Abstract
Esophageal Squamous Cell Carcinoma (ESCC) accounts for a significant portion of esophageal cancer cases worldwide, particularly in high-risk regions like China, Iran, and eastern Africa. It is often diagnosed at advanced stages, leading to poor prognosis and limited treatment options. Image-enhanced endoscopy enables precise visualization of abnormal mucosa, aiding in targeted biopsies for early detection of ESCC and reducing unnecessary sampling. Various techniques have emerged including lugol’s chromoendoscopy, narrow-band imaging, confocal laser endomicroscopy, and high-resolution microendoscopy. This article also discusses advancements in post-processing imaging technology and the novel role of Artificial Intelligence (AI) in improving diagnostic accuracy. While image enhanced endoscopy is promising, challenges such as training, standardization, and long-term validation remain. Implementing image enhanced endoscopy, combined with advancements in AI, has the potential to transform ESCC diagnosis and significantly improve patient outcomes, especially in high-risk populations.
Keywords
Introduction
Definition & Epidemiology of Esophageal Squamous Cell Carcinoma
Esophageal cancer is the sixth leading cause of cancer mortality worldwide, and 90% of these cases are esophageal squamous cell cancer (ESCC), which results in 600 000 cases yearly. 1 The distribution of ESCC exhibits significant geographic variation, with high prevalence regions such as China, Iran, and eastern Africa showing tenfold higher incidence rates than other regions. In terms of gender distribution, ESCC occurs more frequently in males (69%) than in females (31%) and its incidence increases with age. 1 The etiology of ESCC is multi-factorial and strongly population dependent. Risk factors for developing squamous neoplasia include tobacco smoking, heavy alcohol consumption, poor nutrition, dietary deficiencies, thermal injury and exposure to environmental carcinogens. In early stages, patients may be asymptomatic or experience mild symptoms such as dysphagia or chest discomfort. As the tumor progresses, more severe symptoms may develop, including weight loss, worsening dysphagia, regurgitation, gastrointestinal bleeding. However, these symptoms are nonspecific and can be present in other esophageal disorders as well, making early diagnosis challenging.
Importance of Early Detection and Diagnosis
Early detection and diagnosis of ESCC plays a critical role in improving patient outcomes and reducing mortality rates. A significant proportion of ESCC patients are diagnosed at advanced stages, with 5-year survival rates less than 20% in developed nations and dropping below 5% in developing countries where the majority of cases arise.2,3 Furthermore, despite therapeutic advancements, the prognosis for patients identified after symptom onset has remained stagnant over the past few decades (Figure 1) 2 ; conversely, when early-stage carcinoma is managed through endoscopic or surgical intervention, a considerably improved 5-year survival rate of 80% to 90% is observed. 4 At the early stages of ESCC, when the tumor is localized and has not spread to nearby lymph nodes or distant sites, curative treatment approaches such as surgery, radiation therapy, and targeted therapies are more feasible. Thus, the imperative for early detection approaches becomes apparent, as this holds the potential to substantially improve ESCC patient outcomes. Early detection of ESCC allows for the implementation of screening and surveillance programs for high-risk individuals. Individuals with risk factors such as tobacco and alcohol use, family history of ESCC, or a history of precancerous lesions can undergo regular screenings and endoscopic surveillance to detect dysplastic or early cancerous changes. This proactive approach enables the identification of ESCC at its earliest stages when treatment outcomes are most favorable. Furthermore, early diagnosis provides an opportunity for personalized treatment strategies tailored to individual patients. Early-stage ESCC allows for comprehensive assessment, including molecular profiling and genetic testing, which can guide treatment decisions and identify targeted therapies for specific mutations or biomarkers. Additionally, early detection and treatment of ESCC can be more cost-effective compared to managing advanced-stage disease, especially in areas of high prevalence and limited resources.5,6 Timely identification allows for a broader range of treatment options, such as lower cost, minimally invasive therapies, and stresses the importance of implementation of screening programs for high-risk individuals. Investing in early detection efforts is essential to reducing the burden of this aggressive cancer and improving patient outcomes.
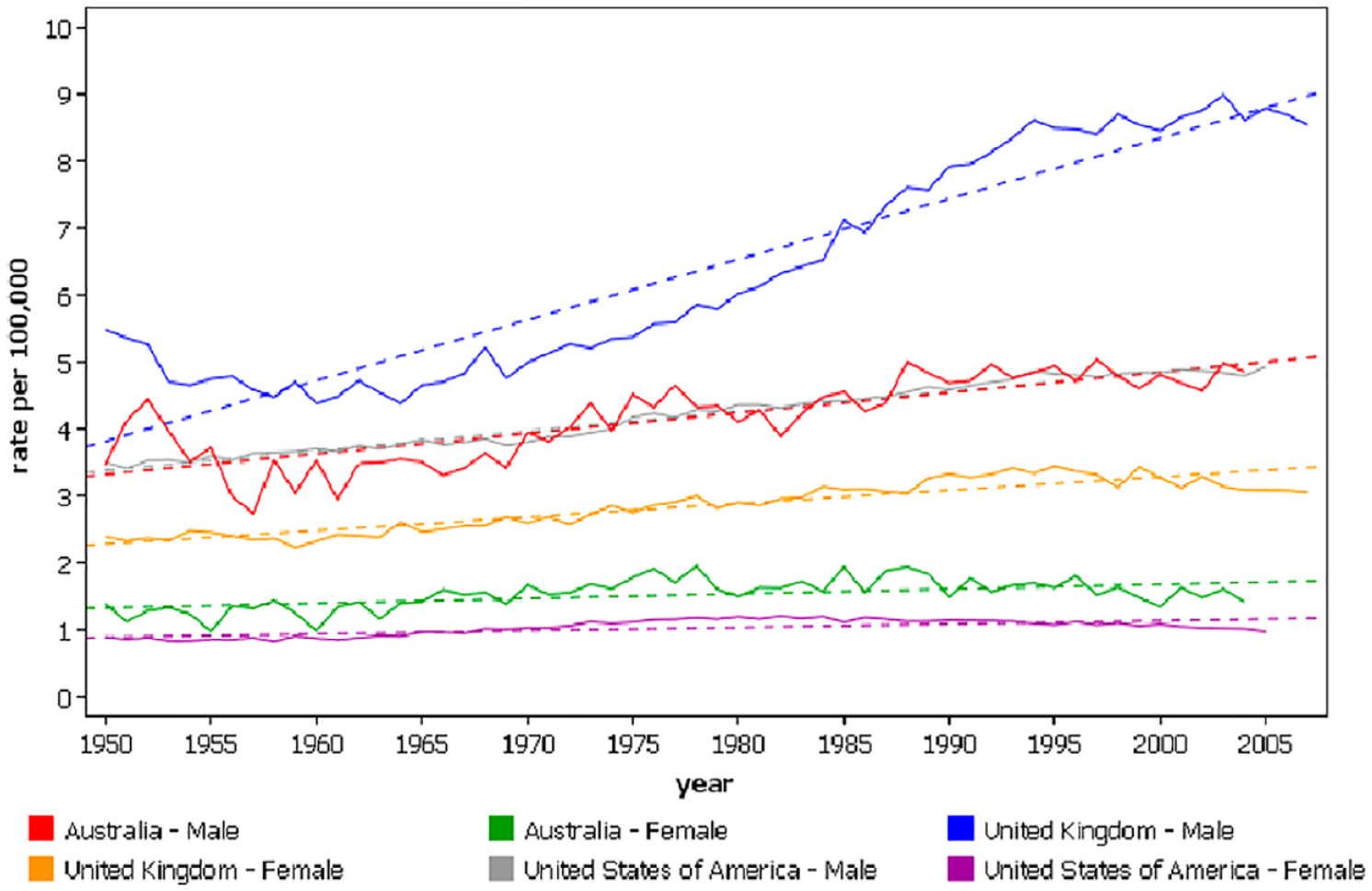
Mortality from esophageal cancer in westernized nations experiencing increased incidence, age-standardized rates (WHO mortality database, International Agency for Research on Cancer).
Overview of Traditional Endoscopy Limitations
Although conventional white light imaging (WLI) exhibits a notable level of precision in identifying ESCC, its sensitivity in detecting squamous dysplasia is comparatively lower due to the minimal vascular alterations observed in comparison to the normal squamous mucosa. 7 More specifically, traditional endoscopy may struggle to detect flat or subtle lesions associated with early-stage ESCC (Figure 2). 8 Furthermore, endoscopic biopsies rely on targeted sampling of suspicious areas, which may introduce a sampling bias. This means that small or focal lesions may be missed during the biopsy process, resulting in false-negative results.
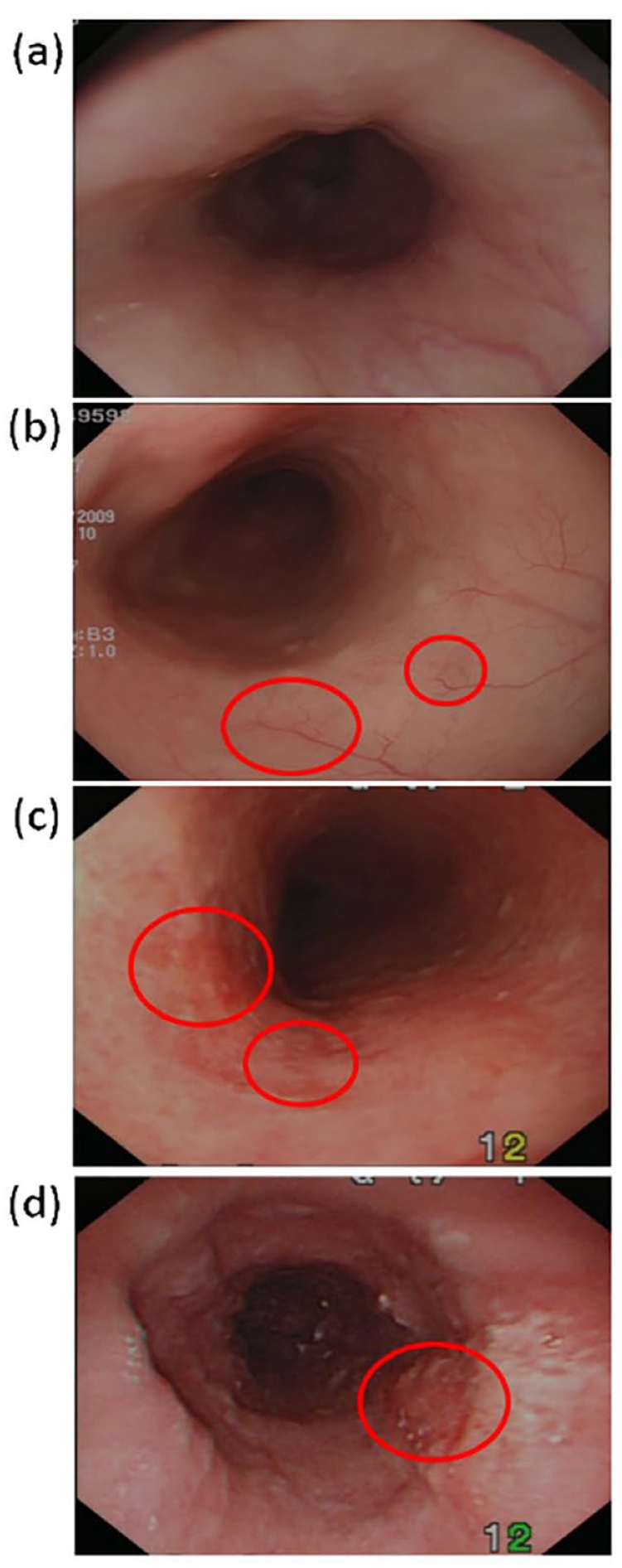
Images of white-light endoscopy: (a) normal, (b) low-grade dysplasia, (c) high-grade dysplasia, and (d) squamous cell carcinoma. Photos courtesy of Wu et al. 8
Indications for Image-Enhanced Endoscopy
Image-enhanced endoscopy has several indications in the evaluation of ESCC and its precursor lesions such as dysplasia. These include the identification and characterization of suspicious lesions, delineation of lesion margins for endoscopic therapy, and surveillance of high-risk individuals. Image-enhanced endoscopy techniques can aid in the detection of subtle or flat dysplastic lesions that may be missed using conventional white light endoscopy alone. During routine non-magnified endoscopy, intra-mucosal cancer typically manifests as flat lesions, exerting minimal influence on the morphology of the mucosal surface. This characteristic renders its identification challenging. 8
Definition and Types of Image-Enhanced Endoscopy
Image-enhanced endoscopy encompasses various modalities that utilize optical filters, chromoendoscopy, or digital processing techniques to enhance the visual contrast and detail of the mucosal surface. 9
Chromoendoscopy
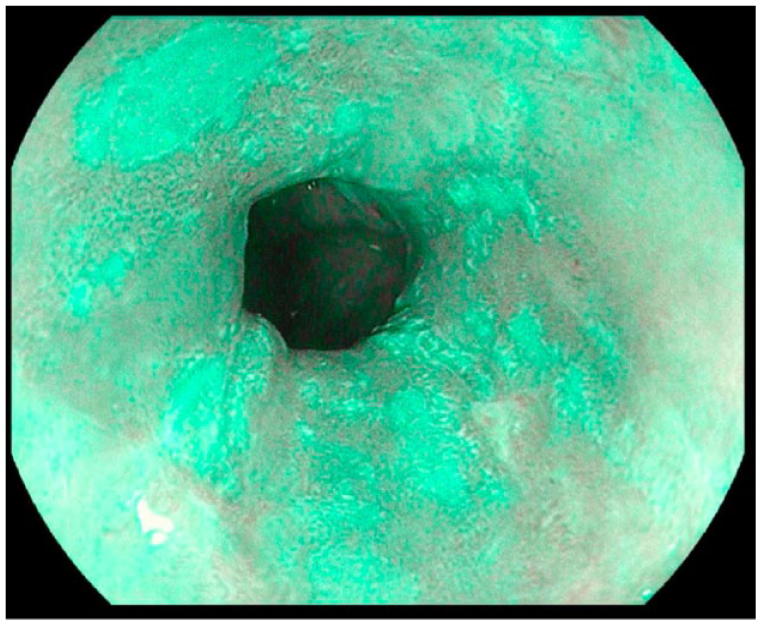
Traditionally, the gold standard for detecting ESCC has been Lugol’s chromoendoscopy (LCE) using iodine to stain the glycogen stored in mature esophageal squamous mucosa. 10 With LCE, abnormal and neoplastic mucosa appears unstained and can be targeted for biopsy, imaging or endoscopic therapy. Unfortunately, LCE can be time-consuming, inefficient (high false positive rate) and can induce mucosal irritation which has limited its use in practice. 11 The application of LCE has demonstrated efficacy in enhancing endoscopic detection and highlighting high-grade dysplasia and early squamous cell carcinoma in the esophagus among populations at high risk. 12 Lugol’s solution comprises a combination of potassium iodide and iodine dissolved in water. Through the administration of a diluted solution (ranging from 0.5% to 3.0%), typically via a spray catheter, the glycogen-containing cells in the mucosa absorb the solution, resulting in a dark green-brown stain (Figure 3). Lugol’s solution has occasionally been associated with discomfort, including retrosternal pain, mild nausea, and, in rare cases, chemical esophagitis which may be relieved by the use of N-acetylcysteine. 11 In a prospective observational study of 326 patients, standard endoscopy was followed by LCE. 13 Two hundred twelve biopsied areas deemed to be “endoscopically normal” by standard endoscopy were biopsied because they were unstained by LCE. A total of 678 biopsies were collected from the 326 patients included in the study, of which 8 lesions of high-grade intraepithelial neoplasia (HGIN) and 6 lesions of low-grade intraepithelial neoplasia (LGIN) were diagnosed. In cases of HGIN & LGIN, standard endoscopy could only detect 55% and 33% of lesions, respectively, while LCE was 100% sensitive. While inexpensive, its use in North America has largely been replaced by NBI for practical purposes. However, it remains a reliable, low cost solution for a majority of the developing world. 14
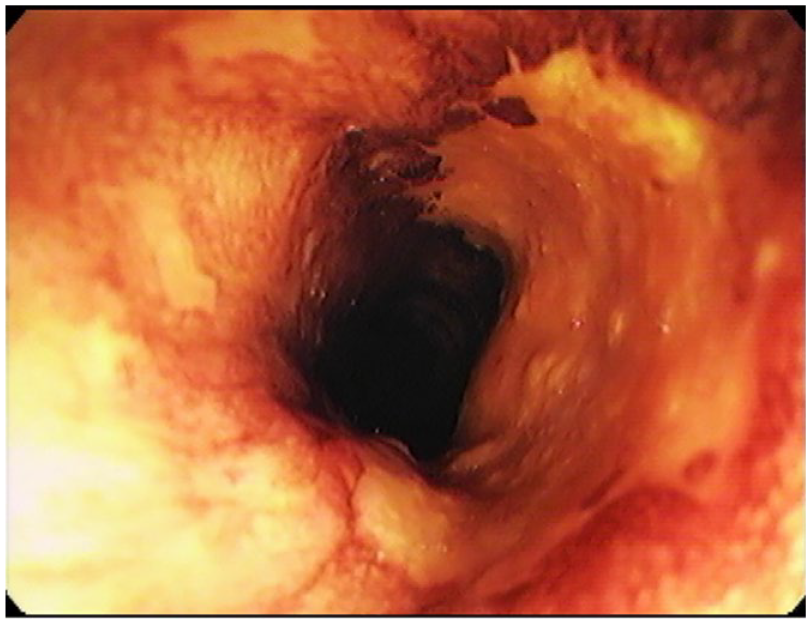
Lugol’s chromoendoscopy showing several glycogen depleted areas suggestive of esophageal dysplasia.
Narrow-Band Imaging (NBI)
NBI, the most researched and commonly used form of virtual chromoendoscopy, employs 2 distinct types of light with specific wavelengths through an optical filter: blue light (415 ± 30 nm) and green light (540 ± 30 nm). By selectively absorbing hemoglobin in capillaries situated on the mucosal surface, the NBI system accentuates capillary vessels (Figure 4). 15 Under NBI, esophageal dysplasia and neoplasia exhibit a brown-black color, while the surrounding healthy tissue appears blue-green. 16 In contrast to Barrett’s esophagus, squamous epithelium does not possess mucosal contours that enable the visualization of pit pattern distortions. Therefore, NBI is crucial for providing high contrast images of the microvascular structure of the esophagus. NBI can visualize normal capillary mucosal vessels consisting of intraepithelial papillary capillary loops (IPCLs) and submucosal vascularity (branching vessels). 17 In cases of squamous neoplastic lesions, IPCLs demonstrate distinctive alterations in morphology. In dysplastic lesions, they become dilated, tortuous, and irregular, while in squamous cell carcinomas, they undergo destruction and replacement by tumor vessels. These progressive atypical alterations inform the endoscopist to anticipate the level of neoplasia, the depth of invasion, and assess the feasibility of curative endoscopic resection as a treatment option. In a landmark 13-year prospective study, squamous dysplasia and carcinoma in situ were the only histological lesions associated with a significantly increased risk of developing ESCC. 18 IPCL changes are the first signs of inflammatory mucosa progressive to dysplasia thus making their early identification with NBI crucial. 19 Numerous studies have proven the benefit of NBI in detecting dysplasia and ESCC. Superficial mucosal changes prevent sensitive detection with WLI, thus NBI serves a special advantage to detect them. In a prospective, randomized controlled trial, 97% of superficial lesions were diagnosed by NBI while only 55% were detected by WLI resulting in a significantly higher sensitivity for the former. 20 In order to decrease mortality in ESCC, an increase in yield of endoscopic exams would be needed which makes studies screening high risk patients of particular interest. In a study of patients with existing head & neck cancer, the sensitivity & specificity of NBI endoscopy for detecting ESCC and HGIN was over 90% and 95%, respectively. 21 Similarly, a prospective study of 69 patients with known head & neck cancer, NBI was particularly helpful for detecting dysplasia with 13 lesions seen as opposed to only 3 with conventional endoscopy. 22 When comparing NBI to Lugol chromoendoscopy, multiple studies revealed that NBI demonstrates improved accuracy and specificity, while maintaining similar sensitivity to LCE. In a propensity-matched prospective trial, while the sensitivity did not differ between the 2 tests, NBI was found to have the superior accuracy (77% to 68%) to LCE. 23 Another prospective observational study on patients with head and neck tumors showed that NBI and LCE were both equally sensitive at detecting ESCC. 24 Furthermore, a large meta-analysis with nearly 2000 patients, NBI and LCE had similar sensitivities and specificities (88%vs 92%; 88%vs 82%) for diagnosing high-grade dysplasia or squamous cell carcinoma. 25 The real advantage NBI can offer is a more efficient and safer diagnosis. Studying 300 randomized patients, Chaber-Ciopinska et al demonstrated that with a lower number of biopsies required, shorter esophagus examination time and better patient tolerance, NBI outperformed LCE in screening high-risk populations for squamous cell esophageal neoplasia. 26 In another prospective trial by Gruner et al NBI showed greater specificity than LCE in patients with previously diagnosed ESCC. 27 Similar to NBI, Blue laser imaging (BLI) is a relatively novel image-enhanced endoscopy technique that has garnered interest for early detection of upper gastrointestinal cancers. BLI has 2 different laser wavelengths that enable narrow band light observation providing enhanced contrast between dysplastic tissue and normal tissue. In a retrospective analysis of 25 superficial ESCC cases, by both subjective and objective measures, BLI detected lesions significantly more than NBI and WLI. 28 BLI maybe useful for detecting superficial and early dysplastic lesions of the esophagus.
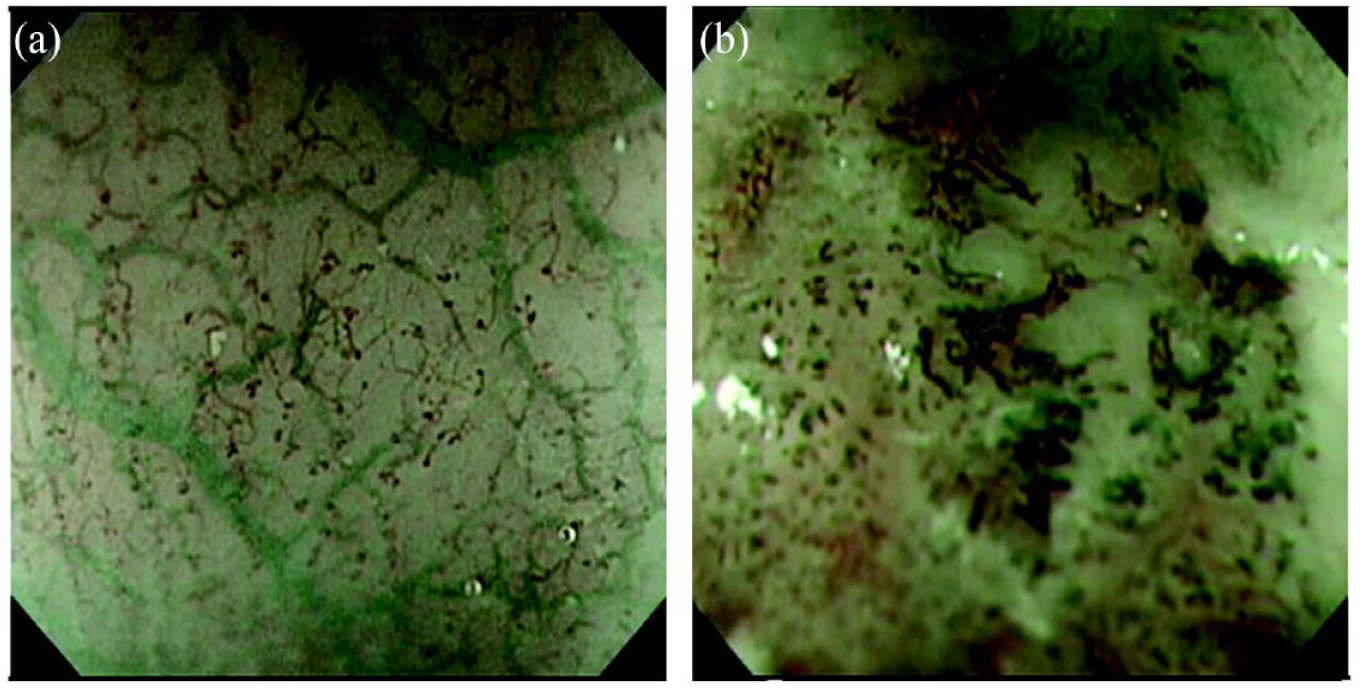
Narrow band imaging with magnification of (a) small, non-dilated looped vessels with no gross abnormality (b) gross vessel dilation with loss of loop structure suggestive of early squamous cell neoplasia. (b) Photos courtesy of Everson et al. 15
High Magnification Modalities
Confocal laser endomicroscopy (CLE) is an imaging technique capable of real-time visualization of the mucosal layer of the gastrointestinal tract with a magnification of up to 1000 times. This sophisticated modality relies on the illumination of tissue using a low-power laser and the capture of fluorescent light reflected back from the tissue through a pinhole located within the same focal plane. By leveraging the high spatial resolution of CLE, it becomes possible to obtain subsurface cellular imaging and examine intricate tissue architecture with remarkable precision. Liu et al demonstrated the ability of CLE to distinguish normal mucosa from cancerous lesions by capturing a higher proportion of irregularly arranged squamous epithelial cells and IPCLs showing its potential for detecting early ESCC. 29 A prospective study by Guo et al validated CLE’s use for performing targeted biopsies of microscopic abnormalities and thus reducing the number of unnecessary biopsies. 30 In a single center study by Huang et al, compared with those of LCE alone, the sensitivity (89.1%vs 95.7%) and negative predictive value (64.3%vs 81.8%) of CLE combined with LCE were significantly improved. 31 The studies described above demonstrate that using CLE can reliably distinguish between the normal esophagus and ESCC, emphasizing the potential of CLE for early detection of ESCC and allowing for timely treatment. However, training is required in operational techniques for locating CLE probes in the area of interest, and working with a pathologist is required to gain a thorough knowledge of mucosal histopathology. In addition, obtaining adequate images of the esophagus is a potential challenge for CLE.
In recent years, high-resolution microendoscopy (HRME) has garnered some interest as a low-cost alternative that can provide subcellular images. Through the endoscope’s accessory channel, a fibro-optic microendoscope can provide a clear delineation of cellular features such as size, crowding, pleomorphism in real time in order to help differentiate neoplastic/dysplastic epithelium from benign tissue (Figure 5). In an international, multi-center, prospective trial, 147 consecutive high-risk patients underwent WLI followed by LCE and HRME.
32
On a per-biopsy analysis, sensitivity of LCE versus LCE + HRME was 96%versus 91% (
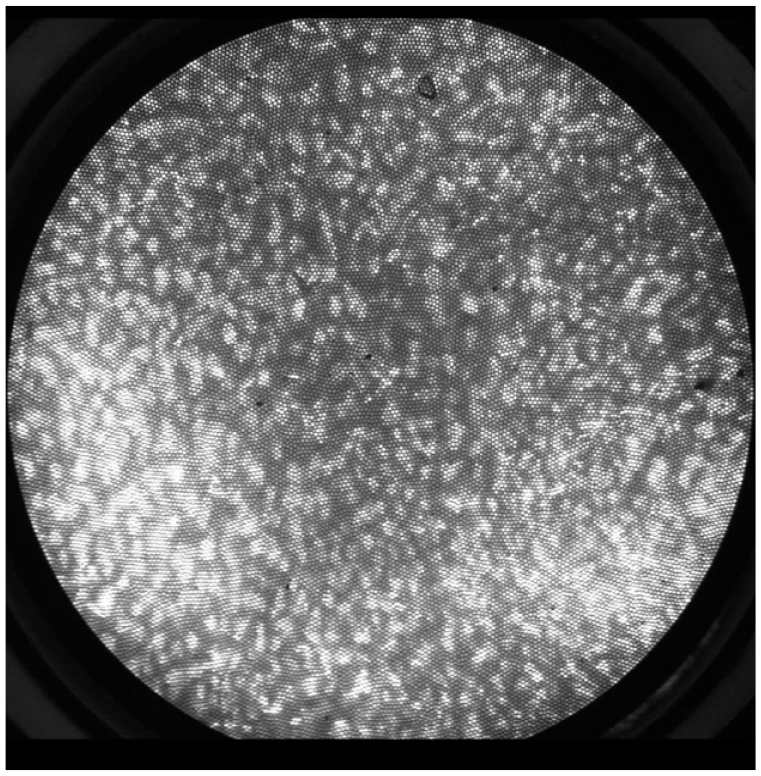
High Resolution Microendoscopy (HRME) with high grade dysplasia characterized by large, crowded, pleomorphic nuclei.
Post-Processing Imaging Technology
Several advancements driven by software have facilitated real-time improvements in mucosal sharpness, image clarity, and image processing. The i-SCAN technology developed by Pentax in Tokyo, Japan, utilizes techniques such as surface enhancement, contrast enhancement, and tone enhancement to enhance the quality of endoscopic images. 33 The i-scan technology incorporates real-time mapping technology within the endoscopic processor through post-image acquisition software. The endoscopic processor is equipped with computer-controlled digital processing capabilities, enabling a resolution of 1.25 megapixels per image to allow for the analysis and modification of luminosity data on a per-pixel basis. Surface enhancement (SE) improves the contrast between light and dark areas, enhancing the visibility of edges and lesion borders. It achieves this by making dark areas appear darker and light areas appear lighter. Contrast enhancement (CE) slightly reduces the red and green components of the white light image, while introducing a subtle blue hue to darker or depressed areas of the mucosa. This adjustment facilitates the detailed observation of subtle irregularities on the mucosal surface, allowing for the identification of “peaks and valleys” in its topography. Tone enhancement (TE) involves the comprehensive analysis of all 3 color components (red, blue, and green) of the white light image. It then selectively suppresses the dominant red component, resulting in an image with enhanced contrast between blue and green. This manipulation enhances the detection of subtle mucosal abnormalities. Through the integration of these algorithms, the i-scan technology offers improved visualization and detailed examination of the mucosal surface, enabling the identification of subtle abnormalities that may have been previously overlooked (Figure 6).
iScan Optical Enhancement showing detailed topography suggestive of superficial early squamous cell carcinoma.
Similarly, the Fuji intelligent chromoendoscopy (FICE) developed by Fujifilm Medical Co., in Saitama, Japan, utilizes spectral estimation technology to improve image quality. 34 This advanced technique involves extracting and processing dedicated wavelength images to generate improved visuals. By extracting red, green, and blue images from the endoscopic image, FICE applies arithmetic processing within the wavelength range of 400 to 695 nm to produce an enhanced image. While both FICE and NBI aim to highlight blood vessels, they employ different approaches. NBI relies on an optical filter, whereas FICE utilizes a spectrum estimation technique. Unlike NBI, which involves irradiating light of specific wavelengths, i-scan and FICE recreate images through computer calculations based on the images acquired from white light endoscopy. This distinction sets i-scan and FICE apart, as they rely on computational algorithms to generate images with enhanced visual characteristics. While current studies on I-Scan and FICE are limited to detection of other diseases such as non-erosive reflux disease, Barrett’s esophagus, gastric cancer and colon adenoma detection, Li et al found FICE to be 93% accurate in detecting ESCC. 35 Further validation and larger scale studies for the diagnosis of ESCC are needed.
Artificial Intelligence
With the emergence of novel endoscopic techniques, the interpretation of multiscale and multidimensional clinical data has become a crucial but challenging task. Furthermore, current modalities for detection of ESCC can be limited by interobserver variability.36,37 Recently, artificial intelligence (AI) systems have gained international interest to solve both clinical and non-clinical solutions. Particularly in the field of gastroenterology, direct visualization of the gastrointestinal tract is ideal for deep learning assisted AI modules such as the GI Genius Intelligent Endoscopy Module (Medtronic). 38 Fukuda et al used over 28 000 endoscopic images to train an AI system for the diagnosis of superficial ESCC. 39 While the specificity of detecting ESCC was lower with AI, the characterization (differentiation between cancer and noncancer) was significantly improved with AI over expert endoscopist review of video images (88%vs 75%). Similarly trained multiple AI systems have been proposed offering equal to or greater than sensitivity in detecting ESCC depth (and, therefore, eligibility for endoscopic therapy) compared to experienced endoscopists.40,41 Using convolutional neural network training, Tokai et al developed an AI system to estimate the depth of invasion compared with values obtained by expert endoscopists and resulted in a remarkable accuracy of 80.9% (225/279). 42 While the exact role of AI in endoscopic diagnosis remains to be seen, it is hard to imagine a future in image-enhanced endoscopy without its implementation. That said, considerable work needs to be done in ensuring safe, ethical and acceptable implementation of AI-based imaging.
Cost Effectiveness & Rationale of Screening Programs
In countries with low incidences of ESCC, such as the United States, implementing screening programs is not cost effective. Therefore, the American Gastroenterology Association guidelines do not advocate widespread screening for ESCC.
43
Nonetheless, in areas where the disease is endemic, targeted screening efforts may be warranted for individuals above a certain age. Additionally, specific high-risk groups, including patients with head and neck cancers, tylosis, or a history of caustic ingestions, may also benefit from screening. In areas with high-risk populations such as China, early diagnosis and treatment may provide significant cost savings compared to diagnosing ESCC in late stage disease.
5
Hur et al showed that HRME could be a cost effective tool for endoscopic screening and surveillance programs in China for average and high risk populations.
44
In another analysis by Yang et al, LCE resulted in substantial reduction in cost when screening in high risk areas of China.
5
Additional considerations need to be taken into account, including the screening frequency, the potential complications associated with endoscopy due to its invasive nature, and the availability of healthcare resources. In regions where ESCC is prevalent in China, a trial involving community assignment and a 10-year follow-up revealed a 33% reduction in ESCC-related mortality in intervention communities.
45
This reduction was observed when adults between the ages of 40 and 69 underwent endoscopy screening with LCE. In the United States, there may be evidence supporting the use of endoscopic screening in individuals with a history of head & neck squamous cell cancers. Su et al demonstrated a higher prevalence of secondary ESCC among head-and-neck cancer patients who underwent routine endoscopies compared to those who did not undergo screening (4.5%vs 3.0%;
Conclusion
ESCC represents a significant global health burden, particularly in high-risk populations. The early detection and accurate diagnosis of ESCC are crucial for improving patient outcomes and reducing mortality rates. In recent years, image-enhanced endoscopy techniques have emerged as valuable tools in the screening, surveillance, and management of ESCC. Proven modalities such as narrow band imaging combined with more recent advances in artificial intelligence have shown great potential in improving the visualization of subtle mucosal changes associated with ESCC. These techniques have enabled endoscopists to identify suspicious lesions with greater precision and accuracy, leading to earlier intervention and improved patient outcomes. Moreover, image-enhanced endoscopy has the potential to guide targeted biopsies, reducing the need for unnecessary sampling and optimizing tissue diagnosis. The ability to accurately differentiate between benign and malignant lesions, as well as to identify high-risk precancerous lesions, allows for tailored management strategies, such as endoscopic resection or close surveillance. This not only improves patient care but also reduces the burden on resource constrained healthcare systems. The implementation of image-enhanced endoscopy techniques in high-risk populations, particularly in endemic areas, has the potential to transform ESCC-related morbidity and mortality. Despite the numerous advantages of image-enhanced endoscopy, several challenges need to be addressed for its widespread adoption. Training and expertise in interpreting image-enhanced endoscopic findings are paramount to ensure accurate diagnosis and appropriate management decisions. Standardization of terminology, classification systems, and diagnostic criteria is essential for consistency across different endoscopic centers and research studies. Long-term follow-up studies are necessary to assess the durability of image-enhanced endoscopy findings and to establish its role in surveillance programs. Future research efforts should focus on validating the efficacy of image-enhanced endoscopy in larger, multicenter trials, and the role of AI in overcoming issues of training and expertise. With ongoing advancements in technology and increasing expertise in interpreting image-enhanced endoscopic findings, these techniques hold great promise for the future of ESCC management.
Footnotes
Author Contributions
S.A. was the senior author of the review. They ensured the preservation of data accuracy and integrity at all stages, agreed to be accountable for all aspects of the article, were in charge of the overall direction and planning of the review, and contributed to the writing of the manuscript. S.V. reviewed and synthesized literature for the manuscript and primarily wrote the review article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
