Abstract
An active and intelligent starch-based biodegradable food packaging system is developed in this work. The growing environmental protection concerns have motivated researchers to look for biodegradable yet sustainable products for food packaging. The packaging is able to resist against undesirable bacterial attack and color-change response due to pH change in food. Polyvinyl alcohol and starch are cross-linked with Glutaraldehyde to improve their mechanical strength. Propolis Extract (PE) and anthocyanin (ATH) were added as an active and intelligent material, respectively. Different film formulations were prepared by varying concentration of PE. All film formulations were characterized by SEM, FTIR and XRD. The results proved good compatibility of the film mixture. The mechanical strength and other physical properties like water vapor transmission rate, moisture retention capability, swelling degree and biological leaching ability were investigated. Films containing 20% PE showed the best results alongside maximum mechanical strength of 15.9 MPa. Furthermore, the same formulation has shown maximum zone of inhibition of 25 mm and 20 mm against E. Coli and MRSA, respectively. Moreover, there was a clear color-change response when films were immersed in different pH solutions ranging from 1 to 14. Finally, to validate the potential application of prepared film formulations, films were tested on pasteurized milk and a color-change response is observed along with the anti-bacterial activity. Hence, this active and intelligent food packaging system is capable of inhibiting and alerting food spoilage.
Introduction
The emerging concept of active and intelligent biodegradable food packaging has taken over the market demands for healthy and safe food with a prolonged shelf life. 1 Active packaging refers to the antimicrobial packaging with the integrated active agent which hinders bacterial growth, protect food and extend shelf life by preserving food. 2 Intelligent packaging refers to on-time monitoring and an indication that any change happened to the pH of food during transportation and handling. 3
Active packaging system aim to prevent food from external microbial attack and retard the growth of bacteria and pathogens. Antimicrobial activity in package material can be acquired by four different modes; (a) incorporating synthetic antimicrobial agent into base polymers (b) surface coating of films with active agent (c) atmosphere modification of package by emitting gases (d) incorporation of natural active agents into the film matrix.4-6 Incorporation of natural active agent mode was preferred for active packaging. The addition of natural active agents like propolis extract improves the physical and mechanical properties of films.2,7 Active agents are divided into two categories (i) conventional chemically synthesized active agents and (ii) naturally extracted active agents derived from plants, animals and micro-organisms. Chemically synthesized active agents include types of organic acids and salts such as potassium sorbate, propionic acid, acetic acid, ascorbic acid, benzoic acid and alcohols. These agents are readily available in the market due to their low prices and affordability. However, naturally extracted active agents have attracted the consumer’s interest due to their low toxicity compared to synthetic agents. 8 Naturally produce active agents are plants extracts (e.g. tea tree oil, rosemary oil) and peptides. 9 Other than natural sources nanoparticles (silver, zinc, titanium and gold) are also incorporated into packaging films for antimicrobial activity. 10 The use of essential oil in food packaging of milk and meat is now limited due to undesirable secretion of an organoleptic agent, which changes color and texture of food. 11 Recently propolis (a by-product of beeswax) has been used for anti-bacterial and anti-inflammatory purposes. 12
Propolis is the sticky resinous compound collected from beeswax, and is composed of 50% resinous flavonoids containing poly-phenols, 30% wax, 10% essential oil and remaining constitutes various organic components and pollen. 13 There are three different chemical compositions of extracted propolis; (i) Ethanol extracts of propolis (EEP) (ii) water extracted propolis (WEP) and (iii) volatile oil from propolis (VOP). EEP is mostly used in food packaging due to its anti-bacterial, anti-viral and antioxidant properties. The chemical composition of flavonoids present in PE mainly includes pinocembrin, quercetin, galangin, chrysin, kaempferol and naringenin. 14 PE can be incorporated into food packaging films as active agent due to its anti-bacterial properties.
Intelligent food packaging can provide all information regarding food storage, transportation and freshness of food. 3 There are sensors, bar codes, time-temperature indicator, gas indicator and pH indicator. pH indicators are particularly applied for intelligent food packaging due to their availability and cost-effectiveness. 15 There are a large number of synthetic pH indicators that have been used for investigating freshness of food. For example, bromocresol green, bromothymol blue and polyaniline are used as pH indicator for checking the quality of food. However, these synthetic pigments have hazardous effect on human health and therefore, not suitable for food packaging. 16 Anthocyanin is a naturally occurring pigment which can be used as pH indicator due to its quick color-change response and non-toxicity. 15 There are many plant sources of anthocyanin extracts such as purple sweet potato, red cabbage leaves and Roselle herb and Roselle anthocyanin.15,17
In this study, the ATH was extracted from red cabbage leaves and used as intelligent material in film preparation. Many base polymers are available to incorporate PE and ATH into the films matrix; such as Chitosan, poly lactic acid (PLA), poly vinyl alcohol (PVA) and starch. 18 PVA is used due to its biodegradable and easily compostable properties as compared with PLA. 19 Starch is the oldest base polymer used for packaging due to its bio-compatibility, bio-degradability and safe to the environment, but lacks in mechanical strength and water-resistant properties. However, these properties can be improved by blending it with other polymers like PVA. 20
PVA is a non-toxic polymer and has good mechanical properties. Starch and PVA blends have the ability to overcome the market demand for economical packaging material. 21 PVA-Starch films are an appropriate substitute for conventional plastic packaging material owing to the excellent film-forming ability, gas and optic barrier, hydrophobicity, bio-degradability, high tensile strength and flexibility of PVA material. 22 However, to broaden the applications of PVA in the food packaging industry, its physical properties can be tailored by adding different cross-linkers. In literature, there are many cross-linkers that have been studied so far such as, malic acid, glyoxal, boric acid, Glutaraldehyde, hexamethylene diisocyanate and citric acid. 23 In this study, Glutaraldehyde was used as cross-linker due to its ability to react in intermolecular spaces and increase the cross-link density. 24 As a result, more hydrophobic and stable films were produced.
Food spoilage is the process in which food is decayed due to attack of external microbes and internally food borne pathogens. They can damage the taste, texture and nutrition of food. Therefore, several techniques are being used for quality control of food. 18 Food spoilage slightly lowers the pH of food, which can be easily measured by pH indicators like anthocyanin. 25 Similarly, the bacterial attack can be inhibited by incorporating active material into packaging films. Propolis extract is naturally extracted active agent which can inhibit bacterial growth without harming the environment. Therefore, anthocyanin and propolis being economical and organic sources are used in these packaging films, which makes them different from conventional synthetic active and intelligent films.
The purpose of this study was to develop and characterize active and intelligent films. Scanning electron microscopy (SEM) was performed to analyze the surface morphology of films. The intermolecular interaction and functional group attachment were investigated by X-ray diffraction (XRD) and Fourier Transform Infrared Spectroscopy (FTIR). Mechanical strength and physical properties of the films were also determined. Such as; tensile strength, % elongation, WVTR, moisture retention capability and swelling degree etc. Furthermore, the colorimetric test and anti-bacterial activity were evaluated to analyze their active and intelligent property. Finally, food spoilage test was performed to evaluate their applicability potential. The results suggest that these active and intelligent films are suitable for food packaging purpose.
Materials
Polyvinyl alcohol (PVA), starch, glutaraldehyde and hydrochloric acid were purchased from Sigma Aldrich USA. The red cabbage was bought from farmers market for anthocyanin extraction. Propolis powder was purchased from Amazon. Nestle Milk Pack was used for food spoilage test. Distilled water was used in all experimental work.
Experimental design and techniques
Propolis extraction
PE was extracted using “ethanol treatment method” as explained by with slight modification. 13 A 50 mL of ethanol/water (7:3) solution was prepared, and 5 g of propolis powder was added to it. Afterward, this solution was further concentrated to half its volume using rotary evaporator at 70°C for 2 hours. The deep orange solution was obtained at the end and stored into the fridge.
Anthocyanin extraction
ATH was extracted from red cabbage leaves using pure ethanol as solvent. 150 g of red cabbage leaves were crushed and mixed into 100 mL of pure ethanol solvent for pigment extraction. Afterward, the mixture was filtered and the filtrate was centrifuged at 400 rpm for 10 minutes for further purification. Later, HCl and NaOH was added to the centrifuged mixture to maintain pH at 2 and was stored for 24 h at 5°C. After 24 h the pH of the solution was maintained at 7 by adding NaOH and stored at 5°C for further use. 26
Films formulation
1 g of PVA in 10 mL of distilled water, and 1 g of starch /10 mL of distilled water solutions were prepared separately. Afterward, PVA/Starch (3:5) solution was mixed and cross-linker GA (0.5 mL) with HCl (0.05 mL) as catalyst was added. Furthermore, to make films active and intelligent different concentration of PE (0.5%, 2%, 5%, 10% and 20%) and 5 mL of ATH were added to form different formulations. Afterward, these film solutions were prepared using solution casting method and vacuum dried at 40°C. Eight possible combinations of film formulations are listed in Table 1.
Chemical composition of active and intelligent films formulation.
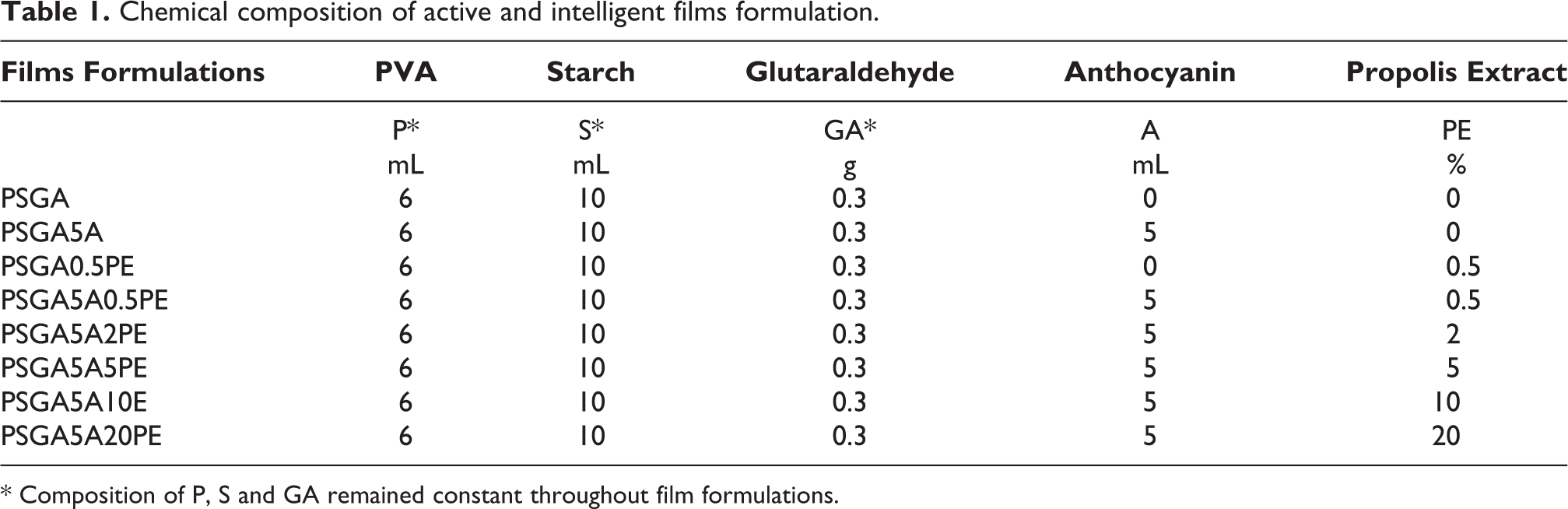
* Composition of P, S and GA remained constant throughout film formulations.
SEM
Scanning electron microscopy (SEM) analysis was performed to reveal the surface morphology of film formulations using JEOL SEM (JSM-64900). All film samples were coated using conductive gold sputter at 10 kV.
FTIR analysis
To study the inter-structural interactions and functional group attachment of formulated films Fourier transform infrared (FTIR) analysis was performed. FTIR Perkin Elmer was used at a resolution of 4 cm−1 and wavenumber was ranged between 4000 cm−1 to 700 cm−1.
XRD analysis
X-ray diffraction (XRD) analysis was performed by using STOE Germany. The 2θ was ranged from 10° to 70°. The step size and scan ratio was 0.5 step−1 and 0.01 sec−1, respectively. The wavelength of Copper-K-α radiation was 1.54 Å.
Mechanical properties
Mechanical properties of active and intelligent films are associated with inter and intra molecular interaction between used material.
Universal testing machine (UTM) was used to calculate tensile strength and % elongation. Cross-head speed and grip separation were 50 mm/min and 25 mm respectively. Samples were cut into rectangular shape with dimension of 40 × 50 mm2.
Tensile strength (TS) was calculated using following formula;

F (N) is applied force and Ai (mm2) is initial area of film sample.
% Elongation (%E) is calculated as;

li (mm) is the initial length of sample film, l (mm) is the final length of sample film.
Water vapor transmission rate (WVTR)
WVTR was calculated by using the standard method followed by the American standard of testing materials. 27 10 mL of distilled water was taken in a media bottle having a diameter of 29.5 mm. The bottle was sealed with film using Teflon tape and the initial weight of the bottle was measured as Wi (g). The bottle was then kept in the oven for 24 h at 40°C and again the weight of the bottle was measured as Wf (g). WVTR was calculated by the formula given below;

A (mm2) is the area of bottle.
Moisture retention capability test
Moisture retention capability test was performed to check the water vapor and moisture resistant properties of films formulations. For this purpose, films were cut into square (1 × 1 cm2) and weighed (Wi, g). After that film samples were placed into the oven for 6 h at 60°C and weighed again (Wf, g). Moisture retention capability was calculated by the formula given below;

Biological leaching test (BLT)
BLT was conducted using UV-visible spectrophotometer GENESYS™ at wavelength 208 nm to measure the amount of additives i.e. ATH and PE that can be leached out of films. Heat methanol treatment method was followed with modifications. 28 Films were cut into square (1 × 1 cm2) and dissolved in 10 mL methanol and continuously stirred for 2 h at 40°C. Afterward these leached out film samples were washed with deionized water and filtrate is then tested for UV spectroscopy.
Normalized swelling degree (NSD)
NSD test was performed to check the performance of cross-linker and polymeric network formed by all components. This test was conducted by following the method used in literature. 29 Films were cut into square (20 × 20 mm2) and then immersed into dimethyl sulfoxide (DMSO) solution for 2 h. After that, this solution is filtered and the insoluble part leftover filter paper is weighed as W1(g). Then this filtered mass is dried in the oven for 6 h at 80°C and weighed again as W2 (g). NSD was calculated by this formula;

A (mm2) is the area of film samples.
Colorimetric test
In order to check the colorimetric response of films formulation pH test was performed using the method followed in the literature. 18 Films formulations were cut into 10 × 10 mm2 square samples. Eight different solutions were made having a pH range from 1 to 14. Afterward, film samples were dipped into different pH solutions and color-change response was captured in the form of images taken by digital camera.
Anti-bacterial test
“Disc diffusion” method was employed to check the anti-bacterial activity of film formulations against two common food born bacterial strains i.e. Escherichia Coli (E. coli) and Methicillin-resistant Staphylococcus Aureus (MRSA). 30 Five discs (6 mm diameter) were cut for each bacterial strain. PE concentration varied (0.5%, 2%, 5%, 10% and 20% PE) for each disc. Afterward, these discs were mounted on Lysogeny broth (LB) medium with positive and negative control. Anti-bacterial activity of each disc was determined by measuring the diameter of zones of inhibition after 24 h and 48 h of each disc. Anti-bacterial activity images were also captured after 48 h.
Food spoilage test
Food spoilage test was performed to check the potential applicability of these active and intelligent films. Pasteurized milk pack (Milk pack) was purchased from the local market. Milk was poured into 10 mL voiles and sealed with square-cut films. The freshness of the milk was checked after 24 h and 48 h. 31
Results and discussion
Surface morphology of films formulations
Figure 1 depicts the surface morphology of formulated films using SEM. All films have different surfaces due to varying films compositions. In Figure 1(a) PSGA has a rough surface with dispersed PVA molecules, which shows immiscibility and poor compatibility of film mixture.31,32 When ATH is added into the film mixture, a partially mixed surface of PSGA5A was observed in Figure 1(b). In Figure 1(c), homogeneous surface is depicted. Whereas, Figure 1(d) showed the smooth homogeneous surface. This could be attributed to the addition of a higher concentration of PE and ATH that facilitated the homogenous mixing of each component. 33 Furthermore, ATH and PE have reduced the intermolecular force between the polymer chains by increasing chain distance resulting in smooth structured flexible films. 32

Surface morphology of different films formulations (a) PSGA, (b) PSGA5A, (c) PSGA5A10PE and (d) PSGA5A20PE.
Analytical properties
FTIR spectra
FTIR analysis of all film formulations is shown in Figure 2(a). Almost identical patterns were observed in all spectra with varying transmittance value.

Analytical properties of films compositions (a) FTIR and (b) XRD.
In all spectra absorption peaks between 3650 cm−1 to 3550 cm−1 of OH group were observed, which was attributed to the presence of PVA. Similarly, absorption peaks at 1150 cm−1 to 1080 cm−1 confirms the presence of C-O group. 34 C-H bond stretching was observed at 2900 cm−1 35 in all spectra. A strong absorption band between 1650 cm−1 to 1550 cm−1 has shown the presence of aromatic ring of ATH and PE. 36 Collectively, it confirms the functional group attachment of PE and ATH to the PVA-Starch film matrix.
XRD analysis
Figure 2(b) depicts XRD analysis of films formulations. All formulations have a similar pattern with slight variations. In spectra PSGA, PSGA5A and PSGA5A0.5PE peaks at 18°, 22.5° and 25° were diffraction peaks of starch. Similarly, the diffraction peaks at 19°, 21° and 45° were attributed to the presence of PVA. 29 In film composition PSGA5A0.5PE, decrease in sharpness of peaks as a result of homogenous mixing of ATH, PE, PVA, Starch and GA was observed. The crystallinity of starch molecules disappeared with addition of PE, ATH and proper interaction of cross-linker GA. 29 Peak shift from 45° to 55° is observed in PSGA5A20PE pertaining to the strong molecular interaction and change in ionic radii. 37 Overall, a decline in crystallinity was observed due to the addition of GA, ATH and PE into PVA and Starch matrix.
Mechanical properties
Tensile strength is the maximum strength measure of a film that can bear external stresses. Whereas, % elongation is the value to which a film can be stretched. 38 Figure 3 depicts the mechanical properties of all film compositions. It was noticed that tensile strength was enhanced by increasing the concentration of PE. On the other hand, percent elongation was decreased by increasing PE concentration. A possible reason for this decrease in % elongation could be the crystalline behavior of the films due to increased PE concentration which made them less flexible. 39 The other possible reason for the decrease in % elongation could be the cross-linking effect produced by increasing PE concentration, which has reduced intermolecular spaces and increased tensile strength. 40 In the literature, increased tensile strength with decreased % elongation has been reported several times with the addition of additives like essential oils, green tea extracts and cinnamon oil.41,42 Overall, PSGA5A20PE film formulation has the maximum tensile strength with minimum %elongation of 15.9 MPa and 4.5%, respectively.
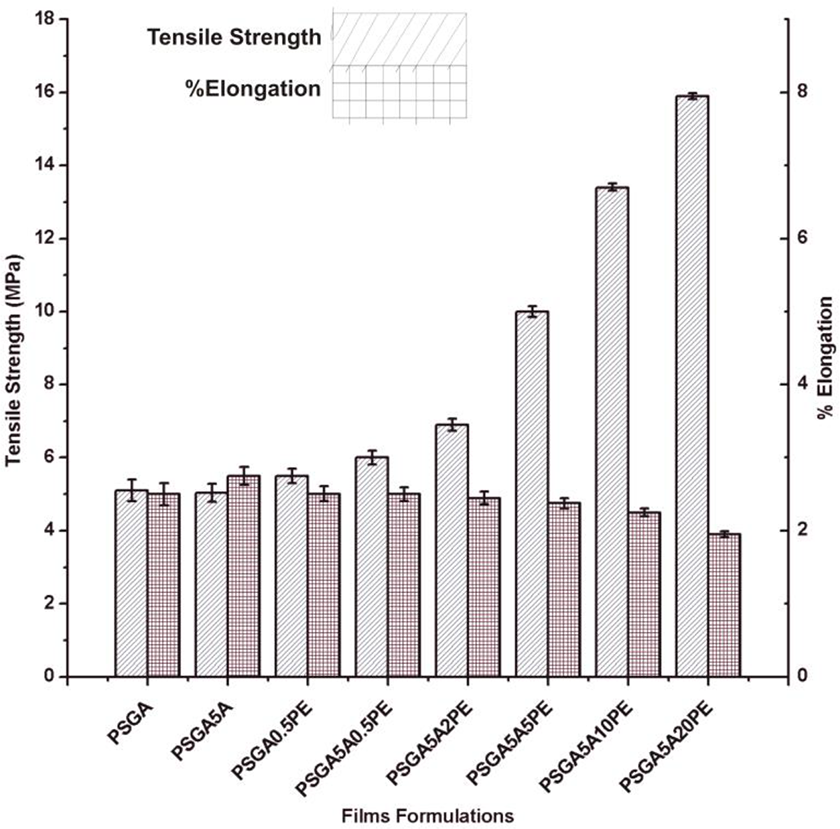
Mechanical properties of all film formulations i.e. tensile strength and % elongation.
Physical testing
Water vapor transmission rate (WVTR)
WVTR is an important aspect in food packaging application as it comprises of water diffusion and absorption. It depends upon the hydrophilic and hydrophobic properties of materials present in packaging films. 43 Base polymers, additives and cross-linkers play an important role for the hydrophilicity and hydrophobicity of films. 44 From results, it is depicted that film formulations without or with low PE concentrations have higher WVTR (Figure 4(a)). PSGA5A20E has the lowest WVTR of 0.0289 g/mm2 hr. The possible reason for this lower rate with higher PE concentration in films could be the presence of phenolic component in PE and ATH fitted into PVA/Starch matrix and enhanced intermolecular interaction and hydrogen bonding. Which consequently, hindered the escape of water vapor molecules from the films.42,45 The other reason for lower WVRT could be the presence of resinous components in PE which are hydrophobic in nature.46,47 It is important to measure WVTR specially in food industry in order to maintain the safety, quality and shelf life of the packed food. 48

Physical properties of films formulations, (a) WVTR, (b) Moisture retention capability, (c) BLT and (d) NSD.
Moisture retention capability test
Moisture retention capability test was performed to check the performance of active and intelligent films formulations. In Figure 4(b), it is shown that PSGA5A20PE has the highest capability to retain moisture. Whereas, PSGA has the lowest capability to retain moisture. Moisture retention is a very important feature for food packaging. The reason behind this retention is the presence of the resinous and waxy component in PE which as a result, produced hydrophobic surface of films formulation. There are many studies reported in the literature in which different organic additives i.e. essential oil, bio-active components and tea tree oil were added to increase hydrophobic features.44,49 Essential oils are soluble in ethers, alcohols and fixed oil but not soluble in water which results in hydrophobicity and helps in moisture retention capability. 50 Same results were obtained, when synthetic active agent potassium sorbate was added and moisture retention capability was enhanced. 51 So, the moisture retention capability of film formulations was enhanced by the addition of higher concentration of PE in formulated films.
Biological leaching test (BLT)
BLT was performed to measure the amount of leached out active and intelligent agents from films into the environment. In Figure 4(c), it is shown that PSGA5A20PE has the highest absorbance value of 4.9 whereas; PSGA has the lowest value of 4. The highest absorbance value of PSGA5A20PE was due to the high concentration of PE used. Also, the chances of leaching augmented, as the amount of PE was increased. As, PE is a natural active agent so it has no effect on the environment, when leached out in minimum amount of absorbance value below 5. 52
Normalized swelling degree (NSD)
NSD test was performed to check the cross-linked density and swelling degree as they have inverse relation. 29 In Figure 4(d), it was observed that PSGA5A20PE has the highest NSD value whereas; PSGA has the lowest value. The reason behind high NSD value could be the addition of ATH and PE. The incorporation of these additives i.e. PE and ATH, result in an increase of chain distance, bond breakage between polymer chains and decrease in intermolecular forces. Resultantly, cross-linked density is decreased and swelling degree increased.29,32
Active and intelligent properties
Colorimetric test
Color-change response was tested using different pH buffer solutions ranging from 1 to 14 pH. Figure 6(a) illustrates the varying color response of film formulations with changing pH and dependence on pH was ascertained. The film used for this test was embedded with ATH and PE (PSGA5A20PE).
At lower pH (acidic) film color changed into pink whereas; when pH increased it was converted into purple. At pH 7, the color of the film was light purple and when pH moved toward basicity the color film changed to blue-green. Finally, at pH 14 the color was changed into yellow.
The pink-red color of the film was due to the presence of flavylium cation, which was shown at lower pH value. The purple color of the film was generated due to carbinol pseudo base at pH 4–6. The blue-purple color of the film was due to the presence of quinoidal base at pH 7–8. 31 The presence of ATH into the films formulations made distinguishable color variations. Such characteristic of films makes them intelligent. In food packaging, food spoilage decreases the pH which as a result changes the color of films. 31
Anti-bacterial activity test
Anti-bacterial activity of films formulations were tested against two common food born bacterial strains i.e. E. coli and MRSA. In Figure 5(a), disc diffusion method was presented in which 6 mm discs of PSGA5A (with 0.5%, 2%, 5%, 10% and 20% PE) were mounted on LB media. Clear zones of inhibition were observed when the concentration of active agent i.e. PE was increased. The highest activity against E coli and MRSA was shown by PSGA5A20PE. In Figure 5(b), diameters of zones of inhibition are presented. The maximum inhibition zone was achieved by PSGA5A20PE i.e. 23 mm and 19 mm against E. coli and MRSA after 48 h respectively. So, it is clear that by increasing the amount of active agent i.e. PE enhanced anti-bacterial activity. The anti-bacterial activity of PE can be explained on the basis of its chemical composition. It contains poly-phenols, flavonoids and aromatic rings. Such components are able to inhibit bacterial growth and kill bacterial cells. 53 As, propolis being organic component it can be used in food packaging due to its less harmful effects to the environment and human health. 54

Active properties; (a) Disc diffusion method for anti-bacterial activity and (b) zones of inhibition diameters (mm).
Potential applications of film formulations
Food spoilage test was performed to check the applicability of prepared active and intelligent films. In Figure 6(b), three different films i.e. PSGA, PSGA5A10PE and PSGA5A20PE were tested using pasteurized milk. Milk has pH 7 at normal conditions but stale milk has pH 2. Therefore, after 48 h at 10°C results were recorded and they suggested that films sample PSGA5A20PE has the maximum anti-bacterial activity. From Figure 6(b), it is clear that blank sample with 0% PE has no anti-bacterial activity. Furthermore, PSGA5A10PE has the minimum anti-bacterial activity. The color of film changed from orange to pink due to decrease in pH of stale milk. PSGA5A20PE has the highest anti-bacterial activity with no spilt milk and color change. Stale milk has slightly lower pH due to the formation of lactic acid produced due to bacterial growth. PE being anti-bacterial agent inhibits bacterial growth and protects milk from bacterial attack. 31

Applicability tests: (a) pH test for color change and (b) food spoilage test.
Active and intelligent films have a broad spectrum of applications in food packaging industries like dairy, fruits and vegetable, meat and poultry and sea food industry. Their unique character of being anti-bacterial and color changing property with different pH make them more reliable option for food packaging.
Conclusion
This study was aimed to prepare biodegradable active and intelligent films, which are further evaluated and characterized by various tests. Firstly, surface morphology proved that increasing concentration of PE increased the homogeneity and smoothness of the surface. FTIR and XRD analysis further show that films mixture at higher concentration of PE had good molecular interaction and functional groups were properly attached. Similarly, by adding cross-linker the mechanical properties of films had been tailored in a positive way. Physical tests like WVTR, moisture retention capability, BLT and NSD had proved that molecules had hydrogen bonding and strong intermolecular interaction at higher concentration of PE. These film formulations had a quick and clear color-change response to pH. Anti-bacterial activity test had exposed a high anti-bacterial activity against both E coli and MRSA bacterial strains with an increase in PE concentration. Finally, food spoilage test had also shown bacterial attack retaliation at higher PE concentration and color-change response as well. Overall, PSGA5A20PE film formulation has depicted better results and is appropriate for biodegradable food packaging applications.
Highlights
➤ Development of bio-degradable active and intelligent films.
➤ Propolis extract used as active agent in films formulation.
➤ Anthocyanin incorporated as intelligent material into film formulation.
➤ Food spoilage test for their applicability in packaging industry.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National University of Sciences and Technology (NUST), Islamabad, Pakistan.
