Abstract


Insulin delivery and glucose monitoring technology in type 1 diabetes (T1D) care has rapidly advanced with the goal of improving glycemia to be closer to that of people without diabetes. Insulin pump and continuous glucose monitor (CGM) sensor integration has progressed from low-glucose and predictive low-glucose suspension of insulin delivery when sensor glucose crosses (or is predicted to cross) the low threshold level to algorithm-driven partially automated insulin delivery (AID) based on sensor readings with manual meal and correction boluses. Further advances have included autocorrection boluses, meal detection, and offering multiple target set points.1-3 One AID system, the iLet Bionic Pancreas (BP), provides adaptive closed-loop algorithms enabling fully autonomous insulin delivery and automatically titrating all therapeutic insulin, including basal, correction, and prandial insulin. Instead of precise carbohydrate counting, the system requires qualitative meal announcements to autonomously determine prandial insulin. The user's body weight is the only input needed to initiate the system because it perpetually learns and automatically refines dosing, no longer requiring manually calculating, programming, and adjusting system settings. 4 Fully autonomous insulin dosing with meal announcements represents the next stage in AID development and is changing the paradigm for clinical management of insulin therapy in T1D care.
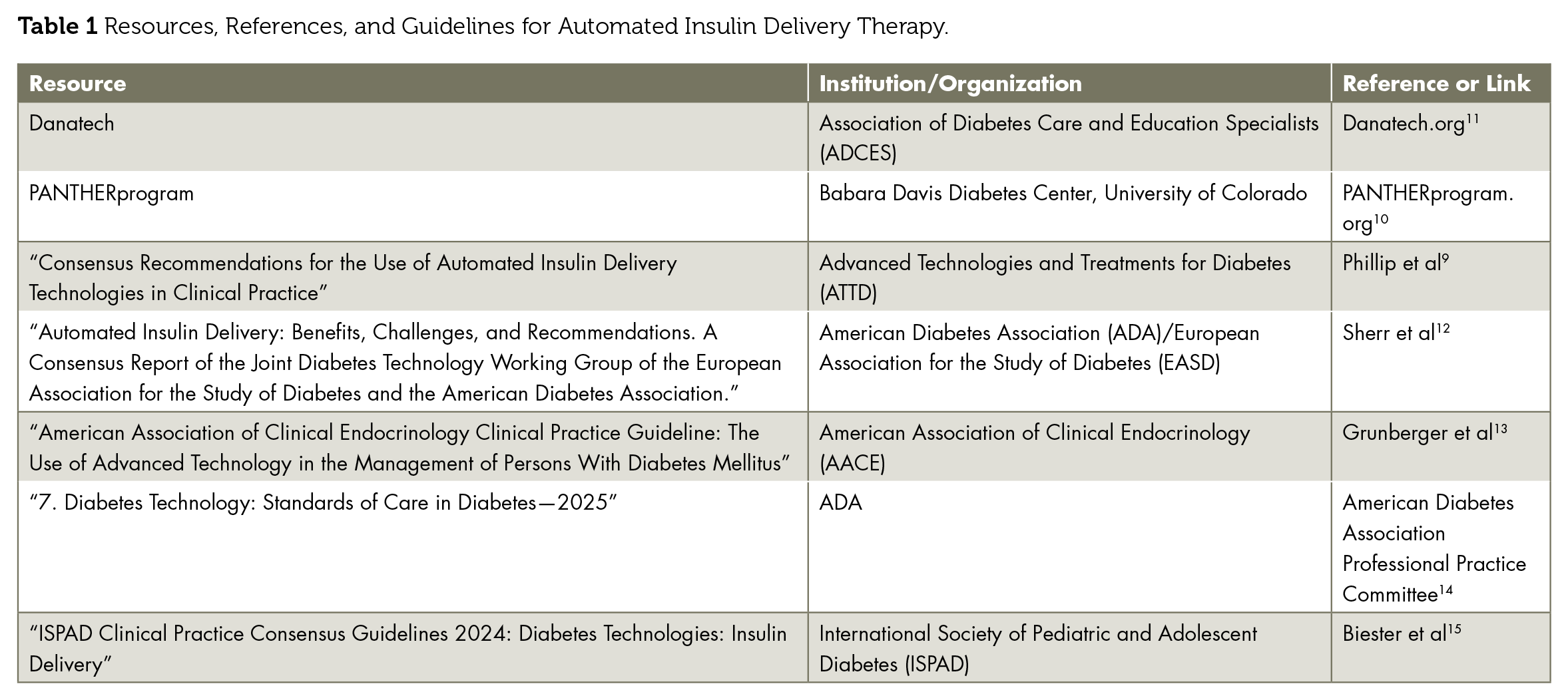
The ability of AID systems to improve glycemia with less effort on the part of users and to compensate for disruptions in daily routines, unplanned physical activity, carbohydrate miscalculations, or missed boluses, and so on is valued by users.5-8 However, continued reliance on an algorithm for glycemic management could lead to forgetting how to carry out key tasks, referred to as “deskilling,” for example, carbohydrate counting, giving injections, checking blood glucose, and so on. This can be a concern in the case of technology unavailability or failure. Thus, it is recommended that individuals using advanced technologies should have an adequate technical understanding of the system to be able to troubleshoot and also have a plan to revert to standard diabetes treatment (ie, nonautomated subcutaneous insulin delivery by pump or injections). 9 They and their clinical team should have remote access to their AID system data to help in determining manual basal, mealtime, and correction doses. For a comprehensive AID comparison table, readers are encouraged to visit PANTHERprogram.org, developed and maintained by the Barbara Davis Center for Diabetes, University of Colorado Anschutz Medical Campus. 10 The ADCES danatech.org website provides comprehensive resources, information, and training on diabetes technology. 11 See Table 1 for a listing of resources and references regarding AID use in clinical practice.
Resources, References, and Guidelines for Automated Insulin Delivery Therapy.
It is also critical to consider the potential disruption in availability of supplies, such as glucose sensors or insulin infusion sets, as experienced during an infectious disease pandemic or natural disasters or during supply chain disruptions or interruptions in data availability. Users are vulnerable when insulin infusion sets or sensors do not last their intended duration or supply interruptions occur because insurance policies change and prior authorizations are required for the necessary AID supplies. 12 Developing backup plans (Table 2) to ensure that the skills and supplies needed for basic diabetes management in these circumstances are available is critical. This includes reverting to conventional pump therapy (in the case of semiautomatic AID systems: MiniMed 780G, t:slimX2 and Mobi Control-IQ, and Omnipod 5) or manual insulin injections, monitoring ketones, and considering whether an insulin infusion occlusion or failure has led to hyperglycemia.
Backup Plan Checklist.
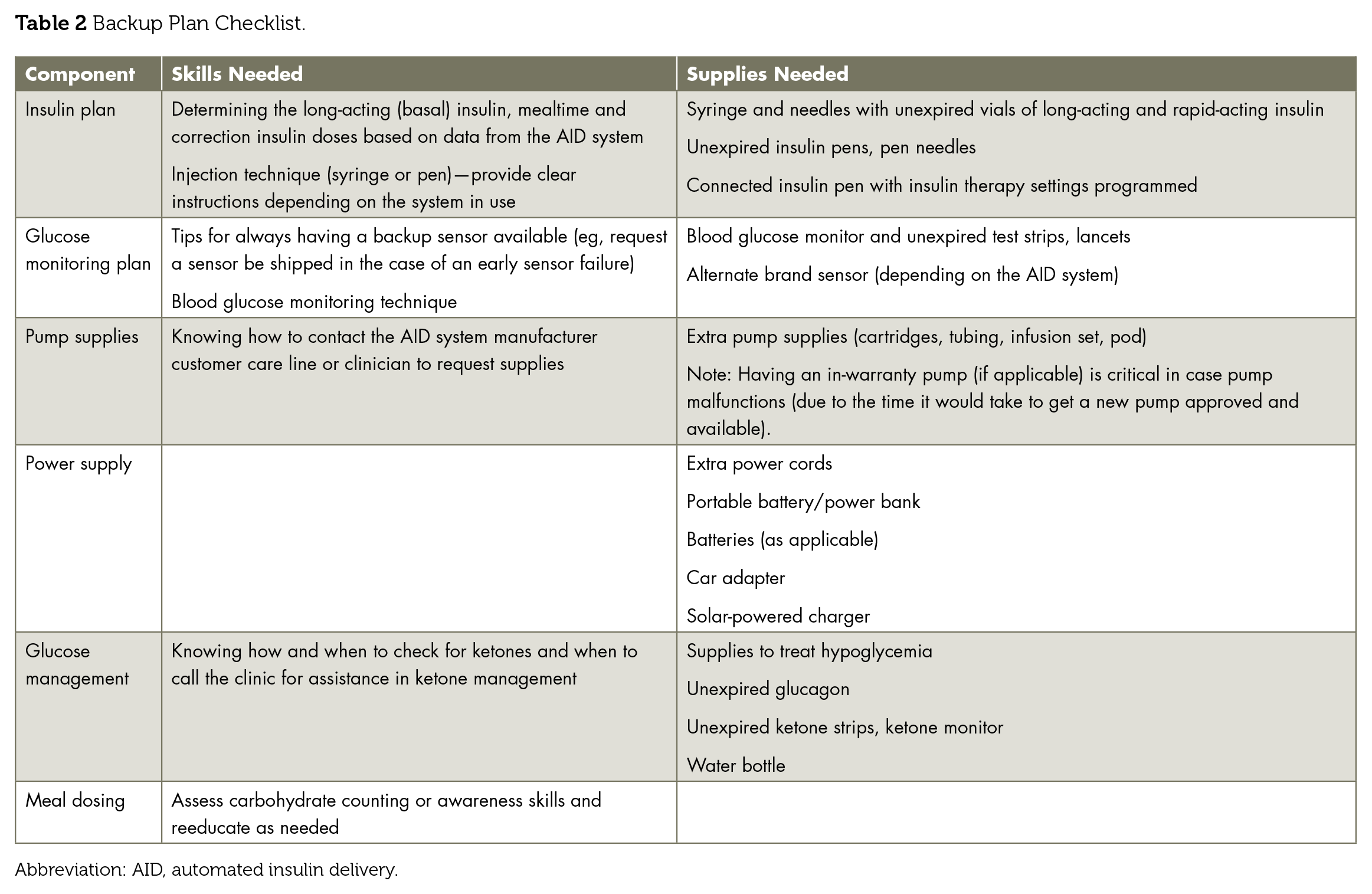
Abbreviation: AID, automated insulin delivery.
There is no option for a manual mode in the fully automated iLet BP. If the BP loses communication with the CGM, it will prompt the user to enter blood glucose values periodically. As long as the user enters blood glucose values into the system, it will continue to automate all insulin delivery based on the entered blood glucose values and previously stored information on the user's basal insulin needs. The BP can operate in blood glucose-run mode for up to 72 hours. Having a blood glucose meter and unexpired test strips and lancets is an important part of the backup plan for BP users. It is also recommended that the user request a replacement sensor from the manufacturer in the event of early sensor failure to assure adequate sensors are available in the event of a supply interruption or early sensor failure. In case of device malfunction, the BP generates and continuously updates backup insulin doses for injection or pump users, including total daily dose, average daily basal, and adapted mealtime doses. These data are readily available for both users and their clinical team to access on demand through the cloud.
The MiniMed 780G system will revert from the semiautomated Smart Guard mode to a safe basal mode if there is a loss of CGM communication or other interruption of CGM data or a concern about sensor accuracy. In this mode, the pump will deliver a basal rate determined by the algorithm but without glucose-dependent basal adjustments and no autocorrection boluses. The user must enter a blood glucose value into the pump before the “time to exit” expires to prevent automode exit. The low-glucose suspend will remain when the system transitions to manual mode. It will be critical to consider altering the low-glucose thresholds and predictive low alerts when not using the AID feature so that the user can manually respond to hypoglycemia.
With the Control-IQ and Control-IQ+ algorithm, there is no limited automation mode. If there is loss of CGM data for 20 minutes or more, the pump will operate in manual mode, delivering the programmed basal rate without glucose-dependent basal adjustments and no auto correction bolus.
The Omnipod 5 has an automated limited mode that will activate if there is no CGM data for 20 minutes or more or if there is an “Automated Delivery Restriction” alarm (if insulin has been suspended too long or if max delivery is too long). The Omnipod will deliver a basal rate determined by algorithm but without glucose-dependent basal adjustments until sensor data return or the user clears the alarm. The “Automate Delivery Restriction” alarm is intended to let the user know to step in and check their sensor, pod, and glucose level. The user acknowledges this alarm by switching to manual mode for 5 minutes or longer.
Contingency planning should include access to batteries, battery pack, charging cables, insulin infusion sets, reservoirs, insulin, syringes (or insulin pens and needles), a glucose meter and test strips, glucagon, ketone test strips, and a backup glucose sensor and transmitter for the CGM system. 13 Putting in place strategies for minimizing supply interruption will also be critical. In addition, a plan with clear instructions for transition to insulin injection therapy or standard pump therapy (if available) and a supply of unexpired insulin pens or vials with rapid-and long-acting insulins should be available for use until a replacement for the AID system is available. It will be important to consider the user's knowledge and skill base, which will vary depending if the user is newly diagnosed (eg, has never used a syringe or a blood glucose meter), has many years with diabetes but is new to pump therapy (eg, may not realize the pump may still work without a sensor), or has many years with pump therapy but is new to AID (eg, may have never used an insulin pen).
It is important to give users guidance about the use of their AID system during illness, when there may be temporarily increased insulin resistance and elevated glucose levels and reduction in oral intake and ketosis without elevated glucose levels. If the user requires use of steroids, the AID system may not be able to respond rapidly enough to account for the increased insulin requirements. 12 Some diabetes centers advocate for providing a connected insulin pen with programmed insulin therapy settings that are updated when changes occur not only as part of initial therapy upon diagnosis but also as part of the backup plans for AID users 16
In the event of hyperglycemia, users may need to return to basic diabetes management with clear instruction on how to restore normoglycemia. The diabetes care and education specialist (DCES) should remind users how to recognize signs of infusion set failure: in cases of sustained hyperglycemia above 250 mg/dL and/or after a bolus insulin correction, the glucose level does not drop by at least 50 mg/dL within 1 hour of treatment. 9 The best advice is “when in doubt, switch it out.” The DCES or designated member of the clinical team should discuss site rotation and regularly assess site and skin health with anyone on insulin therapy. 14
The DCES can take the lead in establishing clinic, practice, or health care system protocols and policies regarding all aspects of backup plans for diabetes technology and advocate for inclusion in standards of care and clinical guidelines. These include the following:
Reinforce the importance of maintaining self-management skills, including blood glucose checking, ketone checking, administering a syringe or insulin pen injection, regular review of basal insulin delivery settings or doses, calculating a correction dose of insulin in case of system malfunction, identifying diabetes emergencies, and carrying supplies to handle them. 17
Demonstrate and verify skills necessary for giving an injection and monitoring glucose without a sensor by return demonstration. This should be conducted at least annually or more frequently as needed.
Automated systems often alleviate the need for precise carbohydrate counting. Assess carbohydrate counting or carbohydrate awareness skills and reeducate as needed. 18
Remind users how to recognize signs of infusion set failure: in cases of sustained hyperglycemia above 250 mg/dL and/or after a bolus insulin correction, the glucose level does not drop by at least 50 mg/dL within 1 hour of treatment. 9
Simulate emergency scenarios where patients practice managing their diabetes without technology and provide resources for emergency assistance.
Integrate smart phrases and protocols into the electronic health record, ensuring backup plan topics are discussed and appropriate supplies are ordered on a timely basis.
Conclusion
Although advanced diabetes technologies offer substantial benefits, backup plans for addressing the associated risk of deskilling are vital. The DCES should proactively equip individuals with the skills and resources needed to manage their diabetes manually. Incorporating regular training, fostering preparedness, and emphasizing the importance of backup plans will ensure individuals relying on advanced technologies can maintain effective glycemia even in the temporary absence of technology. The DCES can lead in their place of practice by putting in place policies and procedures that assure timely backup plans as automation advances.
Footnotes
Author Contributions
Laura Russell and Janice MacLeod wrote the initial outline. Janice MacLeod drafted the original manuscript. Laura Russell and Kelly Postiglione Cook edited the manuscript and made substantial contributions. All authors approved the final draft of the manuscript.
Declaration of Conflicting Interests
Kelly Postiglione Cook is an employee of Beta Bionics. Janice MacLeod is serving as a consultant to Welldoc and Beta Bionics doing medical writing, evidence strategy, and science calendar management.
Funding
There is no funding for this article.
Guarantor Statement
All authors accept full responsibility for the finished work.
