Abstract
BACKGROUND
Lipid alterations have been described in sickle cell anemia (SCA), but their association with the disease severity is not fully understood and their correlation with the nutritional status of this population has not been widely studied.
OBJECTIVES
This study aimed to assess the lipid profile and its relation with the nutritional status, including anthropometric characteristics and daily macronutrient intake, and with the severity biomarkers in adults with SCA in a steady state.
DESIGN
Cross-sectional study
METHODS
This was an analytical cross-sectional study with 55 adults with SCA and 60 members of the control group. Sociodemographic, anthropometric, nutritional, and laboratory data were collected between March 2019 and June 2020. Mann–Whitney test was used to compare groups and Spearman's correlation coefficient was used to test the relation between variables.
RESULTS
The SCA group presented higher calorie, protein, and carbohydrate intake, lower levels of high-density lipoprotein-cholesterol (HDL-c), and a higher triglyceride (TG)/HDL-c ratio than the control. HDL-c showed a positive correlation with hemoglobin and hematocrit (HcT) and a negative correlation with indirect bilirubin and lactate dehydrogenase (LDH). The TG/HDL-c ratio was positively linked with reticulocyte count, LDH, and leukocyte count and negatively linked with Hb.
CONCLUSIONS
The study findings showed hypocholesterolemia in SCA and its independent correlation with macronutrient intake. The links of HDL-c and TG/HDL-c ratio with hemolysis indices indicate that they are easily accessible, low-cost markers capable of predicting SCA severity.
Keywords
Introduction
Sickle cell anemia (SCA) is the most prevalent hereditary hemoglobinopathy in the world 1 and it has an autosomal recessive character, resulting from a genetic variation that leads to the development of an altered hemoglobin (Hb), called HbS.2,3 Its pathophysiology is based on the vaso-occlusion process, possibly related to the hemolytic profile and endothelial dysfunction. Sickle cells have the ability to adhere to the vascular endothelium in a simpler form, resulting in obstruction and chronic inflammation with dysfunction of the affected glands and organs.4,5
Among the endocrine-metabolic alterations in SCA, those that are related to cholesterol and triglyceride (TG) levels really stand out. Although such variations have already been studied, the pathogenesis of hypocholesterolemia is not fully understood. It is suggested that its etiology is multifactorial, with reduced absorption and synthesis of lipids, in addition to the greater demand for cholesterol in the body due to the shorter half-life of red blood cells and abnormalities in the liver.6,7
Studies showed an inverse correlation between the levels of high-density lipoprotein-cholesterol (HDL-c), hemolysis markers, and vaso-occlusive phenomena in the population with SCA, with HDL-c being suggested as a marker of severity in those individuals. 8 The TG/HDL-c ratio, proposed as a marker for coronary artery disease, was higher in individuals with SCA than in the control population. 9 Despite this finding, TG levels and the TG/HDL-c ratio in individuals with SCA still show conflicting and scarce results in the scientific literature.10,11
As the lipidic profile can be influenced by the macronutrient intake and the nutritional status impacts the clinical condition of individuals with SCA, who already have issues that reduce food intake, such as constant hospitalizations, infections, pain crises, malabsorption, and increased catabolism of specific nutrients, 7 it is important to assess the correlation between the macronutrient intake and alterations in cholesterol and TGs. Therefore, this study aimed to assess the lipidic profile and its relation with the nutritional status, including anthropometric characteristics and daily macronutrient intake, and with the severity biomarkers in adults with SCA in a steady state.
Materials and Methods
Study Design
This was a cross-sectional controlled study that involved adults with SCA followed up at the outpatient Hematology Service of the University Hospital of the Federal Sergipe's Hospital, an academic center of regional reference in Aracaju, capital of Sergipe, University Center of Regional Renown in Aracaju, capital of Sergipe, in northeast Brazil.
Sample
This study sample was selected by nonprobabilistic sampling, for convenience, during routine outpatient visits and consisted of patients with SCA aged 19 years or older, all of them homozygous for the sickle mutation confirmed by Hb electrophoresis, without pain or infection in the 4 weeks preceding the blood sampling. Exclusion criteria were pregnancy or breastfeeding, blood transfusion or infection in the month prior to collection, and current use of statins or fibrates. Healthy individuals, academic personnel, resident physicians, and hospital staff with equal or similar age and gender of the sample group made up the control population. The control group underwent Hb electrophoresis to confirm the absence of any hemoglobinopathy or sickle cell trait.
The exclusion criteria applied to the group of patients were also applied to the other groups. Data collection took place between March 2019 and June 2020, with some breaks due to the severe acute respiratory syndrome-coronavirus-2 pandemic.
Data Collection
The informed participants were assessed through a structured collection instrument through the following data gathering process: name, age, gender, associated chronic diseases, such as arterial hypertension or any other chronic pathology, and complications related to the SCA, such as brain stroke, lower limb ulcer, kidney disease, pulmonary hypertension, osteonecrosis, retinopathy, and date of last hospitalization, and through blood collection for the measurement of biomarkers and lipidic profile.
Assessment of Macronutrient Intake
The dietary assessment was performed through the application of a 24-hr reminder (R24h) by a trained nutritionist. The R24h is considered the reference standard and consists of a form enquiring about what was ingested and its quantity in the last 24 hr prior to the interview. The information was collected on the current diet, such as data on all foods, beverages, quantity in homemade measure, way of preparation, time of ingestion, and brand of ingested food. The accuracy and precision of the intake reported by the participants were assessed and, in order to order to avoid information bias, a photographic manual with household measurements, which contains photos of portions, food shapes, and household measurements, was used during the recordatory.
The estimation of energy and macronutrient intake was performed using the Nutwin software version 1.6, the food used came from the Brazilian Food Composition Table and Food Table from the Brazilian Institute of Geography and Statistics, but in the absence of food preparation in the program, the nutritional information of the products described in the preparation recipe was used so that it was possible to measure the food's nutritional content.
Anthropometric Assessment
Participants were assessed for anthropometric data: weight and height. The data was measured using a digital scale (Tanita BF-679) and a wall-mounted stadiometer (Tonelli), according to a standard technique. Then, we calculated their body mass indices (BMIs) using the BMI = weight/height2 formula, expressed in kg/m2.
Laboratory Assessment
To perform the biochemical tests, the research participants were instructed to maintain a regular diet in the days before the testing, fasting for 12 hr, and without strong physical activity in the 24 hr prior to the collection.
Collection of 15 mL of peripheral blood was performed for serum measurements of hematimetric and hemolytic indices such as Hb, hematocrit (HcT), reticulocytes, leukocytes, platelets, lactate dehydrogenase (LDH), and indirect bilirubin (IB) and lipid prolife, such as total cholesterol (TC), HDL cholesterol (HDL-c: high-density lipoprotein), TGs, and low-density lipoprotein-cholesterol (LDL-c: low-density lipoprotein). LDL-c was calculated using the Friedwald formula, according to the calculation LDL-c = CT – (HDL-c + TG/5), if TG < 400 mg/dL. Lipidic levels were measured according to the V Brazilian Guideline on Dyslipidemia and Prevention of Atherosclerosis. 12 As for TG levels over HDL-c levels, a value >3.8 was considered high risk. 13
We used standard automated methods for the clinical analysis of hematological and biochemical tests, with tests performed at the Clinical Analysis Laboratory of the Federal University of Sergipe's Hospital.
Statistical Analysis
The collected data were charted in an Excel spreadsheet and the statistical analysis was performed using the R program, version 4.0.4. Descriptive analysis was used to characterize the distribution of the studied groups. Categorical variables were described through their absolute and relative frequencies.
The central limit theory was used as a parameter for characterizing the sample. Student's t-test was used for comparison between groups. Continuous variables were described through their mean and standard deviation. To assess the correlation between continuous variables, Spearman's correlation was used. To verify the correlation between categorical variables, the chi-square test was used. In all analyses, a significance level of 5% (p < .05) was adopted.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 14
Results
Fifty-five of the 420 patients with SCA followed up at the outpatient Hematology service were assessed, with a mean age of 28.4 (±5.2) years, 29 of whom were women (52.7%). Of the 55 patients, 41 (74.5%) used hydroxyurea and 13 (25.5%) did not. The control group consisted of 60 previously healthy individuals, with a mean age of 28 (±5) years, equally distributed between the genders. Regarding the anthropometric indices, the group of patients with SCA showed a reduction in height, weight, and BMI (Table 1).
Clinical Characteristics of Adults With SCA and Healthy Control.

Abbreviations: SCA, sickle cell anemia; BMI, body mass index; N, number of participants; SD, standard deviation.
*p < .05.
When assessing food intake, a higher rate of calories, proteins, and carbohydrates was detected by the SCA group, with no difference in terms of lipids (Table 2).
Daily Macronutrient Intake of Adults With SCA and Healthy Control.
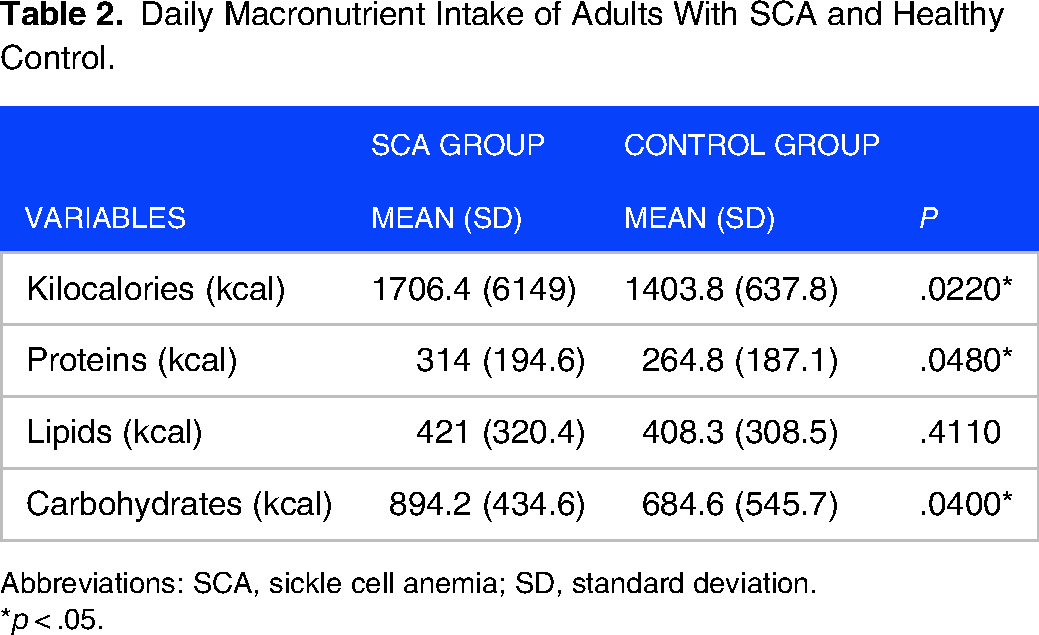
Abbreviations: SCA, sickle cell anemia; SD, standard deviation.
*p < .05.
Regarding the hematimetric indices, the SCA group had lower levels of Hb and HcT and higher levels of leukocytes, platelets, and ferritin. As for the hemolysis markers, individuals with SCA had higher levels of reticulocytes, IB, and LDH. As for the lipidic profile, the levels of TC, LDL-c, and HDL-c were lower in the SCA group, while the TG/HDL-c ratio was higher in this group. It was detected that 76.3% of individuals with SCA had HDL-c levels below the lower limit of normality (>40 mg/dL; Table 3).
Hematimetric Indices, Hemolysis Markers and Lipidic Profile of Adults With SCA and Healthy Control.

Abbreviations: SCA, sickle cell anemia; SD, standard deviation; LDL-c, low-density lipoprotein-cholesterol; HDL-c, high-density lipoprotein-cholesterol.
*p < .05.
No correlation was noted between the lipidic profile and anthropometric data and daily macronutrient intake markers within the SCA group. When verifying the correlation of the lipidic profile with the biomarkers of SCA severity, it was observed that HDL-c was moderately and positively correlated with the levels of Hb and HcT and negatively with IB, LDH, and leukocyte count (Figure 1). The TG/HDL-c ratio showed a weak negative correlation with Hb and a moderate positive correlation with leukocyte count, reticulocyte count, and LDH (Figure 2).
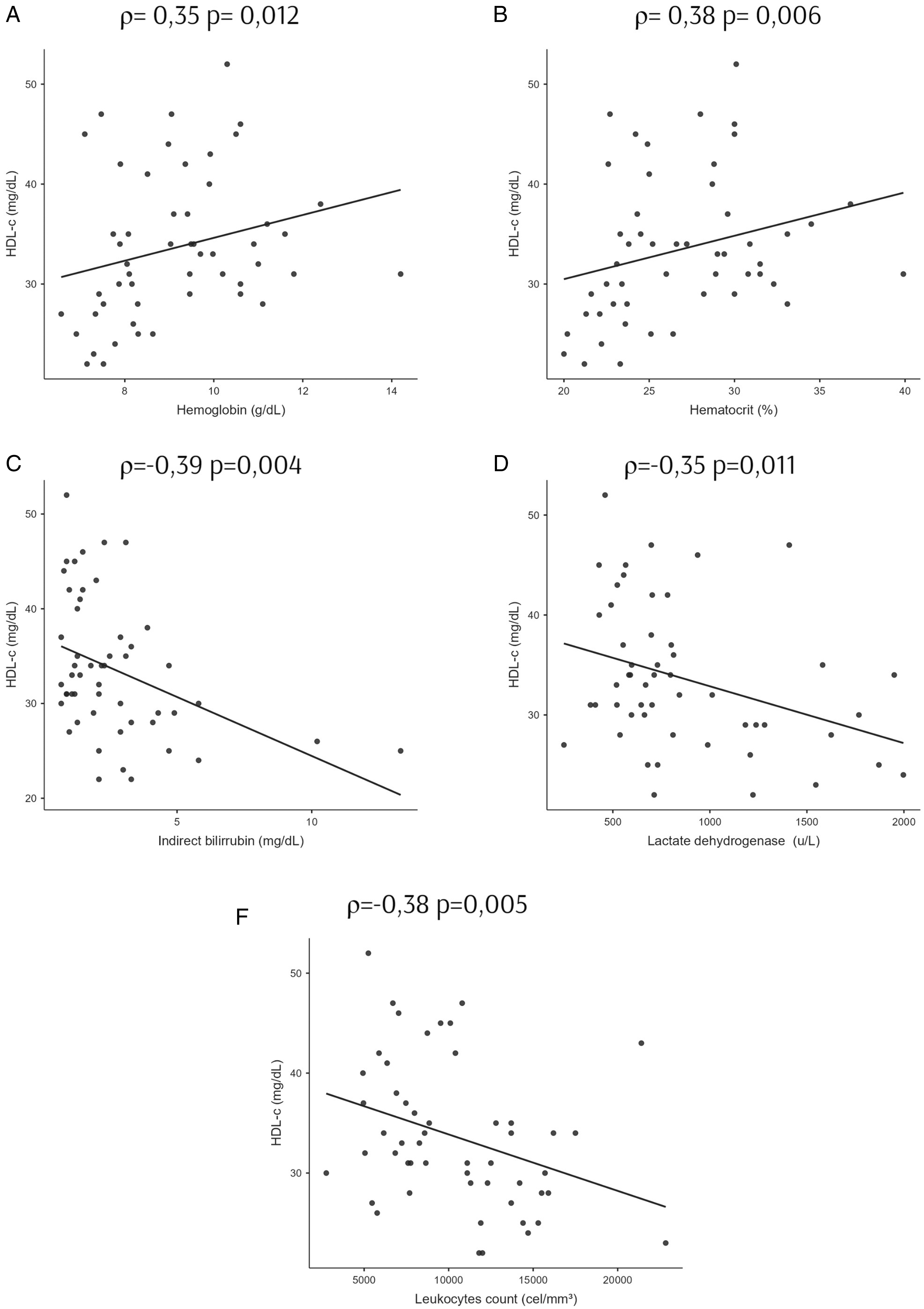
Correlation of high-density lipoprotein-cholesterol (HDL-c) and hematimetric and hemolytic indices of adults with sickle cell anemia. Spearman correlation analyses between HDL-c versus (A) hemoglobin, (B) hematocrit, (C) indirect bilirubin, (D) lactate dehydrogenase, and (E) leukocyte count. ρ is the Spearman correlation coefficient; p < .05.

Correlation of TG/HDL-c ratio and hematimetric and hemolytic indices of adults with SCA. Spearman correlation analyses between TG/HDL-c ratio versus (A) hemoglobin, (B) leukocyte count, (C) reticulocyte count, and (D) lactate dehydrogenase. ρ is the Spearman correlation coefficient; p < .05.
Discussion
This study aimed to assess the lipidic profile and its relation with laboratory markers of severity in adults with SCA.
The pathophysiology of SCA is characterized by a vaso-occlusive process, which involves interactions between sickle erythrocytes, activated leukocytes, endothelial cells, platelets, and plasma proteins. Recurrent vaso-occlusion, the ischemia-reperfusion process, and consequent activation and injury of vascular endothelial cells induce a chronic inflammatory response in individuals with SCA and are related to systemic manifestations that increase morbidity and mortality in this group.4,5,15,16 The assessment of the lipidic profile and its correlation with hemolysis and inflammation can provide data on the vascular risk and clinical severity of patients. 17
In this study, the reduced hematimetric levels findings and the elevated hemolysis markers in individuals with SCA reflect the pathophysiology of the disease and are in line with the scientific literature.18−21 Such changes have resulted from the process of hemolysis, platelet activation, and chronic inflammation that occur in SCA, which triggers a decrease in the Hb levels and an increase in the levels of leukocytes, platelets, and hemolysis markers, such as LDH, reticulocytes and IB.18−21
Although the current findings of lower TC, LDL-c, and HDL-c levels in the SCA group are consistent with previous studies,7,22−24 the scientific literature is unclear on this point, with studies that showed higher HDL-c levels in this group.10,11 The similar TG levels between the SCA and control groups found in this study differ from previous studies, which found lower TG levels in individuals with SCA.10,25 The TG/HDL-c ratio is highlighted as a tool capable of identifying cardiometabolic risk and predicting cardiovascular diseases. 9 This study presents a higher TG/HDL-c ratio in the SCA group compared to the control group, a conclusion consistent with a previous study, which showed a probability of individuals with SCA having a TG/HDL-c ratio 10 times higher than controls. 26 The observed hypocholesterolemia can be explained by the marrow compensatory response with increased production and maturation of red blood cells due to the shorter half-life of erythrocytes from patients with SCA. 6 Another mechanism that explains the reduction in cholesterol levels is the decrease in the volume of red blood cells, which leads to an increase in plasma volume and dilution of lipids and lipoproteins. 7
Although other studies show a decrease in the caloric intake of patients with SCA as a result of inappetence and frequent hospitalizations, associated with pain crises,7,27,28 this study showed an increase in the intake of calories, proteins, and carbohydrates in the group of patients with SCA, such disagreement may be justified by the fact that the group of patients studied was in a stable period of the disease during data gathering. Furthermore, the group of patients with SCA had lower anthropometric indices than the control group, a finding related to weight and BMI in agreement with previous studies.7,29 The findings of increased food intake and reduced anthropometric measurements support the hypercatabolic state of patients with SCA.
Although the individuals with SCA had lower weight and BMI than the controls, there is no evidence that this finding accentuated the difference in their lipid levels, since no correlation was observed between these anthropometric indices and cholesterol levels in any of the groups. This study showed no correlation between macronutrient consumption and lipidic profile in adults with SCA. Studies performed on children and teenagers have shown that, although there is little to no difference in terms of caloric intake, the energy expenditure at rest of patients with SCA is altered, indicating hypercatabolism.28,30 These studies confirm the hypothesis that the differences observed in the lipidic profile between patients with SCA and members of the control group are not only related to the difference in caloric intake, but also to the mechanisms of hemolysis and hepatic alterations previously discussed. 6
The conclusions indicating a positive correlation of HDL-c with Hb and a negative correlation with leukocytes, LDH, and IB, and a negative correlation of the TG/HDL-c ratio with Hb and a positive correlation with reticulocytes, LDH, and leukocytes corroborate previous reports in the scientific literature.23,31 A previous study with children and teenagers with SCA showed a decrease in HDL-c levels in this group and a positive correlation between HDL-c and inflammatory and hemolysis markers. 25 Current results point to more intense hemolytic anemia with lower HDL-c levels and higher TG/HDL-c ratio. The correlations of HDL-c and the TG/HDL-c ratio with the leukocytes found in the study also support the idea of hypocholesterolemia and greater severity of SCA, as leukocyte levels are used as an independent factor in the association with clinical severity in SCA, with leukocytosis associated with shorter life expectancy. 32
Limitations
Some limitations faced during the study were that this was a unicentric study, which involved a sample selected for convenience, the lack of patients on some appointment dates, as most of them reside in other cities and have socioeconomic limitations, and the COVID-19 pandemic, which led to the suspension of data gathering for a 9-month period.
Conclusion
The study results confirm the occurrence of hypocholesterolemia in adults with SCA, with a reduction in TC, HDL-c, and LDL-c levels and a higher TG/HDL-c ratio and point to an independent relation of these changes with the intake of calories, proteins, lipids, and carbohydrates.
The correlation between the alterations in the lipid profile with more intense anemia, characterized by lower levels of Hb, and higher levels of hemolysis markers, with higher levels of reticulocytes, IB, and LDH, points to the role of HDL-c and the TG/ HDL-c ratio as easily accessible and low-cost markers that help predict adverse clinical events related to the severity of SCA in these individuals.
In addition, HDL-c and the TG/HDL-c ratio also contribute to the assessment of cardiovascular risk, which points to the need to consider longitudinal studies that prospectively assess their impact on cardiovascular outcomes and a therapeutic approach, in order to prevent cardiovascular disease in individuals with SCA.
Supplemental Material
sj-docx-1-bdx-10.1177_26348535231193889 - Supplemental material for Lipid Profile, Nutritional Status and Severity Biomarkers in Adults With Sickle Cell Anemia
Supplemental material, sj-docx-1-bdx-10.1177_26348535231193889 for Lipid Profile, Nutritional Status and Severity Biomarkers in Adults With Sickle Cell Anemia by Ingrid Cristiane Pereira Gomes, João Pedro Costa Machado Teles, Anny Catarina Sousa Coelho, Maria Carollyne Passos Cruz, Lindemberg Costa de Albuquerque, Mariana Amaral Carvalho, Paulo Herlan Castro dos Santos, Sérgio Nolasco dos Santos and Rosana Cipolotti in Plasmatology
Footnotes
Acknowledgments
The authors appreciate all patients and control volunteers for their participation in this study and for support from the Health Sciences Graduate Program at UFS, Brazil.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Contributions
Availability of Data and Materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Consent for Publication
Not applicable.
Ethics Approval and Consent to Participate
The project was approved by the Ethics Committee in Research Involving Human Beings of the Federal University of Sergipe (CEP HU/UFS), through serial number 2.897.835. Individuals who expressed their consent by appreciating and signing the Free and Informed Consent Term participated in the study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
