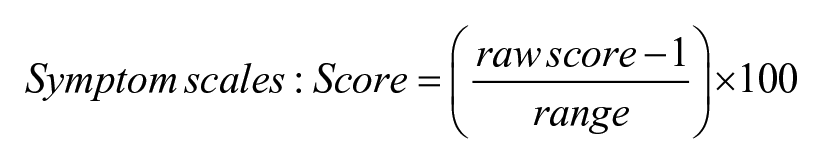Abstract
Objective:
The purpose of this study was to evaluate indications, techniques, and outcomes of patients who underwent delayed esophageal reconstruction.
Methods:
A retrospective chart review was performed for all esophagectomy patients who underwent delayed esophageal reconstruction from 2006 to 2020. Demographics, surgical indication, type of esophageal discontinuity procedure performed, duration between esophagectomy and reconstruction, type and technique of reconstruction performed, follow-up duration, change in body mass index, and overall outcome were assessed.
Results:
Fifteen patients (8 women, 53.3%) with a median age of 57.6 years (IQR 50.5-66.7 years) were included. Etiology necessitating foregut discontinuity included: delayed postoperative complications in 6 (40.0%), iatrogenic injury in 3 (20.0%), strangulated bowel in 3 (20.0%), trauma in 2 (13.3%), and esophageal cancer in 1 (6.7%). Median duration of esophageal discontinuity was 277 days (range 105-1045). Eleven patients (73%) underwent a substernal colon interposition; 2 (13%) substernal gastric advancement; and 2 (13%) small bowel advancement in the posterior mediastinum with jejunum via Roux-en-Y. Complications were noted in 7 (47%) patients. Median decrease in BMI was 4.7 (IQR 1.4-12.4). Overall, 11 (73.3%) patients had a good outcome, 3 (20.0%) patients had a fair outcome, and 1 poor outcome (6.7%). The median follow-up duration since reconstruction was 4.2 (IQR 2.8-5.4) years.
Conclusion:
Delayed esophageal reconstruction can be performed with acceptable outcomes with careful preparation and often require lengthy recoveries.
Keywords
Key Learning Points
Patients who require a period of esophageal discontinuity generally represent a critically ill population.
These patients require tailored treatment from a multidisciplinary healthcare team to be successful.
Reduction in body mass index and a prolonged period of discontinuity is common among these patients.
Background
Esophagectomy continues to be the mainstay therapy for esophageal cancer and end-stage benign esophageal disease. 1 Esophagectomy, whether performed for benign or malignant indications, is typically followed by immediate reconstruction using gastric, colonic, or jejunal conduits. 2 However, reconstruction may be delayed or staged for a number of reasons, including sepsis, poor operative candidacy, or delayed presentation. 3 These patients are left in foregut discontinuity for a period of time, which is a source of both nutritional and psychiatric stress. 4 Additionally, patients undergoing elective foregut surgery may suffer from iatrogenic esophageal injury, resulting in discontinuity. 5 For this subset of patients, reconstruction of their alimentary tract may be best delayed until recovery from the initial surgery, which often involves long periods of critical care and rehabilitation. 5
Strategies for achieving an ideal outcome in these high-risk, low-frequency scenarios are tailored for each patient based on the presenting insult. Few studies have assessed outcomes following esophagectomy with delayed reconstruction. 6 The purpose of this study was to examine our institutional experience with patients undergoing delayed reconstruction to assess for potential commonalities in this challenging, heterogonous cohort of patients.
Materials and Methods
Study Design
This was a retrospective chart review study, approved by the Institutional Review Board (IRB protocol number HSC20150617H), that included all patients from January 2006 to December 2019 who underwent reconstructive surgery to re-establish foregut continuity after a period of discontinuity. Data were collected from the electronic medical record and mailed surveys, with treatment courses broadly classified into 3 stages: Initial presentation (demographics, etiology leading to discontinuity, clinical condition at presentation, and temporizing measures), definitive foregut reconstruction (surgical technique, operative complications, discharge status); and post-operative follow-up (operative re-interventions, assessments during postoperative clinic visits). Nutritional supplementation requirements and body mass index (BMI) were also reviewed at these 3 stages.
Postoperative complications were classified according to the Clavien-Dindo classification system (Table 1). 7 Overall surgical outcome was determined as good, fair, or poor by consensus of the patient care team based on their need for reinterventions (objective) and patient complaints (subjective) as documented during their follow-up (Table 2). Patient-reported quality-of-life outcomes were evaluated using the European Organization for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire—Esophageal Cancer Module (QLQ-OES18), an 18-item validated metric for assessing esophageal-specific QOL (Supplemental Table 1). 8 The QLQ-OES18 covered 10 symptom scales: dysphagia, trouble swallowing saliva, choking when swallowing, eating, dry mouth, trouble with taste, trouble with coughing, trouble speaking, reflux, and pain. 8 Raw scores were standardized via linear transformation using the following formula to create a scale from 0 to 100, with higher scores corresponding to greater symptom burden:
Clavien-Dindo Classification System for Postoperative Complications.

Source. Adapted from Dindo et al. 7
Composite Postoperative Outcome Grades Defined by Objective and Subjective Metrics.

Statistical Analysis
Statistical analysis was performed using GraphPad Prism v9.0 (GraphPad Software Inc, San Diego, CA, USA). Data were non-normally distributed, as determined by a significant Shapiro-Wilk text. Thus, Fisher’s exact tests and Mann-Whitney U tests were used, as appropriate, for bivariate analysis. Kaplan-Meier analysis was used to assess long-term survival. Data were presented as numbers and percentages for categorical data and median and interquartile range (IQR) or range for continuous data.
Results
Patient Characteristics
There were 15 patients (8 women, 53.3%) with a median age of 57.6 years (IQR 50.5-66.7 years) who had a period of foregut discontinuity and subsequently underwent delayed and/or staged reconstruction during the 14-year study period. All reconstructive procedures were performed by a single surgeon. Preoperative comorbidities included hypertension in 8 (53.3%), diabetes in 3 (20.0%), chronic obstructive pulmonary disease in 2 (13.3%), peripheral artery disease in 1 (6.7%), history of tobacco use in 4 (26.6%), and prior radiation in 3 (20.0%). The median BMI at initial presentation was 29.0 (IQR 24.6-33.7).
Initial Presentation
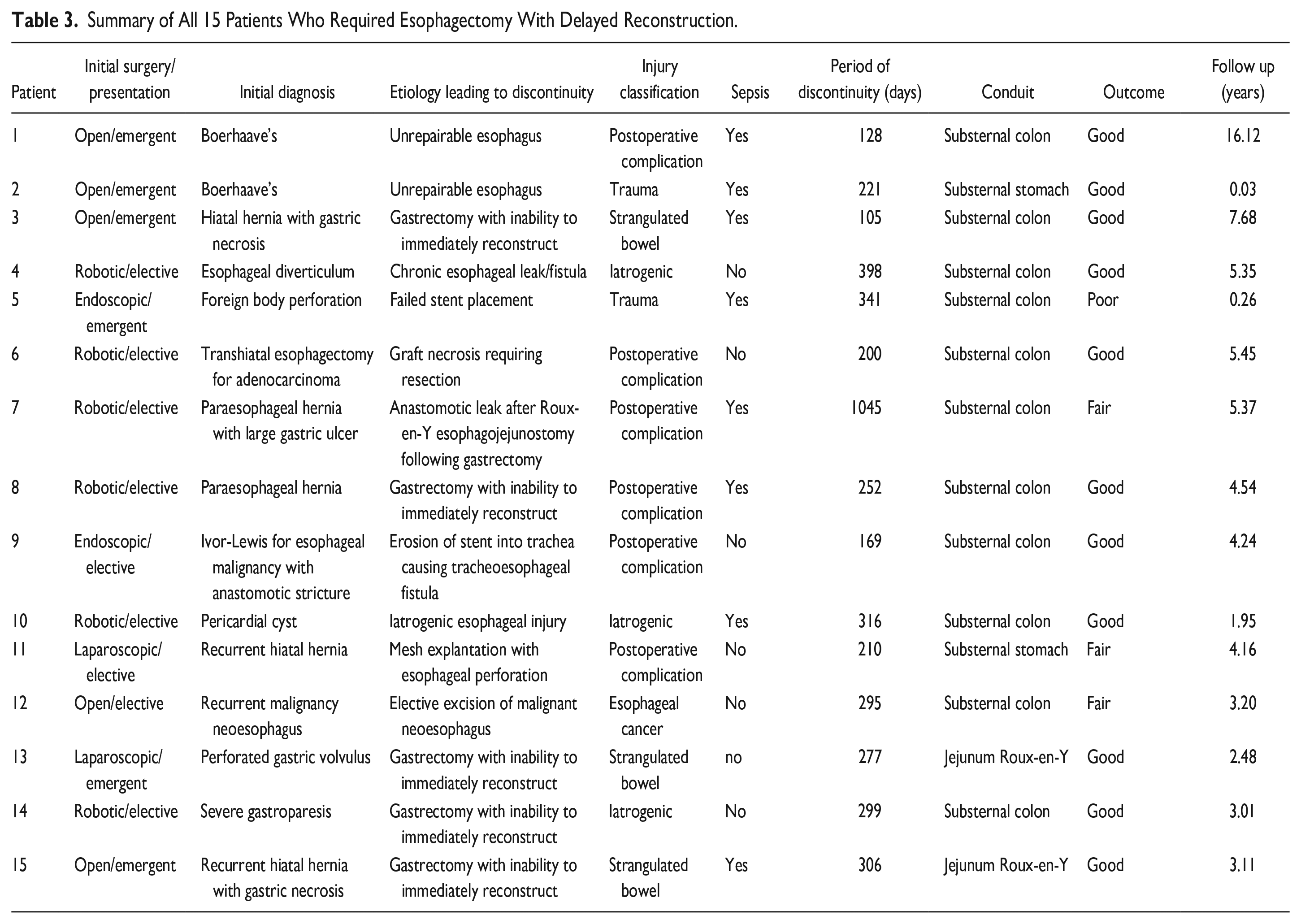
The etiology necessitating foregut discontinuity included: delayed postoperative complications in 6 (40.0%), iatrogenic injury in 3 (20.0%), strangulated bowel in 3 (20.0%), trauma in 2 (13.3%), and esophageal cancer in 1 (6.7%) (Table 3). Eleven (73.3%) of patients initially presented at an outside hospital. Thirteen (86.7%) patients experienced complications during their initial hospitalization, including 10 (67.7%) patients with sepsis. Intubation was required in 6 (40.0%) patients, tracheostomy in 4 (26.7%), esophageal stent placement in 3 (20.0%), esophageal exclusion with staples in 3 (20.0%), a feeding tube in 5 (33.3%), and a thoracostomy tube in 4 (26.7%). One patient (6.7%) required a chest and/or mediastinum washout and 2 (13.3%) patients required an abdominal washout.
Summary of All 15 Patients Who Required Esophagectomy With Delayed Reconstruction.
Discontinuity
All patients underwent unplanned foregut exclusion/diversion procedures, except one (6.7%), who presented with recurrence of malignancy in her neo-esophagus after a transhiatal esophagectomy several years prior, in which a planned staged foregut reconstruction was performed. Twelve (80.0%) patients underwent diversion via a cervical end esophagostomy, 2 (13.3%) via a cervical loop esophagostomy, and 1 (6.7%) via a blind esophageal pouch. The median length of stay during the initial discontinuity operation was 22 days (IQR 16-32 days). The median BMI at discontinuity was 28.0 (IQR 25.0-30.0). Twelve (80.0%) patients received nutritional supplementation via a J-tube, 5 (33.3%) via a G-tube, and 1 (6.7%) via total parenteral nutrition. Eight patients (53.3%) were discharged home following their discontinuity operation, while the remaining 7 (46.7%) were discharged to a skilled nursing facility.
Reconstruction
The median duration of discontinuity was 277 days (range 105-1045 days). Eight (53.3%) patients required at least 1 additional procedure during their discontinuity. The median BMI at presentation for reconstruction was 22.3 (IQR 21.0-24.4). Thus, the median decrease in BMI between initial presentation and reconstruction was 4.7 (IQR 1.4-12.4).
Substernal Colon Interposition
Eleven patients underwent reconstruction using a sub-sternal colon interposition (73.3%; 6 [40.0%] anastomosed to stomach, 5 [33.3%] anastomosed to jejunum via Roux-en-Y). This was often because the stomach was unavailable or was of inadequate length. All colon pedicles were based on the ascending branch of the left colic artery, with ligation and division of the middle colic artery and vein to achieve the required length. The substernal tunnel was chosen because the space was virgin. Although termed a “left colon interposition” because of the origin of its vascular pedicle, the portion of the colon actually reaching to the neck was often the right and transverse colon. The length of the interposition necessary to reach the neck was measured carefully before dividing the colon, preserving as much mesenteric tissue as possible. With the mesentery splayed out, the colon was brought up to the cervical esophagus, maintaining as much of the cervical esophageal length as possible. Early in the series, a hand-sewn end-to-end anastomosis was performed, with more recent patients in the series undergoing a side-to-side, functional end-to-end stapled anastomosis. Once the proximal anastomosis was made, division of the distal interposition was made and chosen to keep at least 5 cm of the colonic interposition within the abdomen. This division stayed very close to the wall of the colon to maintain patency of the marginal artery traveling to the neck. This anastomosis was then performed to either remnant stomach via a hand-sewn end-to-end technique or to small bowel via a side-to-side, functional end-to-end stapled Roux-en-Y.
A surgical pyloroplasty was routinely performed prior to the 1 death in our series, which has since changed our approach to performing a chemical pyloroplasty with Botox. Gastrointestinal continuity was then completed by performing a colo-colostomy, usually stapled side-to-side. Since preservation of the marginal artery to the neck was important, the colo-colostomy was necessarily in close proximity to the distal colonic interposition anastomosis. Tacking sutures were placed between the diaphragm and the graft at the entrance into the substernal space to prevent future herniation. Mesenteric defects were carefully closed, being careful to avoid all major blood vessels.
Substernal Stomach
Two patients underwent reconstruction using a substernal stomach conduit (13.3%). Graft preparation was performed in the usual fashion as when performing posterior mediastinal stomach advancements, preserving the right gastroepiploic artery as the pedicle. The cervical anastomosis and substernal placement were similar to that of the colon. In both techniques, the left hemi-manubrium and costoclavicular joint were removed en-bloc to open the space to the thoracic inlet anteriorly. Removal is recommended because the head of the clavicle protrudes posteriorly, which may impinge on the cervical anastomosis. It is our experience that the substernal route takes more length than the posterior mediastinal route, and for this reason, many stomach grafts may not reach. In addition, many of these patients had jejunal feeding tubes, which tend to cause atrophy of the stomach.
Small Bowel Reconstruction
Two patients underwent reconstruction using a posterior mediastinal small bowel interposition (13.3%). Both patients had most of their esophagus intact, ending in a blind pouch close to the hiatus. One of these patients was able to be reconstructed entirely through an abdominal approach. The other required an additional left thoracotomy secondary to adhesions. This last patient did not have a cervical spit fistula and instead learned to spit out his saliva into a cup during his discontinuity phase. Therefore, we found that a proximal diverting cervical spit fistula is not always necessary. We believe that loop esophagostomies are generally difficult to perform, especially in those with larger diameter necks.
All 15 (100.0%) patients underwent an abdominal incision, 13 (86.7%) underwent a cervical incision, and 1 (6.7%) underwent a thoracotomy incision. Two (13.3%) patients received 1 anastomosis, 2 (13.3%) received 2 anastomoses, 7 (46.7%) patients received 3 anastomoses, and 4 (26.7%) patients received 4 anastomoses. Eight (53.3%) patients underwent at least 1 additional incidental procedure during reconstruction, including 5 (33.3%) appendectomies, 4 (26.7%) cholecystectomies, 1 (6.7%) hiatal hernia repair, and 1 (6.7%) thyroidectomy. The median operative time was 394 (IQR 372-441) minutes.
Post-reconstruction operative complications were noted in 8 patients (53.3%) and included anastomotic leaks in 5 (33.3%; Clavien-Dindo grade I), pleural and/or abdominal fluid collections requiring drainage in 3 (20.0%; Clavien-Dindo grade IIIa), ventilation for more than 24 hours in 2 (13.3%; Clavien-Dindo grade I), supraventricular tachycardia in 1 (6.7%; Clavien-Dindo grade II), reintubation in 1 (6.7%; Clavien-Dindo grade IVa), recurrent laryngeal nerve injury in 1 (6.7%; Clavien-Dindo grade Id), and an esophageal-cutaneous fistula in 1 (6.7%; Clavien-Dindo grade IIIb). None of the patients who underwent colon interposition experienced conduit necrosis, a need for vascular augmentation, or leaks at colo-colostomy, colo-gastrostomy, or colo-jejunostomy anastomoses sites. When stratified by indication for esophageal discontinuity, there were no differences in the overall complication rate (P = .35) or anastomotic leak (P = .41). The median length of stay during reconstruction was 14.0 days (IQR 12.5-21.5 days). Nine (60.0%) patients were discharged on an oral diet. However, all patients were discharged with a J-tube. Fourteen (93.3%) patients were discharged to home, with the remaining patient (6.7%) discharged to an inpatient rehabilitation facility.
Body Mass Index Changes Over Time
The median BMI was 29.0 (IQR 24.6-33.7) at initial presentation, 28.0 (IQR 25.0-30.0) at discontinuity, and 22.3 (IQR 21.0-24.4) at reconstruction. The median decrease in BMI between initial presentation and reconstruction was 4.7 (IQR 1.4-12.4; Figure 1). However, this ranged from positive 1.3 to negative 19.0. There was no correlation between the duration of discontinuity and change in BMI (Pearson’s r = .07, P = .81).
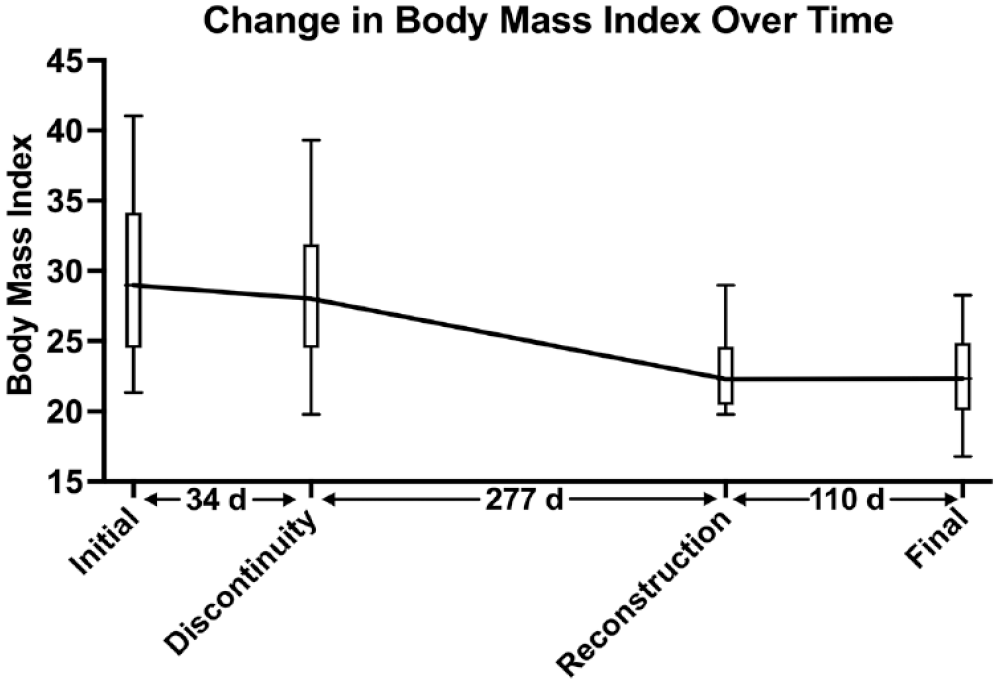
Change in median body mass index for all 15 patients undergoing esophagectomy with delayed reconstruction. Box represents interquartile range and error bars represent 5th to 95th percentile.
Follow-Up
The overall short-term outcome, a composite of patient care team consensus, need for additional procedures, and patient-reported outcomes, was good in 11 (73.3%), fair in 3 (20.0%), and poor in 1 (6.7%). Patients were followed up to 16 years postoperatively, with a median follow-up duration after reconstruction of 4.2 (IQR 2.8-5.4) years. Nine (60.0%) patients required at least one procedure following reconstruction. In addition to the single operative death, two (13.3%) patients died during long-term follow-up (Figure 2), one of which was directly related to their foregut issues (presumed perforated ulcer in excluded gastrum that eroded into his left atrium). One patient was lost to follow-up. No patients remained dependent on tube feeds.
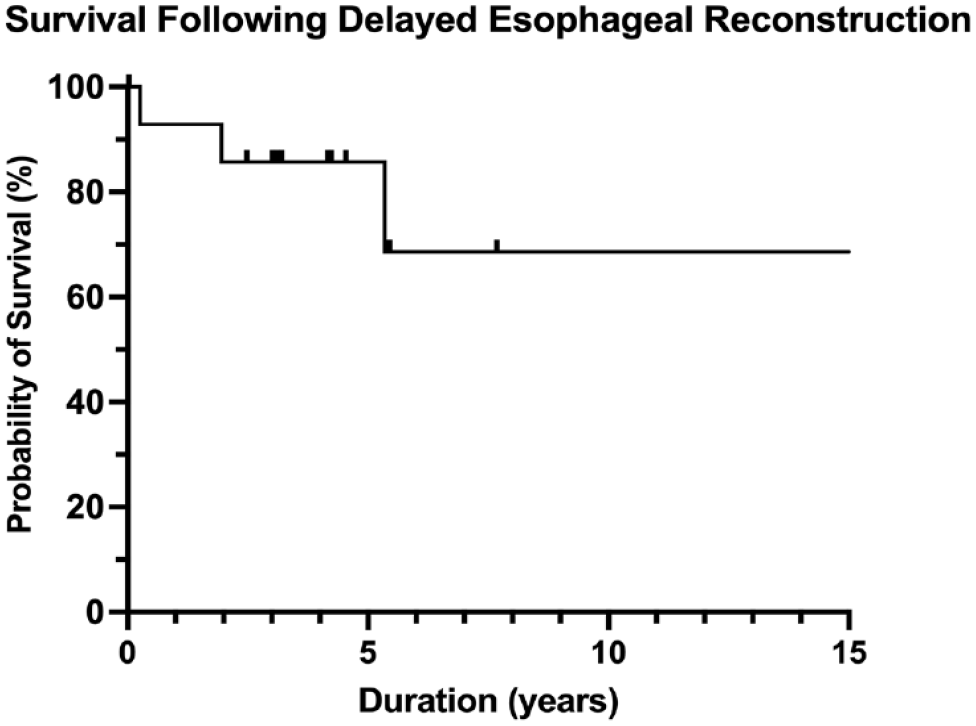
Survival following delayed esophageal reconstruction.
Of the 12 patients without a documented death, the QLQ-OES18 was returned by 5 patients (41.7% response rate). The median duration between reconstruction and survey response was 5.4 (IQR 4.5-7.6) years. Patients generally reported low esophageal symptom burden, with the most common symptoms being reflux (median 66.7, IQR 16.7-100) and trouble coughing (66.7, 33.3-66.7; Figure 3). Trouble eating (62.5, 25.0-75.0) and swallowing saliva (50.0, 0.0-50.0) were also relatively common. However, dysphagia was minimal (0.0, 0.0-67.0).
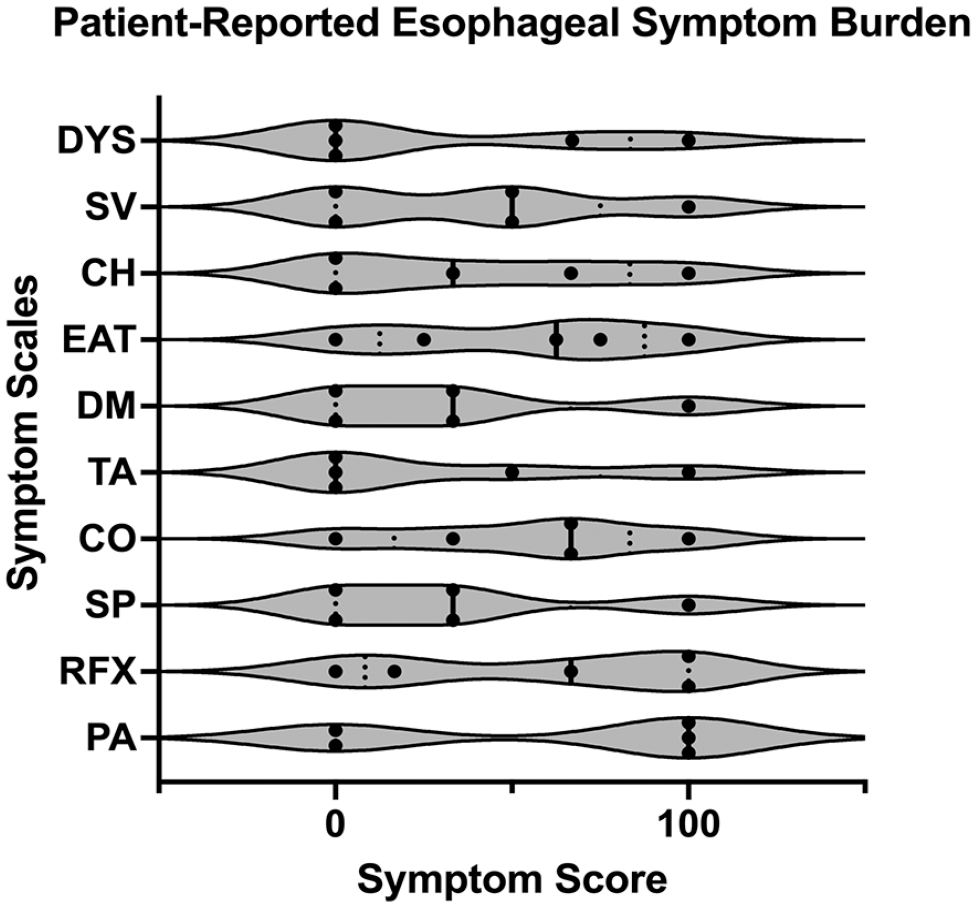
Patient-reported quality-of-life outcomes as assessed by the European organization for research and treatment of cancer quality of life questionnaire—esophageal cancer module. Dots represent each patient score, solid line represents median score, and dotted lines represent quartiles.
Discussion
The prevalence of esophagectomy requiring delayed reconstruction is uncommon and reflected in the sparsity of available literature,2,4,9 with most focusing on acute management.10,11 In this study, we treated approximately one patient per year, which speaks to the relative rarity of these cases. Other published studies have reported an average rate of 2.4 cases 2 and 1.4 cases 4 per year for delayed reconstruction, which likely reflect the referral patterns at these centers. Foregut surgeons may be presented with the difficult prospect of delaying reconstruction after a foregut catastrophe during their career. Depending on their complexity, they may then choose to refer the patient to another facility with more expertise and experience in this area.
There are few studies discussing the timing and management of patients undergoing delayed esophageal reconstruction. Barkley and colleagues from the University of Michigan described a similar series in 2004 of 40 patients over a 29-year period and had similar results, a 68% complication rate, a 20-day average length of stay, and generally good functional outcomes. 4 Orringer also published a narrative review describing technical considerations for these complex and challenging cases. 12 Raymond and Watson also published a brief review to guide clinical decision-making, 13 but there remains a paucity of studies describing outcomes in this rare patient population.
Patients unsuitable for immediate reconstruction will often present with sepsis, nutritional depletion, and poor hemodynamic reserves. 6 Achieving hemodynamic stability can be challenging and may require aggressive measures. Debridement of mediastinal and pleural spaces may be necessary. Patients may be referred with a chronic esophageal leak that has perhaps fistulized after failed attempts at repair, as witnessed in 8 of our patients. These patients may require definitive esophagectomy and diversion procedures to treat their chronic, low-grade sepsis as a first step toward healing. A transthoracic approach provides the dual advantage of good access to the entire length of the esophagus and an opportunity to wash out the thoracic space.
In rare cases, foregut discontinuity may be electively performed as a first-stage surgery, as was performed in one of our patients presenting with a new gastric malignancy in her neo-esophagus following a transhiatal esophagectomy done several years prior. In this case, she was deemed too frail to undergo a prolonged operative procedure to include immediate reconstruction using colon. The excision of the malignant lesion took precedence over having a functional foregut for a period of time in this patient.
Prior to definitive reconstruction, our patients were assessed for readiness using the following criteria: 1) they were free of all infectious processes; 2) they were tolerating tube feeds at goal; 3) they had gained or maintained their body weight; 4) they were off narcotics; and 5) they had a soft abdomen on clinical examination. Meeting these requirements can take considerable time, as evidenced by the median period of foregut discontinuity of 277 days. Therefore, regular encouragement to the patient and deliberate restraint and patience are recommended during this time, which can be a source of anxiety for both patient and surgeon. It may be observed rarely that the advantage from tube feeds plateaus and that the patient is unable to attain what would be considered an optimal nutritional state. This may be the case when tube feeds are poorly tolerated. Having additional anastomoses and dissection adds to the prolonged operative time and surgical stress during reconstruction and underscores the prerequisite for the patient to be as physically fit as possible prior to surgery. Patients generally only have 1 chance at a successful reconstruction, so its timing and conduct must be optimal.
Post-reconstruction, the majority of our patients had a good overall outcome. Our multidisciplinary healthcare team, which includes the foregut surgeon, midlevel provider, and nurse coordinator, provided considerable counseling to the patients and their family members with regard to preoperative requirements, setting reasonable expectations following reconstruction. We believe this to be critical to the high rate of postoperative satisfaction in our patient cohort. It is important to counsel the patient that their swallowing will never be normal again, and despite using the best available conduit, it is never as good as the native esophagus.
A foregut surgeon should not expect to have many of these cases, but they should be prepared to handle them should the need arise. In the event that immediate reconstruction is not feasible, a surgeon’s safe recourse can be to staple off the esophagus proximally and, if possible, bring it out as a spit fistula in the neck. This step will ease the reconstructive surgery by mitigating the need to dissect the esophageal remnant from a scarred space at the time of staged reconstruction.
Patients who require partial or near total esophagectomies but are unable to undergo immediate reconstruction as they are too ill or frail to do so. Fortunately, they comprise only a small population. Even an experienced foregut surgeon will likely only take care of 1 or maybe 2 of these patients in any given year. The vast majority of these patients are not candidates for immediate reconstruction because of sepsis and are the result of elective surgeries gone awry, oftentimes performed minimally invasively. They must survive not only the initial insult but also the reconstruction, which must be specifically tailored to their anatomy.
There are a number of lessons learned over the last 2 decades:
Do what is necessary to overcome the initial insult. If a surgeon is comfortable with reconstructive techniques, then they will be less likely to withhold needed surgical resection of necrotic tissue, although eventual reconstruction should always be kept in mind.
Leave as much length of proximal esophagus as possible. Two of the patients in the series had their intrathoracic esophagus intact and were able to undergo Roux-en-Y reconstructions. Although it is taught that proximal diversion should be performed, it is the authors’ opinion that this is not absolutely necessary, as one of our patients learned to spit their saliva into a cup, avoiding the need for a nasoesophageal tube. There are several techniques used to perform a cervical esophagostomy, such as that described by Raymond and Watson where the stoma is superficially tunneled caudal to the clavicle to maintain length. 13 The stoma placement location is a matter of surgeon preference. However, the authors prefer to bring end cervical esophagostomies out over the manubrium if possible, as this provides for a stable, flat surface for ostomy bags, especially if the patient is thin. Additionally, the esophagus is sometimes shortened because of underlying disease, and the stoma must be brought out through the cervical incision, which is never preferred but sometimes necessary. Blood supply to the esophagus is robust and made possible through submucosal collaterals that can be fed via thyroidal branches. Continuing to exercise the pharyngeal phase of swallowing awaiting reconstruction should be encouraged when possible.
Enteral feeding tubes should be placed at the time of initial resection. Gastrostomy tubes can be placed and are helpful in maintaining trophic feedings to the stomach, especially when the stomach may be used as a possible conduit. Even normal-sized stomachs rarely reach to the neck in the substernal position, which is especially true if it has atrophied during the recovery phase. This is the reason that the vast majority of the patients underwent colon interpositions in our series. Gastrostomy tubes rarely interfere with reconstruction and can be taken down easily.
Surgeons who elect to do this kind of surgery need to be comfortable with neck, chest, and abdominal anatomy and thus should be thoracic and general surgery trained. One patient in the series developed a fistula between her colon and her jejunostomy feeding tube. Her stomach had been resected, and her colon was her only available conduit to reach to the neck. Rather than have a general surgeon repair the fistula, we elected to do this ourselves, as careful manipulation of the colon was essential in maintaining it as a usable conduit. She eventually had a successful substernal colon reconstruction.
Reconstruction should only be done after the patient has completely convalesced from their initial insult, such that they are maintaining weight on tube feeds, are nutritional replete, and are off all narcotics and performing activities of daily living without difficulty. One patient in the series had a cocooned abdomen, precluding placement of feeding tubes at his initial presentation. As a result, he required total parenteral nutrition for several months prior to tolerating a stand-alone jejunostomy feeding tube placement. We elected to do this prior to his reconstruction since his bowels had been at rest, and we were unsure as to how this would affect his reconstruction. After initiating tube feeds, he developed severe hyponatremia and other electrolyte imbalances that took several weeks to overcome, justifying our decision to delay reconstruction.
The reconstruction needs to be perfectly performed, as many require 3 or sometimes even 4 different anastomoses, any of which can leak or stricture.
“Service after the sale” needs to be the mantra of care. Patients may require therapeutic endoscopic procedures such as dilations and stent placements post-reconstruction. The surgeon needs to be versed in performing such techniques, which we find our gastroenterology colleagues to actually view favorably. Many of these patients can take a year or more to fully recover and resume oral diets and have to be counseled as such from the beginning. Their swallowing and eating habits will never again be normal. Having the patient and their family know from the start that their road to recovery will be long and—at times—difficult can be helpful in maintaining realistic expectations.
This study has a few important limitations. First, due to the rarity of these circumstances, the sample size is quite low, limiting our ability to perform sophisticated statistical analysis or to meaningfully compare groups. Thus, our goal was to instead describe our experience with this uncommon clinical scenario. Second, we assessed patient-reported quality-of-life, but only at a single time point, which varied in duration following reconstruction. Future studies would ideally assess quality-of-life in a longitudinal manner. Last, all reconstructions were performed by 1 foregut surgeon at a single institution, which may limit generalizability to other surgeons in different practice settings. However, the aim of this study was to add to the literature describing this rare, highly morbid situation.
Conclusion
Management of patients who require a period of esophageal discontinuity is a multidisciplinary effort that includes the foregut surgical team, the medical intensivist, critical care nursing, dieticians, physical therapists, and rehabilitation specialists which become involved early in the patient’s postoperative course and throughout their recovery. Delayed esophageal reconstruction can be performed with acceptable outcomes with careful preparation and often require lengthy recoveries. Referral to an advanced foregut center should be considered for patients requiring esophagectomy with delayed reconstruction.
Supplemental Material
sj-docx-1-gut-10.1177_26345161231212388 – Supplemental material for Delayed Esophageal Reconstruction: Indications, Techniques, and Outcomes
Supplemental material, sj-docx-1-gut-10.1177_26345161231212388 for Delayed Esophageal Reconstruction: Indications, Techniques, and Outcomes by John A. Treffalls, Christian P. Jacobsen, Nitin A. Das, Rebecca A. Medina, Marc Koch, Daniel T. DeArmond and Scott B. Johnson in Foregut
Footnotes
Authors’ Note
Presented at the 68th annual meeting of the Southern Thoracic Surgical Association, Atlanta GA, Nov 5, 2021.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.