Abstract
Helical structures are ubiquitous in natural and engineered systems across multiple length scales, while they often exhibit nearly uniform radius and pitch. Utilization of biomimetic structure design to develop stretchable and robust 3D helical structures that are shape-preserving and heat-tolerant is of great interest. Herein, we devise flexible 3D helixes by supramolecular self-assembly and high voltage electric field orientation of molecular chains. The 3D helixes not only inherit buffering mechanics with exceptional shape-retention ability against cyclic stretching but also exhibit excellent heat-resistant quality. The results of the experimental tests confirm that the helix can recover its original shape without obvious residual strain and basically withstand after 2000 cycles under force loading of 10 cN. Such robust biomimetic materials hold great prospects in the fields of artificial muscles, wearable materials, and so on.
Introduction
There are many three-dimensional (3D) spiral structures in nature, such as DNA, bacterial flagella, plant tendrils, and sea snail shells. 1 It has been shown that the macroscopical 3D helixes can be regulated by the mechanical balance between surface stresses or internal residual strains and the induced elastic stretching and bending in numerous studies. 2 As one of the key building blocks of the macroscopical 3D helixes, it should retain reliability under substantial degrees of bending, twisting, compression, and stretching deformations.
At present, there has been research and applications on 3D spiral materials. The intrinsic shape of helixes responds to a variety of external stimuli of functional materials, such as hydrogels, liquid crystal elastomers, and shape memory polymers3–5 Carmelo De Maria 6 et al. printed a core–shell bio-based tendril-like structure, by simultaneously depositing a silk fibroin core into a bio-degradable polyhydroxybutyrate-co-valerate (PHBV) shell. With the increase in temperature and humidity, the fabricated spiral showed the capability to self-twist and contract. Cui Yande 7 et al. fabricated tendril-inspired hydrogels by incorporating tunicate cellulose nanocrystals into polymeric networks. These hydrogel muscles exhibited a high actuation rate, large actuation strain, and shape memory property in response to solvents. It has been found that during these external stimulus processes, the shrinkage and crispation of the helixes will usually be guided by the microstructure change of materials. 8
Despite these achievements, researchers have long overlooked two significant functional utilizations of helix structures. For one thing, the coiling structure of the 3D helix is similar to the spring, which can play the role of elastic buffer connection. For another, 3D helix coiling process can shorten or lengthen the axial length, realizing the axial contraction drive.9,10
Besides these 3D helixes with fixed twist shapes, shape-retention materials capable of performing reversible stretching or chiral twisting motions have attracted more and more scientific attention. 10 Shape persistent polymer is a novel type of stimuli-responsive material, for which any given shape that can be reached at an intermediate stage of the process could be preserved.11,12 Fabric shape retention is a crucial property that impacts the durability and usability of materials. To fabricate such shape-retention and heat-tolerant elastic helixes, the essential element is to understand and further mimic bending and chiral twisting motions.
Herein, we devise a shape-retention and heat-tolerant elastic helix by judiciously incorporating chiral molecules into flexible polyimide. The supramolecular assembly helixes readily inherit the functional merits of their counterparts in nature, including the temperature resistance of polyimide (PI) and the ultraviolet light (UV) responsiveness of azobenzene (AZO). The azobenzene-polyimide supramolecular assembly (U-AZO/PI) showed high molecular alignment, which was induced by electric field. The U-AZO/PI ribbons with the S-811 curl into helixes at φ = 45°. Notably, the mechanical properties and temperature resistance of the 3D helixes have improved significantly. The helix can recover its original shape without obvious residual strain and basically withstand after 2000 cycles under force loading of 10 cN. Based on the general reports that such AZO-PI materials were initially impractical for shape persistent applications, the designed 3D helixes maintained shape stable even at high temperature (100°C). Encouraged by mechanical stability and temperature resistance, 3D helixes have the potential application in artificial muscles, sensors, and other biological devices.
Experimental
Materials
2,2′-Bis(trifluoromethyl)-4,4′-diamino biphenyl (TFDB, 99%, Aldrich) and 4,4’−(hexafluoroisopropylidene) diphthalic anhydride (6FDA, 99%, Aldrich) were purified by recrystallization before use. N, N−Dimethylacetamide (DMAc, J&K, 99.5%) was purified by stirring with NaOH and distilled twice from P2O5 under reduced pressure. S−(+)−2−octyl 4−(4−hexyloxybenzoyloxy) benzoate (S−811) (TCI, 98%) (TCI, 98%) was used without previous purification. All other reagents were purchased from Aldrich and used as received.
Synthesis
Synthesis of azobenzene
The 3,3′,5,5′-azobenzenetetracarboxylic acid (AZO) was synthesized by a diazo-reaction. Dry 5-amino-m-phthalic acid (10 mmol) was dissolved in 50 mL (1 mol/L) of sodium hydroxide solution (NaOH), and 1 mol/L sodium nitrite solution (NaNO2) was added to this solution at 0°C. Then, the mixture was added to a certain volume of hydrochloric acid (HCl, 1 mol/L) drop by drop and stirred for 0.5 h at 0°C to form diazonium salt. A mixture of diazonium salt and isophthalic acid in alkaline solution was regulated the pH value (pH = 7) by sodium carbonate (Na2CO3), and then stirred at 0°C for 5 h. The resultant raw materials were filtered, washed with deionized water and then purified by chromatography on silica with dichloromethane/petroleum ether as eluent. Finally, the products were dried in a vacuum (−0.1 MPa) at 60°C. The yield was 73%. 1H NMR (400 MHz, DMSO-d6 δ): 13.35 (s, 4H, COOH), 8.42 (d, 4H, CH), 7.58 (t, 2H, CH). Scheme 1. Reaction process of AZO.
Synthesis of 2-ureido-4[1H]-pyrimidinone (UPy)
Under an N2 atmosphere, 2-amino-4-hydroxy-6-methylpyrimidine (0.025 mmol) was stirring at 25°C, slowly dropping hexamethylene-diisocyanate (0.173 mmol) into the flask with three necks. The reaction mixture was stirred for 0.5 h at 25°C, and then reacted 100°C for 15 h. After the reaction, the product was filtered, precipitated with excess petroleum ether at room temperature and dried under a vacuum at 60°C to obtain UPy. Yield: 89%. 1H NMR (400 MHz, Chloroform-d δ): 13.06 (s,1H, NH), 11.84 (s,1H,CH), 5.85 (m,1H,CH), 3.39 (m, 4H, CH2), 2.56 (d, 3H, CH3), 1.67 (s, 4H, CH2). Scheme 2. Reaction process of UPy.
Synthesis of 6FDA-TFDB fluorinated polyimide (f-PI)
The polyimide precursor (PAA) was synthesized from TFDB and 6FDA with an equivalent molar ratio. The TFDB (5 mmol) in 10 mL DMAc was stable at 25°C, and then 6FDA (5 mmol) in 10 mL DMAc was added dropwise over a period of 30 min under vigorous stirring to enhance reaction efficiency. After the addition of 6FDA, the reaction mixture was stirred at 25°C for 24 h under a nitrogen atmosphere, obtaining the clear and viscous PAA solution. PAA was dried in a vacuum oven at 60°C for 6 h, and then cured at different conditions (80, 150, 250, and 350°C) under nitrogen in a tube furnace, according to the method described in a previous work. 13 As a result, the desired polyimides were performed.
Preparation of 3D elastic helix
The PI, AZO, UPy and chiral dopant S−811 were dissolved and mixed in DMAc uniformly. An appropriate molar ratio of PI: AZO: UPy is 1:25:20. The small amount of the chiral dopant (S-811) was added to the mixture to favor a left-handed twist with molar ratio of 1:6 (PI: S−811). 10 The supramolecular assemblies were coated on the glass substrate by two methods of solvent evaporation and spin coating, and then subjected to thermal curing in a vacuum oven at 50°C for 24 h. The area of the U-AZO/PI film is 4.5 × 4.5 cm2. The films were orientated by high voltage electric field (1.5 kV, 0.5 h). And then, the aligned U-AZO/PI films were cut at an angle of 45° to prepare 3D helixes, according to previously reported in the literature. 10 The length and width of the cutting samples were 2.5 cm and 0.8 mm, respectively.
Material characterizations
The high voltage DC power supply (DW−P503−1AC) was used to induced orientation process of the materials. Fourier transform infrared spectroscopy (FT−IR) were recorded on a Bruker Tensor 650 spectrometer with a disc of KBr. Nuclear magnetic resonance spectra (1H NMR) were measured by a Varian INOVA 500 MHz spectrometer with trimethylsilyl as an internal standard as a reference for chemical shifts. Molecular weights were determined by using a JASCO 860 gel permeation chromatography (GPC, Japan Spectroscopic Co., Ltd.) equipped with a Waters system in reference to a series of standard polystyrenes with tetrahydrofuran (THF) as eluent. X-ray photoelectron spectroscopy (XPS) studies were performed at a power of 450 W using a PHI 1600 surface analysis system equipped with an Mg Ka anode. Polarized ultraviolet–visible (UV–Vis) absorption spectra were recorded by 8453 ultraviolet–visible spectrophotometer (Agilent Technology Inc) at room temperature. Thermogravimetric analysis (TGA, NETZSCH STA 449C, 30–800°C, 10°C min−1 in N2) was carried out to study the thermal properties of the films. The cross-section samples of the U-AZO/PI films were successfully prepared by liquid nitrogen freezing. The microstructures of aligned U-AZO/PI film were observed by a scanning electron microscope (SEM, Phlilps XL30E). The elasticity and helical shape of 3D helical ribbons were studied during 2000 stretching-relaxation cycles at room temperature and treated at 100°C. All experiments were conducted on a dynamic mechanical analyzer (DMA-Q800, TA Instruments). The load capacity was 10 cN.
Results and discussion
The target supramolecular polymeric assembly is constructed by mixing UPy, AZO, chiral molecule, and soluble polyimide, as structurally presented in Schemes 1 and 2. The structures of the assembly are stabilized by both intramolecular hydrogen bonds and steric repulsion.
14
It is worth mentioning that fluorinated polyimide was chosen as the building blocks for constructing supramolecular polymeric assembly because it contained a special molecular structure that facilitated the formation of hydrogen bonds, and exhibited good solubility, flexibility, and heat resistance.
15
The fluorinated polyimide derived from typical diamine and anhydride (6FDA, TFDB) was synthesized according to our earlier studies dealing with polyimide (PI).10,13 More details are presented in the Experimental Section. The synthesized polyimide is characterized by GPC. Figure 3(a) shows the molecular weight distribution of pure PI. The molecular weight distribution of the PI shows only one peak and thus confirms that the unreacted raw materials are completely washed away by organic solvents. The number-average molecular weight (Mn) of PI is 53,834, and the weight-average molecular weight (Mw) of PI is 101,372. As a result, the molecular weight distribution (MWD) is 1.88, indicating that the PI has perfect quality with a narrow molecular weight distribution. (a) GPC of pure polyimide; (b) XPS of pure polyimide and U-AZO/PI.
The element contents of PI and U-AZO/PI.

A comparison of the FT-IR spectra of UPy, AZO, PI, and U-AZO/PI is presented in Figure 4. UPy is synthesized by the reaction of the amino group on 2-amino-4-hydroxy-6-methylpyrimidine and an isocyanate group on hexamethylene diisocyanate, forming an amide bond. As shown in Figure 4, the C≡N stretching vibration absorption peak of the isocyanate group appears at 2270 cm−1. There are amide bond absorption peaks at 1701 cm−1 and 1605 cm−1, which proves that UPy is successfully synthesized.
16
The absorption bands near 1580 cm−1 and 1450 cm−1 indicate the existence of a benzene ring in AZO (Figure 4). Furthermore, the 1690 cm−1 absorption peak represents the stretching vibration of C = O in the carboxyl group, the 1404 cm−1 absorption peak represents the in-plane bending vibration of O-H in the carbonyl group, the 1282 cm−1 absorption peak represents the stretching vibration of C-O in the carboxyl group, and the 914 cm−1 absorption peak represents the out of plane bending vibration peak of O-H.
17
These four peaks indicate the presence of carboxyl groups in the AZO molecule and the AZO was successfully synthesized. The FT-IR spectrum of PI shows important absorption bands at 1665 cm−1 which is attributed to the C = O stretching of amide bonds. The peak at 1418 cm−1 corresponds to the C-N stretching vibration, and the absorption band at 1350 cm−1 is due to the stretching vibration of C−F.
18
FT-IR spectra of AZO, UPy, PI and U-AZO/PI.
The intermolecular hydrogen bonding among PI, UPy and AZO plays a key role in the aggregation of molecules resulting in the stabilization of the molecular network and the uniformity of film. 14 In order to characterize the multiple hydrogen bonds in the U-AZO/PI film, the film is compared with PI, UPy and AZO (Figure 4). Compared with the C = O stretching vibration of PI, the U-AZO/PI of C = O stretching vibration exhibits a distinct red shift from 1665 cm−1 to 1676 cm−1. At the same time, due to the formation of internal hydrogen bonds between N and H, the absorption peak of N-H appears at 1506 cm−1. In addition, fluorine would form H⋅⋅⋅F interactions rather than F⋅⋅⋅F contacts. 10 Similar unusual behavior is observed in the present compound, where the C−F stretching frequency is blueshifted from 1350 cm−1–1354 cm−1, attributing to the intermolecular N−H⋅⋅⋅F−C hydrogen bond. This indicates the formation of intramolecular hydrogen bonds among PI, UPy, and AZO.
To examine the thermal activities of hybrid materials in higher temperature range and their thermal decomposition characteristics, TGA of PI and U-AZO/PI films are carried out (Figure 5). The T5% value of pure PI is 507°C and the residual weight rate is more than 59.1% at 800°C, as shown in Figure 5. The weight decreases continuously from 500 to 650°C. This step of weight loss is mostly due to the conversion of four carbonyl groups (C = O) in the imide part to either CO or CO2. Above 650°C, weight loss begins to decrease as CH4 and H2 start to evolve.
19
In addition, incorporating UPy, AZO and S-811 into polyimide reduces the thermal stability. This indicates that the T5% value of U-AZO/PI film is 500°C, which is slightly lower than pure PI. And a total weight loss of 46.1% during pyrolysis in N2 was observed. All these data indicate that supramolecular polymeric assembly in this study has excellent thermal stability. TGA curves of PI and U-AZO/PI films.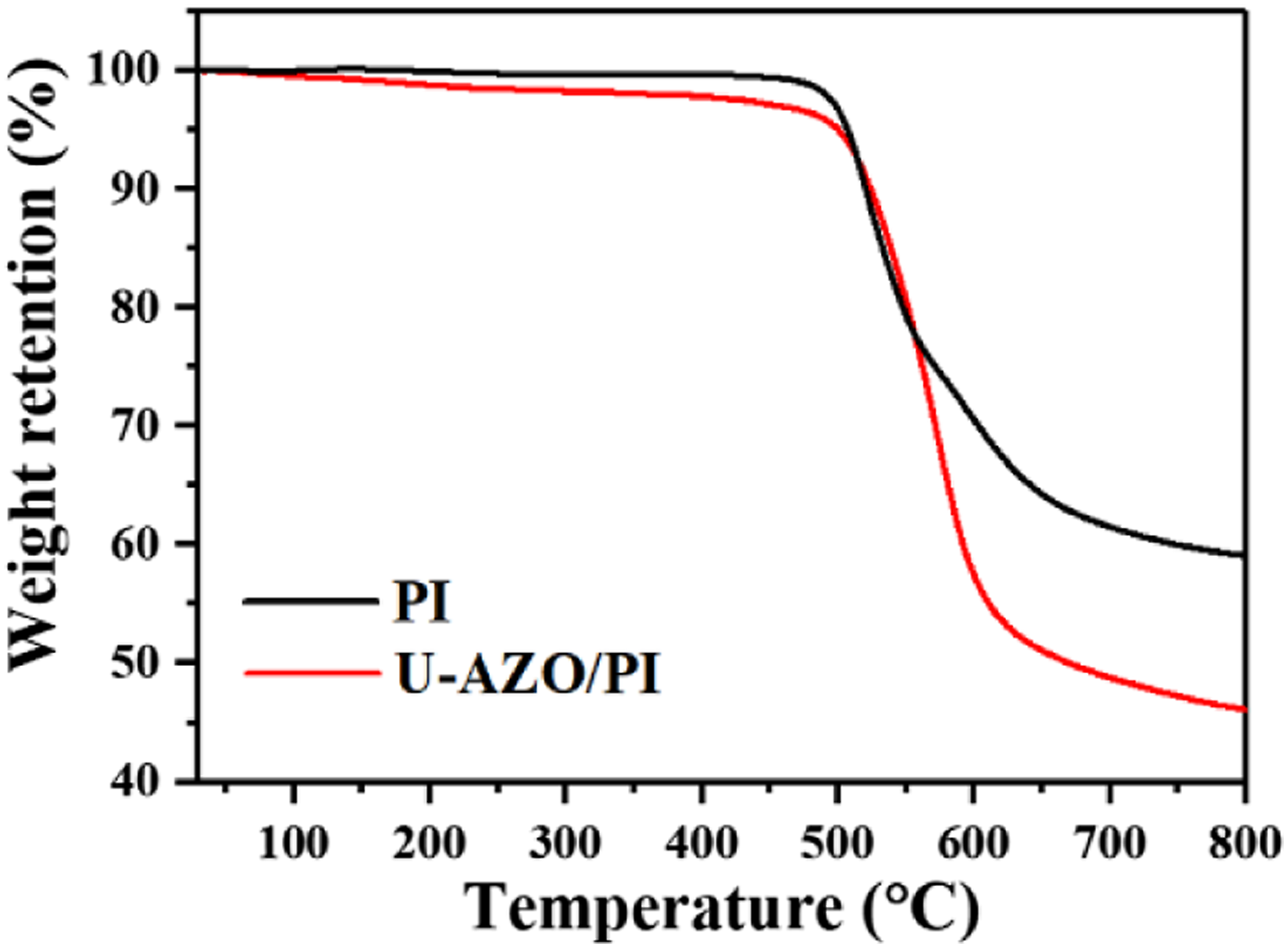
To see whether these supramolecular assemblies treated by high-voltage electric field have ordered structures, the polarized optical properties are studied by polarized UV-Vis absorption spectroscopy. As shown in Figure 6(a), the electric field-oriented U-AZO/PI film has certain absorption in the ultraviolet light range, and its maximum absorption peak is at 337 nm. After being oriented in a high voltage electric field, a large dichroism is induced at the π−π* electron transition of the AZO at about 337 nm. Furthermore, the intensity of the polarized absorption band at 337 nm in the direction parallel to the electric field direction (A//) is much higher than the intensity perpendicular to the electric field direction (A⊥). Optical anisotropy is evaluated by a dichroic ratio (DR) according to equation (1). Polarized UV-Vis absorption spectra of U-AZO/PI film: (a) high-voltage electric field-oriented; (b) without high-voltage electric field treatment.

The DR of the electric field-oriented U-AZO/PI film is 0.12, which indicates the noteworthy anisotropy of U-AZO/PI films. In contrast, the DR of U-AZO/PI film without high-voltage electric field treatment is 0.003, indicating that the film is isotropic (Figure 6(b)).
Based on the above characterization (GPC, FT-IR, TGA, and polarized UV-Vis absorption spectra), we concluded that oriented supramolecular polymeric assemblies with excellent heat resistance were successfully prepared. However, their exact surface morphologies are still unclear. Therefore, the surface and cross-section morphology of U-AZO/PI films are characterized by field-emission scanning electron microscopy (FESEM). Figure 7 shows the morphology of the freestanding film. Obviously, no trace of aggregates or damage can be observed in the films, confirming that the films show large-scale uniformity and smoothly (Figure 7(a)). As shown in Figure 7(b) and (c), it can be found that the cross-section morphology of the film is compact, and the cross-section roughness is believed to be caused by the ductile fracture of film.
20
The image of the cross section indicates that the thickness of the film is 9.8 μm. Furthermore, analysis of the variance method is applied to the study of the film thickness and film uniformity which were influenced by polymer concentration, spin velocity and spin acceleration in the process of spin coating (Figure 7(d)). The average thickness of the films was approximately 9.6 ± 0.32 μm. The surface morphology and thickness of U-AZO/PI films: (a–c) SEM images; (d) thickness of U-AZO/PI films.
Macrochiral helical structures are not only of great value to stimuli responsive polymeric materials but also have potential in high elastic materials.10,21,22 The 3D elastic helixes in this work show extraordinary elasticity, which allows for large deformations without fracture. The helix can bear high tensile force and almost recover its original shape after release of the tensile force. The elastic properties are characterized by dynamic thermomechanical analysis (DMA). Figure 8(a) shows a plot of tensile strength–strain (Rm versus ε) curves of the 3D elastic helixes. It is proposed that three distinct stages are observed during the loading process: the first stage is a linear-elastic deformation stage at ε< 80%, the second stage is slippage and rearrangement, and the third stage is crushing and rearrangement.
23
Based on the spiral structure of the ribbon, the slope of the linear relationship between strain and force corresponds to the purely elastic behavior of the helixes (k). As shown in Figure 8(a), the k of a helix (No.1: pitch = 3.27 mm, diameter = 1.71 mm) is 6.69 × 10−4 N/m, which is greater than k (No.2: k = 5.55 × 10−4 N/m) of a helix with a pitch of 2.86 mm and a diameter of 1.72 mm. It indicates that the elastic constant of the helix is proportional to the pitch length. In particular, the cyclic loading-unloading progresses within the force below 30 cN merely exhibit elastic deformation, and the helix almost recovers its original strain after release of the tension. The helix overwinds under increasing force, and thus we examine the elasticity of 3D left-handed helixes under a stretching force of 10 cN in the remainder of this paper. The variations in helix pitch and strain are shown in Figure 8(b). It can be found that, for all helixes with very similar diameters, as the helix pitch increases, strain gradually decreases. As shown in Figure 8(c) and (d), the helix (pitch = 2.86 mm, diameter = 1.72 mm) shows an elastic deformation up to 34.26% at 10 cN, which is higher than that of the helix with a pitch of 3.27 mm and a diameter of 1.71 mm before cycling (17.14%). Furthermore, the helix can recover its original shape without obvious residual strain (ε) and basically withstand after 2000 cycles under force loading of 10 cN. It is indicated that the helix (pitch = 2.86 mm, diameter = 1.72 mm) can almost recover its original length after 2000 cycles, with 97.1% maximum reversible stress retention, displaying outstanding fatigue resistance (Figure 8(c)). After 2000 cyclic loading-unloading processes, the helix with a pitch of 3.27 mm and a diameter of 1.71 mm can also recover its original structure with a strain of 18.15% and 94.1% reversible stress retention (Figure 8(d)). These results provide direct evidence that the helixes have good shapes and favorable stability. Cyclic force-strain measurements further confirmed that the improved mechanical properties of the helix are associated with the favorable skeleton of the PI matrix. Mechanical properties of 3D elastic helixes: (a) Stress–strain curves of helixes with different pitches; (b) The variation of elongation with pitches; (c) Cyclic tensile strength–strain curves (10 cN, pitch = 2.86 mm, diameter = 1.72 mm); (d) Cyclic tensile strength–strain curves (10 cN, pitch = 3.27 mm, diameter = 1.71 mm).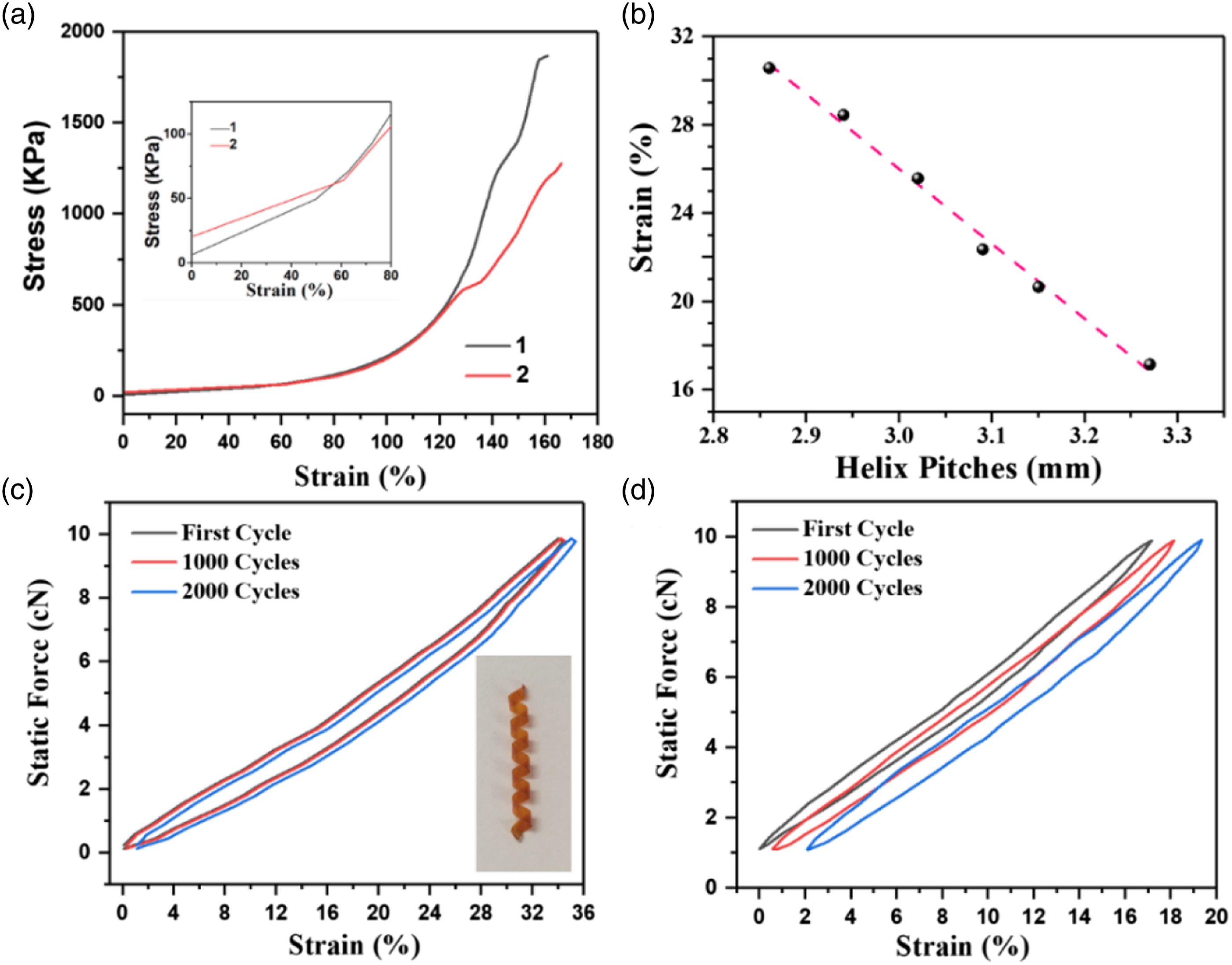
Furthermore, based on the high alignment of the rigid molecular chains and the temperature resistance of PI, the 3D helix can retain its linear-elastic deformation after heat treatment. The shapes of 3D helixes based on U-AZO/PI remain stable as the temperature rises from 25°C to 100°C, indicating the outstanding shape-persistence of U-AZO/PI helixes. To verify the elastic property and shape recovery property, we tested the stress–strain curves of the U-AZO/PI helixes with proper heat treatment. The spring-like structure of U-AZO/PI helixes endows it with excellent deformation recovery ability, as shown in Figure 9(a). The non-heat treated U-AZO/PI helix presents the largest elongation of 35.8% strain and it can recover to its original length without deformation. Compared with the normal helix, the tensile properties of the helix with 100°C thermo treatment also remain stable, while the largest elongation of U-AZO/PI helix is slightly higher (36.2%). Moreover, there was no breakage in the U-AZO/PI helix under the force of 10 cN after 2000 consecutive cycles. Furthermore, the strain of the AZO/PI helix dealed with the temperature of 100°C is slightly increased as the number of cycles increases (Figure 9(b)). The stress–stain curves reveal that the spiral structure of U-AZO/PI helix is stable, while the material has fewer defects and the mechanical performance is stable. Mechanical properties of 3D helixes sealed with the temperature of 100°C: (a) tensile strength–strain curves; (b) the strain at different cyclic numbers.
It has been presented the flexural behavior of 3D helix using U-AZO/PI assembly. The process (Figure 10) demonstrates the applicability and controllability of the 3D helix. As shown in Figure 10, the 3D helix is fixed on a finger. It can always bend with the bending of fingers, and shows amazing bending performance. Moreover, the 3D helix can also be used for the next bending process cycle. The 3D helix is induced to repeatedly bending and straightening up to 100 times. The 3D helix remains undeformed upon 100 cycles. The stability of the bending helix is attributed to the excellent stability of U-AZO/PI assembly. The shape-preserving and heat-tolerant 3D helix can be developed for micro robotics by further optimizing. Photographs show the cyclic process of bending and straightening of the 3D helix.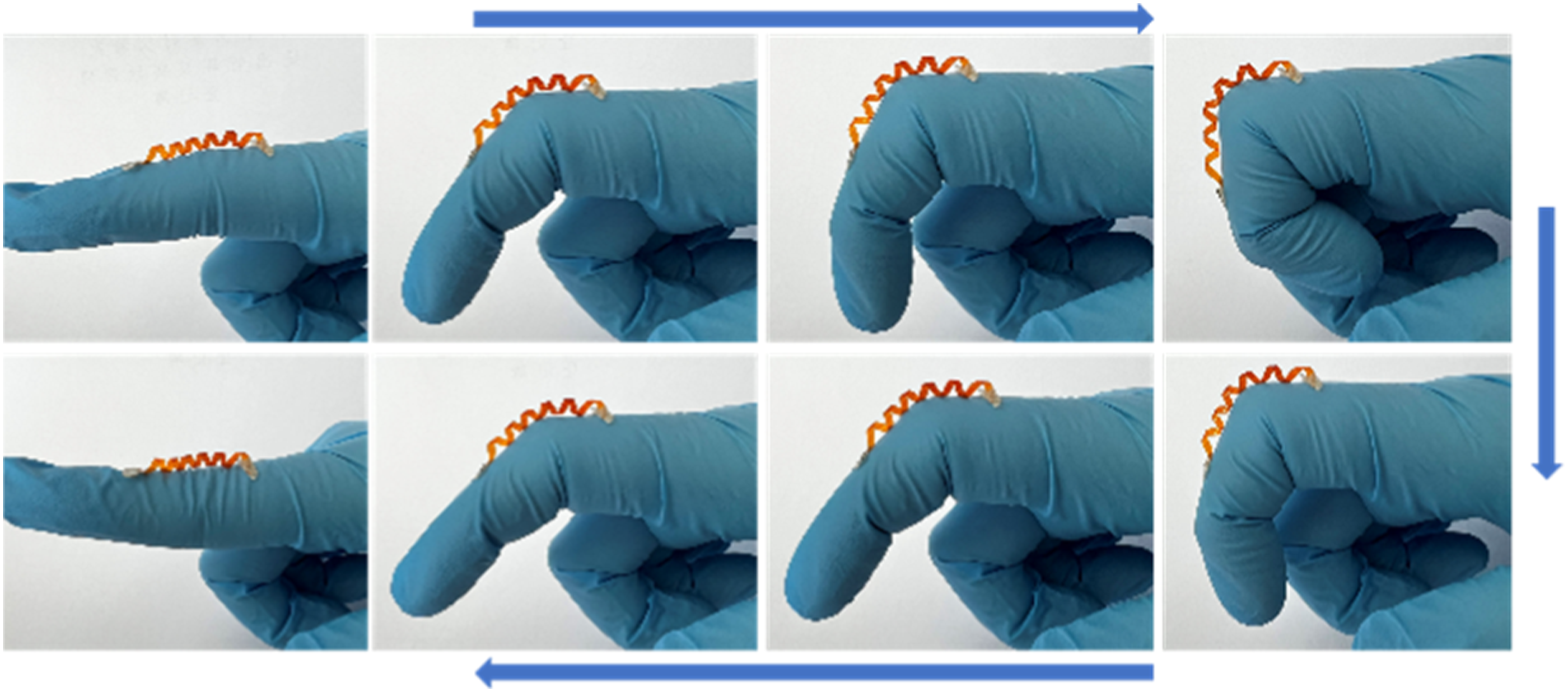
Light has been employed as a convenient energy source to trigger photomechanical response.
24
AZO embedded in a polyimide matrix can be used as a chromophore to harvest light and elicit a mechanical response.25,26 AZO molecules can be supramolecular crosslinked with other AZO molecules, UPy, and CF3 on the polymer chains. The movement of AZO molecules can effectively drive the movement of polymer chains, and ultimately greatly improve the photomechanical response of the entire film. The UV response of supramolecular assembly is further studied in this paper. The 3D helixes were irradiated by 365 nm UV light (LED-200). As shown in Figure 11, the U-AZO/PI helixes exhibit remarkable photo-induced deformations and mechanical behaviors. With the extension of UV irradiation time, the spiral pitches become larger. Furthermore, the slow cis-trans isomerization of AZO is mainly limited by high steric hindrance in the rigid PI matrix. After the irradiation, the Z-isomers in the assembly are stabilized by intermolecular H-bonds and polymer chains. When the UV light is stopped, the macro morphology of the helix remains stable. Thus, electric field-induced orientation of U-AZO/PI assembly with good stability is of paramount importance for fabricating 3D shape-persistent elastic helixes. Optical images of 3D helix under UV irradiation.
Conclusions
The current bionic research efforts on helical structures focus on either the preparation of materials with coiling geometry or the design of orientation microstructures. Through imitating the coiling mechanism, the 3D helix maintained the curl structure of spiral tendrils and successfully inherited the structure-related elastic buffering connection effect for its application. In summary, tendril-like functional polyimide helixes with diameters of 1.5–1.8 mm and pitches of 2.8–3.3 mm have been prepared by supramolecular self-assembly and orientated in high voltage electric field, wherein, the highly oriented U-AZO/PI films were cut at an angle of 45° to prepare 3D helixes. The thermal and mechanical properties of the U-AZO/PI helixes can be tuned with ease due to the temperature resistance and stability of the polyimide matrix. The helical structures provided considerable leeway to extend spring like elasticity with elongation of 35.8% at 10 cN (pitch = 2.86 mm, diameter = 1.72 mm). The helix can recover its original shape after 2000 cycles, accompanied by a small resistance increase of 2.9%. The helixes based on oriented U-AZO/PI also revealed excellent thermal stability, indicating the outstanding shape-persistence of U-AZO/PI helixes at 100°C. The largest elongation of U-AZO/PI helix with 100°C thermo treatment was slightly higher (36.2%) than that of the normal helix (35.8%), while there was also no breakage occurred after 2000 consecutive cycles. This work potentially paths the future cross-disciplinary inquest between bionics and materials science to inspire and stimulate the design of new structures, which makes them competent in applications of wearable materials, soft robotics and biotechnological applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by National Natural Science Foundation of China [Grant Nos. 52003007], Natural Science Foundation of Hebei Province [Grant Nos. E2019409063], Funded by S&T Program of Hebei [NO:216Z1005 G], Natural Science Foundation of Hebei [No. E2019208098] and Langfang top-notch talent [Grant Nos. LFBJ202004].
