Abstract
Background:
Respiratory syncytial virus (RSV) is a leading cause of U.S. hospitalizations in infants. Current guidelines recommend vaccination during pregnancy or immunizing infants after birth to protect infants from RSV. While research has shown pregnant people are in favor of preventative measures for RSV, overall coverage remains relatively low. This study aimed to gather insight into RSV vaccine perceptions, preferred method of prevention, degree of health care providers (HCPs) recommendation, and timing for discussion and vaccination.
Methods:
A 10-minute online survey was administered to pregnant adults and new mothers in December 2024. Data on respondents’ preference for prevention, impact of the strength of HCP recommendation, and preferred timing and location for discussion were collected.
Results:
Among 800 respondents, 65% showed preference for vaccination during pregnancy over immunizing their baby post-birth. A strong HCP recommendation is associated with a higher maternal vaccination acceptance rate (56% “definitely” would vaccinate) compared with a lighter recommendation (47%). Preferred timing for this discussion is during an initial appointment (33%) or a few weeks before they would receive the vaccine (42%). About 84% of respondents prefer having the choice to vaccinate during pregnancy, even if their infants are born outside of the typical RSV season.
Conclusions:
The majority of respondents in our study believed that protecting their infants from RSV was important, and approximately two-thirds preferred vaccination during pregnancy. Higher vaccination uptake is achievable with a strong HCP recommendation, earlier HCP counseling during pregnancy, and flexible vaccination timing outside a typical RSV season.
Background
Respiratory syncytial virus (RSV) is a leading cause of U.S. hospitalizations in infants. In the absence of prevention products, 2%–3% of all infants aged <6 months are hospitalized for RSV each year. 1 Currently, there are two types of RSV prevention options licensed by Food and Drug Administration and recommended by the Advisory Committee on Immunization Practices to the Centers for Disease Control and Prevention. This includes seasonal administration of either a maternal vaccination during 32 through 36 weeks of gestation or a monoclonal antibody administered to the infant after birth.2–4
Several survey studies have reported that most pregnant people and health care providers (HCPs) are in favor of preventive measures for RSV.5,6 In a survey study of adults who were pregnant or trying to become pregnant, Saper and colleagues also found that more than half those surveyed were very likely to get vaccinated during pregnancy, primarily driven by the belief that RSV could be a serious illness. 6 Other research to date among parents and HCPs in countries outside of the United States on attitudes toward RSV prevention also reported similar findings.7–15
A recent study reported an estimated 55.8% of infants are protected against RSV, including a maternal RSV vaccination rate of 32.6%. 16 While these are meaningful coverage rates, there remains a gap between stated preference and actual coverage. Several barriers to vaccine uptake have been identified, including lack of prevention awareness among pregnant women and those with recent births. Lack of provider recommendation for vaccination may be another factor as well, with a recent study noting it to be the most frequently reported reason (16.9%) for eligible women not receiving the vaccination. 16
The current study sought to gain insight into the impact of an HCP recommendation on pregnant individuals’ willingness to receive the RSV vaccine during pregnancy and determine how this varies based on the strength of recommendation. Other potential barriers to vaccine uptake such as potential preferences for timing of discussion regarding RSV and the option to vaccinate outside of the viral season were explored.
Methods
This cross-sectional study consists of a 10-minute, self-paced online survey of individuals who were pregnant or recently had a pregnancy delivery at the time of survey administration, recruited from existing internet market research panels that reached a geographically and racially diverse population. Respondents could enter the survey via survey invitations from their panel provider or via the panel homepage of available studies. Interested participants could then access the screening survey to determine study eligibility. Details of the study topic were not disclosed to potential respondents at the time of survey invitation or screening beyond the broader category of health care. Respondents were blinded to the study sponsor until all questions had been answered, at which time participants were able to request details on the study sponsor, in line with local regulations, should they desire. Upon completion of the survey, participants were compensated through their panel with a $2 honorarium in line with current fair market values for survey-based research.
The study (Protocol # 2024-0499) was deemed exempt according to 45 CFR 46.104(d)(2) Tests, Surveys, Interviews 45 CFR 46.104(d)(3)(i)(A) by Pearl Institutional Review Board (IRB). Informed consent was obtained from all participants prior to participating in the study. Respondents had the ability to remove themselves from study participation at any time without penalty. The survey was fielded from December 3, 2024, through December 27, 2024.
Study population and inclusion and exclusion criteria
This study surveyed pregnant adults and new mothers who met the following study inclusion criteria: (1) being female; (2) aged 18+ years; (3) has a child or children at less than 1 year of age or currently pregnant; (4) being primarily responsible, or shares responsibility, for making decisions about the health of their child(ren); (5) willing to provide informed consent; and (6) willing to consent to adverse event report provisions.
Participants were excluded if they completely opposed to vaccination (for themselves or their children) due to religious, political, or other beliefs or if they were, at the time of survey administration, employed or had a member in the household employed by an advertising agency or market research firm; marketing firm or marketing department in their own firm; a public relations company; a news media company; a pharmaceutical manufacturer, distributor, wholesaler, or retailer; or a medical professional.
Study measures
The survey collected data on sociodemographic factors and the participants’ awareness of various respiratory diseases. For their perceptions of the seriousness of RSV and the importance of protection from RSV, participants selected from a 5-point scale or “Don’t know.” They were then asked about their level of agreement/disagreement with statements related to the management and potential seriousness of RSV. For their willingness to receive maternal RSV vaccination overall and influence on their willingness based on HCP recommendation, the response options were “definitely would get,” “probably would get,” “might or might not get,” “probably would not get,” or “definitely would not get.” Participants were asked for their preference for RSV prevention method by selecting the options of “strongly prefer to receive vaccination during pregnancy,” “somewhat prefer to receive vaccination during pregnancy,” “somewhat prefer to immunize my baby after birth,” “strongly prefer to immunize my baby after birth,” or “I would not at all be open to considering either option.” Participants were then asked for the reasons for their stated preference. Finally, participants were asked about their level of agreement/disagreement using a 5-point scale on the choice to vaccinate during pregnancy regardless of timing of birth relative to the season, preference for timing of discussion of RSV preventives with the options of “initial appointment,” “a few weeks before vaccination,” “at the appointment of the vaccination,” or “I would not get any vaccines during pregnancy,” and the preferred location to receive vaccination with the options of “OB/GYN office,” “hospital,” “family physician’s office,” “pharmacy,” “work,” “health department,” or “other.”
Statistical analysis
Descriptive univariate statistics were used to examine study response. Continuous variables (e.g., age) were examined by mean and median. Categorical variables (e.g., gender, race) were examined with individual totals along with the respective percentages from the total sample size. Respondents were stratified into three subgroups: pregnant—first-time mother (has no other children), pregnant—experienced mother (already has one or more child), or new mother (had at least one pregnancy delivery in the prior 12 months).
Post hoc logistic regression analyses were performed to more clearly evaluate variation of findings by pregnancy status and other demographic covariates. Dependent variables were dichotomized, and all independent variables were included in the model.
The study was conducted in accordance with legal and regulatory requirements, as well as with scientific purpose, value, and rigor and followed generally accepted research practices described in Guidelines for Good Pharmacoepidemiology Practices (GPP) issued by the International Society for Pharmacoepidemiology (ISPE) and Good Practices for Outcomes Research issued by the International Society for Pharmacoeconomics and Outcomes Research.
Results
Among 800 respondents, 300 were first-time pregnant mothers, 300 were experienced pregnant mothers, and 200 were new mothers. Overall, the mean age was 30.4 years, the majority (82%) were married, lived in urban areas (58%), and were employed (54%). Full sample distribution is summarized in Table 1.
Patient Demographics

Percentages may not sum exactly to 100% due to rounding to the nearest full percentage point.
N/A, not applicable; Std. dev., standard deviation.
Awareness and perception of RSV
Overall, 90% of respondents were aware of RSV as a childhood disease with comparable awareness to influenza (89%). New mothers and experienced pregnant mothers reported a high level of awareness of RSV as a childhood disease (93% and 94%, respectively), with slightly lower rates of awareness among pregnant first-time mothers (83%), as shown in Figure 1. A majority (62%) of first-time pregnant mothers believed most RSV cases in babies were easy to treat, as shown in Table 2. However, there is potential for misinterpretation by survey respondents due to the absence of widely approved antivirals and the recent introduction of immunization strategies for RSV, suggesting caution in the interpretation of this finding. In general, there is a trend of first-time pregnant mothers differing from experienced mothers on RSV perceptions, such as being less likely to agree that RSV can cause serious complications in babies (58% compared with 77% of pregnant experienced mothers and 87% of new mothers who are not pregnant), that RSV can be life-threatening in babies (50% compared with 72% and 91%), and that it is important to protect babies from RSV immediately from the moment they are born (55% compared with 74% and 85%). RSV was perceived as a serious condition by more than 90% of respondents across all categories, with similar rates of respondents agreeing that protection from RSV was important to protect babies against such an infection, as shown in Table 3.
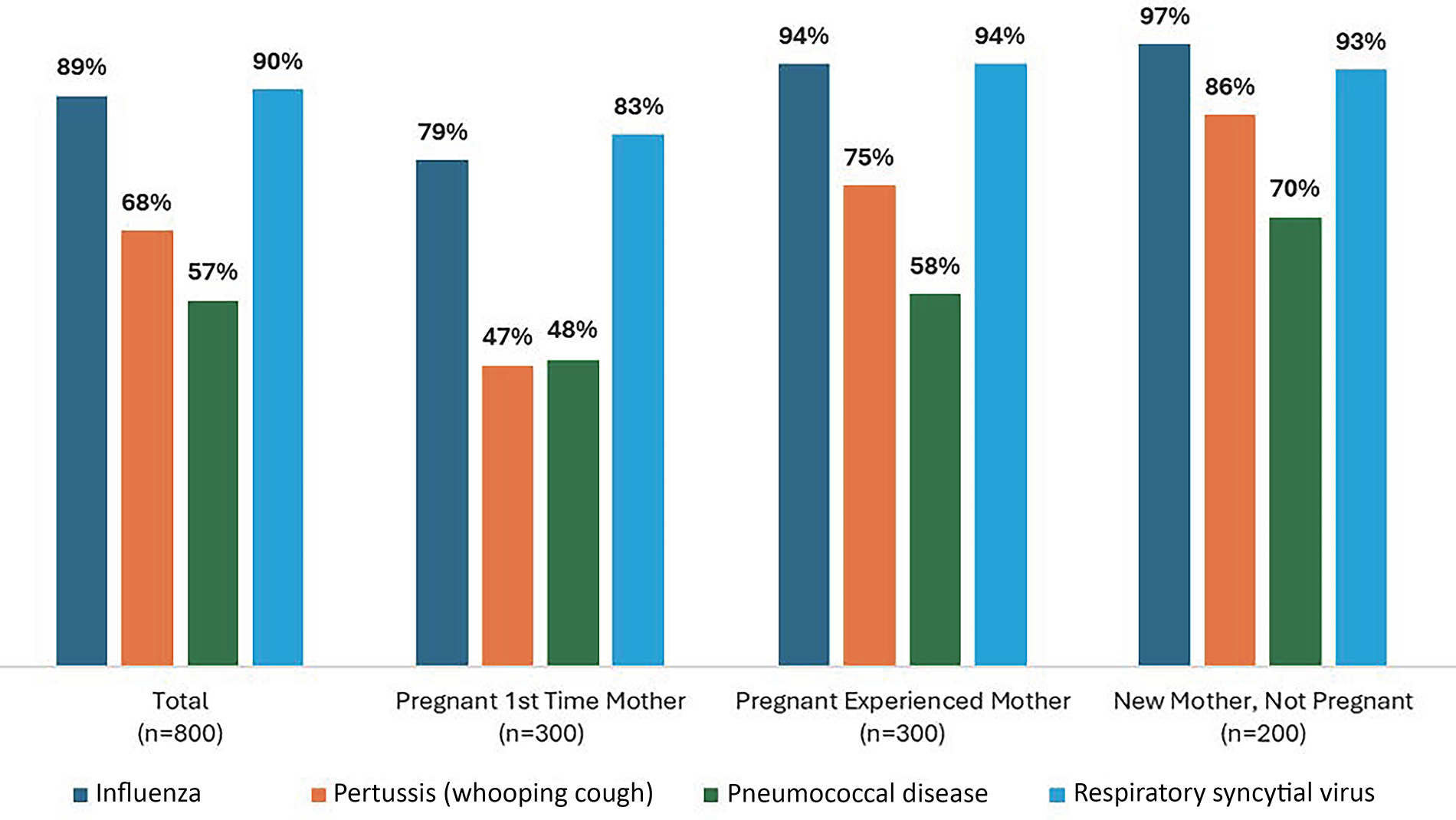
Awareness of childhood diseases—percent of respondents aware of each disease. (Percentages may not sum exactly to 100% due to rounding to the nearest full percentage point.) RSV, respiratory syncytial virus.
Agreement with Statements About Respiratory Syncytial Virus
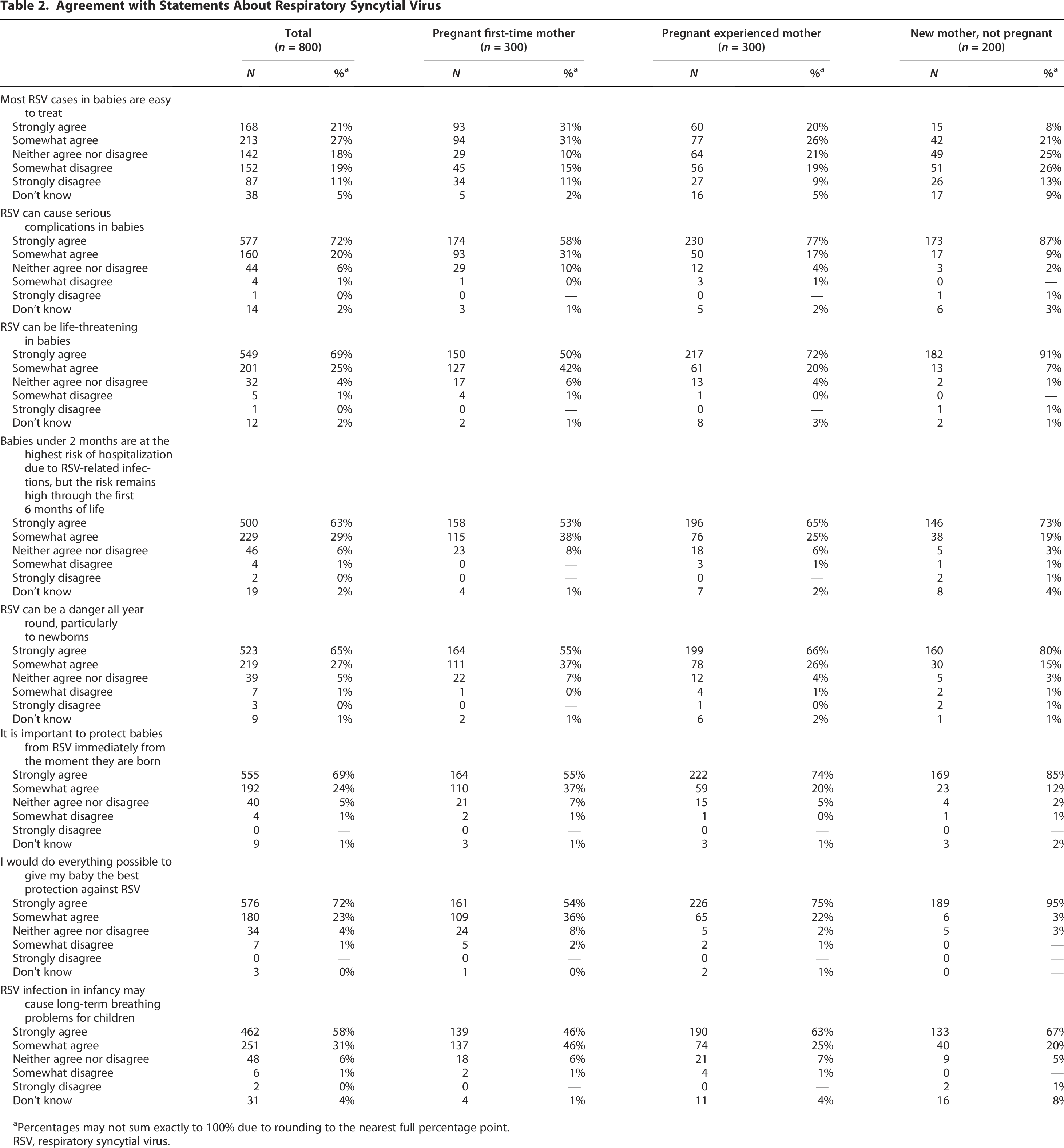
Percentages may not sum exactly to 100% due to rounding to the nearest full percentage point.
RSV, respiratory syncytial virus.
Seriousness of and Importance to Protect from Respiratory Syncytial Virus—Among Respondents Aware of Respiratory Syncytial Virus
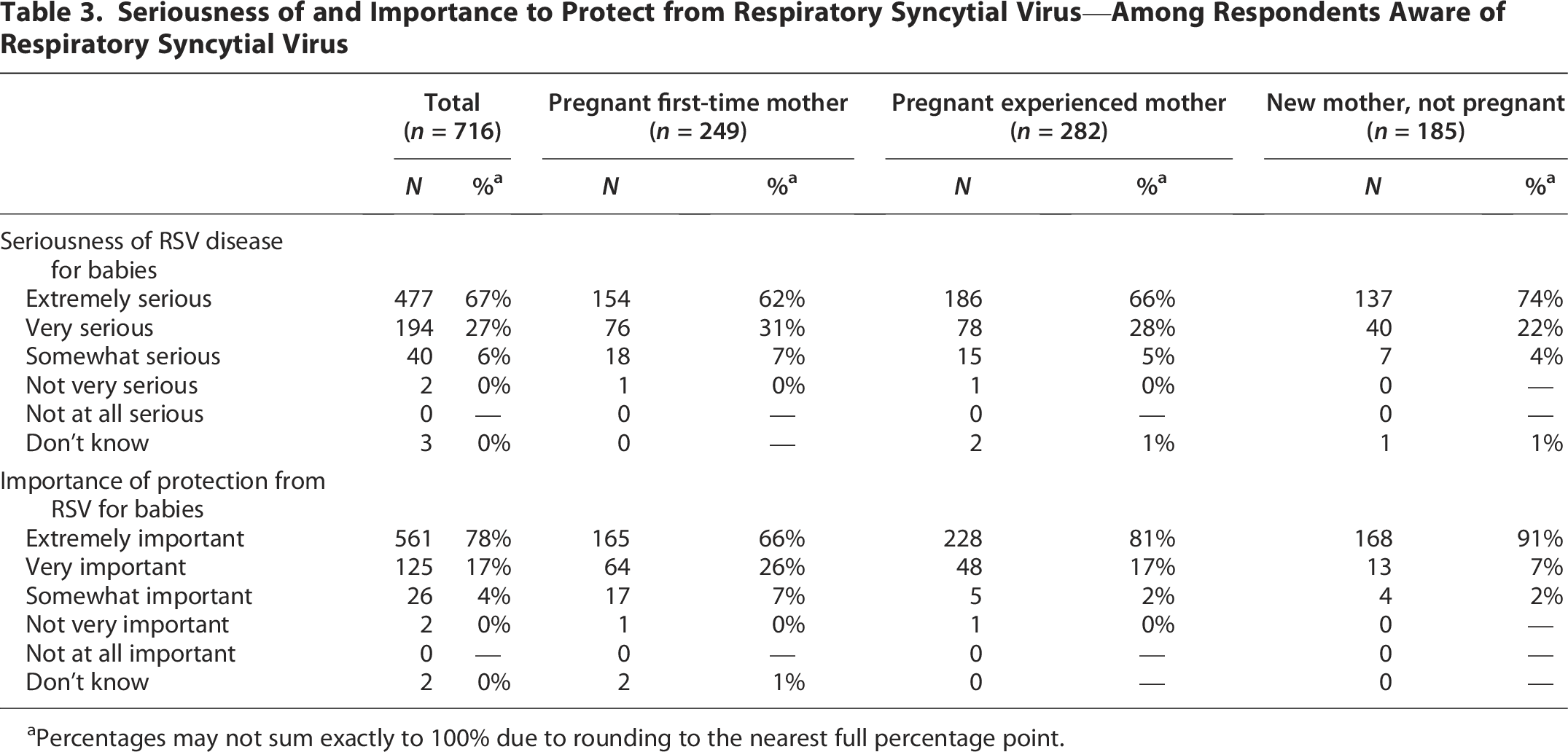
Percentages may not sum exactly to 100% due to rounding to the nearest full percentage point.
RSV prevention
More than 80% of survey respondents expressed willingness to get an RSV vaccine during pregnancy to protect their baby from RSV, as shown in Figure 2. If given the option of a maternal RSV vaccination or immunizing their baby after birth, approximately two-thirds (65%) of respondents opted for vaccination during pregnancy, as shown in Figure 3. Preference for maternal vaccination was highest among new mothers (75%) and lowest among pregnant first-time mothers (58%) but still represented the majority preference for all groups.

Willingness to receive RSV vaccine during pregnancy, overall and based on strength of HCP recommendation. (Percentages may not sum exactly to 100% due to rounding to the nearest full percentage point. Full survey wording: Strongest Recommendation = “RSV is very serious and I strongly recommend you get vaccination,” Moderate Recommendation = “RSV vaccine is recommended and I think it is a good idea to get vaccinated,” Lightest Recommendation = “RSV vaccinate is recommended, here is some information for you, think about it and let me know if you want to get the vaccine.”) HCP, health care provider; RSV, respiratory syncytial virus.

Preference for maternal RSV vaccination versus immunizing baby after birth. (Percentages may not sum exactly to 100% due to rounding to the nearest full percentage point).
Impact of HCP recommendation
Overall, a strong doctor recommendation was associated with greater willingness and intent (56% “definitely” would receive a vaccination) compared with a lighter recommendation (47%). New mothers were most likely to “definitely” receive vaccination based on a strong recommendation (61%), while pregnant first-time mothers saw the greatest increase in likelihood to vaccinate based on the strength of recommendation (13 percentage point increase from lightest to strongest recommendation), as shown in Figure 2.
Post hoc logistic regression analysis
Post hoc regression analyses elucidated few trends across the key metrics measured. New mothers who were not pregnant at the time of the survey were more likely to consider RSV “extremely” serious if contracted by babies (odds ratio [OR] = 1.92, 95% confidence interval [CI]: 1.17–3.15) and more likely to consider protecting babies from RSV to be “extremely” important (OR = 3.74, 95% CI : 2.12–6.58). Those working full or part time were more likely to receive maternal RSV vaccination after an HCP recommendation, no matter the strength (strong recommendation: OR = 2.16, 95% CI: 1.38–3.40; moderate recommendation: OR = 2.02, 95% CI: 1.33–3.09; light recommendation: OR = 2.39, 95% CI: 1.54–3.72). Similarly, those employed full or part time were more likely to receive maternal vaccination over immunization after birth if given the choice to decide (OR = 2.35, 95% CI: 1.65–3.35). Full findings from the regression analysis can be found in Supplementary Table S3.
Timing of vaccination and provider discussion
Most respondents (84%) expressed a desire to be given the choice to receive maternal vaccination, even if their due date was outside of the typical RSV season (Fig. 4). Regarding maternal vaccination, protection immediately from birth was the top reason for preferring this form of RSV vaccination (55%, noticeably higher among new mothers), followed by the vaccine’s safety for the mother, baby, and the pregnancy (45%–48%), and recommendation from their HCPs (44%). Full distribution of these results can be found in Supplementary Tables S1 and S2.

Agreement with desiring the choice to receive maternal RSV vaccination for birth in non-respiratory season. (Percentages may not sum exactly to 100% due to rounding to the nearest full percentage point).
Finally, mothers expressed a preference to discuss recommended vaccines either at their initial appointment after confirming that they are pregnant or a few weeks prior to administration. Only 23% of respondents would prefer to first discuss vaccination on the day they would receive the vaccine. Mothers also prefer to receive vaccination at their OB/GYN’s office or a hospital if it is their first pregnancy. Notably, adding the stipulation that they would need to go to a pharmacy to receive the vaccine did not have a detrimental impact on vaccine willingness. Full distribution of preferred timing and location can be found in Table 4.
Preference on the Timing of Discussion and Location of Vaccination

Percentages may not sum exactly to 100% due to rounding to the nearest full percentage point.
HCP, health care provider; OB/GYN, obstetrician/gynecologist.
Discussion
In our study, pregnant people expressed an overwhelming desire to protect their infants against RSV, and the majority preferred vaccination during pregnancy as the prevention approach. The study findings are consistent with a prior U.S.-based survey study that found that nearly two-thirds would accept RSV protection, either by vaccine or immunization, with side effects being the most notable concern among participants. 17 There is a notably higher rate of interest for vaccination during pregnancy in our study than prior study by Saper and colleagues. 6 This difference may be at least partially attributable to the current study clearly outlining both potential options and soliciting a preference, as opposed to asking about general likelihood without a comparator. This difference clearly highlights the importance of ensuring pregnant adults and new mothers are sufficiently informed of the choices currently available to them.
The awareness levels of RSV reported in this study are notably higher than those in similar studies conducted in the past.18,19 This increase in observed awareness levels could be driven by increased media coverage and advertising over the past few years that accompanied the introduction of new interventions for RSV. Nonetheless, this limits the ability in the current study to evaluate the role of various awareness campaign efforts in enhancing public knowledge of RSV. Additional research that assesses these key efforts may inform future public health education. Similarly, further research should explore the role that prior RSV knowledge may have in the acceptance rates of preventative options for RSV.
The current study builds upon past research by noting the importance of a strong HCP recommendation in patient willingness to accept RSV preventative measures. This highlights the role of HCPs in furthering the protection of infants from RSV infection from birth. Our study results also highlighted the ongoing need for education and awareness among new, or soon-to-be, parents regarding the potential seriousness of an RSV infection. This is consistent with past research showing higher levels of vaccine uptake intention among those who recognize the seriousness of RSV infection,6,10 further supporting that education could increase understanding of disease severity, vaccine interest, and intention to be treated whether during pregnancy or as a new mother.
Finally, parental autonomy emerged as an important factor in vaccine preferences and uptake intentions. In addition to an overall preference for the maternal vaccine as opposed to immunizing infants at birth, respondents also expressed a clear desire for the ability to choose for themselves between the available immunization methods, including the opportunity to opt for the maternal vaccine even if their expected delivery date occurs outside of the typical RSV season to ensure protection for their baby. Similarly, mothers expressed a desire to discuss vaccination with their HCP before the day they would receive the vaccination, affording them time to weigh their options and not feel rushed to decide.
Strengths and limitations
Strengths of the current study include surveying a large sample of respondents across the country and understanding the potential implications regarding HCP practice behaviors on vaccination uptake. The study adds to existing research that already showed people generally agree that HCPs are trusted and reliable sources for RSV information, 17 with our results now suggesting that how this message is delivered may also be crucial.
This study has several limitations. Research questions were descriptive in nature and employed self-report methodology, limiting the certainty with which these study results can be extrapolated to a broader population. Although standard quantitative survey tactics like the inclusion of Likert and gradation scales were used widely, there remains potential for differing interpretation of scale points from respondent to respondent. The current study also relied upon the willingness of respondents to be enrolled in a research panel, which invites the possibility of selection bias, or aligned in some fashion with a patient advocacy group, which invites the possibility of information bias. As the objective of the survey was to explore preferences among those who would consider vaccination, respondents who self-reported opposition to any vaccination were excluded from the study, which may introduce potential selection bias. However, studies estimate this represents less than 10% of potential U.S. respondents. 20 There is also potential for seasonality to impact responses, such as RSV awareness. The study was fielded in December, which coincides with a potential increase in discussions with HCPs about RSV and media exposure stemming from new RSV vaccination brand launches. Furthermore, the study may not collect all pertinent information that influences the participants’ awareness and perception, such as the number of prenatal visits that survey respondents may have had prior to study participation. The absence of this information limits our ability to determine to what extent, if any, participants may be biased toward higher awareness of RSV based on HCP counseling during prenatal visits.
Conclusions
The majority of respondents in our study believed that protecting their infants from RSV was important, and approximately two-thirds preferred vaccination during pregnancy. Higher vaccination coverage is achievable with a strong HCP recommendation, earlier HCP counseling during pregnancy, and flexible vaccination timing outside a typical RSV season.
Writing Support
Medical writing support was provided by E.J.P., MD, at Ipsos Insight LLC and was funded by Pfizer.
Ethics Approval
This study was conducted in full conformance with the Guidelines for GPP published by the ISPE, the Helsinki Declaration of 1964, and the laws and regulations of the countries in which the research was conducted. Informed consent was collected from all participants prior to participating in the study. No identifiable data were collected during the course of this research or are included in this article. The study (Protocol #2024-0499) was approved by the Pearl IRB of Indianapolis, Indiana, on November 26, 2024.
Authors’ Contributions
A.W.L.: Conceptualization, methodology, writing, visualization, and supervision. M.J.H.: Methodology, analysis, investigation, writing, visualization, and project administration. S.J.P.: Writing, visualization, and supervision.
Footnotes
Author Disclosure Statement
A.W.L. and S.J.P. are employees of Pfizer Inc., manufacturer of Abrysvo. M.J.H. is an employee of Ipsos Insight LLC, which was a paid consultant to Pfizer in connection with the development of this article and conducting this research study.
Funding Information
This study was sponsored by Pfizer Inc.
Supplemental Material
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
