Abstract
Previous clinical trials have concluded that trimodality and bimodality therapy are equivalent for treating locally advanced esophageal squamous cell carcinoma (ESCC). However, the use of chemoradiation alone achieves only a 45% to 50% complete response rate. Surgery remains crucial for locoregional control. In ESCC, salvage esophagectomy for locoregional recurrence has increased morbidity compared to planned esophagectomy. Retrospective data suggest that patients requiring cervical/upper chest anastomosis are more likely to undergo salvage esophagectomy rather than completing trimodality therapy. It’s unclear if selective surgery versus an operative approach on all patients after chemoradiation leads to differences in overall outcomes.
Keywords
Background
Esophageal cancer ranks seventh in incidence and sixth in cancer mortality worldwide. In 2020, there were approximately 604 100 new cases of esophageal cancer globally, and 544 076 deaths. 1 Squamous cell carcinoma (ESCC) is the predominant type of esophageal cancer worldwide, accounting for about 90% of all cases. 2 The incidence of ESCC varies significantly across different regions, with the highest rates observed in Asia and Sub-Saharan Africa. While smoking and drinking are significant risk factors in developed countries, dietary factors, including the consumption of foods high in nitrosamine and highly salted foods such as pickled vegetables, beetle nut chewing, and hot foods and beverages, are theorized to play a role in developing nations.3 -5
The treatment of ESCC has undergone notable advancements, with surgery playing a crucial role in locoregional control as part of multimodality treatment. Upfront surgery has largely been abandoned due to earlier experience of R0 resection rate of 60%. 6 This failure of adequate locoregional control translated to poor overall survival (OS) of 15 months (median), with 30% to 31% locoregional and 40% to 51% distant recurrence. 6 The landmark CROSS trial 7 demonstrated significant improvements in outcomes for locoregional esophageal cancer treatment. The preoperative chemoradiation group achieved a favorable R0 resection rate of 92% compared to 69% in the upfront surgery group. Median survival significantly improved to 49.4 months in the chemoradiation group compared to 24 months in the upfront surgery group. Notably, squamous cell pathology showed high sensitivity to chemoradiation, with a 49% pathologic complete response rate. These findings were further supported by the NEOCRTEC5010 trial, 8 which included 451 ESCC patients who underwent either surgery or chemoradiation plus surgery. The chemoradiation plus surgery group achieved a pathologic complete response rate of 43.2% and a median OS of 100.1 months, compared to 66.5 months in the surgery-only group.
Several European trials have compared definitive chemoradiation (bimodality) to chemoradiation plus surgery (trimodality) and concluded that there is no survival benefit with the addition of surgery. However, this belief that surgery does not play a role in ESCC has had an impact on the quality of cancer care in our community. Recent studies have shown that in adults aged 65 and older, half receive definitive chemoradiation, while only 6% receive trimodality therapy.9,10 Disparities in socioeconomic class, regional differences, patient fitness for surgery, and tumor location all likely contribute to this imbalance. Nonetheless, the concept of salvage esophagectomy has emerged for patients who have failed definitive chemoradiation. To discuss salvage esophagectomy in ESCC, it is necessary to dispel misconceptions about the outcomes of these trials and explore how surgery can be used selectively to resect ESCC patients who have failed chemoradiation.
Bimodality Versus Trimodality Therapy
Two European randomized controlled trials have compared bimodality with trimodality treatment strategies for esophageal cancer. The trial conducted by Stahl et al, 11 involved patients who received induction chemotherapy, followed by either definitive chemoradiation (≥65 Gy) or chemoradiation (40 Gy) followed by surgery. After a median follow-up time of 6 years, the 2-year survival rates were 35.4% (95% CI: 25.2%-45.6%) for the bimodality group and 39.9% (95% CI: 29.4%-50.4%) for the trimodality group. Based on predetermined trial parameters, the authors concluded that the 2 treatment arms were equivalent. However, concerns have been raised regarding the methodology and interpretation of the trial results. The primary concern revolves around the randomization scheme, which occurred prior to the initiation of chemoradiation therapy. Only 66% (57/86) of patients in the trimodality group underwent esophagectomy, while 6% (5/86) of patients in the bimodality group underwent esophagectomy after showing no response to chemoradiation, resulting in significant patient crossover and treatment effect contamination. Additional criticisms include a high perioperative mortality rate of 11% in surgical patients,12,13 as well as statistical considerations such as unrealistic equivalence limit estimation, appropriateness of the one-sided log-rank test, and lack of disclosure regarding adaptive design criteria, which may have led to an underpowered sample size calculation.12,13
The Stahl trial predetermined the need for surgery prior to chemoradiation, but we now practice a selective surgery strategy as an effective organ-sparing approach to patients treated with chemoradiation. 14 The decision for trimodality or bimodality is heavily influenced by treatment response to chemoradiation, particularly in cases where there is no evidence of residual disease. Trimodality patients are those who are fit for surgery, who either had a good response to chemoradiation or did not respond but still had resectable disease. Many of these patients undergo a delayed esophagectomy to allow for recovery before they can tolerate surgery. The bimodality group comprises a mixed population; including patients who achieved a complete response to chemoradiation and opted for surveillance, patients who were not fit enough to undergo surgical resection, patients who were diagnosed with metastatic disease during surveillance, and patients who experienced locoregional recurrence during surveillance and subsequently underwent salvage esophagectomy (Figure 1). Therefore, a relevant question arises regarding patients who achieve clinical complete response after chemoradiation—should they undergo esophagectomy or continue surveillance? This is what the French FFCD 9102 trial 15 aimed to investigate. In this trial, a mostly ESCC cohort received chemoradiation. Those who had complete or partial response were randomized to either surgery or surveillance. The evaluating process for treatment response involved an esophagogram, abdominal ultrasound, chest X-ray and possible endoscopic ultrasound. Patients who did not show a treatment response were not included in the trial and were offered surgery as an alternative option. Ultimately, the survival outcomes in the French trial indicated similar survival rates between its 2 arms, leading the authors to conclude that bimodality and trimodality therapy yield equivalent survival rates.
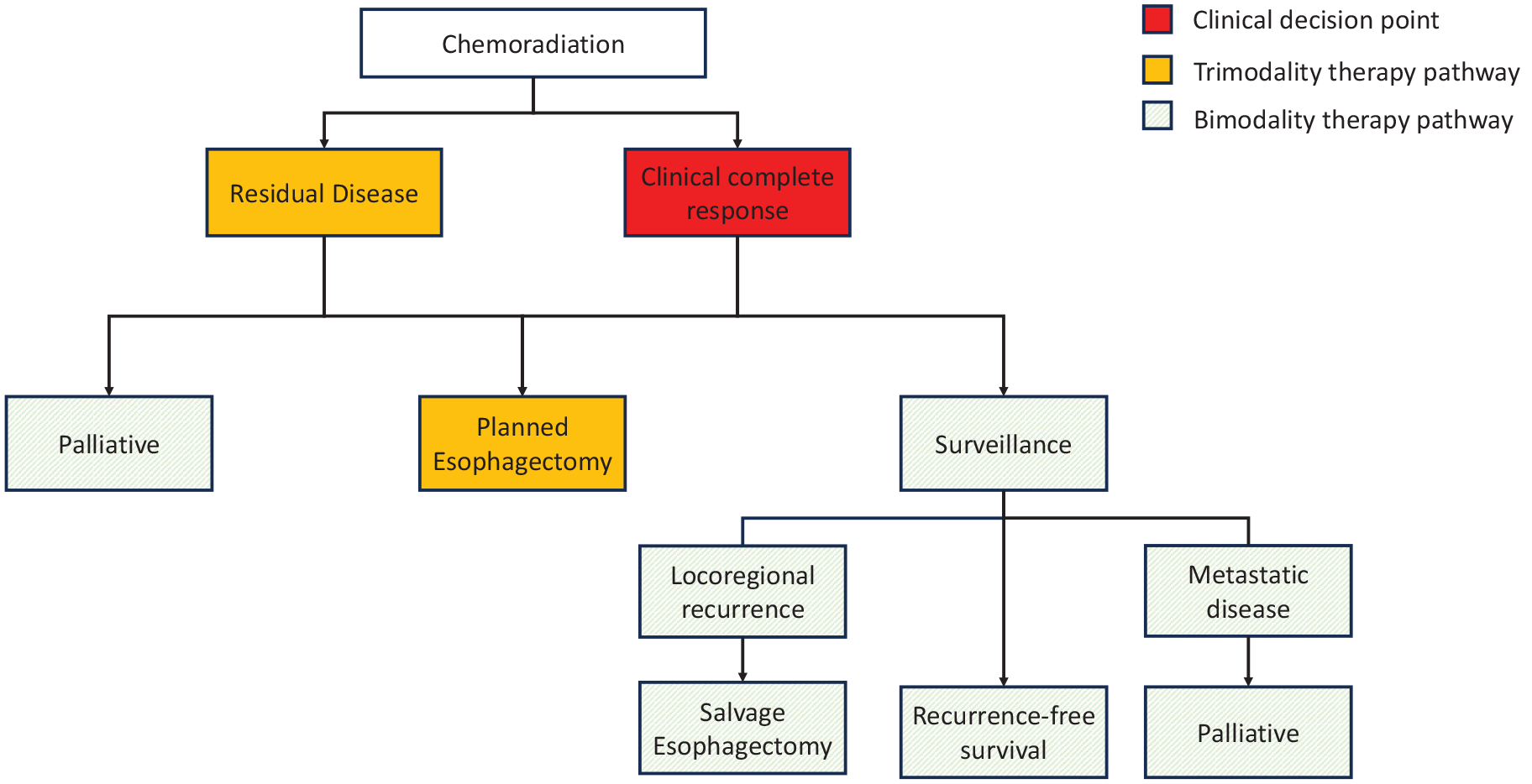
Selective surgery treatment algorithm for locally advanced esophageal squamous cell carcinoma.
The trial’s surgical outcomes exhibited several inconsistencies. Notably, the surgery arm had a 75% R0 resection rate, which was significantly lower than the more contemporary rates of 92% observed in the CROSS trial 7 and 98% in the NEOCRTEC trial. 8 Furthermore, there was a 9% perioperative mortality rate after esophagectomy, contrasting to the lower rates of 2% in CROSS and 0.5% in NEOCRTEC.7,8 The low R0 resection rate likely attributed to 33.6% locoregional recurrence rate in the surgery arm within 2 years. 15 This failure rate is higher than the 3.3% locoregional and 10.8% locoregional plus distant recurrence in the CROSS trial, 16 as well as 9.8% locoregional, 4.3% locoregional plus distant recurrence in NEOCRTECT5010 with more than 3 years of follow up. 17 Despite randomizing after achieving clinical complete response, only 85% of the allocated patients in the surgery arm actually underwent the intended surgical intervention. As for patients who did not achieve a good response after chemoradiation, they underwent surgery and achieved a survival rate equivalent to those on the trial (17 months), in contrast to non-responders who did not undergo surgery (5.5 months). 18 Despite all the flaws, the 2 European studies set the tone for the treatment of locally advanced ESCC.
Across the world, the Japanese experience provided some counter-evidence suggesting survival advantage of trimodality compared to bimodality therapy in locally advanced ESCC. Nomura et al 19 compared patients in the trimodality arm of the JCOG9907 trial 20 and those in the bimodality arm of the JCOG9906 trial. 21 Their analysis demonstrated improved OS in patients treated with trimodality therapy compared to bimodality therapy. Important to note that there was only a 0.6% perioperative mortality in the trimodality arm. Contemporary U.S. retrospective studies also favor trimodality for OS and DFS.22,23 While the debate for whether trimodality or bimodality is superior continues, patients who had residual disease still benefitted from a surgical resection. This raises the question of patient selection: who has residual disease, and who can tolerate an esophagectomy?
Evaluation for Complete Response
While approximately 45% to 50% of patients achieve a pathological complete response after esophagectomy following completion of chemoradiation,7,8 significant efforts have been made to differentiate patients with residual disease from those who have achieved a complete response. Most centers utilize a combination of endoscopic procedures including endoscopy with biopsy, endoscopic ultrasound (EUS), and metabolic cross-sectional imaging such as PET-CT. The accuracy of this combined strategy ranges from 46% to 79%, with a negative predictive value of 31% to 74%.24 -26 However, none of the available tools can perfectly identify residual disease within the esophagus, and this residual disease can often persist beyond the mucosa, involving the submucosa and even the surrounding muscle stroma which is why mucosal biopsy is usually non-remarkable even when there is residual, mural disease. 27 Not surprisingly, the accuracy of restaging methods decreases as the size of residual disease diminishes.
In the preSANO trial, 28 EGD with biopsy alone carries a high false negative rate of 56% for TRG3 and 18% for TRG4 disease. A bite-on-bite biopsy strategy was implemented in this trial in attempts to improve diagnostic yield by obtaining tissue samples beyond the mucosa. While the use of bite-on-bite biopsy was reported to have decreased the rate of false negative biopsies from 31% to 11%, a separate trial by the Netherlands group on using biopsies from the SANO trial 29 showed that the improved residual tumor detection was due to an increased number of samples taken rather than the depth of the biopsy. Nevertheless, the trial used a combination of endoscopic bite-on-bite biopsy, EUS, PET-CT imaging, and a two-stage clinical response system (Figure 2) to assess the presence of residual tumor based on tumor response. These methods demonstrated a sensitivity of 77%, specificity of 72%, positive predictive value of 45%, and negative predictive value of 92% for detecting residual carcinoma (≥1%). This strategy is being utilized in the ongoing SANO trial (NTR6803), which will compare esophagectomy and active surveillance in patients who achieved clinical complete response after 2 rounds of restaging (Figure 2). The results of the SANO trial, along with preSINO (NCT03937362), and ESOSTRATE (NCT02551458) are eagerly awaited as they may potentially reshaped the treatment approach for locally advanced esophageal cancer.
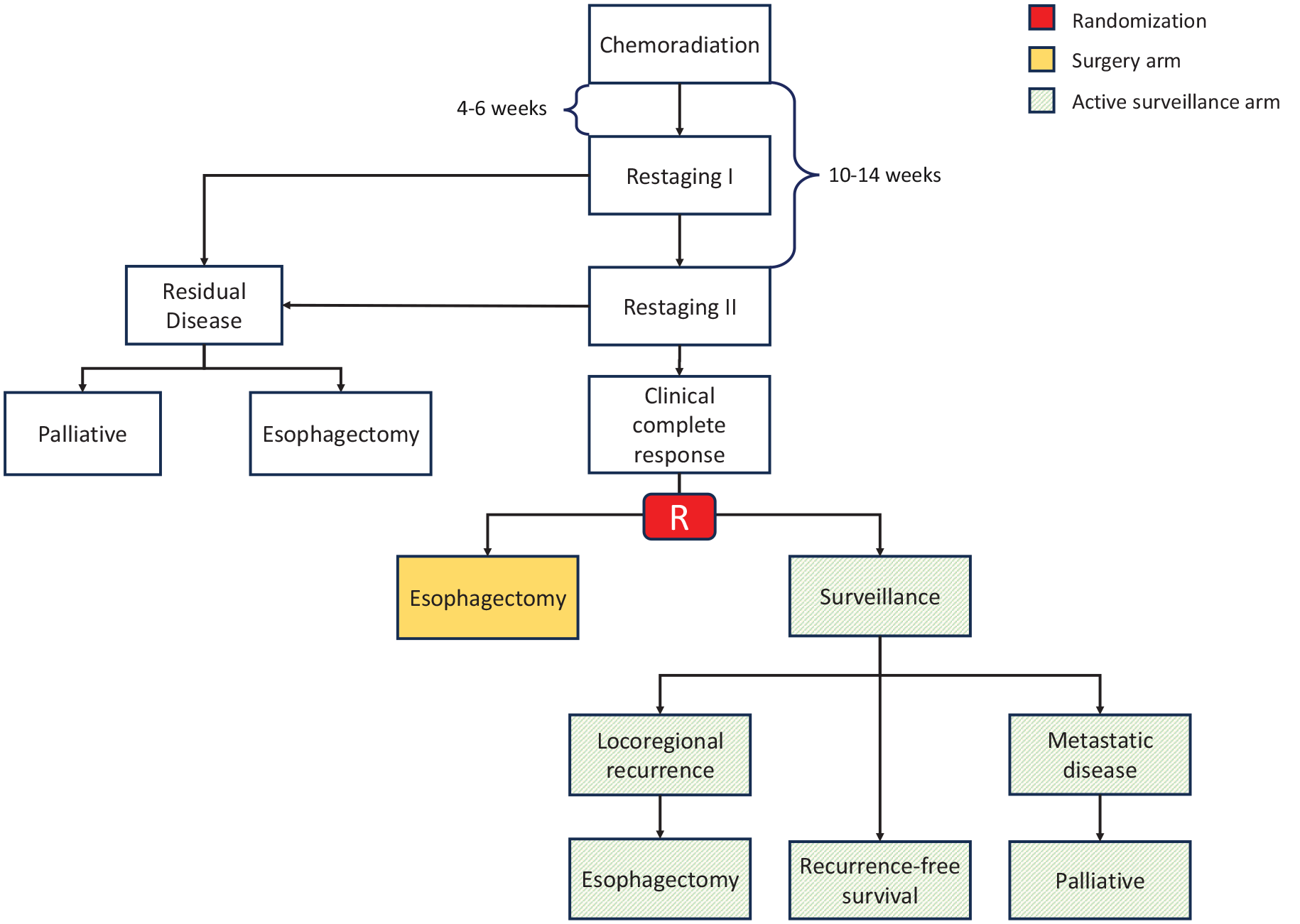
Randomization schema for the SANO trial (NTR6803), restaging involves endoscopic bite-on-bite biopsy, EUS, and PET-CT imaging.
Circulating tumor DNA (ctDNA) show some promise in early detection of residual disease after chemoradiation. Azad et al, showed that in patients treated with chemoradiation, ctDNA alone detected 71% of residual disease, metabolic imaging alone detected 57%, and combining metabolic imaging with ctDNA detected 100% of residual disease. Detection of ctDNA also preceded radiographic imaging by 2.8 months. 30 ESCC were also found to shed ctDNA 7 times higher than esophageal adenocarcinoma. Although promising, the utility of ctDNA in detecting loco-regional disease remains to be seen. Larger studies will need to be conducted to determine the use of ctDNA in esophageal cancer.
Salvage Esophagectomy
The concept of salvage esophagectomy was popularized in the west by Swisher et al, 31 aiming to perform esophagectomies in patients who experienced locoregional failures following definitive chemoradiation and were considered physically fit for surgery. This specific patient group presented surgical challenges that developed months after completing definitive treatment. Additionally, a higher proportion of these patients had upper esophageal lesions, reflecting the initial preference for definitive chemoradiation to avoid the need for laryngectomy, considering their poor baseline physical health. Salvage esophagectomy patients with ESCC experienced longer operative times, extended hospital stays, and higher rates of anastomotic leaks compared to planned esophagectomies (38% vs 7%). Moreover, a perioperative mortality rate of 16.7% (2/12) was observed. 31 However, a subset of patients achieved long-term survival exceeding 5 years.
Currently, there is a growing body of literature on salvage esophagectomy (Table 1). Since most studies have a limited number of patients, the focus has been on systematic reviews and larger retrospective reports. However, there is significant selection bias in retrospective analyses, and the definition of salvage varies between different studies. 32 Studying this population retrospectively without bias is challenging due to difficulties in distinguishing salvage from delayed resections and differentiating between early recurrence and persistent disease. Salvage esophagectomy outcomes were reportedly comparable to trimodality therapy patients from a European cohort, 33 however, more contemporary data from the Memorial Sloan Kettering group 34 showed salvage to have higher odds of postoperative complication and shorter overall and disease-free survival. Both of these studies combined esophageal adenocarcinoma (EAC) and ESCC patients in their analyses. Nonetheless, it appears to be a consensus that salvage esophagectomy for ESCC is associated with significant perioperative morbidity. In total, 35% to 76% of salvage patients experience major morbidity, with major pulmonary complications reported in 23% to 43% of cases, major cardiovascular events in 3% to 49%, anastomotic leak rates of 11% to 25%, and 30-day mortality rate of 2% to 8% (Table 1). The differences in perioperative outcomes between salvage and planned resection for ESCC is notable, as juxtaposed to the salvage experience for EAC that demonstrates comparable perioperative mortality and long-term survival rates to planned resections. 35 There seems, however, to be a strong component of selection bias in these retrospective reviews, where patients were selected to undergo observation for many different patient derived variables such as co-morbidity, perceived fitness after chemoradiation, location of tumor and need to do more extensive surgery, to name a few.
Selected Series on Salvage Esophagectomy After Bimodality Therapy.
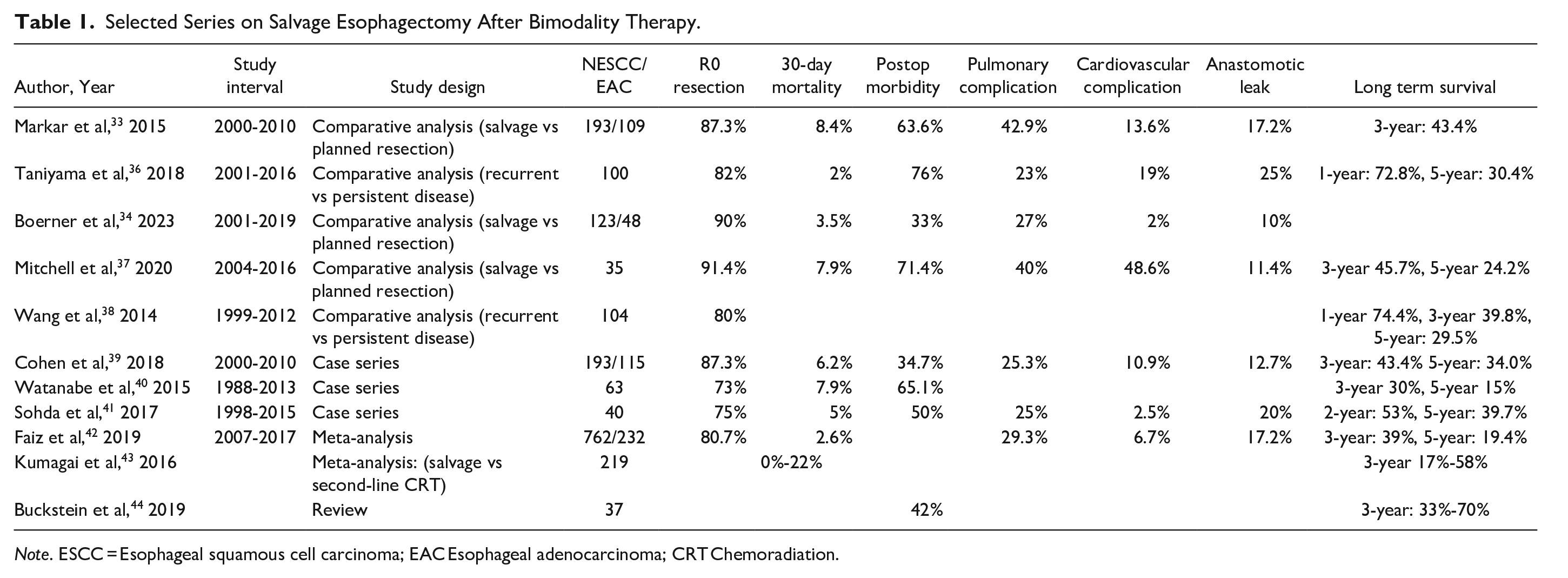
Note. ESCC = Esophageal squamous cell carcinoma; EAC Esophageal adenocarcinoma; CRT Chemoradiation.
There were several observed differences between salvage and planned esophagectomy patients. In our experience, salvage resections were performed a median of 6 months after chemoradiation, compared to 2 months for planned resections, more 3-field esophagectomies were performed in salvage cases, and the location of the anastomosis tended to be in the neck or upper chest. 37 We find that disease behind the airway presents a significant challenge due to potential involvement of the tracheobronchial tree. The selective surgery strategy in this group of patients can lead to a delay in timely surgery. As radiation fibrosis intensifies over time, salvage surgery can become increasingly difficult for disease recurrence in this area. Considering the risk factors associated with ESCC, these patients are often suboptimal surgical candidates, and we are operating on them as an attempt at locoregional control. Until more accurate methods of residual disease detection become available, we advocate for a planned resection in this group of patients if deemed suitable surgical candidates. Patient selection remains crucial, as the risks and benefits of salvage esophagectomy are specific to each individual, and the decision to pursue this option should involve shared decision-making. Referral to high-volume centers for evaluation is recommended for centers without adequate experience in caring for these complex patients.
Quality of Life
There is a lack of specific quality of life (QoL) data for salvage esophagectomy. Nevertheless, when examining existing studies focused on QoL after esophagectomy and chemoradiation, the long-term outcomes appear to be similar between the 2 treatment modalities.45,46 However, short-term trends differ. 46 Within 3 to 6 months after surgery, esophagectomy patients exhibit worsened dyspnea and cough, but these differences disappear by the 2-year mark. In contrast, bimodality patients experience a progressive worsening of dyspnea and cough over the same 2-year period. Both groups encounter worsening symptoms of dysphagia at 2 years, likely due to an increase in endoscopic strictures. Blazby et al 47 demonstrated that for patients who survived beyond 2 years, their QoL returned to preoperative levels at 9 months, whereas those who succumbed within 2 years of surgery never regained their previous QoL levels.
Conclusion
The standard treatment approach for ESCC has been chemoradiation with or without surgery. However, the claims of equivalence between bimodality and trimodality therapy in 2 European randomized controlled trials are confounded by their methodology and surgical outcomes, which may not fully reflect the outcomes in a more contemporary patient population. With the advancement of technology for accurate detection of residual disease in patients who have undergone chemoradiation, surgeons are now faced with the challenge of selectively identifying patients with ESCC who would benefit from surgical resection. It is important to note that salvage esophagectomy in this patient population is associated with increased morbidity compared to planned resection. These patients undergo surgery at a significantly longer interval after completing chemoradiation therapy, and they often require 3-field surgeries with higher levels of anastomosis. Based on our clinical experience, we favor planned esophagectomy for surgically appropriate ESCC patients who tolerated neoadjuvant chemoradiation. Special consideration for planned esophagectomy should be given to those with disease located near the airway, rather than waiting for disease recurrence and performing salvage surgery. Careful patient selection and individualized treatment decisions are crucial in this context. There is also significant selection bias in all the retrospective analyzed data. As we await further advancements in the detection of residual disease after chemoradiation, it is important for surgeons to assess each patient's unique circumstances and consider the risks and benefits associated with salvage esophagectomy versus planned resection. Referral to experienced high-volume centers for evaluation and management of these complex patients may be beneficial for centers without sufficient expertise in caring for such cases.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by generous philanthropic donations from the Mason Family Research Fund.
