over time (baseline vs. 4 months) were analyzed using linear mixed-effects models (LMMs). Each model included group, time, and the group versus time interaction (group vs. time) as fixed effects, with a random intercept for each participant to account for within-subject correlation. The group × time interaction represented the difference-in-differences and served as the primary effect of interest. Model residuals were inspected for approximate normality using the Shapiro–Wilk test. When model assumptions were violated, generalized linear mixed-effects models (GLMMs) with appropriate distributions (e.g., gamma with the log link) were employed. Model selection was based on the Akaike information criterion (AIC). Statistical significance was set at p < 0.05.
Results
Fifty-three individuals were eligible, and 29 were allocated to the supervised IMT program, while 24 were assigned to the control group (CG). None of the patients dropped out of the study. Participants in the IMT group underwent 12 weeks of IMT. The flowchart illustrating the setup and monitoring process of the telemonitored IMT program is presented in Figure 1. Both groups presented similar baseline characteristics in terms of anthropometric data, dyspnea symptoms, MIP, endurance inspiratory muscle test, and functional capacity (Table 1). However, patients in the IMT group had more severe conditions during the acute phase of COVID-19 than those in the CG. The overall hospital stay for all participants was 25.4 ± 15.7 days, with the IMT group staying longer than the CG (32.7 ± 18.1 vs. 19.1 ± 8.0 days; respectively, p = 0.001). Additionally, the IMT group had worse lung function compared with the CG at baseline (p < 0.05; Table 1). Eighty-two percent of participants in the IMT group underwent orotracheal intubation during hospitalization compared with 25% in the CG (p < 0.05). There were more male participants in the control group (65%) compared with the IMT group (51%).

Flowchart of the telerehabilitation inspiratory muscle training (IMT) setup and monitoring process. Stepwise workflow of patient recruitment, setup, and monitoring during the 12-week supervised IMT program delivery via telerehabilitation. IMT, inspiratory muscle training.
Demographic, Clinical, and Functional Characteristics at Baseline
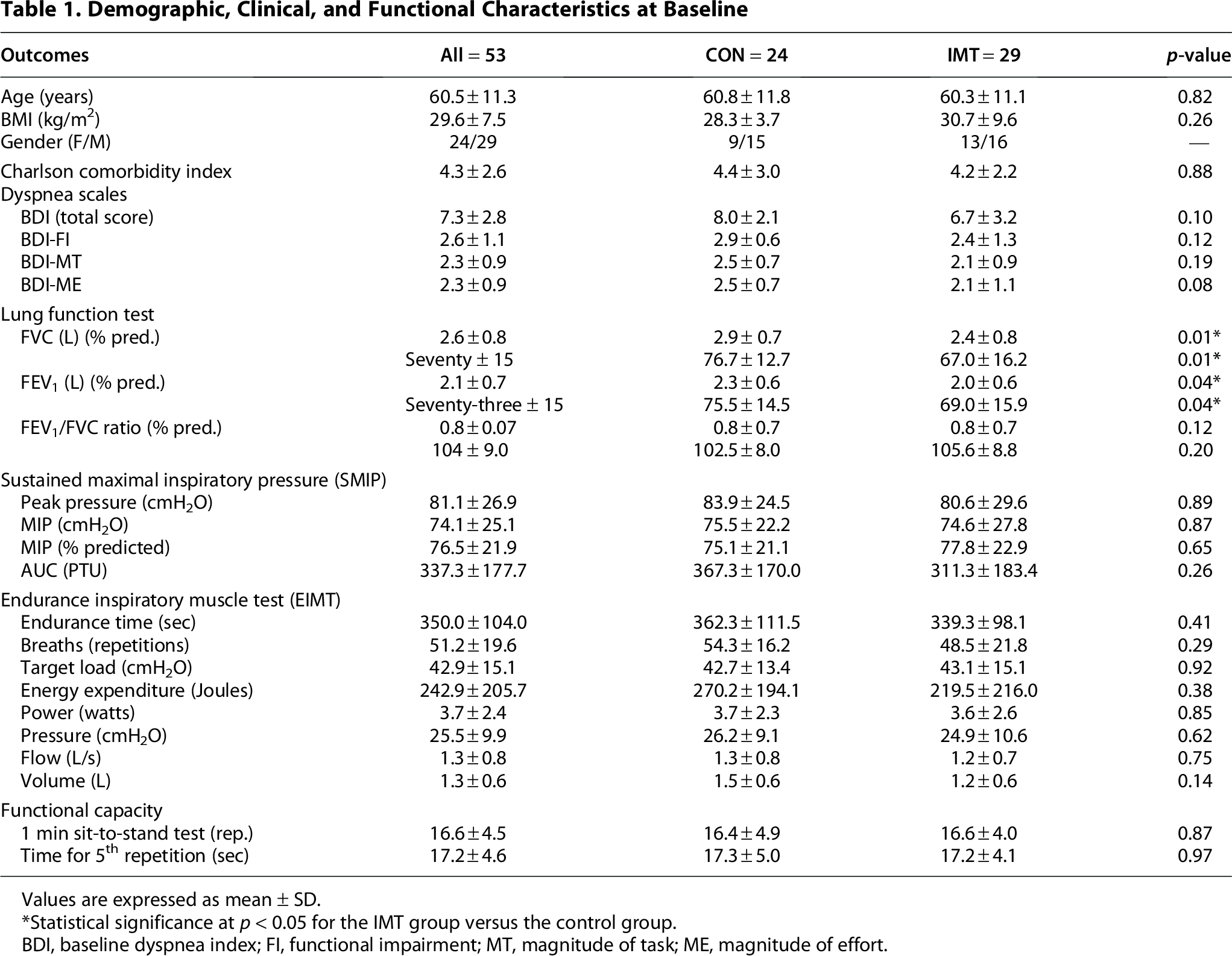
Values are expressed as mean ± SD.
Statistical significance at p < 0.05 for the IMT group versus the control group.
BDI, baseline dyspnea index; FI, functional impairment; MT, magnitude of task; ME, magnitude of effort.
Changes in dyspnea symptoms
Because dyspnea (TDI) is an ordinal outcome, it was analyzed separately using ordinal logistic regression, including the PS as a covariate. The intervention effect was expressed as an OR with 95% CI, representing the likelihood of greater improvement in dyspnea for participants in the IMT group compared with controls after adjustment for baseline imbalances (Tables 2 and 3). The results for all TDI domains are shown in Figure 2.

Changes in dyspnea associated with improvement in activity of daily living between study groups measured using the TDI, after 12-week supervised IMT program delivery by telerehabilitation. Distribution of the TDI questionnaire results in two groups of patients: The box represents the interquartile range (25th to 75th percentiles) of the data, with the central line indicating the median (50th percentile). The whiskers extend to outlier values. TDI, transition dyspnea index.
Adjusted Marginal Means and Between-Group Differences from Linear Mixed-Effects Models (Adjusted for Propensity Score)

Values are estimated marginal means (95% CI), adjusted using a mixed-effects model with a propensity score (including hospital stay, orotracheal intubation, baseline FVC, and FEV1).
Note that the symbol ΔΔ represents the difference-in-differences (group × time interaction). It quantifies the net intervention effect, computed as: (Change in IMT [Post − Baseline]) − (Change in Control [Post − Baseline]).
Significant results (p < 0.05) are shown in bold.
IMT, inspiratory muscle training; FVC, forced vital capacity; FEV1, forced expiratory volume in 1 sec; AUC, area under the curve; CI, confidence interval; PTU, pressure-time units.
Ordinal Logistic Regression for TDI Components Adjusted by Propensity Score

Odds ratios (95% CIs) indicate the likelihood of higher TDI scores for the IMT group relative to controls. Higher odds ratios represent greater improvement.
IMT, inspiratory muscle training; OR, odds ratio; CI, confidence interval; TDI-MT, transition dyspnea index motor domain; TDI-ME, transition dyspnea index mental domain; TDI-FI, transition dyspnea index fatigue domain.
Changes in sustained inspiratory muscle pressure and the inspiratory endurance muscle test
After 12 weeks, the IMT group demonstrated greater improvements in inspiratory muscle performance compared with the control group. Continuous outcomes, including inspiratory muscle strength, endurance, and pulmonary function, were analyzed using LMMs, also adjusted for the PS. The detailed results are presented in Tables 2 and 3. After PS adjustment, the IMT group demonstrated significant improvements in MIP (t47.4 = –3.92; p < 0.001), PIP (t48.3 = –3.74; p < 0.001), and most inspiratory muscle endurance metrics, including endurance time (t48.2 = –1.98; p < 0.05), energy (J) (t48.4 = –3.46; p < 0.01), power (W) (t48.3 = –3.12, p < 0.01), pressure (cmH2O) (t48.4 = –3.51, p < 0.001), volume (L) (t46.3 = –2.21, p = 0.032), and number of breaths (t48.6 = –2.62; p = 0.011), compared with the control group. A significant improvement was also observed in the FVC % of predicted (t47.4 = –2.29, p = 0.026). No between-group differences were detected for functional capacity (1-min sit-to-stand [STS] test) or other pulmonary function outcomes (FVC absolute, FEV1 absolute, and FEV1 percentage of predicted; Table 2).
Furthermore, a linear association was observed between the improvement in inspiratory muscle strength and improvements in functional and TDI impairment domains. Consistently, TDI scores showed moderate correlations with changes in PP (r = 0.55; p < 0.001), MIP (r = 0.58; p = 0.03), AUC (r = 0.63; p < 0.001), inspiratory power output (r = 0.60; p < 0.001), and inspiratory pressure (r = 0.52; p < 0.001), reinforcing the relationship between improved respiratory muscle performance and the reduction of dyspnea.
Telerehabilitation IMT intervention
Figures 1 and 3 provide a detailed overview of IMT outcomes over the 12-week program. Panel A illustrates the progressive decline in dyspnea perception, measured by the Borg scale, across the training period. Panel B depicts the weekly variation in the average number of inspiratory efforts performed during sessions. Panel C demonstrates the gradual increase in the training load over 12 weeks, whereas Panel D shows the reduction in the session duration as the program progressed, from an average of 21 min at the beginning to 15 min by the end.
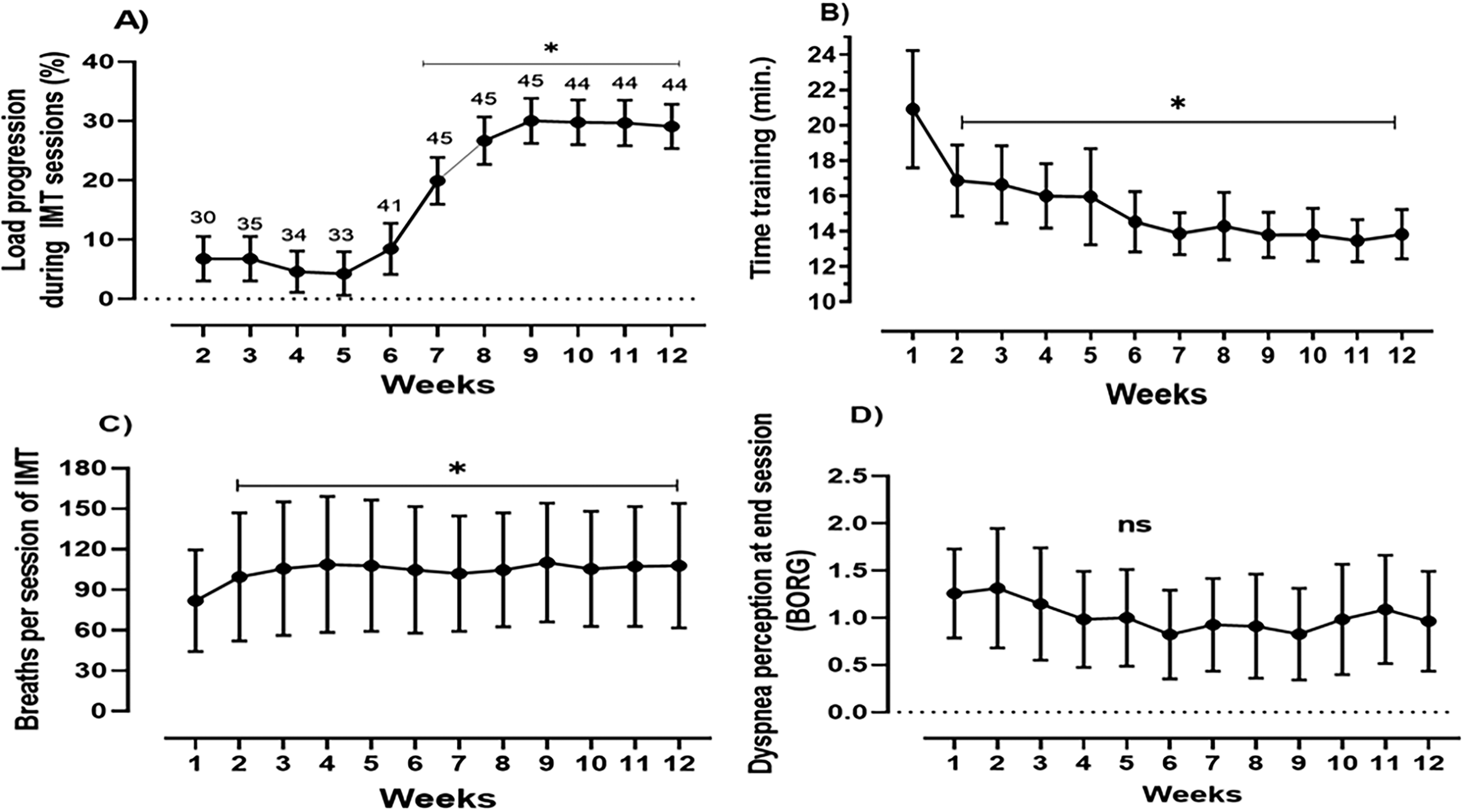
IMT progression through a telerehabilitation program over 12 weeks.
Discussion
This study demonstrated that a 12-week supervised IMT program delivered through telerehabilitation significantly improved inspiratory muscle function and reduced dyspnea in survivors of severe COVID-19. Ordinal regression analyses confirmed a strong and clinically meaningful effect on dyspnea, with ORs exceeding 12 across all domains of the TDI. The median changes in the IMT group (+2 units) surpassed both the improvement observed in controls (+0.5 units) and the established MCID of +1 units. However, this reduction in dyspnea did not translate into a significant improvement in functional capacity, as measured by the 1-min STS test, suggesting that the physiological effects of IMT on inspiratory muscles did not meaningfully extend to lower limb performance in this cohort.
Baseline differences (hospital stay, intubation, FVC, FEV1) were incorporated into a PS, which was included as a fixed covariate in all LMMs and ordinal logistic regression models to adjust for baseline imbalances. The consistent improvement observed in PIP, MIP, and endurance metrics reinforces the ability of remotely delivered IMT program capacity to strengthen and improve the endurance of inspiratory muscles. Moderate correlations between increases in inspiratory performance (PP, MIP, AUC, inspiratory power, and inspiratory pressure) and higher TDI scores highlight the close physiological link between respiratory muscle function and dyspnea perception. These findings support the interpretation that strengthening and improving the endurance of the inspiratory muscles may directly reduce breathlessness and, consequently, improve participation in daily activities, even if these benefits are not fully captured by brief functional performance tests. Our findings align with previous studies in post-COVID-1912,33,34 and indicate that IMT, even when delivered at moderate intensities through telerehabilitation, yields meaningful physiological and clinical benefits, reinforcing its role in long COVID rehabilitation.
Importantly, a key strength of our study is the use of an IMT-only protocol in patients with long COVID who were uniformly severely affected during the acute phase. This contrasts with previous studies that included patients with milder disease or those who had never been hospitalized and who therefore exhibited higher baseline inspiratory strength, pulmonary function, and functional capacity.1,12,14,34,35 Our results extend the potential applicability of IMT to a more clinically compromised population.
The intervention produced a robust effect on inspiratory muscle strength. Linear mixed-effects modeling revealed that MIP improved by 20.5 cmH2O (95% CI: 9.95–31.0; p < 0.001) more in the IMT group than in the control group. The within-group improvement in IMT (29.6 cmH2O, from 78.6 to 108.2) exceeded the MCID of 18 cmH2O proposed by Del Corral et al. 12 This substantial improvement likely reflects the protocol emphasis on deep, complete inspirations performed under progressively increasing loads up to 70% of individual MIP. The intermittent structure (1 min of training, followed by 1 min of rest, repeated across six sets) was designed to facilitate high-quality breaths while limiting excessive fatigue.
In contrast, functional capacity showed no significant between-group difference in the 1-min STS test (p = 0.437), with an estimated intervention effect of 0.91 repetitions (95% CI: −1.43–3.25). The magnitude of within-group improvement in IMT (2.2 repetitions) did not exceed the MCID of three repetitions. 32 Differences from the findings of Del Corral et al., 12 who reported larger improvements, may stem from their cohort’s substantially lower baseline performance. Although IMT can improve functional capacity in healthy populations by attenuating the inspiratory metaboreflex and reducing the neural respiratory drive, 36 the magnitude of transfer in long COVID survivors may depend on baseline impairment, training load, and the presence of persistent systemic limitations that lead to altered ventilatory mechanics.
Regarding pulmonary function, the results were mixed but physiologically coherent. A significant improvement was observed in FVC % of predicted (p = 0.026), whereas absolute FVC, absolute FEV1, and FEV1 % of predicted remained unchanged. The isolated increase in FVC % of predicted suggests that IMT improved the ability to perform deeper inspiratory maneuvers, likely through diaphragm strengthening, without altering airway mechanics or parenchymal injury. This interpretation is supported by prior evidence demonstrating a direct relationship between MIP and FVC % of predicted. 34 The absence of changes in absolute spirometric values reinforces the notion that IMT is a muscle-specific intervention and not a treatment that reverses structural pulmonary abnormalities following severe COVID-19.
Endurance outcomes further support the presence of positive adaptations. Our standardized endurance assessment protocol, which used a fixed 60% of the MIP load, differed from variable load protocols in prior studies 12 ; yet, both approaches yielded improvements in endurance. The IMT group showed significant increases in the endurance time (difference: 62.4 sec; p < 0.05), inspiratory power (2.13 W; p < 0.01), energy (210 J; p < 0.01), and number of breaths (14.7 repetitions; p = 0.011). These improvements may be partially explained by structural adaptations in the inspiratory muscles, including a greater proportion of type I fibers in the external intercostals, as previously described. 37
Adherence was high throughout the program, reaching 100% during the intensive supervised phase and 91% during the maintenance phase. In contrast, unsupervised IMT protocols often report lower adherence and higher dropout rates, 34 underscoring the value of professional supervision. Future studies should investigate whether supervision exerts long-term effects on adherence, dose response, and clinical outcomes.
Patient satisfaction with telerehabilitation was also high in this cohort, with previous reports from our group 38 showing that satisfaction increased from 65.6% at baseline to 93.1% at the end of a 12-week IMT program. 38 These findings suggest that telerehabilitation is feasible and acceptable for individuals recovering from severe COVID-19 and may help overcome barriers related to mobility, fatigue, or apprehension toward technology.
This study has limitations. The primary limitation is the absence of proper randomization, inherent to its quasi-experimental design. This decision reflected the ethical and logistical reality of the early pandemic, when prioritizing rehabilitation for the most severely affected patients was imperative. Due to sequential assignment, the IMT group ultimately included individuals with more severe baseline impairment, including higher intubation rates, longer hospital stays, and lower pulmonary function. These differences may have introduced residual selection bias. To reduce this risk, we employed a rigorous statistical approach that incorporated a PS derived from all significantly unbalanced covariants (hospital stay, intubation status, FVC, and FEV1) in all linear mixed-effects and ordinal models. Although these adjustments strengthened internal validity, some residual confounding cannot be entirely excluded. Additionally, the absence of a sham IMT group limits our ability to distinguish intervention effects from potential placebo responses.
In conclusion, a 12-week supervised IMT program delivered through telerehabilitation significantly improved inspiratory muscle strength, endurance, and dyspnea in patients with long COVID following severe disease. These findings support IMT as a structured, safe, and accessible rehabilitation strategy suitable for remote implementation. Although the improvement in functional capacity was modest, the physiological and clinical improvements reinforce the role of IMT as a part of the long-term rehabilitation of this population.
Authors’ Contributions
V.C.I., C.T.J., and C.R.R.C. conceived and designed the study. V.C.I., C.T.J., C.F.S., A.C.L.P., M.B.d.O., R.R.O., and C.R.F.C. collected the data. V.C.I., E.F.P., J.E.P., N.A.C., and D.M.C. analyzed and interpreted the results. V.C.I. and C.R.F.C. drafted the article. All authors critically revised the article for important intellectual content and approved the final version for submission.
Footnotes
Acknowledgments
The authors acknowledge HaB International (Latin America) and NCS for providing the training equipment and consumables, including the mouthpieces used in this study.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
We thank the São Paulo Research Foundation (FAPESP) (Grant number: 22/01769-5).
