Abstract
Background:
Within the spectrum of gastroesophageal reflux disease (GERD), pathologic reflux applies to the subset of patients with either erosive esophagitis or abnormal esophageal acid exposure on pH-metry, consequences of a dysfunctional antireflux barrier (ARB).
Methods:
The American Foregut Society (AFS) tasked a 13-member working group of expert foregut surgeons and esophagologists (The ARB Cooperative) to develop a white paper on ARB function, dysfunction, and mechanisms of action of antireflux surgery through discussion of relevant literature summarized herein.
Results:
The ARB Cooperative concluded that pathologic reflux is a consequence of the interplay between progressive anatomical distortion of the ARB and physiology. Factors contributing to ARB dysfunction include (1) separation of crural diaphragm from the lower esophageal sphincter with widening of the hiatus and diminished crural diaphragm sphincteric function; (2) loss of the intra-abdominal lower esophageal sphincter segment with complete disabling of the flap valve component of the ARB; (3) axial hiatal hernia leading to reflux during swallow-induced lower esophageal sphincter (LES) relaxation, LES hypotension, inspiration related reflux, a lowered threshold for eliciting transient LES relaxations, and increased compliance of the gastroesophageal junction leading to greater diameter of sphincter opening during transient LES relaxations. With regard to antireflux surgery, the objectives include: (1) reduction of hiatal hernia and restoration of the intra-abdominal esophageal segment; (2) repair of the dilated hiatus; (3) restoring flap valve function by modifying gastroesophageal anatomy; and (4) restricting gastroesophageal junction opening during periods of relaxation.
Conclusions:
This ARB Cooperative white paper supports the concept of there being 3 major inter-related mechanisms promoting ARB competence: the LES as an intrinsic sphincter, the crural diaphragm as an extrinsic sphincter, and the gastroesophageal valve, a mechanical 1-way valve. Pathological reflux occurs with progressive anatomical disruption of the ARB which in turn leads to physiological dysfunction, the severity of which parallels the extent of anatomical disruption. The corollary of this is that the primary mechanism of antireflux surgery is to restore the ARB by eliminating or compensating for its anatomical disruption. It is the hope of the cooperative that understanding the proposed framework will help clinicians and researchers in improving antireflux procedures.
Keywords
Introduction
Gastroesophageal Reflux Disease (GERD) is a common digestive disorder with millions of afflicted individuals in the US.1,2 However, as currently understood, GERD has a spectrum that includes reflux esophagitis, non-erosive reflux disease, extra-esophageal GERD, reflux hypersensitivity, and Barrett’s esophagus. 3 Within this wide spectrum, the dominance of a defective antireflux barrier as a primary pathophysiologic determinant differs widely, being greater for entities with mucosal damage and non-erosive reflux disease with quantitatively abnormal esophageal pH-metry. Within the realm of esophagitis, high-grade esophagitis (Los Angeles grade C or D) implies greater antireflux barrier dysfunction than low-grade esophagitis (Los Angeles grade A). 1 In the assessment of esophagitis, it is imperative that endoscopy be interpreted in the context of patients’ symptom profile and history of proton pump inhibitor usage. Within the realm of pH-metry, it is essential that this be conducted without ongoing antisecretory therapy recognizing that the distinction between “normal” and “abnormal” is not dichotomous; the greater the degree of abnormality, the greater the degree of associated antireflux barrier dysfunction. For the purposes of this discussion, these entities (reflux esophagitis and non-erosive disease with quantitatively abnormal pH-metry) will be defined as pathological reflux. To date, there is lack of consensus regarding mechanisms leading to pathologic reflux. Similarly, there is no consensus on the necessary elements of antireflux surgery to correct pathological reflux. Within that context, the American foregut Society (AFS) convened a 13-member working group of esophagologists (n = 6) and foregut surgeons (n = 7) to critically evaluate the relevant literature and formulate a white paper on the dominant mechanisms causing pathologic reflux and on the restoration of an effective antireflux barrier with antireflux surgery. The group met virtually to review and discuss the issues with all authors reviewing each draft. The literature was critically reviewed to confirm support for the group position. This paper summarizes the output from that working group—the AntiReflux Barrier Cooperative.
The Dynamic Antireflux Barrier
The antireflux barrier is a complex entity representing a fascinating interplay between anatomical structure and physiology. Functionally, the antireflux barrier serves to prevent the reflux of gastric fluid into the distal esophagus during recumbence, in the face of abrupt increases in intra-abdominal pressure, during normal respiration, or during swallow-induced lower esophageal sphincter (LES) relaxation. However, at other times it is permissive of venting gas from the stomach or vomiting. Moreover, the antireflux barrier reacts to abrupt increases in intra-abdominal pressure with an equally abrupt contraction. In brief, it is a very dynamic entity.
Historically, 3 major concepts have been proposed that promote antireflux barrier competence: (1) the LES as an intrinsic esophageal sphincter, (2) the crural diaphragm as an extrinsic sphincter, and (3) the gastroesophageal valve wherein the distal half of the LES (which is normally intra-abdominal) enters the saccular stomach at an oblique angle forming the angle of His (Figure 1). 4 Notably, these mechanisms are interdependent and not mutually exclusive. All 3 are important, each with its own functional profile. The LES, with its muscular architecture inclusive of the gastric sling fibers forms a “noose” around the gastroesophageal junction both maintaining gastroesophageal junction closure in the absence of crural diaphragm contraction and maintaining the angle of His when the gastroesophageal complex is in its normal subdiaphragmatic location.5,6 The crural diaphragm with the rapidly reactive properties of skeletal muscle and under independent neural control by phrenic nerve branches contracts to angulate and pinch the proximal part of the LES during inspiration and abdominal straining.7-11 Notably, the inspiratory contractions of the crural diaphragm are maintained during swallow-induced LES relaxation resulting in a pulsatile bolus flow into the stomach and preventing swallow-induced reflux. 12 The gastroesophageal valve is a 1-way valve that opens when intra-esophageal pressure exceeds intragastric pressure as occurs during peristaltic transport and closes when intra-gastric pressure exceeds intra-esophageal pressure as occurs during gastric distention.13-15
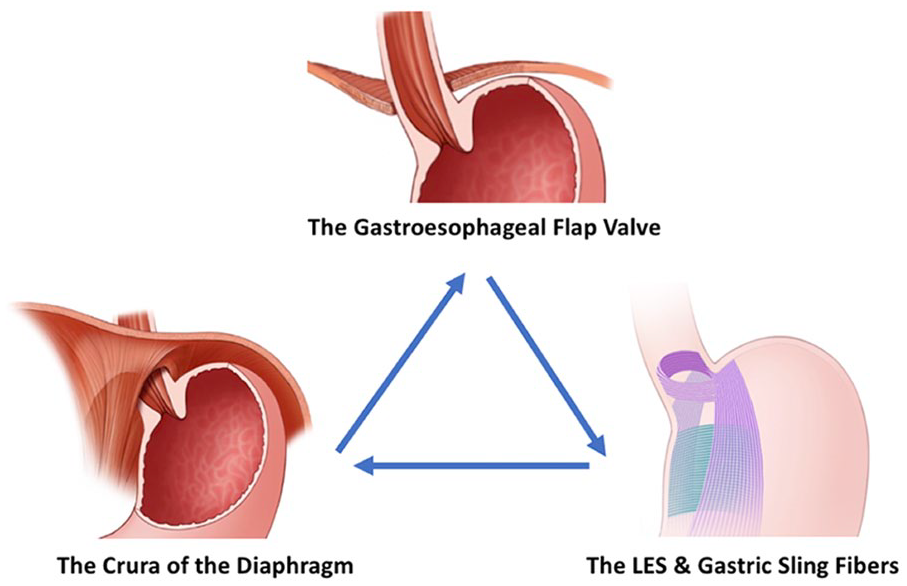
Components of antireflux barrier: the crural diaphragm, the gastroesophageal flap valve, and lower esophageal sphincter (LES) including the gastric sling fibers. Note that the 3 components of the antireflux barrier are interdependent, each dominating antireflux barrier function under specific physiological challenges. The LES maintains the angle of His with its unique myoarchitecture and maintains closure in the absence or crural diaphragm contractions. The crural diaphragm is rapidly reactive skeletal muscle and pinches off the distal esophagus during acute increases in intra-abdominal pressure. The gastroesophageal valve prevents antireflux barrier opening with increased intragastric pressure.
As alluded to above, the antireflux barrier is dynamic; in addition to preventing fluid reflux, it is permissive of venting gas from the stomach, that is, gastric belching. Belching occurs by a mechanosensitive vago-vagal reflex, initially termed as a transient LES relaxation, which not only disables the antireflux barrier, but facilitates the occurrence of gastroesophageal reflux with only a minimal pressure gradient (2-4 mmHg) between the stomach and distal esophagus.16-19 However, because of the minimal intraluminal pressure, transient LES relaxations are normally associated with minimal gastroesophageal junction opening which is ultimately governed by the compliance of the esophageal wall. This effectively restricts the flow of fluid while facilitating the venting of gas from the stomach because the flow rate into the esophagus is proportional to the luminal opening diameter of the gastroesophageal junction (to the fourth power) and inversely proportional to fluid viscosity, which is 56-fold greater for water than air.20,21 The physiological components of a transient LES relaxation are: (1) complete and prolonged LES relaxation, (2) complete and prolonged inhibition of the crural diaphragm, and (3) contraction of the longitudinal muscle of the distal esophagus pulling the gastroesophageal junction through the diaphragmatic hiatus into the mediastinum and transforming its anatomical configuration from that of a gastroesophageal valve to that of an inverted funnel.17,22,23 The stimulus for transient LES relaxation is gastric distention which can be experimentally induced by gas insufflation into the stomach.24,25
Anatomical Distortion of the Antireflux Barrier
Dominant epidemiological variables associated with the development of pathological reflux are advancing age and abdominal obesity. 26 Both of these are also associated with an increased prevalence of hiatal hernia, the common understanding of which is that the anatomical gastroesophageal junction (the LES) has migrated from its native location, within and just below the diaphragm, to an abnormal location some distance at or above the crural diaphragm. However, in the context of antireflux barrier function, this is an over-simplification because it ignores both the dynamic nature of the gastroesophageal junction and how each component of antireflux barrier function might be compromised. Consider the anatomical perturbations potentially associated with hiatal hernia: (1) dilatation of the diaphragmatic hiatus; (2) attenuation of the phrenoesophageal ligament thereby misaligning the esophagus and hiatus; (3) loss of the intra-abdominal location of the distal LES; and (4) repositioning the LES from the abdomen (positive inspiratory pressure) into the mediastinum (negative inspiratory pressure). Some of the physiological consequences of these perturbations are: (1) diminished sphincteric function of the crural diaphragm during inspiration and abdominal straining; (2) loss of the crural diaphragm’s ability to prevent reflux during swallow-induced LES relaxation, most evident during recumbency; (3) complete disabling of the valve mechanism which requires that the gastroesophageal valve to exist in an intra-abdominal pressure environment; (4) weakening of the intrinsic LES which is now challenged rather than bolstered by the pressure changes during the respiratory cycle; (5) increased compliance of gastroesophageal junction during transient LES relaxations leading to greater dimensions of LES opening and loss of its ability to restrict the associated reflux to gas as opposed to both gas and liquid; (6) allowing the gastric contents below diaphragm to move into the herniated portion of stomach above the diaphragm during the expiratory phase of respiratory cycle (relaxed crural diaphragm and lower pressure in the herniated stomach within the mediastinum) and then allowing the contents within the herniated stomach into the esophagus during inspiratory phase of respiratory cycle (contracted crural diaphragm and hypotensive LES), thus making hiatus hernia a 2-stage pump for gastroesophageal reflux; and (7) a reduced threshold for eliciting transient LES relaxations.20,24,27-29 These mechanisms are summarized in Figure 2.
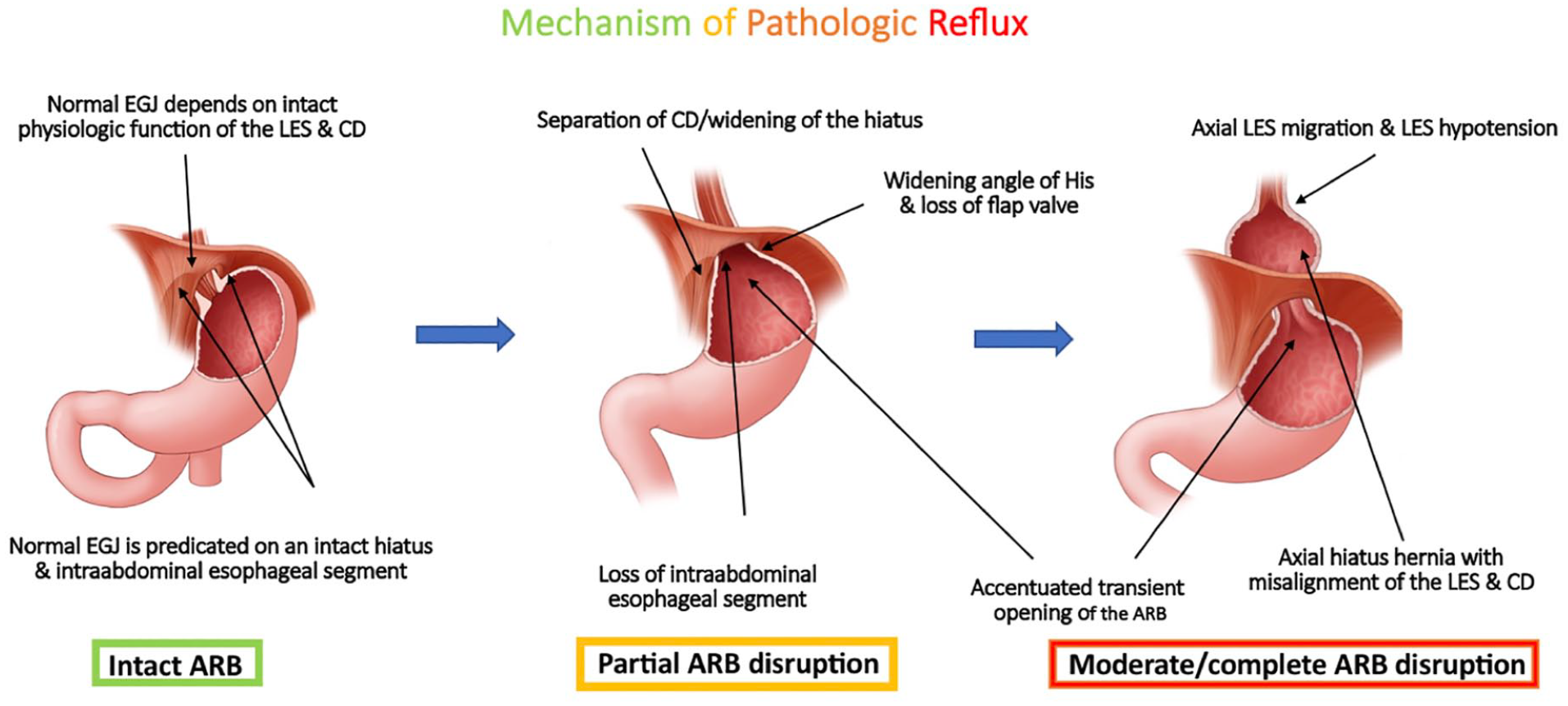
AFS consensus on the mechanism of pathologic reflux. The primary determinant of pathologic reflux is attributable to anatomic and physiologic defects of the esophagogastric junction. Along with antireflux barrier (ARB) disruption comes physiological dysfunction due to of loss of the intra-abdominal segment of the lower esophageal sphincter (LES), progressive degrees of hiatal dilatation, development of hiatal hernia, and LES-crural diaphragm (CD) separation.
Implied in the above, disruption of the antireflux barrier exists along a spectrum of severity, much of which is reflected in anatomical distortion of the native esophagogastric junction (EGJ). A recent initiative by the AFS aimed to stratify the degree of EGJ disruption based on a novel endoscopic assessment of EGJ integrity (Figure 3). 30 The AFS Hiatus Grade differs from prior such assessments, most notably the Hill classification, in several ways: (1) it specifies that the endoscopic technique should employ substantial intragastric insufflation and an endoscopic maneuver of applying gentle traction from the lesser curve side during retroflexion to elicit a hiatal hernia that may not be otherwise evident, (2) it characterizes EGJ integrity in 3 domains, the length of axial herniation in cm (L), the diameter of the diaphragmatic hiatus, gauged by comparison to the diameter of the endoscope (approximated as 1 scope equals 1 cm, D) and the presence (+) or absence (−) of the gastroesophageal flap valve (FV), and (3) it grades the hiatus on a scale of 1 through 4 based on its most disrupted element, be that FV—making it a Grade II (partial disruption), either L > 2 cm or D > 3 cm making it a Grade IV (complete disruption), or with intermediate findings thereby constituting Grade III (moderate disruption). Only AFS Hiatus Grade I is intact (L0, D1, FV+).
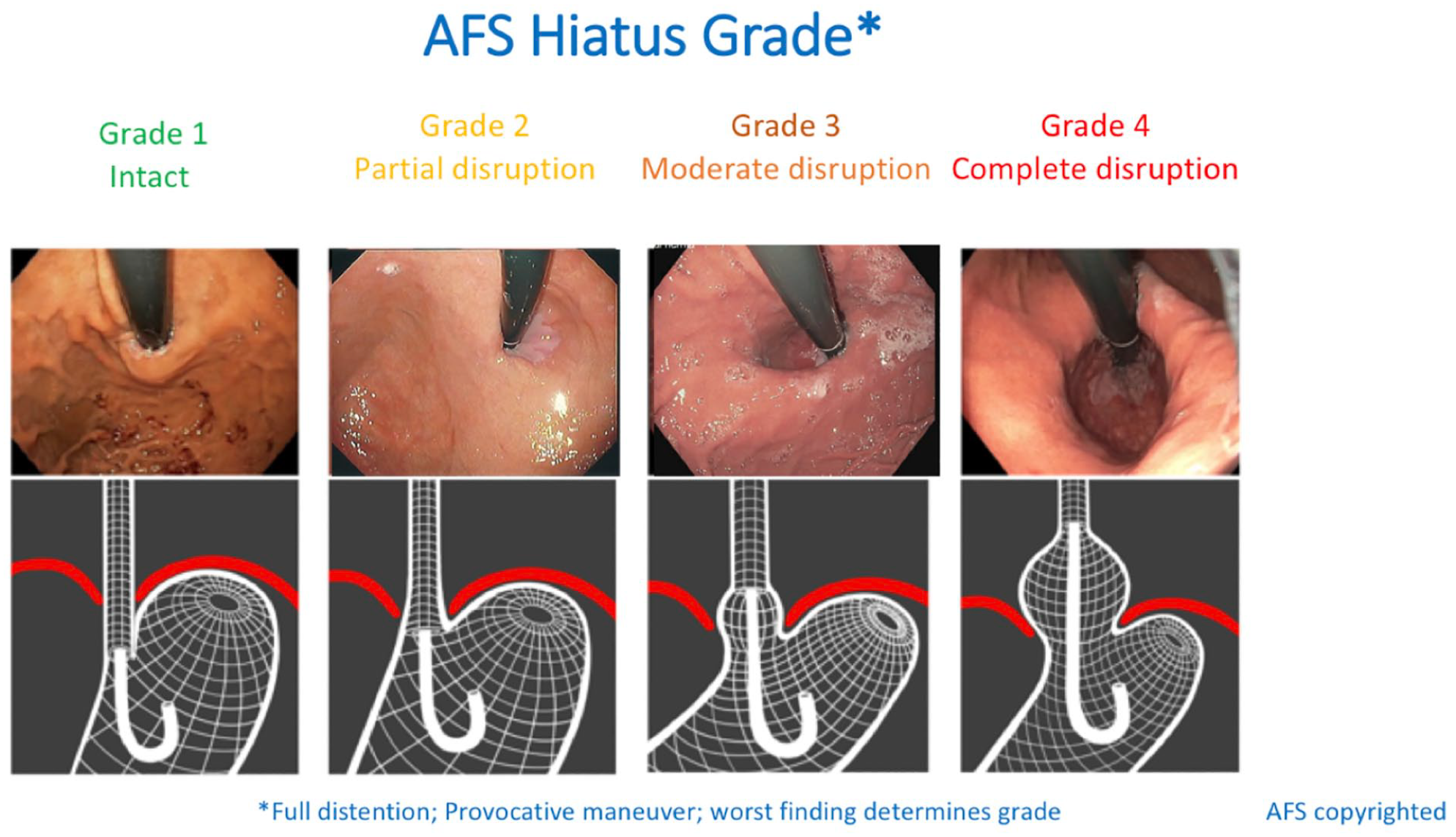
The American Foregut Society (AFS) Hiatus Grade.
Mechanism for Pathologic Reflux and Antireflux Surgery
Selected consensus publications addressing proposed pathophysiology of GERD are summarized in Table 1.31-36 Each of these identifies EGJ dysfunction as a key mechanism and most identify anatomic factors and/or hiatal hernia as being a key component of this. Numerous potentiating factors have been identified: abdominal obesity with an increased abdominal-thoracic pressure gradient, poor esophageal clearance, delayed gastric emptying, impaired or altered esophageal mucosal integrity, and esophageal hypersensitivity. However, in trying to encompass the multidimensionality of the “GERD” diagnosis, these publications focus more on the secondary consequences of antireflux barrier dysfunction and factors exacerbating it rather than on its root cause. Our Antireflux Barrier Cooperative narrowed the task by focusing only on well-defined pathologic reflux which can only occur with antireflux barrier dysfunction. We concluded that the severity of antireflux barrier disruption parallels the AFS Hiatus Grade classification.30,37 Research is ongoing to ascertain the accuracy of this novel classification as a predictor of pathologic reflux.
Selected Societal Consensus Publications on GERD Pathophysiology.

Note. TLESR = transient lower esophageal sphincter relation; LES = lower esophageal sphincter; EGJ = esophagogastric junction.
Along with antireflux barrier disruption comes physiological dysfunction due to of loss of the intra-abdominal segment of the LES, progressive degrees of hiatal dilatation, development of hiatus hernia, and LES—crural diaphragm separation as summarized in Figure 2. The corollary of this is that the primary mechanism of antireflux surgery is to restore and augment the antireflux barrier by eliminating these functional distortions, be that with conventional antireflux surgery as illustrated in Figure 4a or with newer procedures such as the magnetic sphincter augmentation procedure combined with hiatal hernia repair, or the combined transoral fundoplication which adds hiatal hernia repair to the transoral (incisionless) fundoplication.38-41 Notably, a major driving force for innovation in the realm of antireflux surgery is seeking to restore and augment the ARB while preserving the ability to belch, loss of which (along with associated bloating) is a common consequence of conventional antireflux surgery. Future research should focus on calibrating the repair to create the optimal gastroesophageal flap valve to restore the antireflux barrier while maintaining the ability to belch.
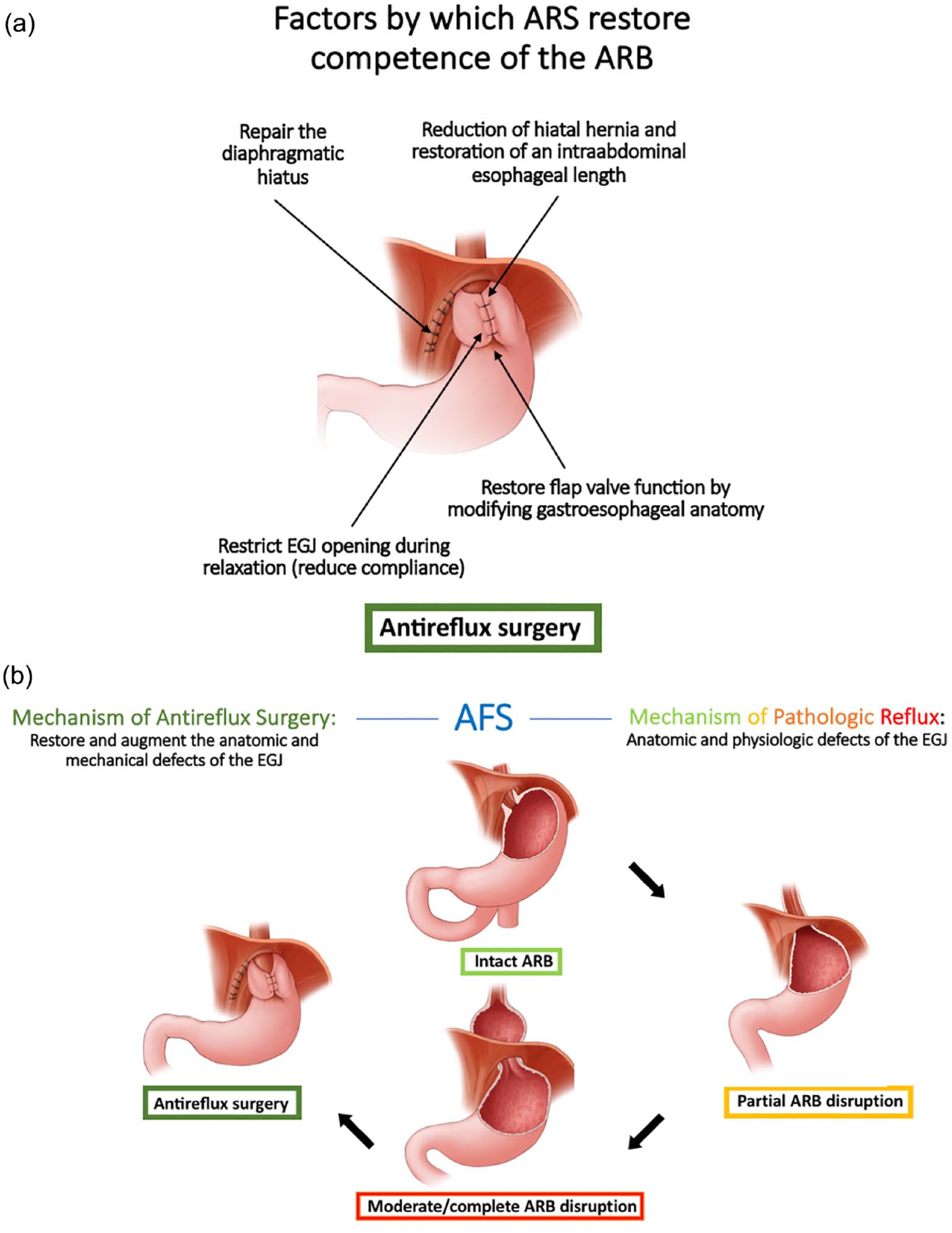
(a) AFS consensus on the mechanism of antireflux surgery (ARS) which is to restore the competence of the antireflux barrier (ARB) and (b) summary of the mechanism of pathologic reflux and mechanism of ARS.
There have been many mechanisms of action proposed for antireflux surgery that are summarized in Table 2.13,42-56 The majority of these revolve around augmenting the LES in some way: increasing LES pressure and/or length, creation of a flutter valve mechanism, mechanical augmentation of the LES or reducing transient LES relaxations. Other proposed mechanisms include the mechanical effect of the wrap, restoring crural diaphragm function, and restoration of the gastroesophageal flap valve.42-56 Based on the expert group discussions, the Antireflux Barrier Cooperative concluded that the objectives of antireflux surgery are to restore and augment the anatomic and mechanical defects of the antireflux barrier and that antireflux surgery can be tailored to the individual with an understanding that all 3 antireflux barrier components are integrally related and work as a unit (Figure 4b). Optimal intra-abdominal esophageal length should be 2-3 cm with an understanding that, generally speaking, more is better.56,57 Hence, extensive esophageal mobilization should be emphasized. Second, crural diaphragm repair should result in the esophagus being snug against the crural diaphragm which will reduce the compliance of the gastroesophageal junction and reduce the diameter to which it opens during relaxation. In a study quantifying factors essential to the integrity of the EGJ during antireflux surgery, Stefanova et al apply the Endoflip technology at baseline, after crural repair, and after fundoplication and found that diaphragmatic crural repair has a greater effect on EGJ compliance than sphincter augmentation suggesting that antireflux procedures should address both for optimization of EGJ physiology. 55 Additionally, failure of the crural repair is the dominant mechanism for reflux recurrence after antireflux surgery. 58 Finally, it is important to modify gastroesophageal anatomy in some way to simulate the function of the native flap valve; this can be done by approximating the gastric cardia and fundus around the distal esophagus as exemplified by a fundoplication (Figure 5).
Selected Publications Detailing the Mechanisms of Action of Antireflux Surgery.

Note. TLESR = transient lower esophageal sphincter relation; GEFV = gastroesophageal flap valve; LES = lower esophageal sphincter; EGJ = esophagogastric junction; SAGES = Society of American Gastrointestinal and Endoscopic Surgeons.
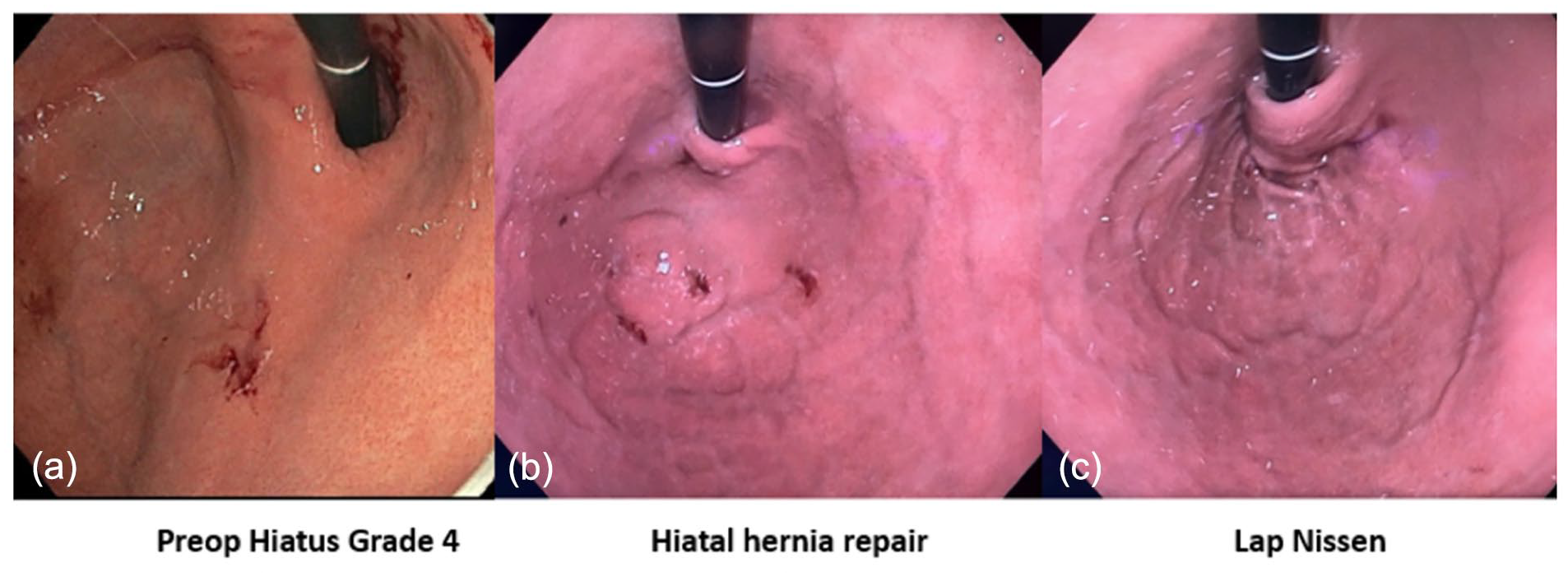
Endoscopic retroflex view of (a) the gastroesophageal junction (GEJ) with >2 cm axial hiatal herniation, classified as an AFS Hiatus Grade 4 (left panel), (b) the GEJ after laparoscopic crural repair with restoration of intraabdominal esophageal length showing a flap valve (middle panel), and (c) the GEJ view after augmentation of the flap valve with a laparoscopic fundoplication (right panel).
The above framework on factors contributing to the antireflux barrier can be applied to mechanistically assess current and future antireflux procedures. For example, transoral incisionless fundoplication was conceived primarily to restore the gastroesophageal valve without addressing crural diaphragm dysfunction. Since its introduction in 2010, clinical practice has transitioned from transoral incisionless fundoplication to primarily combined transoral fundoplication with recognition that hiatal repair and restoration of intra-abdominal esophageal length are often necessary to restore antireflux barrier competence. 39 The combined transoral fundoplication procedure rather than transoral incisionless fundoplication procedure is now a recognized mainstay therapy in the management of patients with pathologic reflux. Similarly, the magnetic sphincter augmentation procedure was originally performed with minimal dissection and without a crura repair. However, clinical studies have since shown improved reflux control in patients with hiatal hernia repair in combination with magnetic sphincter augmentation compared to magnetic sphincter augmentation alone. 39 It is the hope of the Cooperative that the proposed framework will help ensure that all components of antireflux barrier dysfunction are addressed in the development of novel antireflux procedures. As a corollate to this, the above framework on factors contributing to the antireflux barrier can also be used to predict worsening of reflux when one or more of the antireflux barriers are disrupted. Sleeve gastrectomy is an excellent operation for the treatment of morbid obesity. One of the adverse consequences of this operation is development or worsening of reflux symptom. The proposed mechanism for the development of GERD after sleeve gastrectomy is related to the disruption of the LES, its gastric sling fibers and elimination of the gastroesophageal valve by division of the stomach at the level of the angle of His. In a study of 225 patients who underwent sleeve gastrectomy, the DeMeester score increased from 16.7 to 42.9 after sleeve gastrectomy with 79.5% of patients developing de novo GERD. 59
Conclusions
The antireflux barrier is a complex entity exemplifying the interdependence of anatomy and physiology. This Cooperative supports the concept of there being 3 major mechanisms promoting antireflux barrier competence: the LES as an intrinsic sphincter, the crural diaphragm as an extrinsic sphincter, and the gastroesophageal valve wherein the intra-abdominal LES enters the stomach at an oblique angle creating a mechanical 1-way valve. The Cooperative also recognizes the normal physiological function of transient LES relaxation is to completely disable the antireflux barrier in order to facilitate gas venting from the stomach. In essence, this term may be better expressed as transiently disabling or opening of the antireflux barrier rather than just relaxation of the LES. Pathologic reflux in adults is attributable to the progressive anatomic deterioration of the antireflux barrier with loss of the intra-abdominal segment of the LES, enlargement of the diaphragmatic hiatus, and axial herniation of the LES leading to its physiological dysfunction with an excessive number of acid reflux events, greater volumes of fluid reflux (often experienced as regurgitation), and a reduced ability to selectively vent gas from the stomach unaccompanied by gastric juice. We also summarized the mechanisms by which antireflux surgery restores and augment the antireflux barrier, the essential elements being eliminating hiatal hernia, restoring the anatomic integrity of the crural diaphragm, restoring a segment of intra-abdominal LES, and recreating some form of the gastroesophageal valve. Future development of novel endoscopic and surgical therapies in the management of pathologic reflux research should utilize the above framework with the goal of effective restoration of all components of the antireflux barrier.
Footnotes
Authors’ Note
This AFS position paper has been approved by the AFS board.
Author Contributions
Ninh T. Nguyen: study design, acquisition and analysis of data, draft of paper, review of article, revision of article. Barham Abu Dayyeh: study design, acquisition and analysis of data, draft of paper, review of article, revision of article. Kenneth Chang: study design, acquisition and analysis of data, draft of paper, review of article, revision of article. John Lipham: study design, acquisition and analysis of data, draft of paper, review of article, revision of article. Reginald Bell: study design, acquisition and analysis of data, draft of paper, review of article, revision of article. Francis Paul Buckley: study design, acquisition and analysis of data, draft of paper, review of article, revision of article. Christy M. Dunst: study design, acquisition and analysis of data, review of article, revision of article. Ravinder K. Mittal
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Ninh T. Nguyen receives honoraria as a speaker for Endogastric Solutions and Olympus. Kenneth Chang receives honoraria from Apollo, Boston Scientific, C2 Therapeutics, Cook Medical, Covidien, Endogastric Solutions, Erbe, Mederi, Medtronic, Mauna Kea, Olympus, Ovsco, Nine Point, Pentax, and Torax. Barham Abu Dayyeh is a consultant for Endogenex, Endo-TAGSS, Metamodix, and BFKW; consultant and grant/research support from USGI, Spatz Medical, Endogastric Solutions, Boston Scientific, Medtronic; grant/research support from Apollo Endosurgery, Cairn Diagnostics, Aspire Bariatrics; speaker roles with Olympus, Johnson and Johnson. Peter J. Kahrilas is a consultant for Reckitt and speaker for Phathom Pharmaceuticals. Rena Yadlapati has served as a consultant for Medtronic, Ironwood Pharmaceuticals, Phathom Pharmaceuticals, StatLink MD and Medscape. She has received research support from Ironwood Pharmaceuticals and served on advisory boards for RJS Mediagnostix with stocks. Nirav Thosani is a consultant for Boston Scientific Corporation, Pentax America, Advisory board member for Iterative Health and speaker for Abbvie.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This position paper was entirely an AFS initiative with no outside financial support.
Ethical Approval
Ethical approval is not required for this manuscript.
Use of Artificial Intelligence
We did not use AI in the writing of this manuscript.
