Abstract
Endoscopic evaluation after antireflux surgery is a challenge, due to the complexity of anatomy, variations in anatomical repairs, and the various patterns of failure. Studies have demonstrated that endoscopy reports of postoperative examinations are often inaccurate and inconsistent. The key to consistent high quality endoscopic examinations of the integrity of an antireflux surgery is a sound foundation in the native anatomical gastroesophageal junction and an understanding of the anatomy of the various postoperative configurations. This review will clarify the critical details necessary to perform a detailed endoscopic evaluation of the patient with suspected gastroesophageal reflux, and highlight key features to distinguish the intact repair from the dysfunctional one. It first explores the anatomical components of the native gastroesophageal junction and the unique geometric architecture that manifests in the physiologic reflux barrier. Then details the essential structures to evaluate and techniques to perform in an endoscopic examination prior to an antireflux surgery. It then systematically examines the altered anatomy and postoperative changes in endoscopic appearance after Nissen, Toupet, and Dor fundoplication, Collis gastroplasty, magnetic sphincter augmentation, and transoral incisionless fundoplication. Finally, to aid in the endoscopic diagnosis of specific dysfunction, it discusses the various patterns of failure after antireflux surgery, their characteristic endoscopic appearances, and the most useful adjunct testing modalities to augment the endoscopic examination when diagnosis is unclear.
Introduction
The gastroesophageal junction (GEJ) is arguably the most complex anatomic and physiologic segment of the gastrointestinal tract. It functions as the chief barrier to reflux, yet accommodates deglutition and venting. The competency of this barrier predominantly depends on lower esophageal sphincter (LES) function, the crural diaphragm, and the geometric architecture of the GEJ. 1 Reconstruction of this anatomy and restoration of its function are the main goals of all antireflux surgeries. There are several modalities that can aid in the assessment of the GEJ preoperatively, but postoperative anatomy is best assessed endoscopically. However, given the multitude of ways anatomy can be re-engineered or augmented during antireflux surgery, the endoscopic evaluation of postoperative anatomy is proven to be complex. Recognition of postoperative endoscopic anatomy is necessary to understand dysfunction or failure of the surgical repair and is crucial in the assessment of patients with recurrent symptoms. However, studies have shown that the overall accuracy in describing a specific type of fundoplication dysfunction is only 45% and as many as 68% of reports fail to recognize a previous fundoplication on endoscopy altogether.2,3 Therefore, there is a pressing need for additional guidance in postoperative endoscopic examination. This manuscript will review the structural components of the native GEJ, detail the essential components of endoscopic examination after antireflux surgery, and discuss the endoscopic appearance of common postoperative complications and dysfunctions to guide recurrent reflux management and decision-making for revisional surgery.
Native Gastroesophageal Junctional Anatomy
A comprehensive understanding of the anatomy of the intact native reflux barrier provides the necessary foundation to recognize variation after antireflux procedures and dysfunction both before and after surgery. The LES is not an anatomical sphincter, but a functional sphincter, which corresponds to the manometric high pressure zone. 4 The fibers of the outer longitudinal muscle layer of the distal esophagus run parallel with the long axis of the stomach and continue onto the longitudinal axis of the lesser and greater curvatures of the stomach. However, on the anterior and posterior aspects of the stomach, these fibers only continue along the longitudinal axis until they pass the location of the LES, at which point they turn 90° superolaterally toward the fundus.
In contrast to the outer longitudinal esophageal muscle fibers, which are consistent in number along the distal esophagus, LES, and proximal stomach, the inner circular fibers become more numerous, forming an oblique ring of increased fiber aggregation at the LES. Further by progression from cephalad to caudad direction, the fibers of the inner muscle layer change from 360º circular fibers to semicircular muscular loops or “clasps” of 180°. These clasp fibers interdigitate with each other to form a stack of broken rings, which continue along the lesser curvature. The clasp fibers are likely responsible for most of the muscle tone of the intrinsic LES in the resting state. Along the greater curvature, the parallel clasp fibers are replaced by oblique “collar sling” fibers (Figure 1). It is the orientation of the sling fibers which contributes to the asymmetry of pressure at the level of the lower esophageal sphincter, with greater pressure along the greater curvature. These sling fibers maintain the angle of His. The geometric architecture of the clasp and sling fibers has them in asymmetric tonic opposition to one another, with greater tension on the left side, creating an overlap between a portion of the distal esophagus and the gastric cardia, comprising the “flap valve,” visible during endoscopy in a retroflexed view of the GEJ. This geometric architecture and the angle of His are crucial components of the reflux barrier and are best assessed during endoscopy. 4
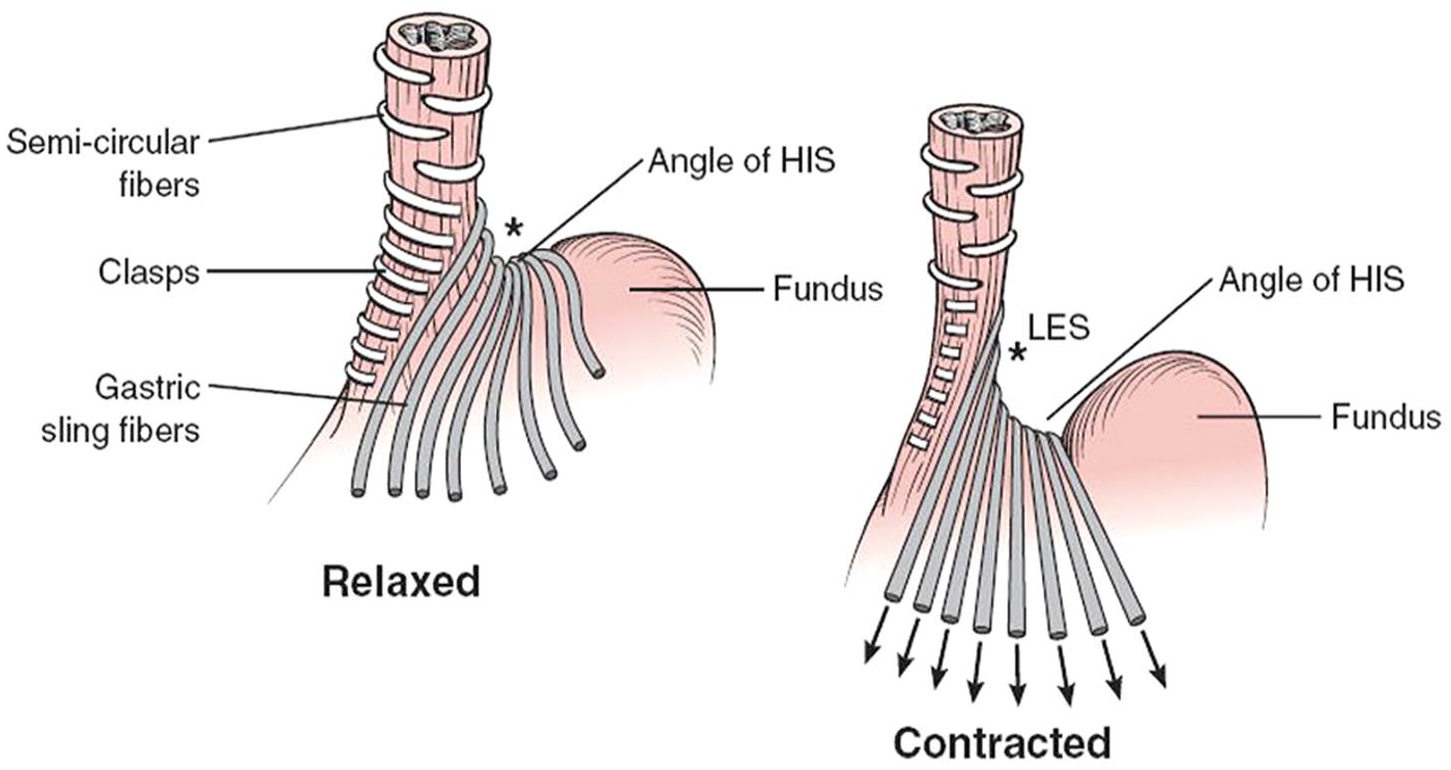
Geometric architecture of the inner muscle layer fibers of the gastroesophageal junction. Note the different orientation of the clasp and sling fibers, the latter of which contract to sharpen the angle of His. The transition from semi-circular fibers to clasp and sling also shows an increase in the number of fibers, corresponding to the location of the LES (*). Reproduced with permission from Fischer et al.
The extrinsic components of the GEJ include the crura and phrenoesophageal ligament. The right crus of the diaphragm consists of superficial and deep components and envelops the esophagus at the GEJ. The superficial component lies to the right of the hiatus, while the deep component lies on the left. The left crus is smaller and also lies to the left of the esophageal hiatus. The phrenoesophageal ligament arises from the inferior diaphragmatic fascia and connects to both the distal esophagus and the gastric cardia, allowing independent movement of the esophagus and diaphragm, as is seen during respiration.
Preoperative Endoscopic Assessment
Guidelines for optimal endoscopic examination technique and documentation are available from several societies, including the American Foregut Society.1,5,6 However, studies have demonstrated that endoscopy reports do not universally include the essential elements of a preoperative endoscopic examination.
7
A complete examination should include a description of the squamocolumnar junction (SCJ) appearance, an identification of mucosal integrity, an evaluation of the GEJ during insertion and from the retroflex view, and assessment of the crural impression.
6
The SCJ is defined as the transition from esophageal mucosa, which is pale in color and contains esophageal palisade vessels, and proximal end of the pink salmon-colored columnar mucosa. The SCJ can be regular, irregular, or when Barrett’s esophagus is present, can be assessed using the Prague classification of the circumferential and maximal extents of columnar lined esophagus. An assessment of mucosal integrity includes the presence and grade of esophagitis using the Los Angeles Classification. LA grade
Upon retroflection, optimal visualization of the GEJ is achieved through insufflation for 30 to 45 seconds until there is flattening but not complete elimination of the gastric rugal folds. Provocative maneuvers can also aid in the assessment of the hiatus; pulling the endoscopic tip along the lesser curvature can induce sliding herniation and open the hiatal aperture so that subtle hiatal hernias can be observed. 1 The endoscopist should also look for a portion of the fundus that is compressed by the crural impression or herniated above the gastroesophageal junction, signifying a paraesophageal hernia (PEH). This can sometimes appear similar to an additional lumen to the side of the GEJ in cases of large PEH. An important step in the retroflex evaluation is classification of the geometric integrity of the GEJ. The Hill grade classification system has been used since 1996 to describe 4 characteristic appearances of the increasingly disrupted geometry of the GEJ. It was established by Hill et al by taking measurements of the pressure required to cause gastroesophageal reflux in a cadaver model both before and after suture reinforcement and lengthening of the geometric flap valve and comparing the results with the endoscopic appearance of patients with and without GERD. 9 Follow up studies have shown an association between grade of the geometric flap valve and the presence of reflux disease and its complications, prompting widespread adoption of the Hill classification. 10
However, the Hill et al classification has been criticized by some investigators as imprecise and subjective. 9 Therefore, the American Foregut Society (AFS) recently described a new classification system to expand on the Hill classification, increase precision, and reduce subjectivity. 1 The AFS classification is similarly scored from 1 to 4, but incorporates the hiatal axial length, hiatal aperture width, and flap valve description in its graded system.
Endoscopic Evaluation of the Anatomically Intact Antireflux Surgery
The first endoscopic evaluation after an anti-reflux surgery should occur in the operating room and after construction of the fundoplication. This vital portion of the procedure is often neglected; however, it provides vital information on the quality and integrity of the repair, need for correction during index surgery and sets a new baseline for comparison in subsequent endoscopic examinations. To transform surgery from art to science, this postoperative examination must utilize a standardized conceptual framework with consistent terminology so that endoscopic reports tell a continuous narrative regardless of who holds the scope.
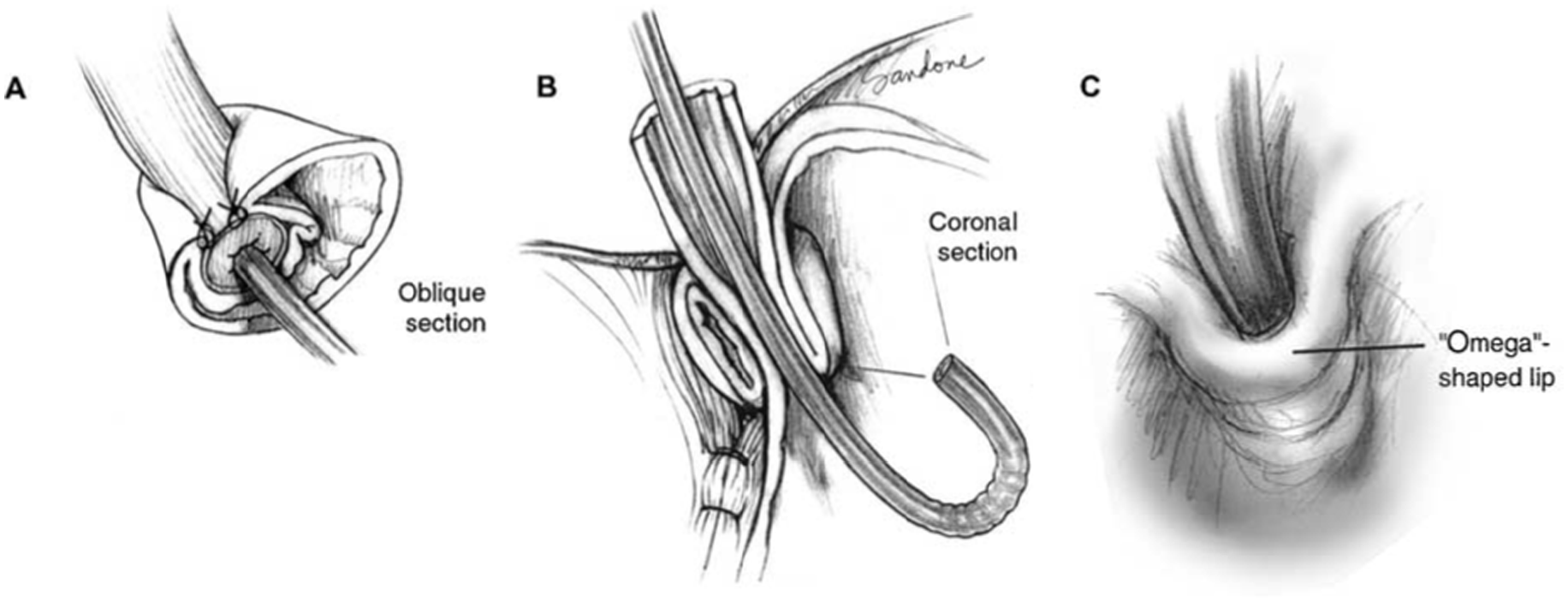
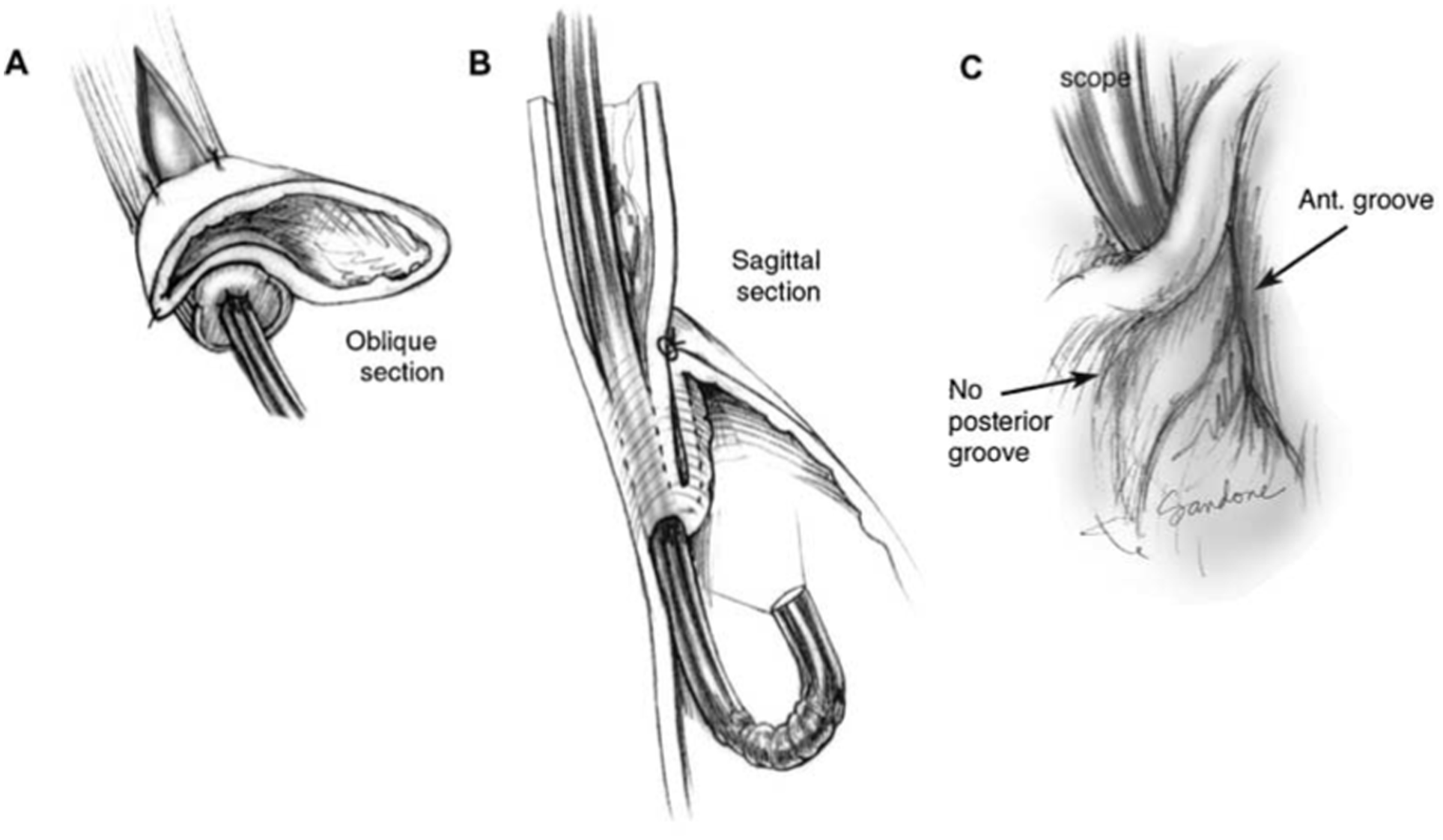
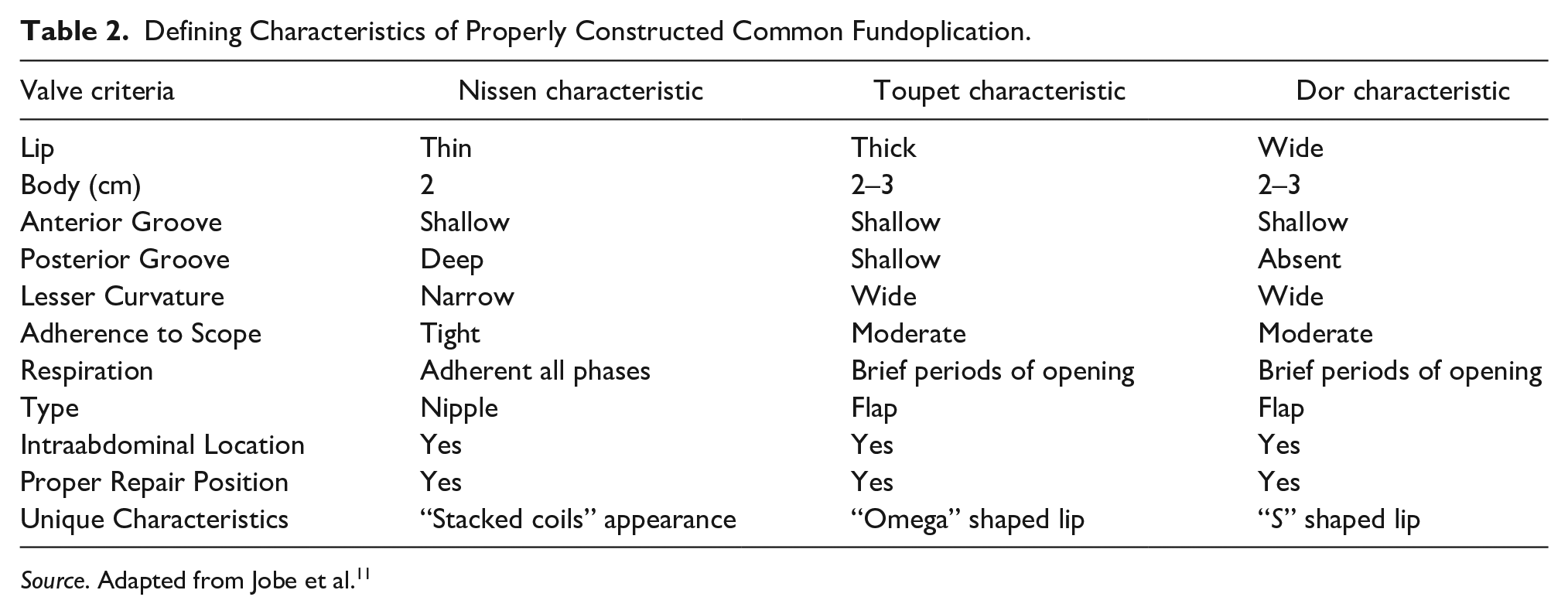
An expert panel of surgeons and gastroenterologists published the first systematic description of the endoscopic appearance of various fundoplication repairs in 2004. The authors intention was to create a standard descriptive lexicon to ensure endoscopic examination quality and reproducibility. 11 The panel reviewed postoperative endoscopic images from asymptomatic patients, defined descriptive terms, and created a consensus examination and reporting system. There were 11 separate criteria used to describe the fundoplication, as shown in Table 1. The entirety of the repair lying distal to the crural impression, with the squamocolumnar junction visible 1 to 3 cm proximal to the lower “lip” of the fundoplication is an indispensable component of an anatomically intact repair. The authors noted that the Nissen fundoplication performs similarly to a native geometric flap valve during gastric distention, compressing from the greater curvature side up against the lesser curvature side. Unlike the native valve, the Nissen does not efface with sufficient pressure, as the distal esophagus is anchored to the fundoplication. The illustration and description of the characteristic appearance of an anatomically intact Nissen fundoplication, Toupet fundoplication, Dor fundoplication, and Nissen fundoplication with Collis gastroplasty are shown in Figures 2 to 5, respectively. The specific criteria used to describe the Nissen, Toupet, and Dor fundoplications are shown in Table 2.
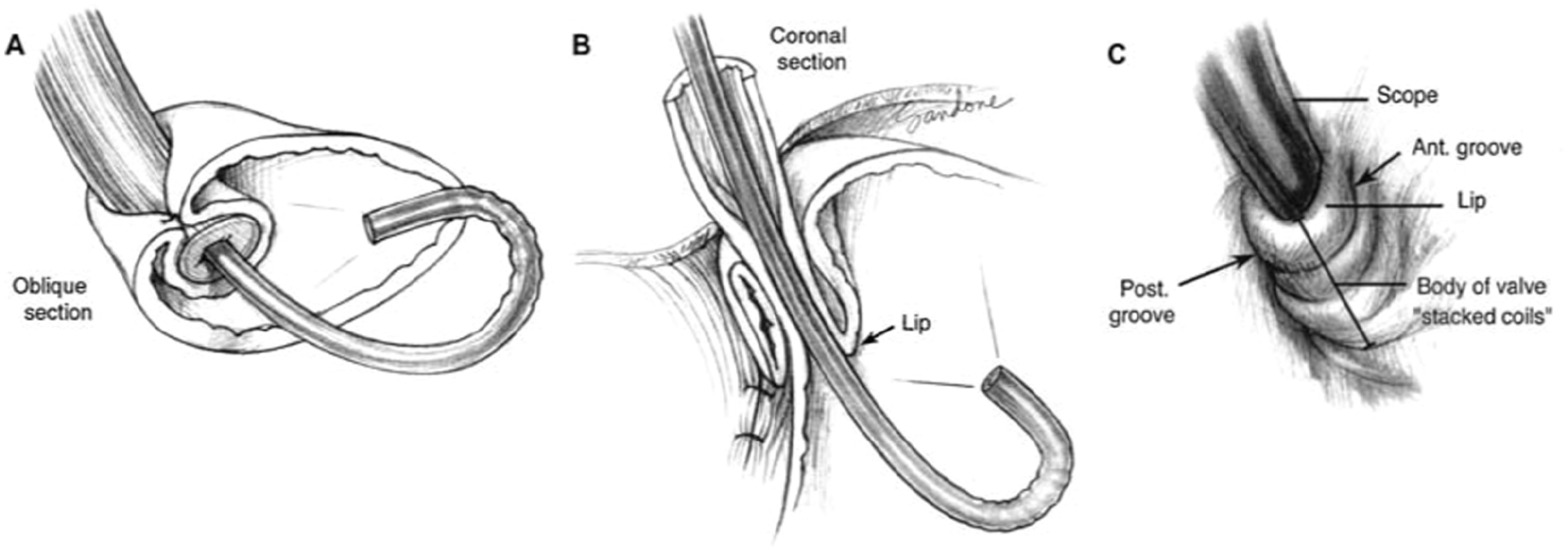
Nissen fundoplication: Oblique (A) and coronal (B) sections and endoscopic retroflex (C) view. The Nissen valve body is characteristically arranged like “stacked coils” in alignment with the long axis of the endoscope, indicating a “floppy” fundoplication around the gastroesophageal junction. The lip should be 2 to 3 cm from fundal apex, thin and tightly adherent to the scope during all phases of respiration. The anterior limb of the fundoplication rests at 11 o’clock and has a relatively shallow groove. The deeper posterior groove is due to the posterior passage of the right limb of the fundus. Reproduced with permission from Jobe et al. Copyright Wolters Kluwer Health, Inc.
Toupet fundoplication: Oblique (A) and coronal (B) sections and endoscopic retroflex (C) view. The 270 degree wrap around the posterior esophagus with an anterior opening creates a characteristic “omega” (Ω) shapes lip. The lip should be 3 cm from fundal apex, thin and tightly adherent to the scope, with no opening during respiration. The anterior and posterior grooves are shallow with a wide lesser curvature due to the placement of the sutures. Reproduced with permission from Jobe et al. Copyright Wolters Kluwer Health, Inc.
Dor fundoplication: Oblique (A) and coronal (B) sections and endoscopic retroflex (C) view. The Dor fundoplication has a characteristic
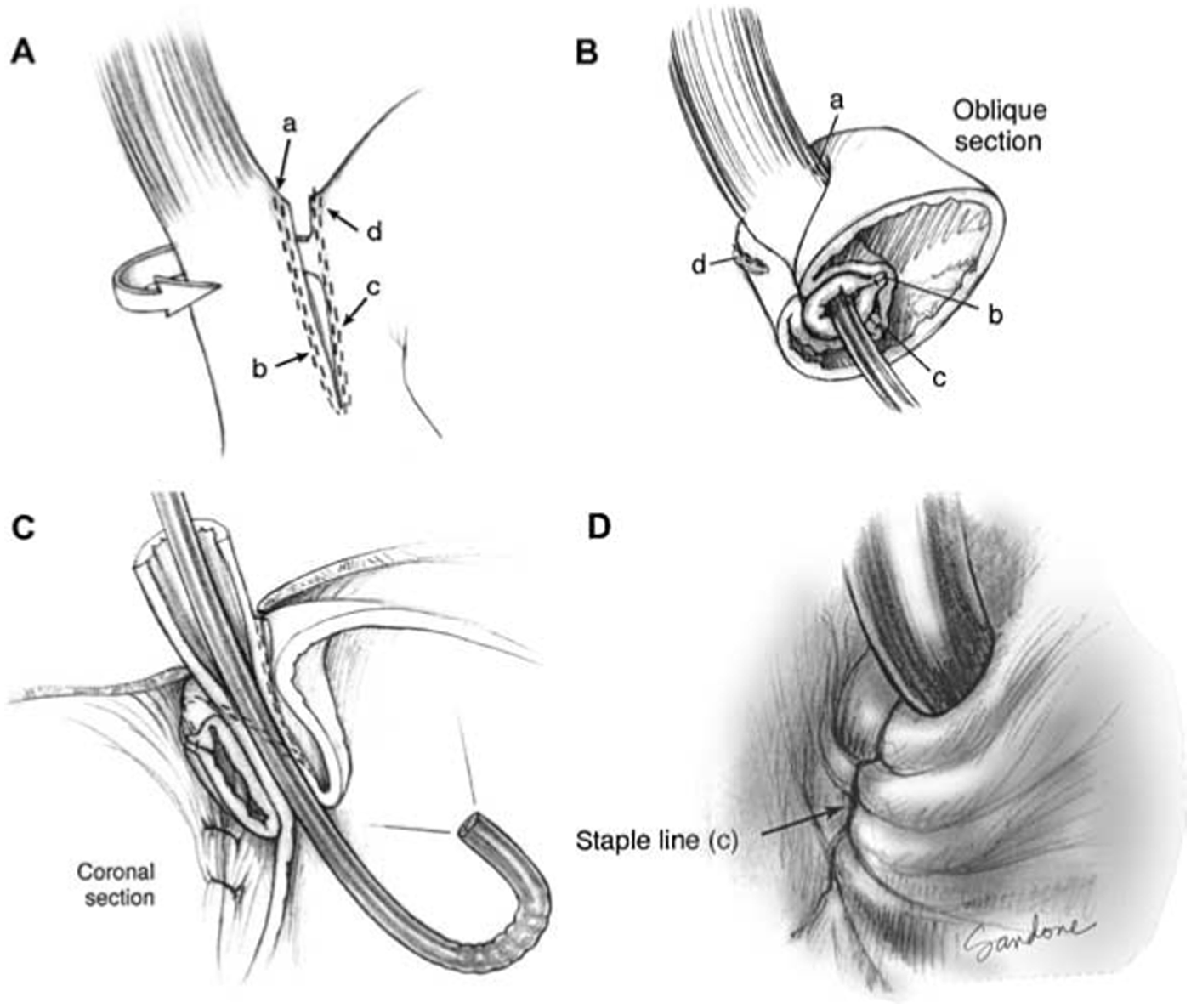
Collis-Nissen Fundoplication: The lower case letters depict the proximal and distal points of the left and right suture lines before (A) and after (B) construction of the fundoplication. Coronal (C) and endoscopic (D) views. Collis gastroplasty creates a characteristic spiral appearance due to the torque of the fundoplication and the rigidity of the staple line, which should be apparent on the body of the valve. Additionally the neoesophagus will be more pliable. The lip should be 3 cm from fundal apex, thick and tightly adherent to the scope, but with periods of opening during inspiration (absent with positive pressure ventilation). The anterior groove will be absent and the posterior groove should be shallow with a wide lesser curvature. Reproduced with permission from Jobe et al. Copyright Wolters Kluwer Health, Inc.
Defining Characteristics of Properly Constructed Common Fundoplication.
Patterns of Fundoplication Failure
There are several patterns of failure, which can be broadly categorized into malposition of the fundoplication and disruption or abnormal fundoplication morphology. Herniated fundoplication is the result of the crural repair failure, where the fundoplication lies above the diaphragm. Endoscopically this will appear as an “hourglass” stomach with a narrowing at the GEJ, a ballooning of the proximal cardia, and a second narrowing at the hiatus. A slipped fundoplication, in which the fundoplication has migrated distally on to the cardia, can also appear as an hourglass. As with the preoperative evaluation, identification of the SCJ, GEJ, and crural impression are essential to differentiate these 2 clinical entities. In a herniated fundoplication, the SCJ, and GEJ will be at or below the fundoplication with a distal crural impression. By contrast, in a slipped fundoplication, the GEJ, and SCJ will be above the pressure zone of the fundoplication with a proximal, and the crural impression at the level of the GEJ. One of the mechanisms of function of the fundoplication is maintaining an intra-abdominal GEJ. This function can succeed, while the hiatal repair fails, resulting in a portion of the stomach herniating into the thoracic cavity while the fundoplication, and GEJ remain intra-abdominal, like a type II hiatal hernia. A retroflex evaluation with adequate insufflation is necessary to evaluate for this type of recurrent paraesophageal hernia.
A fundoplication can be too tight or too loose. A tight fundoplication will provide increased resistance when passing the endoscope. This finding may be due to fibrosis or a twisted fundoplication. Fibrosis may appear otherwise unremarkable on endoscopy. There are 2 types of twisted fundoplication: the spiral valve, where inadequate fundal mobilization was pulled tightly around the esophagus and subsequently unwound, and the 2-compartment stomach, where a point too low on the greater curvature was brought up to the fundoplication, isolating a pocket of fundus from the antrum. 3 A twisted fundoplication can be recognized by asymmetry between the anterior and posterior “tucks” of the fundoplication on retroflection. A loose fundoplication can be partially or completely disrupted. Partial disruption can be recognized by observing that 1 or both tucks are shallow. If the tucks are absent, there is complete disruption of the fundoplication (Figure 6).
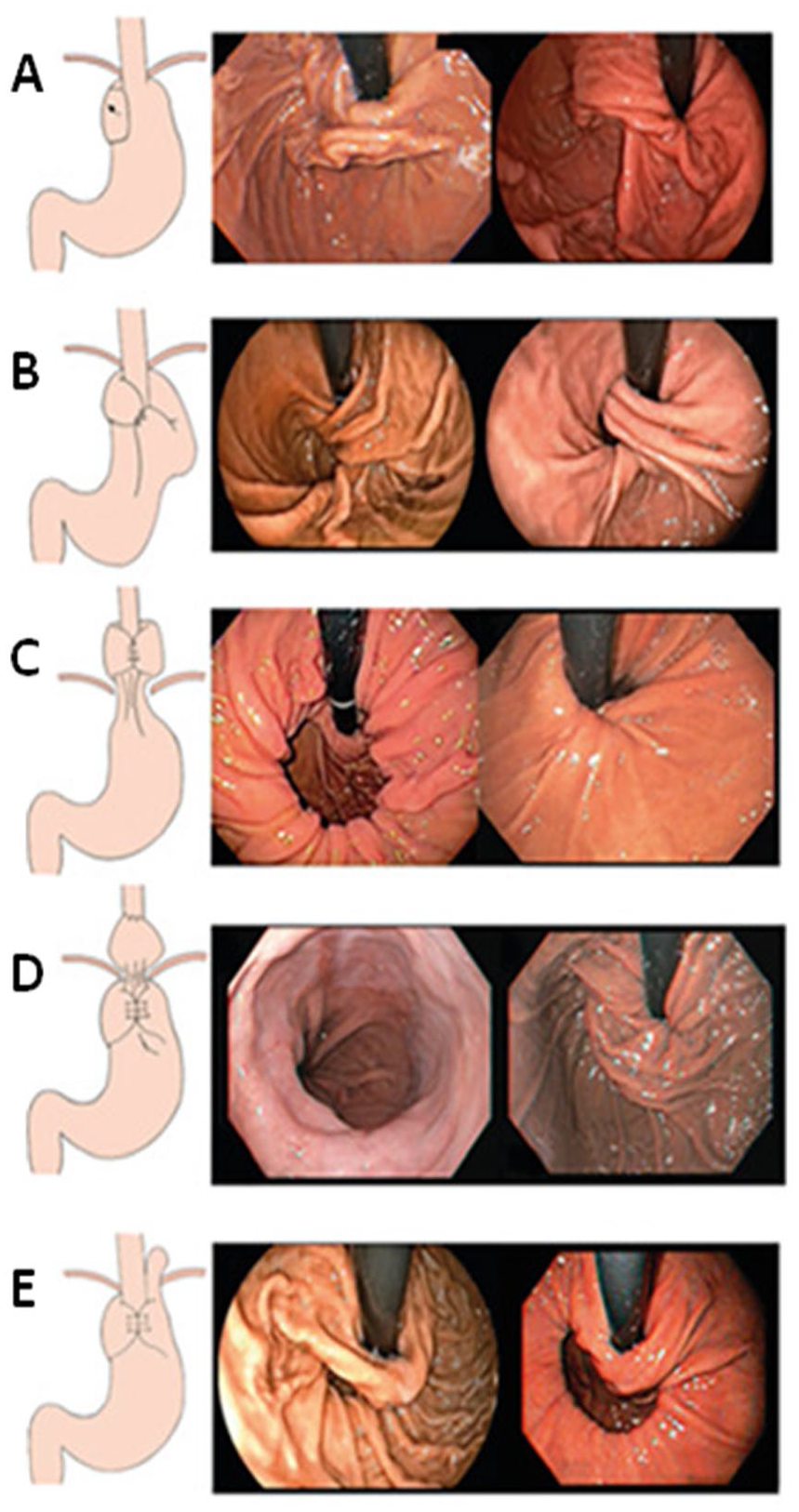
Depiction and endoscopic appearance of characteristic fundoplication failures. (A) Disrupted fundoplication is defined by shallow or loss of 1 or both of the grooves. The transverse gastric fold does not involve the endoscope and may not even be perceptible. (B) Twisted fundoplication has characteristic oblique gastric folds, not parallel with the scope. Additionally there is a craniocaudal displacement. (C) Herniated fundoplication has hourglass shape with proximal GEJ and distal crural impression. (D) Slipped fundoplication has hourglass shape with proximal crural impression and distal GEJ. (E) Paraesophageal hernia with intraabdominal fundoplication. Reproduced with permission from Martins et al. Copyright.
Several studies have attempted to standardize the classification of fundoplication failure by placing them into subcategories. One such classification system is based on different types of hiatal closure and fundoplication failure. Endoscopic findings using this classification system correlated correctly with intraoperative findings in 78% of cases. 12 Additionally, correlation with symptomatology has been inconsistent and external validation has not yet been done. Furthermore, many patients had multiple mechanisms of failure, and were difficult to categorize. 12 Therefore, a detailed description of endoscopic findings with photographs remains the most reliable way to effectively communicate fundoplication dysfunction.
Description of Magnetic Sphincter Augmentation Anatomy
Magnetic sphincter augmentation (MSA) consists of a ring of magnetic beads placed circumferentially around the GEJ. This procedure is standardized with minimal to no technical variations between surgeons. As a result, the endoscopic appearance is relatively straightforward and standardized: an entirely intra-abdominal circumferential impression near the GEJ (Figure 7). The device should allow for sufficient gastric distention to obtain a retroflexed view with partial effacement of the rugal folds. The
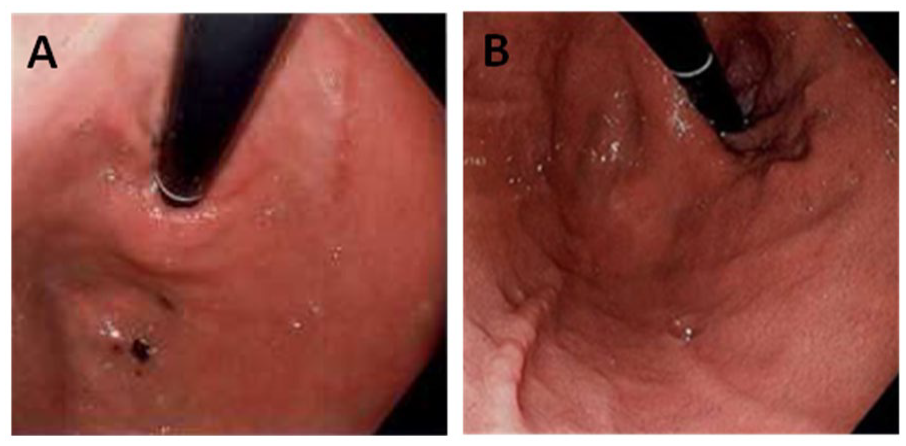
Magnetic sphincter augmentation (A) with characteristic circumferential impression. (B) herniated repair.
Classifying Dysfunction After Magnetic Sphincter Augmentation
The categories of herniation, slipped, and disruption also apply to MSA. Additionally, there is a risk of device erosion in 0.3% to 0.5% of cases.
13
These conditions have characteristic endoscopic presentations. A herniated device may be properly placed in relation to the GEJ, but will be intrathoracic, with the crural impression caudad to the GEJ. A slipped device will show an impression caudad to the GEJ. Erosion is readily apparent on endoscopy. Disruption of the device is more difficult to definitively diagnose on endosopy alone. The endoscopic view of the device may no longer show a complete ring, or there may be no abnormalities visible. Useful adjunctive studies in this situation are esophagram to observe the flow of contrast and the position of the device relative to both the diaphragm and the GEJ. Disruption of the ring is relatively easy to identify on either chest
Our center has previously described patterns of recurrence of hiatal hernia after magnetic sphincter augmentation. 14 Our recommendation for the recurrent hernia with a device correctly positioned at the GEJ is hernia repair without removal. Migration of the device proximally or distally in the setting of a PEH requires removal and either replacement with a new device or conversion to fundoplication. In the setting of a small sliding hiatal hernia with proximal migration of the device, balloon dilation, and a short course of steroids is frequently effective. 14
An endoscopically unremarkable examination in a patient with recurrent symptoms does not rule out failure or preclude the necessity for device removal. The most common indications for device removal are dysphagia and persistence of GERD symptoms, which are present at 2% and 4% of implants, respectively. 15 In absence of evidence of an endoscopic or radiologic anatomical failure, a study from our center suggests that management and decision regarding revisional antireflux surgery should be tailored to the presenting symptom. 16
Transoral Incisionless Fundoplication
The Transoral Incisionless Fundoplication (TIF) is a purely endoscopic antireflux procedure which creates a 300° fundoplication with the shape of the Greek letter Omega. The opening of the Omega shape is located medially along the lesser curvature rather than anteriorly, as in the Toupet fundoplication (Figure 8). This geometry creates opposition between the gastric cardia and the distal esophagus in a similar manner as a normal geometric flap valve. Nguyen et al., 2021 17 due to its novelty there is paucity of data on TIF failures or their endoscopic appearance. More studies are needed to describe in detail how to identify and categorize TIF dysfunctions.
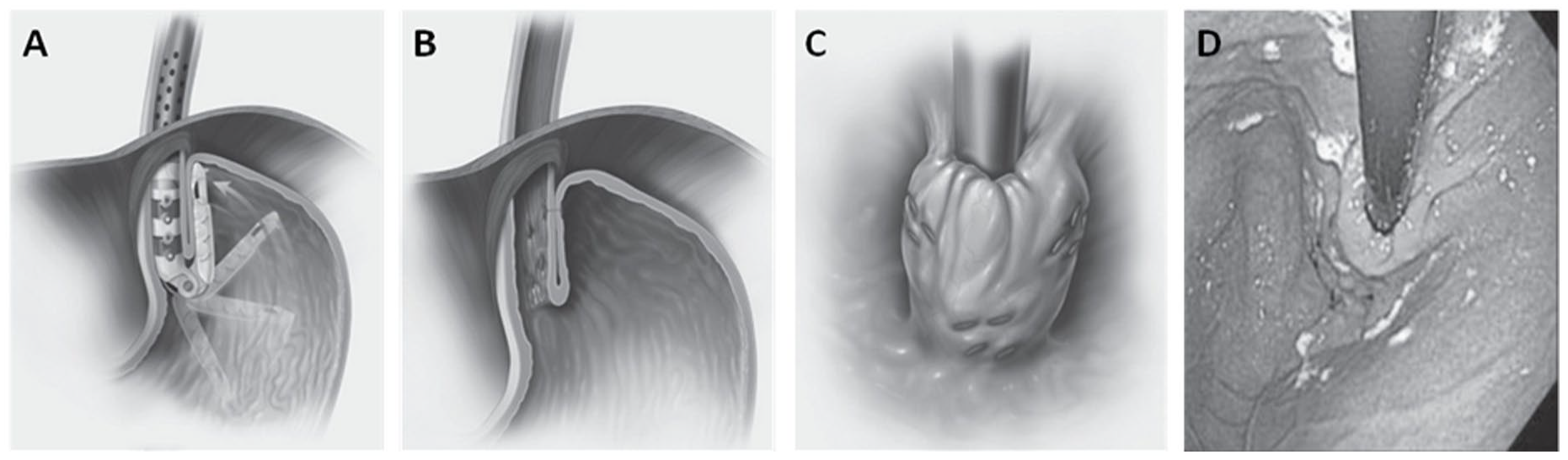
Transoral Incisionless Fundoplication (TIF): Coronal views of plications created with Esophyx device (A and B) and endoscopic retroflex view (C and D). The 270° wrap around the right side of the esophagus creates a characteristic “omega” (Ω) shapes lip. The anterior and posterior grooves are deep. Reproduced with permission from Karim Trad. Copyright.
Adjunct Studies to Assess Fundoplication Dysfunction
Direct endoscopic evaluation may not always clearly reveal dysfunction after antireflux surgery. Fortunately, there are testing modalities that provide additional information to aid the interpretation of endoscopic findings. The functional lumen imaging probe (FLIP) utilizes impedance planimetry to provides physiologic information during endoscopy. 18 The FLIP device measures the pressure and diameter of a balloon at 1 cm intervals as it traverses the GEJ. The smallest diameter divided by the pressure is the distensibility index (DI). A high DI indicates that a fundoplication stretches easily and may raise suspicion of a loose or disrupted fundoplication. Su et al collected FLIP data from 175 patients during laparoscopic fundoplication and hiatal hernia repair and assessed outcomes at 1-year. 19 They found that a DI less than 2 was associated with significantly more gas bloat and dysphagia at 1-year, and a DI between 2 and 3.5 had the lowest incidence of GERD-related symptoms. Turner et al found that a decrease in the minimum diameter of 0.15 mm or less, or a decrease in cross-sectional area of 1.5 mm2 or less was associated with persistent heartburn after fundoplication. 20 Impedance planimetry can also be used to aid the detection of a hiatal hernia. Carlson et al used FLIP to identify the presence of a hiatal hernia and found an 84% agreement with kappa statistic of 0.667 between high resolution manometry and FLIP for presence of hiatal hernia. 21
Another useful technique for fundoplication evaluation is endoscopic ultrasound. Gopal et al described the appearance of the layers of a fundoplication and crural diaphragm on ultrasound. 22 The normal fundoplication configuration has 5 distinctive layers: the esophageal wall, the space between the esophagus and fundoplication, the inner gastric wall of the fundoplication, the gastric lumen, and finally the outer gastric wall of the fundoplication (Figure 9). The authors described specific ultrasonographic criteria which could be used to identify fundoplication dehiscence, an overly tight Nissen, or a slipped fundoplication. Similar findings were reported in a study by Chang et al of 7 symptomatic and 9 asymptomatic patients who underwent endoscopic ultrasound evaluation 1 to 6 years after fundoplication. They found that endoscopic ultrasound was capable of identifying slipped fundoplication in 2 patients and a hypotensive lower esophageal sphincter in another 2 patients. 23
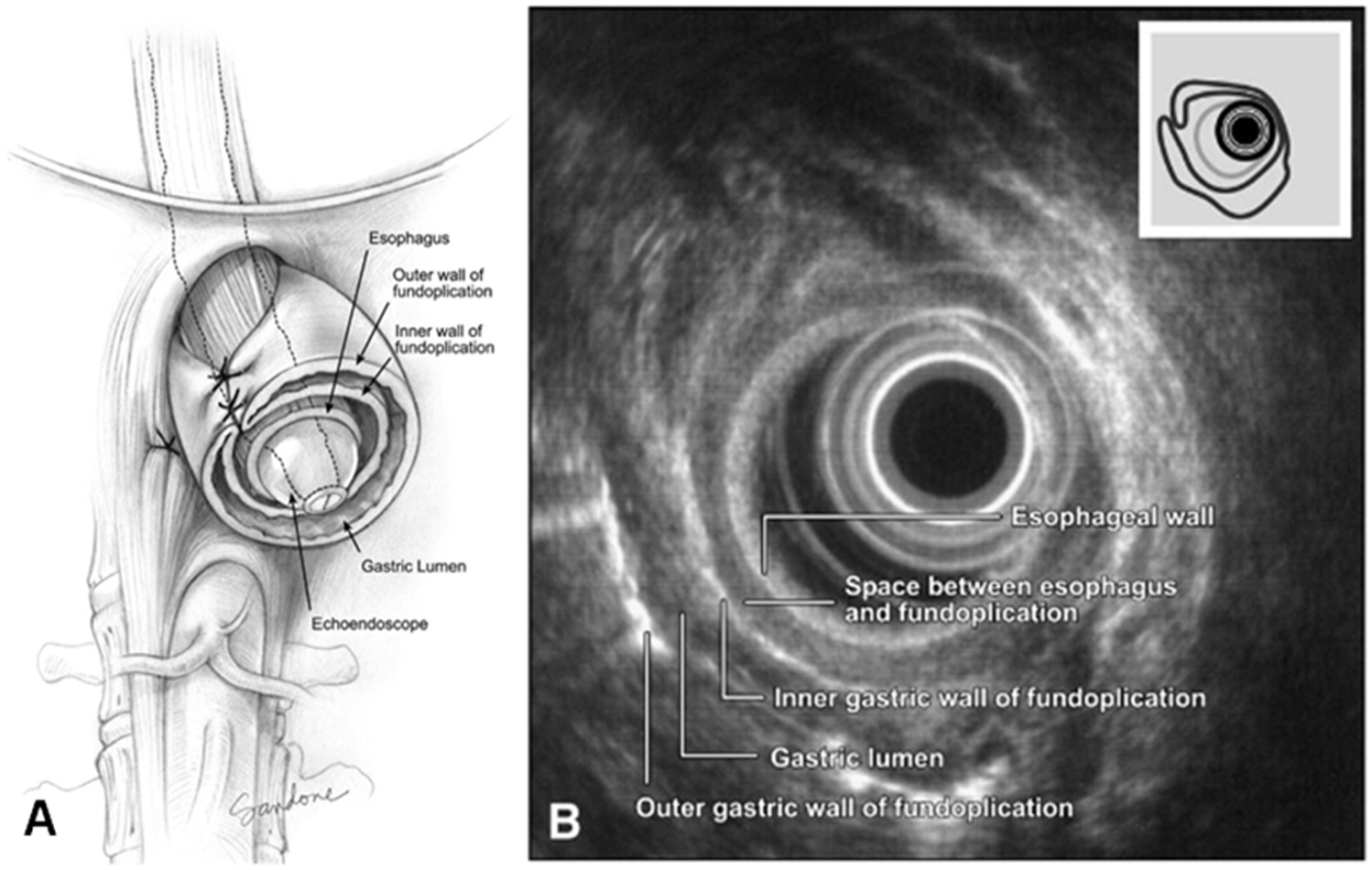
Multiple layers of the fundoplication (A) including the esophagus, space between esophagus and fundoplication, inner fundoplication wall, gastric lumen, and outer wall, and their endoscopic ultrasonographic appearance (B). Reproduced with permission from Gopal et al. Copyright American Society for Gastrointestinal Endoscopy, Published by Mosby.
Another important adjunct is the barium esophagram. This can identify structural lesions such as strictures, hiatal hernia, paraesophageal hernia, or major anatomic alterations associated with motility disorders such as achalasia or distal esophageal spasm. Esophagram is especially useful in the assessment of postoperative dysphagia, providing precise information on the location of the fundoplication in relation to the diaphragm. Several pathologies are evident on esophagram. A tight fundoplication will result in ballooning of the epiphrenic ampula, with retrograde escape of barium, signifying that the pressure gradient across the wrap is greater than the pressure of the primary wave peristalsis. A type 3 hiatal hernia will demonstrate a portion of the fundoplication superior to the crural diaphragm as well as the appearance of gastric rugal folds above the diaphragm. A slipped Nissen may demonstrate a 2 compartment stomach. A disrupted fundoplication can also be observed through passage of the barium tablet and caliber of the distal esophageal lumen. Recurrence of gastroesophageal reflux during the study is a finding that alone is insufficient to diagnose failure but can serve as an adjunct to other findings in support of suspicion for ARS failure.
Conclusion
A thorough understanding of the anatomy of the GEJ from both the endoscopic and surgical point of view is necessary before embarking on antireflux surgery. A detailed endoscopic assessment must be conducted before, during, and after surgery. There are several patterns of fundoplication dysfunction, and few classification systems have tried to categorize them. However, correlation with symptomatology has been inconsistent and external validation has not yet been done. Furthermore, many patients had multiple mechanisms of failure, and were difficult to categorize. Therefore, a detailed description of endoscopic findings with photographs remains the most reliable way to effectively communicate fundoplication dysfunction. Relatively novel anti-reflux procedures such as TIF and MSA must be similarly studied in depth to characterize successful and failed procedures. Adjunctive studies can complement endoscopy, and are each superior at particular aspects of defining the anatomy. More widespread adoption of standardized protocols and classification systems will avoid misdiagnosis and improve efficiency of care.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

