Abstract
In this research, nanocomposite films based on starch were developed with the addition of hydroxyapatite nanoparticles as a mineral filler. Hydroxyapatite was synthesized by a chemical method using calcium nitrate and diammonium hydrogen phosphate. Various concentrations of hydroxyapatite nanoparticles were mixed with starch, and the developed films were evaluated in terms of physical, mechanical, and morphological properties. The highest values of mechanical parameters (tensile strength and elongation at break) were determined for the starch/hydroxyapatite film at 15 wt.% hydroxyapatite nanoparticles concentration (3.03 MPa, 37.41%, respectively). As hydroxyapatite concentration was increased from 0 to 20 wt.%, the solubility in water of the films decreased, whereas the solubility in acid increased. The crystalline structure of hydroxyapatite decreased the transparency of film and increased transparency value. Thus, a biodegradable film could be obtained with the addition of hydroxyapatite as a reinforcement filler up to 15 wt.%. It could be developed as a sustainable alternative for packaging industry.
Introduction
In recent years, environmental pollution originating from using polymeric materials and problems arising from their recycling (digging in soil and burning) have drawn great attentions.1-5 Most of packaging films are formed from polymers like polyethylene. 6 Degradation of such materials needs many years. Using degradable polymers instead of their undegradable counterparts is a practical solution to solve this problem.7,8 Attempts in the filed have led to introduction of some degradable polymers like polycaprolactone9,10 and polylactic acid.11,12 The most important disadvantages of the latter polymers are their high synthesis cost which limits their extensive application. On the other hand, starch is a biodegradable polymer which can be used in packaging industries with profit gain. Nearly 50% of raw starch is used in nonfood applications and thus can be used in packaging industries. 13 Starch is known for its ability to form films in food packaging applications. 14
Low price and high natural degradation rate of starch have extended its application in production of biodegradable materials. However, starch-based thin films suffer from some significant problems which hinder their application. High water affinity of starch,15,16 high sensitivity of starch-based films to moisture, 17 and its low flexibility comparing with synthetic polymers are some disadvantages of starch which limit extensive application of this material. 18
Modified starches can also be used in film production. 19 Addition of nanofillers to starch is a strategy to overcome these drawbacks. 20 Nanoparticle size, interfacial area, and aspect ratio of fillers are parameters which determine extent of mechanical properties enhancement upon addition of nanofillers.21-23
Hydroxyapatite, Ca10(PO4)6(OH)2, is the most stable specie of calcium phosphate family regarding human body temperature and pH. 24 Addition of nano-sized hydroxyapatite to starch will produce starch/hydroxyapatite nanocomposite where its mechanical properties are improved in comparison with pure polymer. Hydroxyapatite is a mineral filler that its incorporation into starch results in development of a material which can be used for production of food, medical, and cosmetic grades thin films.
Hydroxyapatite is synthesized through various methods and its mechanical properties are highly dependent on synthesis method. In fact, different methods introduce particles with different sizes and chemical structure. 25 As far as we know, no research has been carried out on synthesis of hydroxyapatite by precipitation method and its addition into starch for producing nanocomposites. In the current study, hydroxyapatite was added to starch and the physical and mechanical properties of the resultant nanocomposite were evaluated.
Experimental
Materials
The following chemicals were utilized in the present research:
Corn Starch ([C12H22O11]n, humidity content: 12%, Glucozan, Ghazvin, Iran), Citric Acid (C6H8O7, Merck, Germany), Glycerol (C3H8O3, Merck, Germany), Calcium Nitrate (Ca(NO3)2.4H2O, Merck, Germany), Diammonium Hydrogen Phosphate ((NH4)2H(PO4), Merck, Germany), and Ammonia (NH3, Merck, Germany).
Preparation of hydroxyapatite
To prepare hydroxyapatite (Hap), calcium nitrate and diammonium hydrogen phosphate were used as the sources of calcium and phosphate ions, respectively. These salts were weighted accurately and dissolved in double deionized water. In all samples, Stoichiometric molar ratio of Ca/P was considered 1/67. Calcium nitrate solution was gradually added to diammonium hydrogen phosphate solution while the mixture was stirred by a magnetic stirrer at 4000 rpm and at room temperature for 15 h. By using ammonia, the solution was adjusted to pH 10–12. A white sediment was formed in the reaction and separated using a centrifuge (Sigma 2–16P). For the removal of ammonia, the sediment was washed five times with distilled water. Next, the product was dried at 90°C for 24 h and hydroxyapatite powder was obtained.
Preparation of starch/citric acid/hydroxyapatite film
At first, a solution of citric acid (CA) and water (0.8/100, w/v) was added to hydroxyapatite as it solvent. Then, 100 mL distilled water was added to 10 g starch (S). For producing Starch/Citric Acid/Hydroxyapatite mixtures, Hydroxyapatite was added to each solution at loadings 0, 3, 5, 15, and 20 wt.% and homogenized by using an ultrasound (Power-SONI SONI410, 100 W) to separate nanoparticles for 30 min. After that, the solution was heated for 15 min at 75°C while stirring. The obtained gel-like material was cooled at the room temperature. Next, 10 mL glycerol (G) was added to it as a plasticizer. Subsequently, the mixture was mixed gently to homogenization by using a shaker (IKA-Werke KS-501 Digital) at 140 rpm for 10 min and was cast onto a polystyrene mold and dried for 72 h at the room temperature. The obtained film was peeled off and used for further tests. The films were labeled as “FH-,” followed by digits “0” to “20,” which denoted the loading (wt.%) of Hydroxyapatite added. 0% was referred to reference film.
Methods
Fourier transform infrared spectroscopy (FTIR)
The FTIR analyses of the used raw material in synthesized hydroxyapatite were performed in Spectrum 65 Perkin Elmer. Also, functional groups and covalent bonds were evaluated by using it. After preparation of the samples, all spectra were recorded in the range of 450–4000 cm−1.
Morphology characterization
The morphologies of the synthesized hydroxyapatite and biocomposite films were observed using a scanning electron microscope, (SEM), (LEO435VP, Philips) at an accelerating voltage of 20 kV. Before scanning, the specimens were installed on an aluminum surface and coated with gold.
X-Ray diffraction (XRD)
D8ADVANCE XRD instrument (Bruker Corporation, Germany) was used to obtain XRD patterns of all films. It was carried out in a diffractometer using Cu Kα radiation (λ = 1.54 Å) in the 2θ range of 10–50. The instrument was set at 40 Kv and 30 mA. The test speed was 1°/min and the test steps were 0.03°. XRD patterns investigated crystalline structure of the nanocomposite films.
The size of the hydroxyapatite particles was measured by using Scherrer equation (1)
Equation definitions.

Film thickness
The thickness of the film was measured using micrometer (Helios, H-103) with a precision of ± 0.01 mm. Measurements were done at five random positions on each film to obtain the mean thickness and were reported.
Mechanical properties
The mechanical properties of films were determined by using a tensile tester (Santam STM-5, Iran) in reference to ASTM- D88283. 26 The dumbbell-like specimens (100 mm * 25.4 mm) were conditioned at RH = 55% for 24 h. The initial grip separation and cross-head speed were 50 mm and 5 mm/min, respectively. Tensile strength (TS), elongation at break (EB), and toughness were measured in three replicates for each specimen.
Light transmission and transparency
A UV-Vis spectrophotometer (Meager UV-9200) was used to measure the light transmittance and absorbance of films at the UV wavelength 600 nm, as suggested by Yan et al. method.
27
The samples were cut into rectangles with an approximate area of 1 cm * 4 cm and their thickness was measured in five different points. After that, the sample was placed in the quartz cell of the instrument. Each obtained absorbance was used to calculate the transparency value of the samples using the Equation transparency 2
28
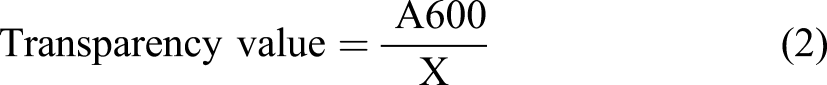
Symbols of the equation have been determined in Table 1. Greater transparency values represent lower film transparency.
Solubility in water
For each film, two circles (2 cm diameter) were prepared and placed for 2 weeks in desiccator to dry. Dry samples were weighed and subsequently transferred to test tubes filled with 50 mL distilled water. After 24 h immersion in water, samples were dried for 24 h at 105°C and weighed. Solubility was calculated using equation (3)
29

Symbols of the equation have been determined in Table 1.
Solubility in acid
For each film, three circles (2 cm diameter) were cut and placed for 2 weeks in desiccator to dry. Dry samples were weighed and subsequently transferred to test tubes filled with 50 mL hydrochloric acid (1 N). After 24 h immersion in hydrochloric acid, samples were dried for 24 h at 105°C and weighed. Solubility was calculated using equation (3). 30
Results and discussion
Fourier Transform Infrared Spectroscopy
In the Figure 1, the FTIR spectra of calcium nitrate, diammonium hydrogen phosphate, and hydroxyapatite are represented. Fourier transform infrared spectroscopy spectra of Calcium nitrate (a), Diammonium hydrogen phosphate (b), and Hydroxyapatite (c).
In case of hydroxyapatite, the absorption band at 568 and 606 cm−1 can be attributed to phosphate groups stretching vibrations. Also, the absorption band at 1422 cm−1 can indicate carbonate groups. Wide peaks at 1639 and 3570 cm−1 are related to the water which exists in the hydroxyapatite structure. 31 The FTIR spectra confirm that hydroxyapatite can be formed by the precipitation method.
Morphology characterization of hydroxyapatite and films
Figure 2 shows micrographs of hydroxyapatite with a cauliflower structure. This structure is formed when hydroxyapatite is synthesized by chemical precipitation method. Hydroxyapatite micrographs at 1000x (left) and 5000x (right) magnification.
Figure 3 shows micrographs of the films FH3, FH5, FH15, and FH20. In the graphs, cauliflower structure of hydroxyapatite is visible. Moreover, uniform surface indicates compatibility of starch and hydroxyapatite. Figures 3(a) and (b) show that aggregation of the particles is not observed due to low concentration of nanoparticles. As the hydroxyapatite concentration in the FH films increases, the aggregation of hydroxyapatite particles increases in the matrix and their clusters are shown in Figures 3(c) and (d). They present SEM images of the surface of the nanocomposite films at two different regions. They show the surface with using highly concentrated hydroxyapatite. The formation of cauliflower-like hydroxyapatite nanoparticles has been indicated on the surface of starch matrix that dispersed uniformly in the matrix. Scanning electron microscope micrographs of the nanocomposite films: FH3 (A), FH5 (B), FH15 (C), and FH20 (D).
X-Ray diffraction (XRD)
Figure 4 shows the XRD pattern of hydroxyapatite which is very similar to the obtained sample by Hanawalt et al.
32
The average particle size of the hydroxyapatite can be calculated as 14.01 nm by using Scherrer equation (1). X-Ray diffraction pattern of hydroxyapatite.
The XRD patterns of the films were recorded in the 2θ = 10°–50°. In Figure 5, hydroxyapatite peaks are located in the 2θ = 25°–40°. As can be seen, low and strong intensity peaks are appeared when hydroxyapatite concentration is low and high, respectively. Figure 5 A shows pure starch without hydroxyapatite. Figure 5 B contains only 5% hydroxyapatite and its crystallinity has little effect on the nanocomposite film. In the XRD pattern of nanocomposite C, small peaks are observed, which indicate the low crystallinity of hydroxyapatite in the prepared nanocomposite and its presence in the sample. Sample D shows stronger peaks with increasing hydroxyapatite concentration (20%). The X-Ray diffraction patterns of nanocomposite films: FH0 (a), FH5 (b), FH15 (c), and FH20 (d).
The comparison of Figures 4 and 5 shows that the crystallinity of hydroxyapatite is reduced in the nanocomposite and the crystallinity of the nanocomposite increases with increasing hydroxyapatite concentration that may be due to interaction of starch OH− groups with Ca2+ ions in the nanocomposite.33,34
Film thickness
Effect of hydroxyapatite content on the thickness and mechanical properties of films.

Mechanical properties
The mechanical properties of films were characterized by tensile measurements at room temperature. The dependence of tensile strength and elongation at break on hydroxyapatite content has been presented in Table 2.
Crosslinking agents and nanoparticles increase the mechanical properties. Citric acid is a crosslinking agent and hydroxyapatite is a nanoparticle. Therefore, both of them improve the mechanical properties.
Generally, the tensile strength increases and the elongation at break decreases with hydroxyapatite content in the starch/citric acid/hydroxyapatite films. The starch-based film exhibits the lowest tensile strength. The addition of hydroxyapatite nanoparticles into starch matrix results in increase of tensile strength because the load is transferred to the polymer matrix/nanoparticle interface.
As can be seen in Table 2, the systems containing higher hydroxyapatite content exhibit higher value of the tensile strength and lower value of the elongation at break. The tensile strength is 1.49 and 1.54 MPa for FH3 and FH5, respectively. Also, the elongation at break is 28.5% and 23.37% for them in which show this trend. On the other hand, the tensile strength of FH20 (2.50 MPa) is lower than FH15 (3.03 MPa) and the elongation at break FH15 (37.41%) is higher than FH20 (28.83%). Pramanik et al. investigated the starch–hydroxyapatite composite with different hydroxyapatite concentration from 0% to 90%. The thin film had shown the best tensile strength (3.03 MPa), elongation (21.5), as well as modulus (15.69 MPa), while the pure starch had shown the tensile strength (0.28), elongation (76.18), as well as modulus (5.024 MPa). Also, the mechanical properties have been reduced by increasing hydroxyapatite concentration due to the agglomeration formation during processing of composite above 40 w% hydroxyapatite in starch. 35 Menzel et al. investigated the molecular structure of citric acid cross-linked starch films and showed that curing the films at a high concentration of citric acid (30 pph) at 150°C extensively increases cross-linking reaction. 36 It has been reported that increasing the citric acid content up to 30 pph extensively promotes the cross-linking reaction and makes no toxicity.36,37 The crosslink formation by citric acid and hydrogen bonds between hydroxyapatite and starch will show effect on the mechanical properties of films. These lead to an increase in the tensile strength and the reduction of the elongation at break. That can suggest that the sample with hydroxyapatite 15 w% reduces crosslink bonds formation and subsequently an increase in the polymer chains’ mobility, which improved the film’s extensibility. Moreover, in FH15, hydrogen bonds improve the tensile strength. While, in FH20, hydroxyapatite content reduces hydrogen bonds formation and its effect is higher than crosslink bonds formation, which decreases the tensile strength and the elongation at break. Therefore, the optimum mechanical property has obtained for FH15. Mirab et al. investigated that the esterification was occurred by reacting the carboxyl group of citric acid and the hydroxyl group of the starch. They indicated that the completely compressed scaffold rapidly recovered its initial shape in 7 seconds after unloading. In fact, the crosslinking of starch had led to the high elasticity of the scaffold. 38 This phenomenon was referred to the shape-memory property of the scaffold. 39
Light transmission and transparency
In the packaging industries, transparency of the films is vital. The film can prolong the shelf life of fat-based food products by absorbing UV light and thus suppress oxidative degradation and highly transparent films can reduce antimicrobial activity.40,41 A high degree of transparency is usually accepted because the food can be seen in its packaging. 42 However, different degrees of film transparency may be also useful, depending on the specific application.
Effect of hydroxyapatite content on transparency value, solubility in water, and solubility in acid of the films.
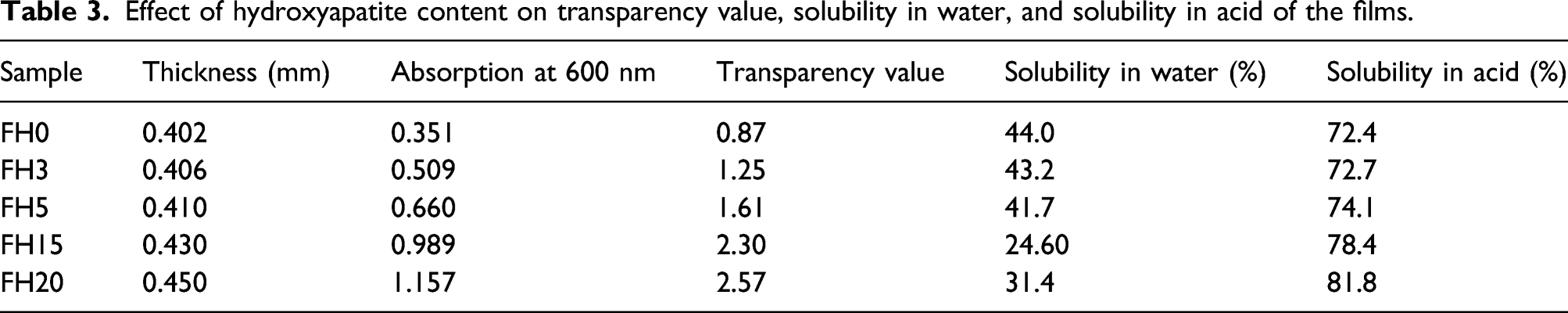
Domene-López et al. found the effect of thermoplastic starch films made from potato, corn, wheat, and rice starch on the opacity. The transparency values were 0.61, 1.56, 0.93, and 0.9 for potato, corn, wheat, and rice starch, respectively. 43 Also, Basiak et al. 44 and Dai et al. 45 obtained the same conclusion. In any case, a certain degree of opacity may be suitable for applications in which protection against incident light is important; for example, for wrapping products which can be degraded by light-catalyzed reactions. 46 Nevertheless, at macroscopic scale, these differences in transparency (0.87–2.57) are almost negligible. Moreover, the comparison of the light transmittance percentage with other studies, the barrier against the UV radiation of these FH films was reportedly higher than that of the synthetic polymer films. For example, low-density polyethylene (LDPE) and oriented polypropylene (OPP) range from 4.6% to 67.5%, in addition to the rest of the tested wavelengths.
Solubility in water
The solubility in water of the films as a function of hydroxyapatite content is shown in Table 3. Generally, the solubility decreases with increasing hydroxyapatite content. The solubility in water depends on the presence of hydrophilic groups, free volume, as well as crosslinking density. Because hydroxyapatite is a hydrophobic material, the higher hydroxyapatite content exhibits the lower solubility that is revealed in Table 3. Also, the solubility in FH20 is higher than the solubility in FH15 while hydroxyapatite content in FH20 is higher than FH15. The higher concentration of hydroxyapatite is shown the lower crosslinking density with citric acid that can result in the addition of the solubility in water. The results confirm the obtained results of mechanical properties. Ai et al. investigated the swelling percentage of chitosan-starch composites (0.135–0.02%) filled hydroxyapatite. They indicated that the swelling percentage was decreased gradually with increasing filler percentage (0–40%). 47 Mehyar and Han found film solubility in water of 44.4% and 32.0% for rice starch and pea starch films, respectively. The glycerol concentration used in those experiments was 50% of starch weight. 48 Also, Domene-López et al. found the effect of the amylose content on the hydration properties of the films. The water solubility values of the films were around 28–32% for all the samples 43 that are similar or even lower than the values found in the literature.49,50 The solubility in water was greatly influenced by the molecular weight of the starch.
Solubility in acid
Table 3 indicates the solubility in hydrochloric acid of the films. As can be seen in Table 3, the systems containing higher hydroxyapatite content exhibit higher value of the solubility in acid because its solvent is the acid. So, the higher hydroxyapatite content shows the higher solubility in acid.
Conclusions
Biodegradable films based on starch were obtained by using hydroxyapatite nanoparticles and citric acid. The effect of hydroxyapatite content on physicochemical properties of prepared films were evaluated. The XRD and morphological studies showed that the films were formed uniformly. The mechanical tests revealed that the highest tensile strength and elongation at break were determined for FH15 film (3.03 MPa and 37.41%, respectively). The transparency value, the solubility in water, and the solubility in acid importantly depended on hydroxyapatite nanoparticles content. The value of the transparency and the solubility in acid increased generally with hydroxyapatite nanoparticles amount in the system. They were noted for FH15 (2.30 and 78.4%, respectively) and FH20 (2.57 and 81.8%, respectively). Moreover, the value of the solubility in water decreased with hydroxyapatite nanoparticles amount, which obtained 24.6% for FH15.
It could be concluded that the biodegradable starch-based films containing 15 wt.% hydroxyapatite exhibited the most promising physicochemical properties, which suggested protection of the product against UV radiation, if used in packaging industry. Also, biodegradable films could decrease environmental pollution.
Footnotes
Acknowledgment
The authors gratefully acknowledge the Islamic Azad University, Shahreza Branch for financially supporting the present research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

