Abstract
Primary non-Hodgkin's lymphoma of the bone is an unusual presentation of Hodgkin's lymphoma, with primary cranial vault lymphoma (PCVL) being the rarest of bone lymphomas. We describe a case of an immunocompetent elderly patient with new onset gait imbalance and cognitive delays. Brain imaging showed a large extra-axial mass with osseous invasion. An initial misdiagnosis of meningioma led to delays in management. Further work up included a brain magnetic resonance image showing a large right transcalvarial parietal mass. The mass was surgically removed, and pathology revealed B-cell lymphoma with low-grade features thus establishing the diagnosis of PCVL. Management included subtotal resection of the tumor followed by adjuvant radiation therapy. The patient has achieved a durable response for over a year. Meningioma and PCVL have similar imaging and clinical presentations. This case report addresses differing features of the tumors and explores therapeutic options for management of PCVL.
Introduction
Primary cranial vault lymphoma (PCVL) is a rare primary non-Hodgkin's lymphoma (NHL) of the bone; it comprises approximately 0.3% to 1% of all bone NHL. Primary bone lymphomas are described as solitary lesions with no disease involvement at other distant sites or metastasis within 6 months of tumor detection. We reported a case of an immunocompetent elderly patient presenting with altered mental status, ataxia, and a sizeable extra-axial mass in the right posterior frontal region with osseous invasion and extension to the right temporal bone. The patient described in the case presentation was initially misdiagnosed with meningioma. Meningioma and PCVL have similarities that can lead to the misdiagnosis of primary bone lymphoma as meningioma. For instance, both meningioma and PCVL clinically present as scalp masses, in addition can cause mass effect symptoms such as seizures and headaches. Meningioma and PCVL may present as enhancing masses that involve the dura on brain imaging. 1 Distinguishing between meningioma and PCVL includes clinically assessing growth pattern of the tumor, evaluating brain imaging, and pathological analysis of tumor tissue after tumor resection. This report aims to describe the clinical manifestations, unique diagnostic features, and management of PCVL, thus minimizing delays in diagnosis and treatment.
Case Presentation
The patient is an 81-year-old female with a medical history of hyperlipidemia and recurrent pyelonephritis requiring left-sided nephrectomy that presented with 1 month of malaise, loss of balance, and decline in mental status. Abnormal presentation labs included thrombocytopenia of 99,000/μL with other labs within normal range. Magnetic resonance imaging (MRI) of the brain showed a large extra-axial mass with osseous invasion extending and involving the squamosal portion of the right temporal bone with adjacent extracranial extension. Neurosurgery was consulted and the patient was presumed to have a meningioma. Neurosurgical intervention was deferred due to thrombocytopenia. The patient was discharged with outpatient neurosurgery follow up.
The patient presented 2 weeks after discharge with worsening confusion and left-sided weakness. Computed tomography (CT) of the head showed a large transcalvarial mass in the right frontal and parietal region with a large intracranial component, with no interval change (Figures 1-3). Repeat MRI showed a large right-sided parietal cranial lesion with intracranial, epidural, and extracranial component; the intracranial extension was leading to mass effect on the frontal lobe (Figure 4). The brain imaging findings were consistent with a malignant brain lesion. The patient was started on treatment with intravenous Decadron. Neurosurgery was consulted, and patient underwent right frontoparietal craniotomy, microdissection, and removal of tumor. The intra-operative report described the tumor as extending through the bone of the skull and spreading into the subperiosteal for several centimeters over the temporal region of the skull; underneath the bone, the tumor invaded the dura extensively and formed a thin layer of tissue extending across the convexity down to the sylvian fissure and up to the midline for a width of approximately 6 cm. The patient's postoperative period was complicated by generalized seizures and controlled with anti-epileptics. Electroencephalogram showed severe diffuse encephalopathy and right frontocentral epileptogenicity with overlying skull defects.

Axial CT scan of the head: large transcalvarial mass (6.2 × 6 × 3.3 cm) in the right frontal and parietal region with a large intracranial component (left); a large mass involving the right frontal and parietal bones, with components in the anterior cranial fossa and in the extra cranial soft tissues (right). Abbreviation: CT, computed tomography.

Both bone CT windows show a large mass involving the right frontal and parietal bones, with components in the anterior cranial fossa. Abbreviation: CT, computed tomography.

Sagittal CT of the head; large mass in the right frontal and parietal region with a large intracranial component. Abbreviation: CT, computed tomography.
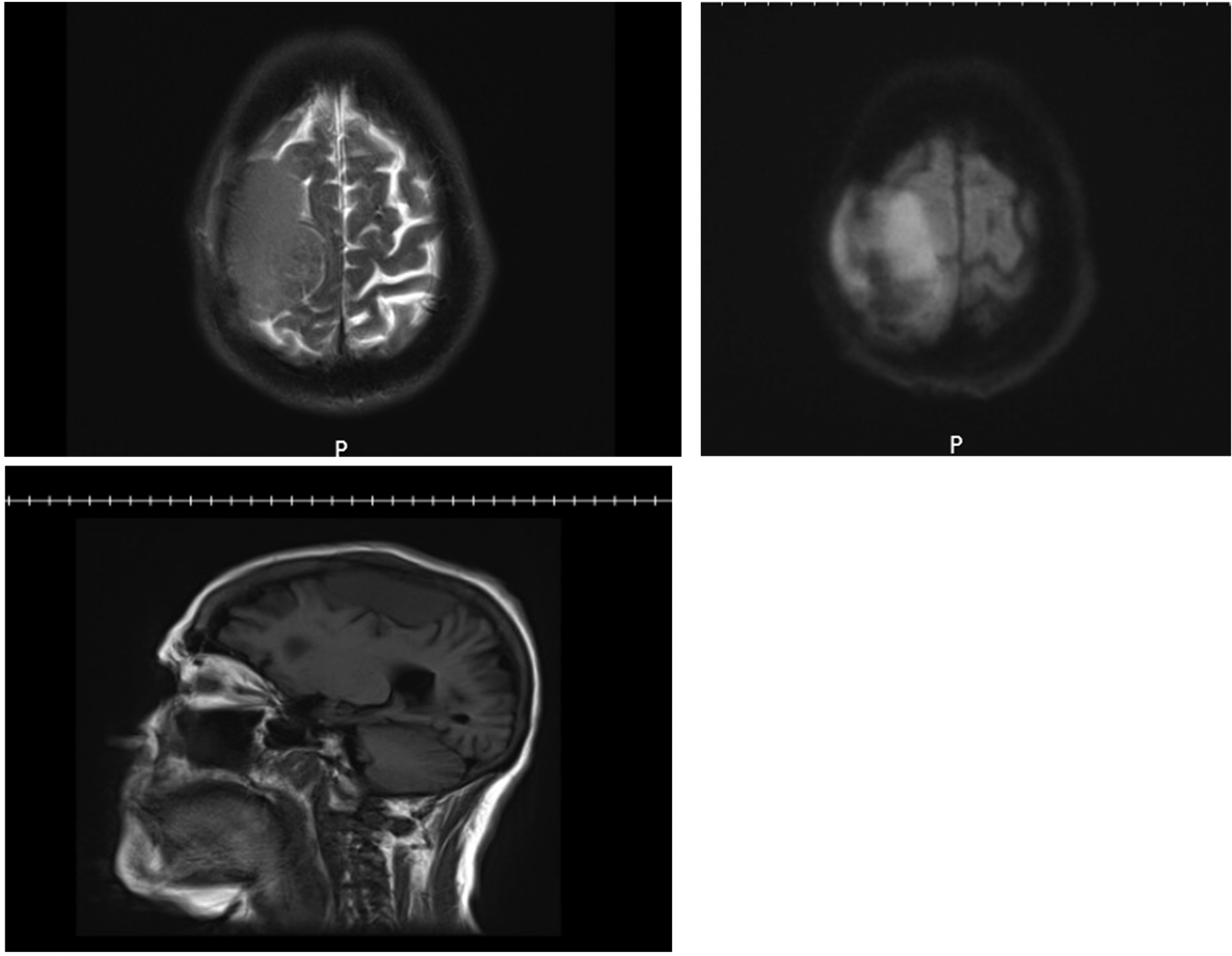
Axial T2-MRI of the brain (left), axial DW-MRI of the brain (right); and sagittal T1-MRI of the brain. The images show a large right high parietal calvarial lesion with an adjacent intracranial and extracranial homogeneous enhancing and diffusion restricting soft tissue component. There is prominent mass effect on the adjacent frontal lobe. The findings are suggestive of a densely packed malignant lesion. Abbreviation: DW-MRI, diffusion-weighted magnetic resonance imaging.
The pathology report of the resected cranial mass showed a lymphoproliferative process involving the peri-cranial and epidural tissues; brain tissue was not identified in the resected tumor. The cells were consistent with small lymphocytes and Ki67 was approximately 40% (Figure 5). Fluorescence in situ hybridization (FISH) analysis did not detect translocations involving t(11;14), t(14;18), b cell lymphoma locus 1, BCL2, BCL6, immunoglobulin heavy chain, or mucosa associated lymphoid tissue translocation 1. Laboratory analysis showed lactate dehydrogenase 344, uric acid 6.3, and beta-2-microglobulin 3.3. Urine and serum protein electrophoresis with immunofixation, and kappa and lamda quantitative free light chain ratio was within normal limits. The patient was human immunodeficiency virus (HIV) negative. The pathology report was consistent with B-cell lymphoma with low-grade features (Figures 6-11). A positron emission tomography CT after craniotomy showed moderate focal increased tracer uptake in the right posterior inferior frontal lobe above the temporal lobe with standard uptake value of 10.7 with mild diffuse tracer uptake along the skull base and no fluorodeoxyglucose avid lymphadenopathy. There were no bone lesions or presence of disease elsewhere in the chest, abdomen, or pelvis, thus, bone marrow biopsy was not performed. A multidisciplinary approach was used to manage the patient postneurosurgery. The decision was made to defer adjuvant systemic chemotherapy given localized disease, patient's advanced age, and frail status. She received adjuvant intensity-modulated radiation therapy at a dose of 40 Gy in 20 fractions over 1 month. The patient tolerated radiation treatment. The patient has been in remission for over a year.
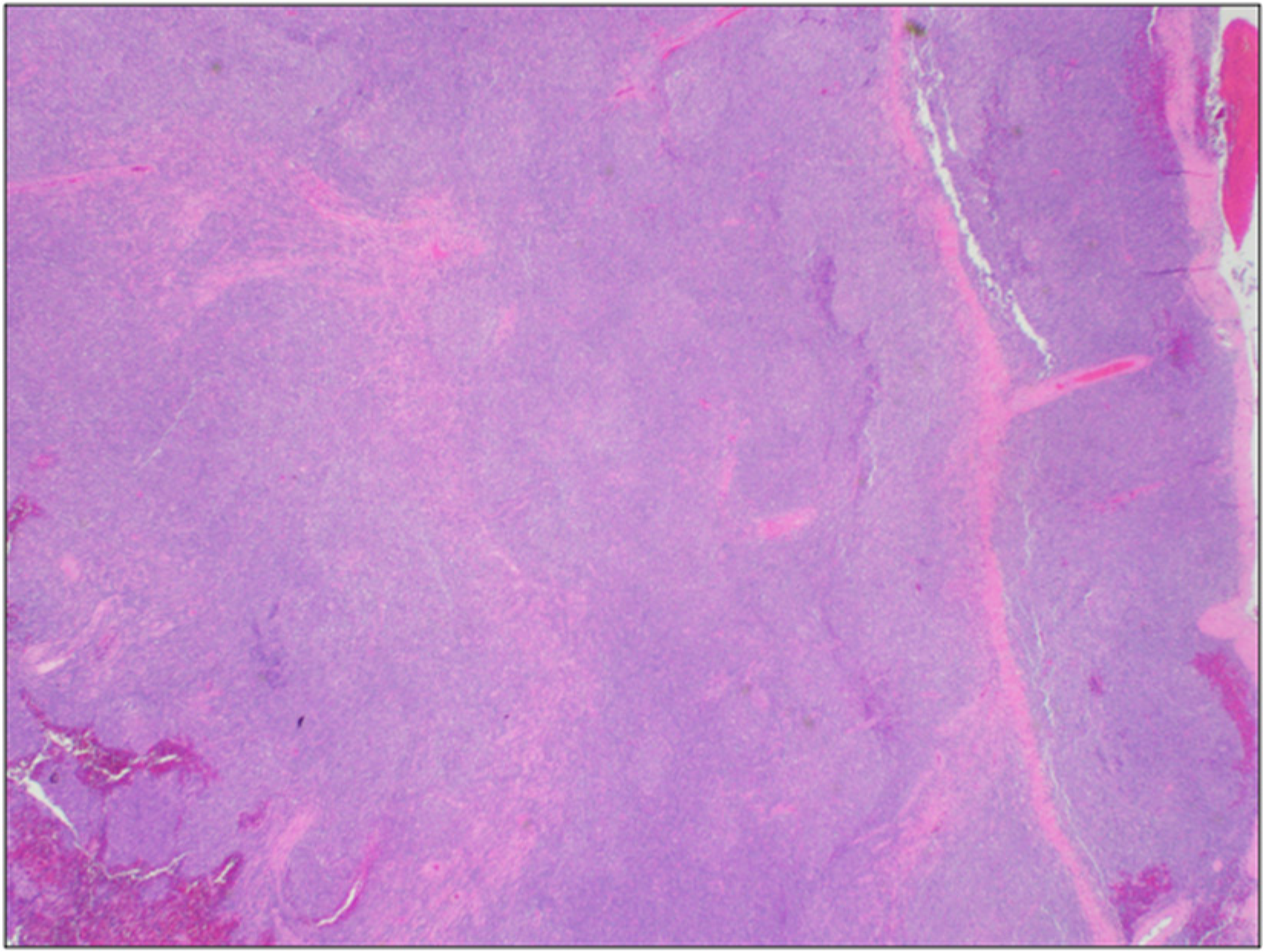
Low power (2X) view shows monotonous appearing blue cells adjacent to dura.

Low power (4X) shows bony fragment and dura on top left of the picture with lymphoma cells on the bottom.

Medium power (10X) view shows monotonous appearing small-to-medium-sized lymphoma cells adjacent to dura.

High power (40X) view shows monotonous appearing small-to-medium-sized lymphoma cells with inconspicuous nuclei and pale cytoplasm.

The lymphoma cells show positive membranous staining with CD20 indicating they are of B-cell origin.
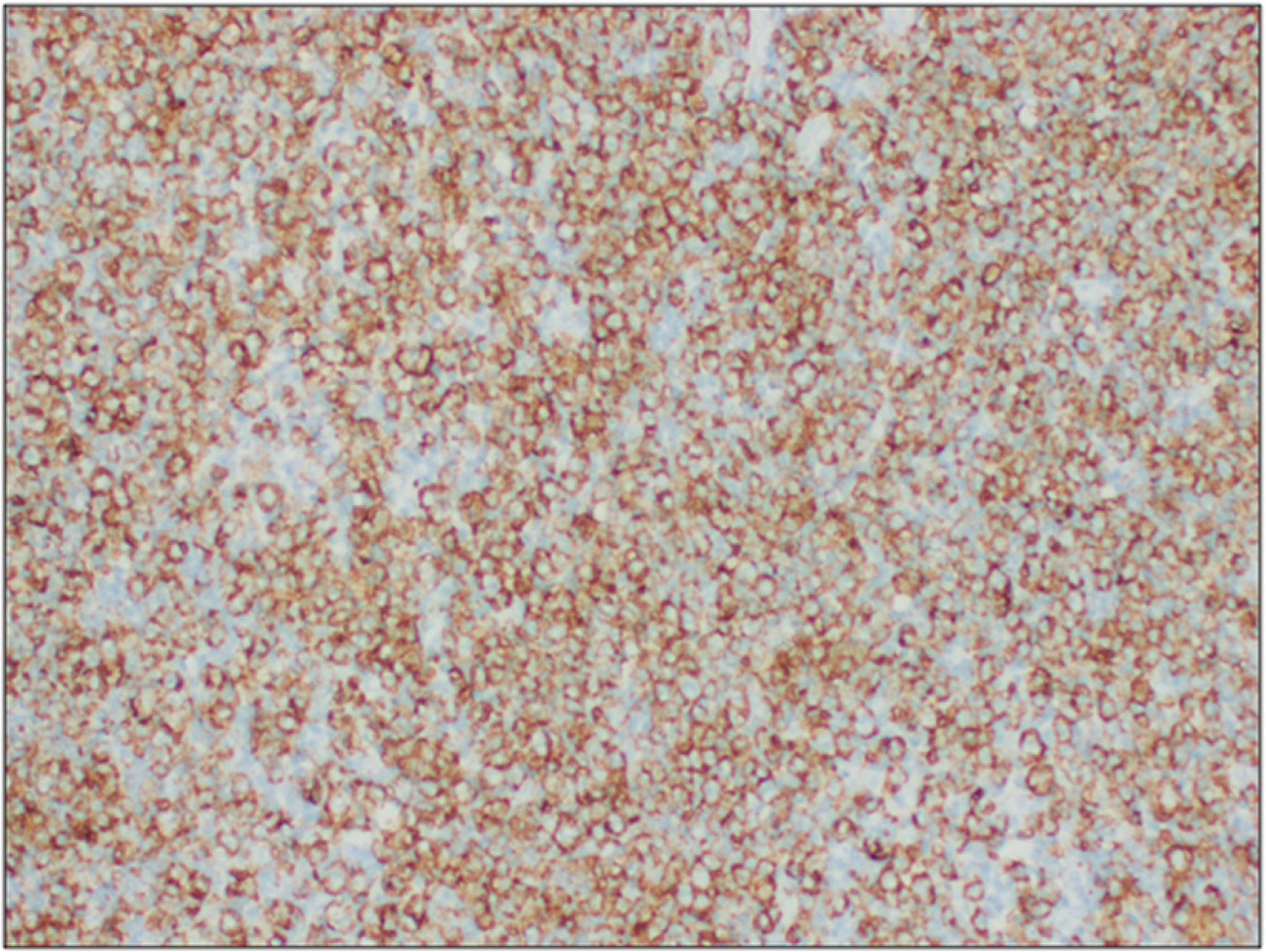
The lymphoma cells show positive nuclear staining with PAX5 further confirming B-cell origin of the lymphoma. Abbreviation: PAX5, paired box 5.
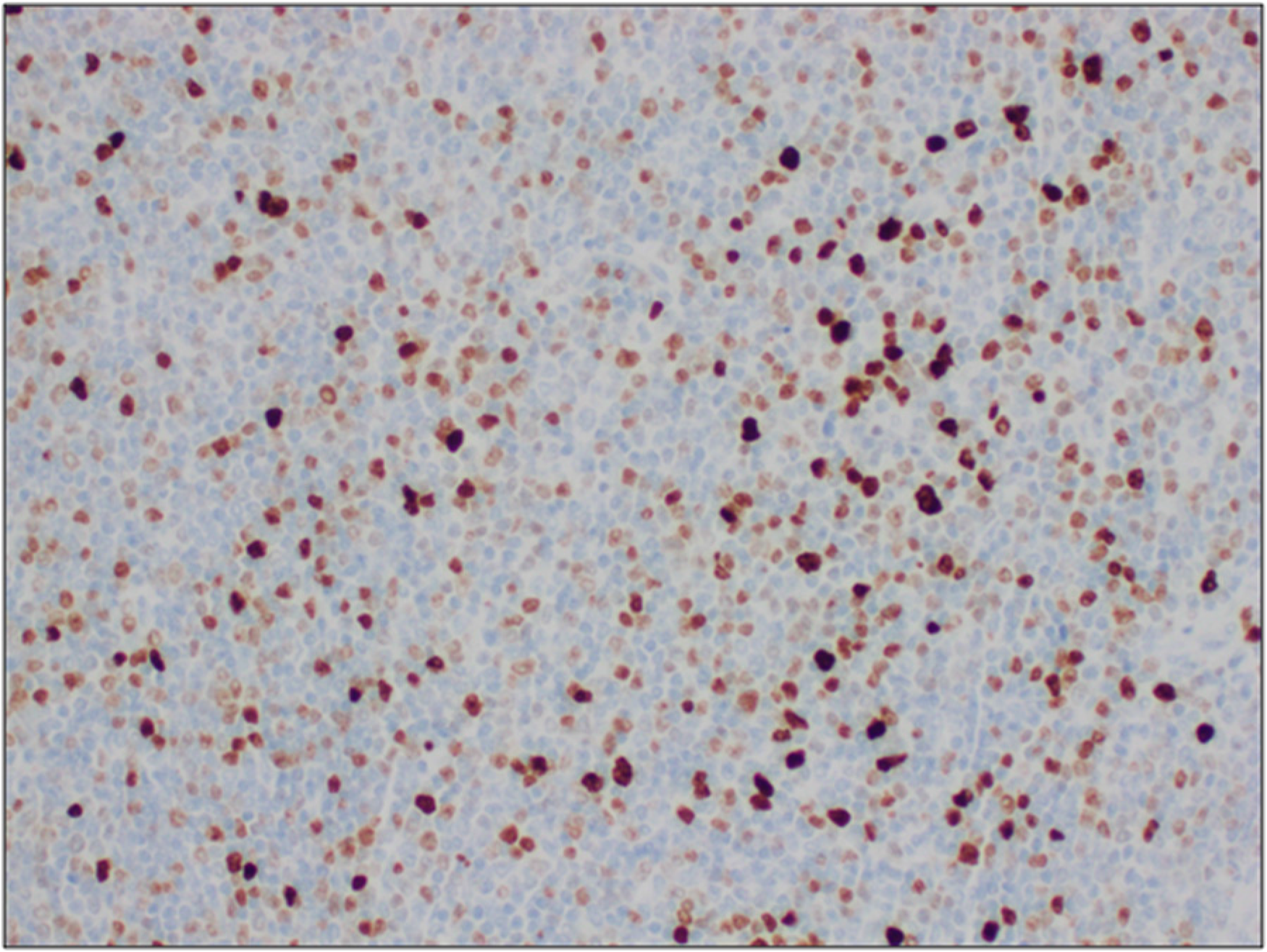
The Ki67 proliferation index is approximately 40% indicating it is a low-grade lymphoma.
Discussion
Primary NHL of the bone is extremely rare and accounts for <1% of cases of NHL.1,2 Common sites for bone lymphomas include the femur (50%), humerus (20%), and 30% involve the spine, ribs, mandible, and scapula. PCVL is even rarer and accounts for <1% of primary bone lymphomas. PCVL commonly effects older patients above the age of 60 and effects both sexes equally.1,3,4 A review by el Asri et al 1 evaluated 39 patients with PCVL; the most common clinical manifestations were a subcutaneous scalp mass (90%), headaches (30%), and focal neurological deficits (20%). The least common presentations included seizures (10%) and proptosis (10%).4–8 Neurological symptoms can arise if PCVL infiltrates the brain cortex either by leptomeningeal involvement or direct infiltration. PCVL can involve all bones of the cranial vault, but the most common is the parietal bone, which accounts for approximately 37% of cases. Most of the reported cases of PCVL are in immunocompetent patients; only a minority of patients are immunocompromised with HIV.1,4
PCVL can be misdiagnosed as an atypical meningioma, as was the case for our patient. Both PCVL and meningioma can present as nontender subcutaneous scalp masses with intracranial and extracranial extensions. 9 A difference between PCVL and meningioma is the timeline of tumor growth and presentation of mass effect symptoms. The tumors seen in PCVLs grow more rapidly with symptom presentation within 5 months of growth. In comparison, meningiomas are benign slow growing tumors with rare presentation of neurological deficits from the neoplasm. A noninvasive cerebral angiogram is a procedure that can be used to differentiate meningiomas from primary cranial vault lymphoma; meningiomas are highly vascular due to supply from the external carotid artery while PCVLs have minimal to no vascularization.9–12 Although nonspecific, serum soluble interleukin 2 receptor levels (sIL2R) is a lymphoproliferative marker that can be used to differentiate between meningioma and PCVL; levels > 2000U/mL may suggest the presence of a lymphoid malignancy as opposed to meningioma.12,13 The majority of the PCVL cases have a permeative or moth-eaten growth pattern both intracranially and extracranially involving a large soft tissue component with mild-to-moderate osteolytic changes seen on brain imaging. In contrast, in metastatic lymphoma to the bone purely sclerotic bone lesions are more commonly found. In addition, meningiomas have been shown to have a hyperostotic bone involvement and no brain invasion; brain invasion has been reported in a case of PCVL. 9 In the case of our patient, brain imaging showed a permeative bone lesion with osseous invasion.1,3,6,14
The histological subtypes in about 94% of patients with primary cranial vault lymphoma are of B-cell lymphoma and about 6% of T-cell lymphomas. 1 The appropriate treatment for PCVL is determined after histological subtype is confirmed. In our patient, the use of surgical resection of the tumor, followed by adjuvant radiation therapy at a dose of 40 Gy in 20 fractions was employed. With this regimen our patient has achieved a durable response and had prolonged survival for over a year (see Figure 12 for complete timeline of patient events). The few cases reported show that extra-nodal disease localized to a region is best managed with either surgery or radiotherapy. In one case report a patient developed cranial hematoma from tumor biopsy, which resulted in death from complications with hematoma evacuation. 1
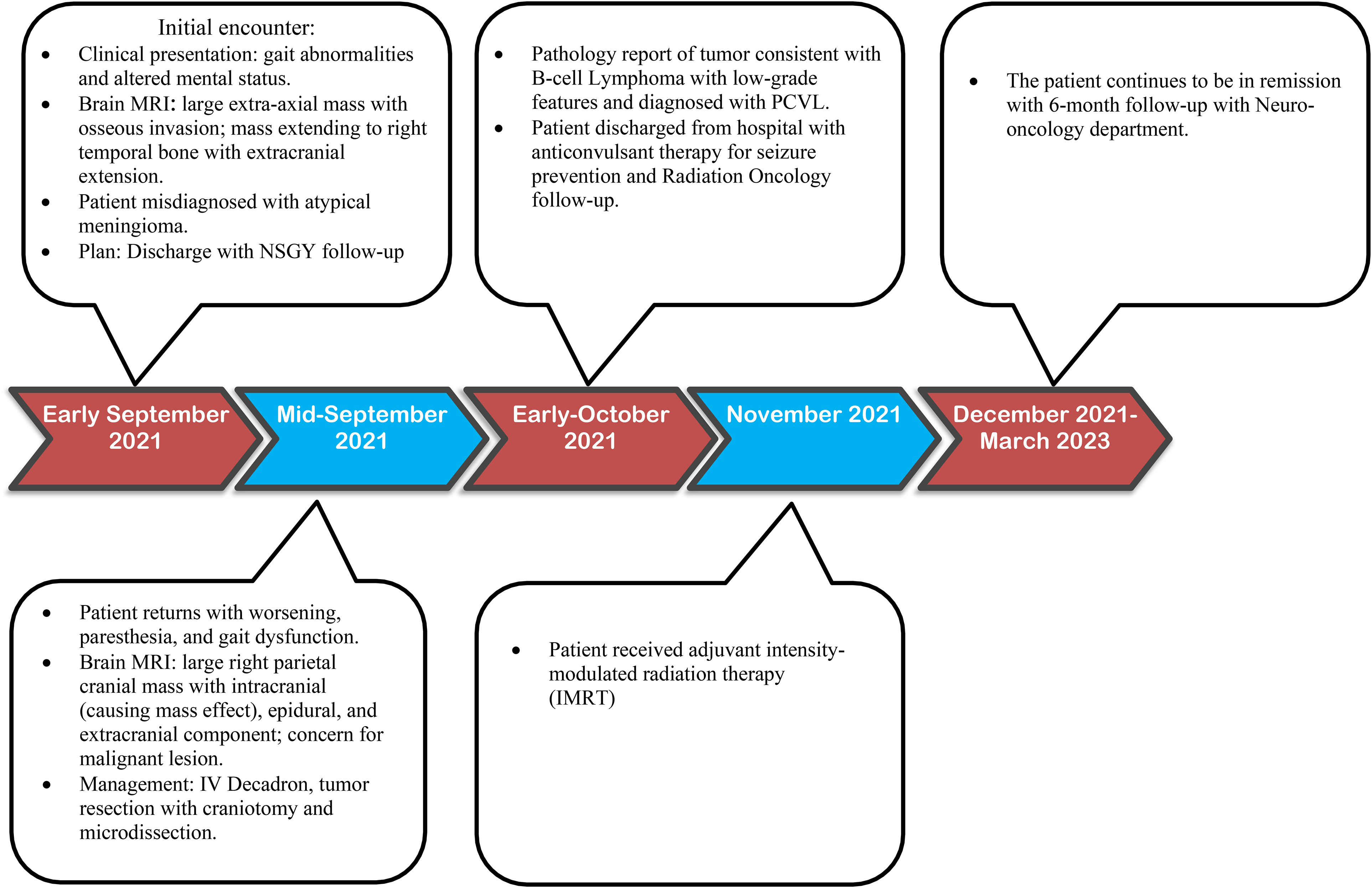
Timeline of patient events. Abbreviations: NSGY, neurosurgery; IV, intravenous; PCVL, primary cranial vault lymphoma.
Other treatment options include surgical resection followed by adjuvant chemotherapy with rituximab, cyclophosphamide, doxorubicin, vincristine, prednisone chemotherapy (cyclophosphamide: 750 mg/m2 on day 1; doxorubicin: 50 mg/m2 on day 1; vincristine: 1.4 mg/m2 on day 1; and prednisone 40 mg/m2 by oral route from days 1-5), plus rituximab (375 mg/m2 on day 1) every 21 days for 4 to 6 cycles for B-cell subtype. 15 For patients that are advanced age, frail, or have focal disease, R-mini-CHOP can be considered. 16 Studies on the best treatment regimen for PCVL, especially regarding the role of radiation and chemotherapy, are lacking. Review of reported cases show that surgical resection followed by radiation therapy as the first-line treatment of choice for extra-nodal disease.2,14,17 Radiation therapy is recommended for leptomeningeal involvement or extensive dura lesions. 1
A few cases adopted the strategy of radiological observation without chemotherapy or radiation therapy when complete resection is achieved; no follow-up studies on these patient groups are available to evaluate the outcome and efficacy of this approach. 1 Growing data has shown durable response with the combination modality with chemotherapy and radiation following surgical resection. 2 Our patient continues to have well-controlled disease with surgical resection and radiation therapy with yearly brain imaging surveillance without the need for chemotherapy.
Conclusion
The diagnosis of PCVL is challenging due to the rarity of the lymphoma and clinical plus radiographical similarities with meningioma. In this case report, we describe the differing features that can be used to distinguish PCVL from meningioma. These include the rapid extensive pattern of growth for PCVL (cranial, extracranial, and intracranial) and limited vascularity seen in cerebral angiogram of the tumor. The mainstay first-line treatment is resection of the tumor. Limited studies exist on the role chemotherapy and radiation therapy have on overall survival of patients; thus, future retrospective studies must be done to assess the most efficacious management for patients with primary cranial vault lymphoma.
Footnotes
Acknowledgment
The authors thank Dr Anindita Ghosh for providing relevant pathology images for the case.
Author Contribution(s)
Ethics Approval and Consent to Participate
Patient's consent was obtained for the case report.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
