Abstract
Background
Population pharmacokinetics (popPK) has been reliably leveraged to generate individual PK in hemophilia patients. Specific popPK models are suited to predict individual PK under a variety of scenarios that may not be captured by clinical trials, allowing for individualized prophylactic treatment. The Web-Accessible Population Pharmacokinetic Service-Hemophilia (WAPPS-Hemo) project generates individually predicted pharmacokinetic profiles relying on concentrate-specific popPK models used for Bayesian forecasting.
Objective
Specification of a popPK model for the plasma-derived factor VIII (FVIII) concentrate Koate-DVI and its suitability for pharmacokinetic estimation in low-dose scenarios.
Methods
A popPK model was developed for Koate-DVI WAPPS-Hemo PK data in combination with the existing WAPPS-Hemo Fanhdi/Alphanate model derivation dataset using nonlinear mixed effects modelling, and was validated via cross-validation and prediction-corrected Visual Predictive Checks (pcVPC). Bootstrap and PK outcomes between the Fanhdi/Alphanate and the Fanhdi/Alphanate/Koate models were compared, and the relative error distributions from a limited sampling analysis (LSA) under the latter model for low and normal doses (10 vs 50 IU/kg) and inclusion/exclusion of pre-dose measurements.
Results
A Fanhdi/Alphanate/Koate model was derived (126 patients, ages 1–71 years) after deeming a Koate-brand covariate not clinically significant, resulting in similar parameter estimates than the Fanhdi/Alphanate model with satisfactory goodness of fit, cross-validation and pcVPC results. Low-dose predictions resulted in a higher accuracy and slightly lower precision of half-life (
Conclusions
The Fanhdi/Alphanate/Koate model is appropriate for Bayesian forecasting in the WAPPS-Hemo platform, providing a comparable prediction capability for low and normal dosing regimens (10 vs 50 IU/Kg).
Introduction
Hemophilia is a bleeding disorder caused by a hereditary deficiency of plasma coagulation factors VIII or IX (FVIII, FIX). Tailored prophylaxis for severe hemophilia patients, who present factor plasma levels below 0.01 IU/mL, is an effective strategy to increase the net clinical benefit of treatment.1,2 Population pharmacokinetics (popPK) employs statistical methodology to describe PK and can be subsequently leveraged to reliably estimate individual pharmacokinetic parameters based on a reduced number of plasma factor level observations and patient's characteristics. 3 Specific popPK models are therefore suited to predict individual PK profiles under a variety of scenarios that may not be captured by clinical trials, allowing for individualized prophylactic treatment.
The Web-Accessible Population Pharmacokinetic Service – Hemophilia (WAPPS-Hemo) 4 project provides a comprehensive popPK application for hemophilia health-care providers (www.wapps-hemo.org), generating individually predicted pharmacokinetic profiles, hence assisting in individual treatment optimization. At its core lies a set of popPK models for commonly used factor concentrates that incorporate the patterns of variability that are identified in the underlying population. 3 Such popPK models are used as priors in a Bayesian estimation methodology that provides predictions of individual pharmacokinetic profiles.
Koate-DVI (Koate, Grifols Therapeutics Inc. NC, US) is a plasma-derived factor VIII concentrate containing von Willebrand factor (vWF) indicated for the treatment of hemophilia A. Koate provides a temporary clotting factor replacement to control or prevent bleeding episodes and may be used in anticipation to surgery. 5 Koate contains naturally occurring vWF, which is co-purified during its manufacturing process. Koate is a sterile, stable, purified, freeze-dried concentrate, treated with tri-n-butyl phosphate (TNBP) and polysorbate 80 and heated in lyophilized form in the final container at 80 °C for 72 h. Koate is purified from the cold insoluble fraction of pooled fresh-frozen plasma by modification and refinement of the methods described in Hersgold, Pool, and Pappenhagen. 6 It contains purified and concentrated FVIII. Chozie et al. reported the clinical benefits of low-dose tertiary prophylaxis with Koate compared to on-demand treatment for severe hemophilia A in children, which was associated with significant reduction in bleeding frequency and better joint function. 7
The popPK models currently available in WAPPS-Hemo include standard and extended half-life FVIII and FIX concentrates. 3 To date, no specific model describing Koate pharmacokinetics has been developed. In order to increase the range of concentrate-specific popPK models presently available in WAPPS-Hemo, this study describes the development and validation of a popPK model that includes the plasma-derived FVIII concentrate Koate. The WAPPS-Hemo dataset used to derive the existing plasma-derived FVIII Fanhdi/Alphanate model 8 was used as leverage in order to compensate for Koate data sample size and density limitations. Ensuring the suitability of a combined Fanhdi/Alphanate/Koate dataset for modeling and individual prediction implies answering the question of whether the previously developed Fanhdi/Alphanate popPK model would differ if Koate infusions were included. A secondary objective was to use the resulting Fanhdi/Alphanate/Koate model to assess whether Bayesian estimation had similar precision between low versus normal dose treatments under various sampling times and number of factor activity levels.
Material and Methods
Derivation dataset
Koate FVIII activity (one-stage) versus time profiles for 34 patients from six WAPPS-Hemo participating centres were extracted on June 6, 2017 (117 observations; samples/patient, median: 4, range: 2–7). The majority of the patients (n = 25) came from the Hemophilia Center in Indonesia. Two patients had a history of inhibitors, but none had them at the time of sample collection. Koate data were combined with the derivation dataset of the Fanhdi/Alphanate popPK model, 8 resulting in a total of 126 patients (503 observations, extracted on February 16, 2018). Individual information for modeling was extracted, including but not limited to, dose and duration of infusion; body weight (BW); age; height (HT); endogenous (baseline) FVIII activity as well as measured plasma FVIII levels and times of blood samples. There were no missing data in the Koate dataset.
Fanhdi/Alphanate/Koate model building and internal validation
The model building strategy for the Fanhdi/Alphanate/Koate data followed the workflow outlined in McEneny-King et al. 9 Nonlinear mixed effects modelling as well as Bayesian predictions were completed in NONMEM and PDx-Pop (v 5.10; ICON Development Systems, Ellicott City, MD, US), using the first order conditional estimation with interaction (FOCEI) method. 10 Briefly, a naïve pooled analysis for the exploration of model structure and mean estimates of pharmacokinetic parameters preceded the determination of the number of compartments that would define the base model structure through graphical techniques and goodness of fit assessments. Between subject variability (BSV) on pharmacokinetic parameters as well as various residual unexplained variability (RUV) error models were explored. Prior to covariate incorporation, a graphical assessment of the relationships between covariates and estimated random effects was performed. Covariates likely to be significant were included into the model in a forward selection fashion and formally tested. Covariates involved age (y), total body weight (kg), fat-free mass (kg),11,12 height (cm) and brand (Koate or Fandhi-Alphanate). Blood group was not available for every patient as it is not currently a mandatory input on the WAPPS-Hemo platform, and therefore not included as a potential covariate. A correlation term was assessed between the pharmacokinetic parameters that included BSV. Goodness of fit was assessed through plots of model based predictions against observed values, conditional weighted residuals (CWRES) and estimated random effects.
The overall predictive capability of the final model was assessed through a hold-out cross-validation assessment as in Hajducek et al.
3
Succinctly, the hold-out cross-validation method is employed to assess the model's ability to predict derived pharmacokinetic outcomes (half-life: post-distribution or
As a second model validation tool, a prediction-corrected Visual Predictive Check (pcVPC) 13 was implemented to assess the extent in the ability of the Fanhdi/Alphanate/Koate model to recreate the central tendency and variability of the Fanhdi/Alphanate/Koate factor level observations. Factor activity versus time simulations under the model consisted of a total of 500 repetitions. Observed factor levels, their median, and upper and lower fifth percentiles were plotted against time and compared to the same percentiles of each model simulation-based predictions, with bands representing 90% inter-percentile ranges within pre-specified time windows. As a visual tool, the predictive capability of the Fanhdi/Alphanate/Koate model is considered satisfactory if the observed outcomes fall reasonably well within the corresponding simulation-based 90% interquartile bands.
Comparison with Fanhdi/Alphanate model
Since the Fanhdi/Alphanate/Koate model resulted in the same structure, covariate and variance relationships than the Fanhdi/Alphanate model, 8 parameter estimates of the two models were deemed similar based on the overlap between their bootstrap (with 1000 repetitions) confidence intervals (CIs). Cross-validation results were compared through the relative errors distributions for estimates of half-life, time to 2% (TAT2%) threshold, population central clearance (CL) and volume of distribution (V1).
Individual half-life estimates and their variability under each model were derived by first using the estimated popPK parameters in conjunction with conversion to scaling values of terminal decay 14 appropriate for half-life estimate calculation for a 2-compartment model and second, by incorporating individual random effects as simulated, normally distributed random variables with model-based estimated variability. To replicate the true variability of half-life estimates, a total of 10 000 datasets of 6600 patients of ages between 2 and 50 years were simulated with related body weight and height based on the NHANES database. Boxplots of the distribution of half-life estimates by age were compared.
Dose dependency of predictions under Fanhdi/Alphanate/Koate model
A Limited Sampling Analysis (LSA)
15
is routinely employed for WAPPS-Hemo popPK model evaluation to assess pharmacokinetic prediction accuracy during Bayesian forecasting in sampling scenarios of various sample-densities.
3
In addition, the purpose of LSA in this study was to compare the dose dependency of predictions under the Fanhdi/Alphanate/Koate model with low and normal dose administrations and to assess the effects of the number and timing of samples. The following steps were used:
Simulation of dense pharmacokinetic data. Pharmacokinetic profiles from 1000 virtual patients with 17 factor level observations were simulated based on the Fanhdi/Alphanate/Koate model with sampling times at: {pre-dose, 0.5, 1, 2, 4, 6, 12, 18, 24, 30, 36, 42, 48, 54, 60, 66 and 72 h post-dose}. The covariate space was simulated based on the combined Fanhdi/Alphanate/Koate dataset (n = 126), with a median FFM and age as in Table 1. Pharmacokinetics for each individual were simulated following prophylaxis treatment with low and normal doses (10, 50 IU/kg) administered 3 days a week (Mon-Wed-Fri), for 4 weeks to achieve steady state. Pre-dose concentrations were recorded as the factor level activities simulated at 47.5 h after the preceding infusion (administered at 48 h prior to the Friday administration of the fourth week). Pre-dose activities lower than 0.01 IU/mL were set as below limit of quantification (BLQ) and endogenous FVIII levels were set at 0.005 IU/mL. Individual pharmacokinetic-derived outcomes (half-life, TAT2%) based on the 17 simulated observed data points were estimated through Bayesian forecasting and used as reference. Extraction of sparse pharmacokinetic data. Sparse pharmacokinetic profiles based on various sampling designs and covariate information for each patient were extracted from the dense data simulated in (i), with sampling time points selected according to each of 5 to 3-, or 2-sampling designs, constituting subsets of the sampling time points generated in (i): {2, 4, 6, 12, 48 h}, {2, 12, 48 h} and {2, 48 h}, including or excluding a pre-dose observation. Patient-specific pharmacokinetic-derived outcomes based on Bayesian predictions from each sampling design were recorded. Evaluation. Bayesian estimates (
Patient demographics in Fanhdi/Alphanate/Koate dataset (n = 126).

BW: body weight; BMI: body mass index; CV: coefficient of variation; FFM: fat free mass; HT: height; Min: minimum; Max: maximum; SD: standard deviation.
Ethics
The WAPPS user agreement allows reuse of the data for modelling and other research purposes, as described in the WAPPS study protocols, approved by the research ethics boards at McMaster University and the University of Waterloo and registered in clinicaltrial.gov (NCT02061072, NCT03533504).
Results
Derivation dataset
Statistical summaries of the combined Fanhdi/Alphanate/Koate dataset comprising 126 patients are presented in Table 1 (92 and 34 from the Fandhi/Alphanate and Koate, respectively), involving body weight (BW; Kg), height (HT; cm), age (y), body mass index (BMI; kg/m2) and FFM (Kg). The histograms in Figure 1 show the overlap in their distributions within the Fanhdi/Alphanate and Koate datasets. Inclusion of the Koate data shifted the median age from 25 to 16 years, while the FFM typical values remained similar (FFM median 50.5 vs 48.4 Kg).
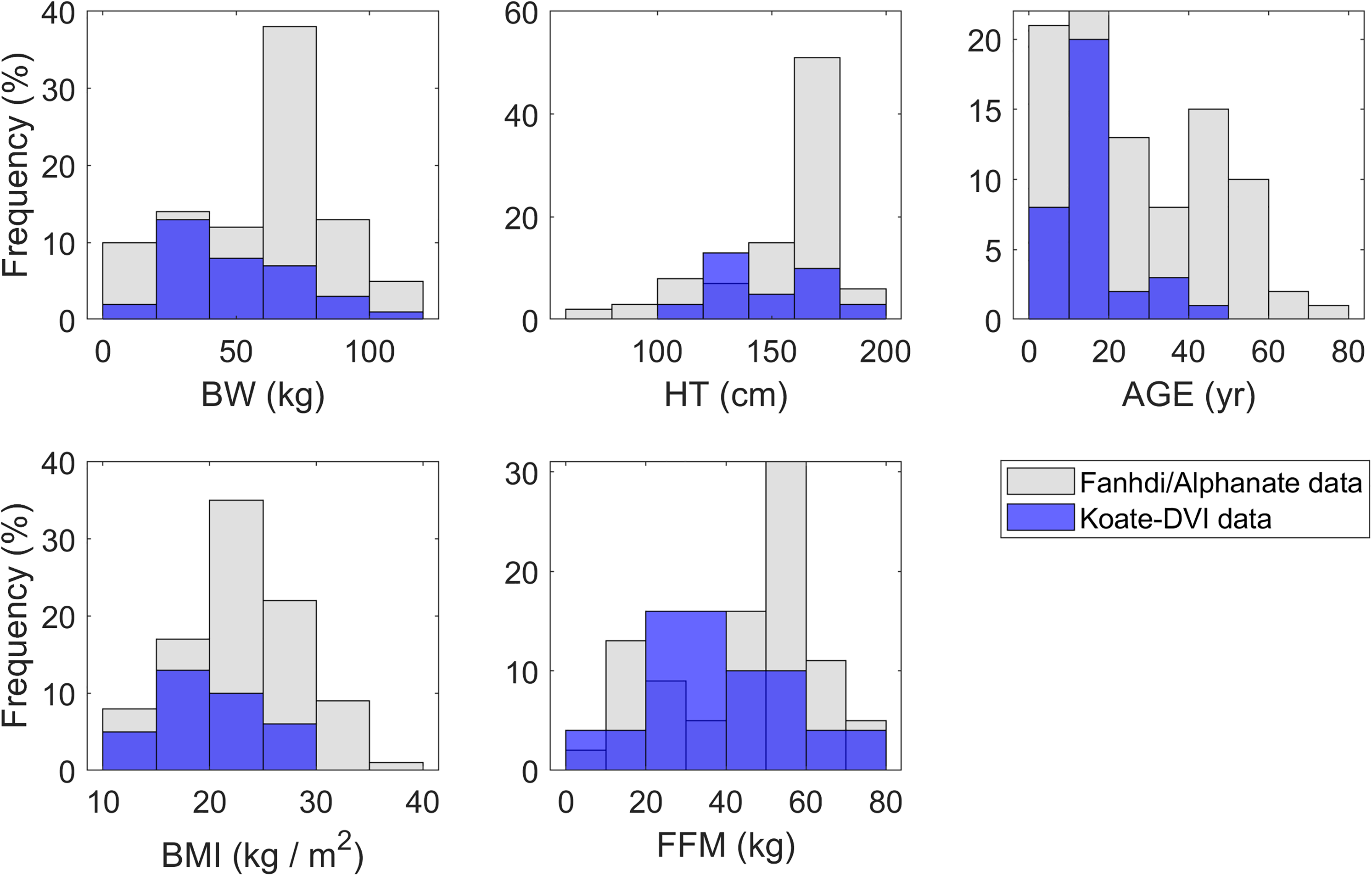
Distributions of covariate values in Fanhdi/Alphanate/Koate model derivation dataset.
Factor activity observations from Fanhdi/Alphanate/Koate data are presented in Figure 2. As the figure shows, Koate measurements tend to cluster below the median of the Fanhdi /Alphanate data at early time points, while most of the later observations lie within the lower and upper percentiles. This does not have a clinically significant implication in terms of the effect of a brand covariate for Koate, as discussed in the next section.

Fanhdi/Alphanate and Koate PK observations versus time after dose, linear and semi-log scales. Red lines: Fanhdi/Alphanate sample median (solid); Fanhdi/Alphanate fifth and 95th sample percentiles (dashed).
Fanhdi/Alphanate/Koate model building
The final Fanhdi/Alphanate/Koate model was an IV infusion 2-compartment model with FFM and age as covariates and was attained after exploring 1-,2-,3-compartment structures and additive, proportional and combined RUV specifications. In cases where the proportional and combined specifications gave similar parameter estimates, the additive component of the latter resulted in negligible values. In other cases, the additive specification had unstable estimates or presented numerical difficulties. The 3-compartment models with the proportional and combined RUV specifications gave negligible parameter estimates for inter-compartmental clearance (Q) and peripheral volume (V2). Consequently, the 2-compartment model with proportional RUV was further explored by inclusion of BSV terms, giving better goodness of fit results when placed in central clearance (CL) and volume of distribution (V1).
Both body weight and FFM resulted highly significant; however, as has been the case for WAPPS-Hemo models, 3 FFM gave a substantially lower objective function value than body weight (log-Likelihood Ratio: 130 vs 93). As a potential source of variability, a brand covariate was included in the model and its association with respect to individual log-CL and log-V1 was explored through an indicator variable (KOATE = 1 for Koate, 0 for Fanhdi/Alphanate) in the form of linear fractional change (1 + θKOATE *KOATE), resulting in statistical significance for V1 (log-Likelihood Ratio: 14.9, P < .001). Koate patients had an estimate for the population central volume of distribution (V1pop) which is 1.32 (95% bootstrap CI: 1.16–1.52) times that of Fanhdi/Alphanate patients. The estimated total volume of distribution (Vpop = V1pop + V2pop) was 45.6 and 57.8 mL/kg for Fanhdi/Alphante and Koate patients, respectively, compared to 48.2 mL/kg for all patients after excluding the brand covariate. Nevertheless, the magnitude of change in the BSV for V1 was deemed not clinically significant (only increased by 2%) implying that this covariate does not explain a substantial portion of the variability on V1. In addition, bootstrap confidence intervals (CI) estimates in Figure 3 show that exclusion of the brand covariate does not result in substantial changes in the distributions of the remaining estimates, especially those representing the variability in V1 (CVωV1).

Bootstrap CIs for Fanhdi/Alphanate 8 and Fanhdi/Alphanate/Koate models with and without brand covariate on V1 (centered at median fat free mass 50.5 kg and age 25 years; Central and inter-compartmental clearance CLpop, Qpop: L/h; Central and peripheral volume of distribution V1pop, V2pop: L).
Table 2 presents estimation results for the Fanhdi/Alphanate/Koate model. Model parameters are: population clearance (CLpop), population central distribution volume (V1pop), inter-compartmental clearance (Qpop), peripheral volume (V2pop), FFM association effect on CL, V1 and V2, an age association effect on CL, BSV on, and correlation between, CL and V1, and a proportional variance RUV term. The model is expressed in the following equation, where 
Model and bootstrap confidence interval estimates for the Fanhdi/Alphanate/Koate model.

RSE: relative standard error; CI: confidence interval, CV: coefficient of variability, Cor: correlation, FFM: fat free mass, RUV: residual unexplained variability. CLpop, V1pop, Q2pop, V2pop: population clearance, central volume of distribution, inter-compartmental clearance and peripheral volume.
Observed versus predicted values under the Fanhdi/Alphanate/Koate model are shown (a) in Figure 4, followed by distribution and scatter plots of CWRES (b) and random effects estimates (c). This model yields satisfactory predictions and the distributional assumptions of normality of the residuals and random effects are reasonably met.
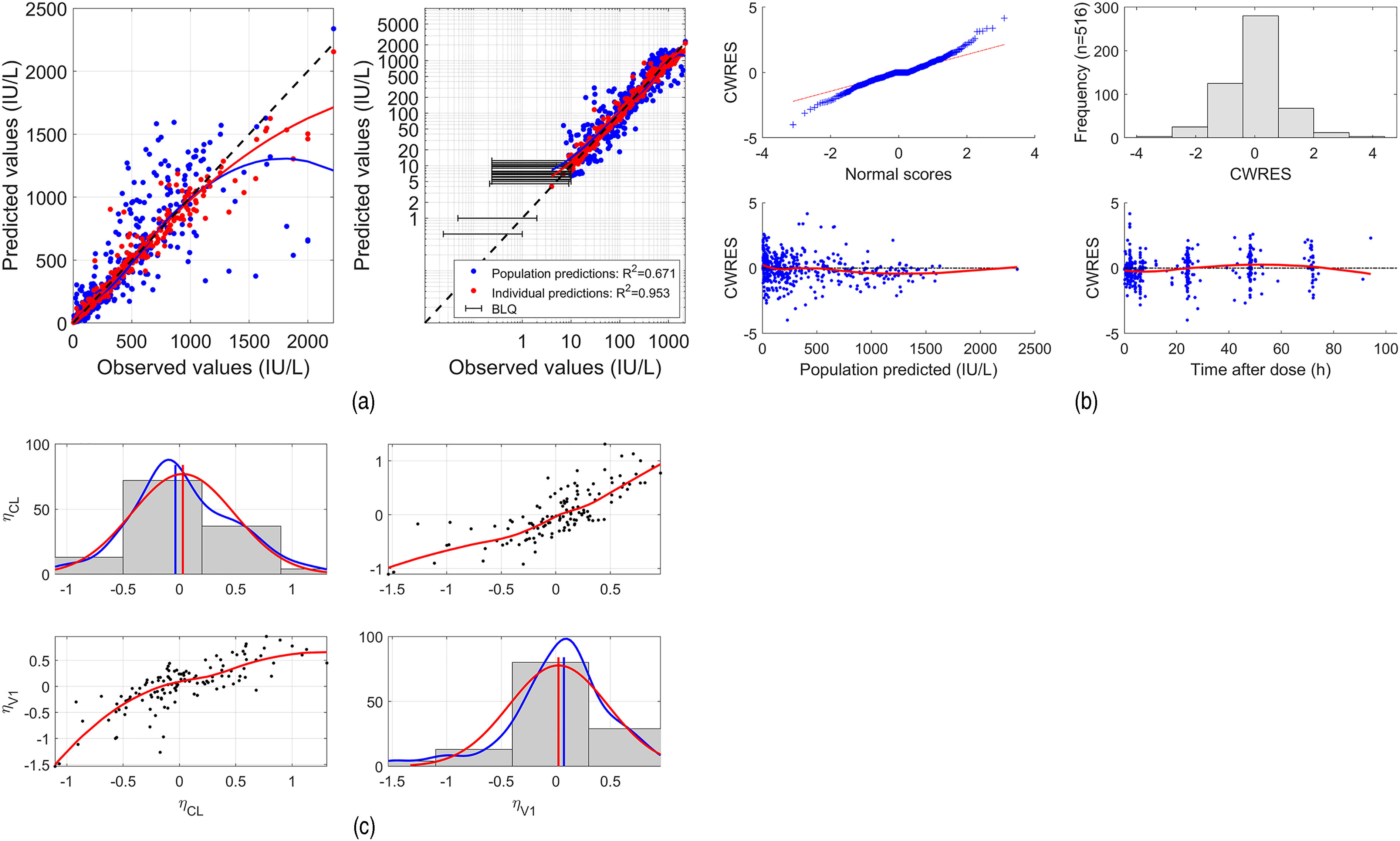
Fanhdi/Alphanate/Koate model goodness of fit assessment. (a): Predicted versus Observed, linear and logarithmic scale; (b): Conditional Weighted Residuals (CWRES); (c) estimated random effects (
Medians and 95th percentiles of the absolute values of the Fanhdi/Alphanate/Koate cross-validation relative errors (%) for estimated parameters are presented in Table 3, showing that medians and 95th percentiles were not higher than 0.66% and 3.43%, respectively. A pcVPC assessment of predictions under the Fanhdi/Alphanate/Koate model is presented in Figure 5, where observations fall reasonably well within the 90% interquartile bands.

Plots for pcVPC. Top: linear scale, bottom: semi-logarithmic scale. Dots:Fanhdi, Alphanate and Koate (prediction-corrected) observations; red dashed lines: 25th, 50th and 95th sample percentiles; solid lines: the corresponding percentiles obtained from 500 simulated prediction-corrected Fanhdi/Alphanate/Koate model-based replicates; colored bands: their 90% interpercentile predictive ranges (simulated BLQ values imputed as medians).
Median [95th percentile] of absolute values (%) of cross-validation relative errors.

TAT2%: time to 2% threshold; CL: clearance; V1: central volume of distribution.
Comparison with Fanhdi/Alphanate model
Comparison between the existing Fanhdi/Alphanate and the new Fanhdi/Alphanate/Koate model is presented based on their bootstrap CIs, cross-validation outcomes and the distribution of their half-life estimates. Bootstrap CIs for both models are quite similar (Figure 3), the latter yielding a substantial lower spread for the FFM effect on V2, indicating an improved estimation stability. This model also yields cross-validation RE with a lower median and 95th percentile (in Table 3) and slightly higher typical half-life estimates (
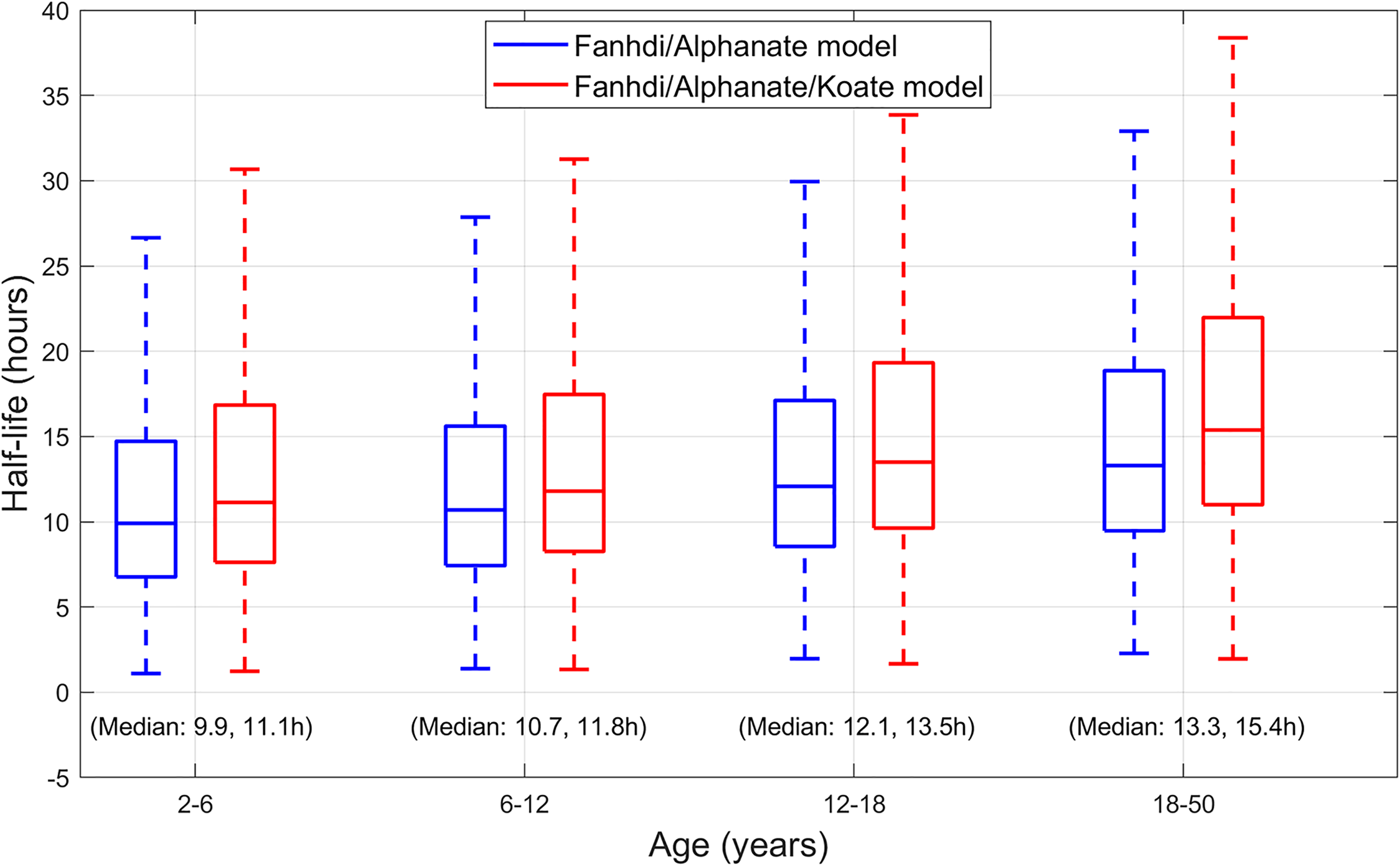
Simulated half-life estimates (
Dose dependency of predictions under the Fanhdi/Alphanate/Koate model
Boxplots in Figure 7 show LSA results of the RE distributions under the Fanhdi/Alphanate/Koate model and Table 4 shows a summary of absolute bias and precision (IQR) for half-life and TAT2%. Designs that included pre-dose had an overall 10% RE spread (whiskers in boxplots) and a 2% to 3% IQR reduction. Precision remained virtually unchanged between 5- and 2-sample designs, regardless of dose and pre-dose inclusion (maximum IQR reduction: 1.8%). Low-dose predictions resulted in a higher accuracy and slightly lower precision of half-life and TAT2% estimates than normal dose (eg, median absolute bias for half-life: 0.12 vs 0.5%; median IQR: 11.79% vs 9.95%).

Relative error (%) distributions of Limited Sampling Analysis results for Fanhdi/Alphanate/Koate model: half-life (
Absolute bias and interquartile range (IQR) of relative error distributions (%), by dose and design for LSA half-life (

Discussion
Motivated by requests within WAPPS-Hemo, a popPK model has been developed for use under treatment with Koate, resulting in a diversification of popPK model usage. Low-dose FVIII tertiary prophylaxis using Koate has been shown to be effective in treating children with hemophilia, 16 and the model will support informed low-dose prophylaxis options of vWF containing FVIII concentrates in low-resource settings, especially beneficial for developing countries. The developed Fanhdi/Alphanate/Koate model was not significantly different from the existing WAPPS-Hemo Fanhdi/Alphanate model, in structure or parametrization, and also had satisfactory outcomes at the validation stage. Relative errors obtained under the hold-out cross validation of the Fanhdi/Alphanate/Koate model gave values not greater than 3.43% and the pcVPC assessment provided a satisfactory representation of the data. This model can be used in the WAPPS-Hemo platform to produce Bayesian forecasting for individualized treatment.
The study demonstrates that Bayesian forecasting is possible for a brand with limited pharmacokinetic data when leveraged by a derivation dataset employed for a similar popPK model, thus increasing access to dose optimization. A study by McEneny-King et al. 17 demonstrated that a generic FVIII popPK model with FVIII-technology as covariate (B-Domain Deleted, Recombinant, Plasma-derived) could well describe the pharmacokinetics of brands not used for model development. This is primarily a result of standard-acting FVIII models being similar in structure and parametrization. 3 Considering this, the conclusion of the study may be applied to other plasma derived FVIII SHL concentrates. The limited sample size of the Koate data and its representativity within the Fanhdi/Alphanate/Koate dataset and model will most likely be overcome in time by future Koate use and data collection within the WAPPS-Hemo platform, which will allow for re-assessment and calibration of the model. Information on blood group was not available for every patient it since was not set as a mandatory input in the WAPPS-Hemo platform. Hence, the study of its effect on the PK behaviour of this plasma derived FVIII concentrate containing vWF was not performed; however, it remains of interest upon data availability.
In developing countries, prophylaxis even in low doses is not recommended as a standard treatment, however, is used as daily practice or research. Chozie et al. 7 evaluated pharmacokinetics of severe hemophilia A patients receiving low dose (10 IU/Kg, twice weekly), through WAPPS-Hemo and the Dr Cipto Mangunkusumo Hospital in Indonesia. Evaluation of efficacy and safety of low-dose FVIII tertiary prophylaxis showed a reduced frequency of joint bleeding and improved musculoskeletal function. 16 This result, in conjunction with individual clinical interpretation of pharmacokinetic profiles, will be recommended to encourage prophylaxis in Indonesia. The World Federation of Hemophilia Guidelines for the Management of Hemophilia, published in 2020, states that all forms of prophylaxis, including low dose, provide superior benefits over episodic treatment. 18
The Fanhdi/Alphanate/Koate model can be implemented for Bayesian forecasting on the WAPPS-Hemo platform with dosing regimens of 10 and 50 IU/Kg, and was evaluated based on the hypothesis that an increased presence of BLQs, more likely to be observed in low-dose scenarios, will reduce pharmacokinetic prediction accuracy. For instance, in the dense dataset that was generated in LSA for reference, the low versus normal dose scenarios had 16% versus 4% BLQ observations and 20% versus 4% predicted BLQ. The hypothesis was false from a clinical perspective as the Fanhdi/Alphanate/Koate model provided a comparable prediction capability for low and normal dosing regimens. Of course, the burden of BLQs will increase with lower doses thus progressively affecting prediction accuracy; however, at the low-dose tested, this burden was not reached. LSA results on RE for half-life (
An ethical implication of this study is that the use of small datasets may enable the incorporation of brands into the WAPPS-Hemo platform, which may in turn aid in dose-optimization for a wider number of brands and patients. This low-dose effectiveness consideration has motivated a survey of dosing guidance from FVIII monographs in Europe, Canada and the United States, which showed that the lowest bound was 15 IU/kg, below which doses were administered in approximately 4% of WAPPS-Hemo infusions from these countries between January 2017 and December 2019. This prompts an interest to pursue an investigation on low-dose treatments under other brands. The outcome of this assessment is that the Fanhdi/Alphanate/Koate model may be implemented on the WAPPS-Hemo platform for low or normal doses.
Footnotes
Acknowledgements
This study was supported by a research service agreement between the University of Waterloo, McMaster University and Grifols SA. Funds were used to partly support investigator meetings and data analysis. The study protocol was prepared by the authors, who are solely responsible for the analysis of data and interpretation of results.
DECLARATION OF CONFLICTING INTERESTS
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FP and NC are study investigators and received funding from the manufacturer of Koate® (Grifols SA). RM is employee of Grifols. AI's Institution has received project-based funding via research or service agreements with Bayer, CSL, Grifols, NovoNordisk, Octapharma, Pfizer, Roche, Sanofi, Sobi, Spark, and Takeda. ANE has received speaking fees from Bayer. All other authors have nothing to declare.
FUNDING
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by a research grant from Grifols SA.
Author Contribution
ANE, FP, NC, RM, AM, and AI made substantial contributions to the study design and data acquisition, DH performed the writing, data analysis and modeling. All authors drafted and critically reviewed the manuscript and approved the final version.
