Abstract
Background
No systematic summary exists on childhood physical activity and later-life multimorbidity risks. We primarily investigated the association of physical activity in childhood and adolescence and the development of multimorbidity in adulthood. Secondarily, we examined whether physical activity level differ in children and adolescents with and without multimorbidity and whether there is a cross-sectional association between physical activity and multimorbidity.
Methods
Following Cochrane Handbook guidelines and adhering to PRISMA recommendations, we included cross-sectional, case-control and longitudinal studies that investigated the association between physical activity in children and adolescents and development of multimorbidity. Results were summarized narratively and we assessed the certainty of the evidence using the GRADE approach. The protocol was registered in PROSPERO, CRD42023407063.
Results
Of 9064 studies identified, 11 were included in 13 papers. Longitudinals studies suggested that being physically active in childhood and adolescence was associated with a lower risk of multimorbidity in adulthood. Three out of five studies reported lower physical activity level in children and adolescents with multimorbidity compared to those without, and two did not find a between-group difference. Cross-sectional evidence on the association between multimorbidity and lower physical activity was uncertain. Overall, the evidence certainty for all outcomes was considered low due to the indirectness and inconsistency in findings.
Conclusions
Childhood and adolescence physical activity appeared to be linked with a reduced risk of later-life multimorbidity but the certainty of the evidence is low. These results support the promotion of physical activity during childhood and adolescence.
Introduction
Insufficient physical activity poses a significant global health problem and has been called a pandemic. 1 It ranks as the fourth leading cause of death worldwide, accounting for 7.2% of all-cause mortality 2 and an economic burden of international $53.8 billion globally. 3 Worryingly, physical inactivity is increasingly affecting children and adolescents, 4 which represent 24% of the world’s population. 5 Data suggest that most children and adolescents are insufficiently active.6,7 Thus, the development of strategies to promote a physically active lifestyle in youth is more relevant than ever, and may provide benefits even in adulthood.8,9
Several systematic reviews and large longitudinal cohort studies have been conducted to investigate the benefits and harms of physical activity in children. Engaging in physical activity at a young age has been associated with different health indicators including body composition,10,11 reduced cardiometabolic risks, 10 higher bone health, 12 reduced risk of depression, 13 better mental health,11,14 greater brain development 15 and overall higher quality of life.16,11 Additionally, physical activity in childhood and adolescence seems to provide benefits later in adulthood by lowering the risk of developing cardiometabolic risk factors17,18, single chronic conditions such as arterial hypertension, type 2 diabetes mellitus 19 , depression 20 , and all-cause mortality. 21
However, no systematic reviews have investigated whether physical activity in young ages prevent the development of multiple chronic conditions (multimorbidity) later in life. Addressing this critical research gap is important, given the strong association between multimorbidity and age. 22 Thus, the main aim of our study was to investigate the longitudinal association between physical activity in childhood and adolescence and the development of multimorbidity later in adulthood. Secondarily, we examined whether physical activity level differ in children and adolescents with and without multimorbidity and whether there is a cross-sectional association between physical activity and multimorbidity.
Methods
This systematic review was performed according to recommendations in the Cochrane Handbook 23 and the reporting was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines. 24
The protocol was registered in PROSPERO (CRD42023407063) Available from: https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42023407063).
Eligibility criteria
Type of Participants
We used the definition from Sawyer et al. to define different age ranges,
25
that is children (6-9 years old), young (10-14 years old), middle (15-19 years old), and late (20-24 years old) adolescents. Participants in longitudinal studies could have been healthy or with a single chronic condition at baseline. Multimorbidity was defined as the occurrence of at least two or more
22
of the following chronic conditions: - Pulmonary conditions (chronic obstructive pulmonary disease, asthma, cystic fibrosis) - Musculoskeletal conditions (osteoarthritis, osteoporosis, back pain, rheumatoid arthritis) - Cardiovascular conditions (hypertension, coronary heart disease, heart failure, stroke, arrhythmias, congenital heart disease, cerebral apoplexy, claudication intermittent). - Metabolic conditions (obesity, hyperlipidemia, metabolic syndrome, polycystic ovarian syndrome, type 2 diabetes, type 1 diabetes) - Cognitive conditions (dementia, Alzheimer’s disease, Parkinson’s disease, multiple sclerosis) - Mental/psychiatric conditions (depression, anxiety, stress, schizophrenia, attention-deficit/hyperactivity disorder (ADHD)) - Cancer (any type).
The rationale for focusing on these conditions is that physical activity is associated with the development and progression of those chronic conditions. 26 The conditions could be self-reported or diagnosed via medical report of clinician.
Exposures
There was no restriction on the type of physical activity and the assessment method (subjective or objective). Physical activity was defined as “any bodily movement which requires energy expenditure, including during leisure time, for transport to get to and from places, as part of a person’s work and as part of paid or unpaid domestic tasks around the home.” 27 Included studies reported data to evaluate the association between physical activity in childhood and adolescence and multimorbidity later in life (primary aim), and the cross-sectional association between physical activity in childhood and adolescence and multimorbidity at these ages (secondary aim).
Information sources and types of studies
We included published observational studies (prospective and retrospective longitudinal cohorts, case-control and cross-sectional). We did not apply language restrictions for the searches, in line with the Cochrane handbook recommendations, but full-text articles were included if they were reported in English, Danish, French or Italian. We did not apply restrictions to publication date. A systematic literature search was performed in MEDLINE via PubMed, EMBASE via Ovid, CINAHL (including preCINAHL) via EBSCO supplemented with citation tracking in Google Scholar and reference screening of the included studies. Initial search was up to 21th March 2023.
Search strategy
The search was initially performed in MEDLINE (via PubMed) then translated by SR accelerator polyglot search translator 28 (https://sr-accelerator.com/) to EMBASE and CINAHL. AB and ACL checked and adapted the output of the tool before performing the search in EMBASE and CINAHL. Validated filters were used to guide the search for observational and cross-sectional studies. 29 All terms were searched, if possible, both as keywords (MeSH) and as text words in titles and abstract (ti/ab). The full search strategy is available in Table S1A.
Selection process
Two review authors (LS and ACL) independently screened titles and abstract for inclusion from all the potentials studies identified in the searches. We then sourced full texts and both review authors (LS and ACL) independently read them to confirm eligibility; in the event of exclusion, we documented the reasons. Disagreements between the two reviewers were discussed until consensus was reached. Another author (AB) was consulted to solved disagreement between (LS and ACL). The processes were facilitated by Covidence systematic review software (Veritas Health Innovation, Melbourne, Australia), and allowed us to easily record the selection process with a PRISMA flow diagram.
Data items and collection process
Two reviewers (LS and ACL) extracted data from eligible articles: authors name, author contact information, publication year, design of the study, country, total sample size, sample characteristics (e.g., age, % female), physical activity characteristics (e.g., assessment methods, age at assessment, duration of assessment) and chronic conditions characteristics (e.g., number, diagnosis, severity), outcomes characteristics (the number of individuals who were physically active during childhood and developed multimorbidity in adulthood, the number of individuals who were physically inactive during childhood but still developed multimorbidity in adulthood, the number of individuals who were physically inactive during childhood and did not develop multimorbidity in adulthood, the number of individuals who were physically active during childhood but did not develop multimorbidity in adulthood). When the outcome characteristics were not reported in the original studies, we prioritized extracting the risk ratio, but if not present we extracted the odds ratio, or any other measure presented. If both unadjusted and adjusted estimates were presented, we used the adjusted estimates. Additionally, we contacted the corresponding author of the study with a checklist, including the data we aimed to obtain, in line with the Cochrane handbook guidance on retrieving missing data from the authors of the studies included 30 . If the email bounced back, we contacted the second author and so forth. After three days, we sent a reminder including the last author of the paper. After seven days of the first email, we re-email the corresponding and last author. A second reminder followed ten days after the first email. We considered the data as missing after not receiving any communication from the authors 15 days after we sent the first email. 31 This approach has been followed in systematic reviews on multimorbidity. 31 – 33
Risk of bias assessment
We assessed risk of bias of the included study using the Newcastle-Ottawa Scale for cohort studies and the modified Newcastle-Ottawa Scale for cross-sectional studies 34 (Supplemental material S1B -S1C). Newcastle-Ottawa scale addresses eight items distributed in three dimensions: selection, comparability and outcome or exposure for cohort studies and cross-sectional studies, respectively. Two review authors independently applied the tool to the included studies and recorded supporting information and justifications for judgements of risk of bias for each domain. Any discrepancies in judgements of risk of bias or justifications for judgements were resolved by discussion to reach consensus between the two review authors, with a third review author acting as an arbiter if necessary. Before the assessment, a minor training session was performed, assessing the first five studies, and comparing the assessment between LS and ACL. For cohort studies, eight items were scored with one or two stars, for a maximum total of nine stars, leading to an overall judgement as serious concerns (0-4 points), some concerns (5-6 points), no serious concerns (7-9 points). For cross-sectional studies, seven items were scored with one or two stars, for a maximum total of 10 stars, leading to an overall judgement as serious concerns (0-4 points), some concerns (5-6 points), no serious concerns (7-10 points).
Synthesis method
Meta-analyses as well as subgroup and sensitivity analyses were not considered appropriate due of the substantial clinical heterogeneity between patients’ characteristics and outcomes variables. Instead, we summarized the results narratively following the synthesis without meta-analysis guidelines. 35 We used the vote counting method based on direction of association and the statistical significance (p-value <0.05) of the findings. 36 For the individual studies, a beneficial (positive) association was interpreted when physical activity was associated with decreased risk of multimorbidity (cross-sectionally or longitudinally). Overall, we interpreted the associations as positive, negative, or no association when most of the studies (≥50%) reported statistically significant positive, negative, or no association findings, respectively.
Certainty assessment
The overall certainty of the evidence was assessed using the GRADE recommendation for prognostic studies, as we included observational studies. 37 The certainty of the evidence from observational studies started as high quality and can be downgraded or upgraded. Downgrading is based on five domains focusing on methodological flaws in the included studies; study limitations, inconsistency, indirectness, imprecision, or indications of publication bias, whereas upgrading is based on two domains of methodological strengths; a large effect or a dose-response relationship. Using the GRADE approach, we evaluated the certainty in the findings as either; high, moderate, low, or very low. 38
Amendments to initial protocol
The articles retrieved through the initial protocol were deemed too few (k=2) for conducting an informative systematic review. Therefore, we expanded our search to include cross-sectional studies, allowing us to incorporate two additional outcomes: the association between physical activity and multimorbidity in childhood, and the physical activity level comparison between children and adolescents with and without multimorbidity. These were considered secondary outcomes and did not change the conclusion or interpretation of the primary outcome. We added minor modification to eligibility criteria, by enlarging the age range to 5 to 24 years old according to the Sawyer et al. definition. 25
Results
Study selection
A total of 10640 records were identified in the three databases during the initial search (16 March 2023). Studies screening and removing of duplicated were facilitated by Covidence systematic review software (Veritas Health Innovation, Melbourne, Australia). After removing duplicated, 9064 records were screened for abstract and title, including seven articles retrieved through a manual search of references and citation tracking. We assessed 109 full text records and excluded 96 articles. Overall, 11 studies reported in 13 articles39–51 were included in this systematic review. The study selection process is summarized in the PRISMA flow diagram (Figure 1). Flow chart of the study selection process. *Consider, if feasible to do so, reporting the number of records identified from each database or register searched (rather than the total number across all databases/registers). **If automation tools were used, indicate how many records were excluded by a human and how many were excluded by automation tools. From: Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021;372:n71. doi:10.1136/bmj.n71. For more information, visit: https://www.prisma-statement.org/
Study characteristics
Nine studies39–42,44–46,48–51 were cross-sectional and two were longitudinal (one retrospective and one prospective).43,47 In one longitudinal study 43 physical activity was evaluated from 7 to 55 years old participants through face-to-face interviews administered paper-based questionnaire. Multimorbidity, defined as co-occurrence of at least two chronic conditions was diagnosed by medical examination or parent-reported in childhood and self-reported at 55 years old. 43 Another longitudinal study, 47 retrospectively assessed physical activity in childhood and adolescence through self-reported questionnaires. Multimorbidity was self-reported as the co-occurrence of obesity and hypertension or type 2 diabetes mellitus at a mean age of 46 years old. 47
In cross-sectionals studies, the MY LIFE cohort39,40 included children, with functional limitations, aged 2 to 16 years old with physician-diagnosed chronic physical illness for ≧12 months. Physical activity was assessed by accelerometer worn over the hip for seven days and multimorbidity was defined as mental illnesses comorbidity diagnosed by the MINI-KID Questionnaires. In the PETALE cohort,44,45 comprised of childhood cancer survivors with a mean age 22 years old, physical activity was assessed by 3 months recall questionnaire (Minnesota Leisure Time Physical Activity). Multimorbidity in this context were defined of having different comorbidities (anxiety, depression, obesity) diagnosed following clinical visit.44,45 The Early Developmental Stages of Psychopathology cohort study, 51 included children with mental disorders assessed the cross-sectional association between physical activity (questionnaire-based) and multimorbidity (clinically diagnosed) during childhood at two different time-point (14-17 years old and 18-24 years old). 51 One study 50 focused on children aged from 6 to 20 years old with congenital heart diseases who had undergone surgery. Physical activity were 12 months retrospectively evaluated by questionnaire (The modified Youth-Adolescent Activity Questionnaire) and divided in three classes: total physical activity, vigorous activity (>6 METs) and inactivity. Obesity was diagnosed as BMI ≥95th percentile within two years as a comorbidity. In a large sample of children with ADHD, 49 aged from 6 to 17 years old, a parent or guardian was interviewed about the past 12-month physical activity to dichotomize those not meeting or meeting “the Healthy People 2010” guidelines. Parents reported BMI with obesity defined as BMI ≥95th percentile, considered as comorbidity. Another study, 46 enrolled a large sample of school-aged children (6-18 years old) with parent-reported diagnosis of either ADHD or asthma. Participation in organized sport during the last 12-months and daily physical activity behaviors were parent-reported. Obesity defined as BMI ≥95th percentile was considered as comorbidity. A study in Turkish children 41 aged 2-18 years old and diagnosed with ADHD, recorded physical activity hours per week based on reports from parents, teacher, personal trainer or occupational therapist. Height and weight were assessed during clinical visits, with obesity defined as BMI ≥95th percentile (comorbidity). Conversely, a study 42 enrolled obese children (6-19 years old) defined with a clinically assessed BMI ≥95th percentile. Parent-reported days physically active per week, defined as 30 minutes or more of purposeful physical activity were considered. ADHD, considered as comorbidity, was parent-reported and confirmed with patient medical record. Finally, one study 48 assessed physical activity in young individuals aged 16-24 years old through survey assessments, transforming data into MET value based on IPAQ criteria to indicate active and inactive adolescents. Multimorbidity was defined as the co-occurrence of two or more conditions in a single individual and was self-reported and confirmed by a physician.
Summary of the included studies in the systematic review.
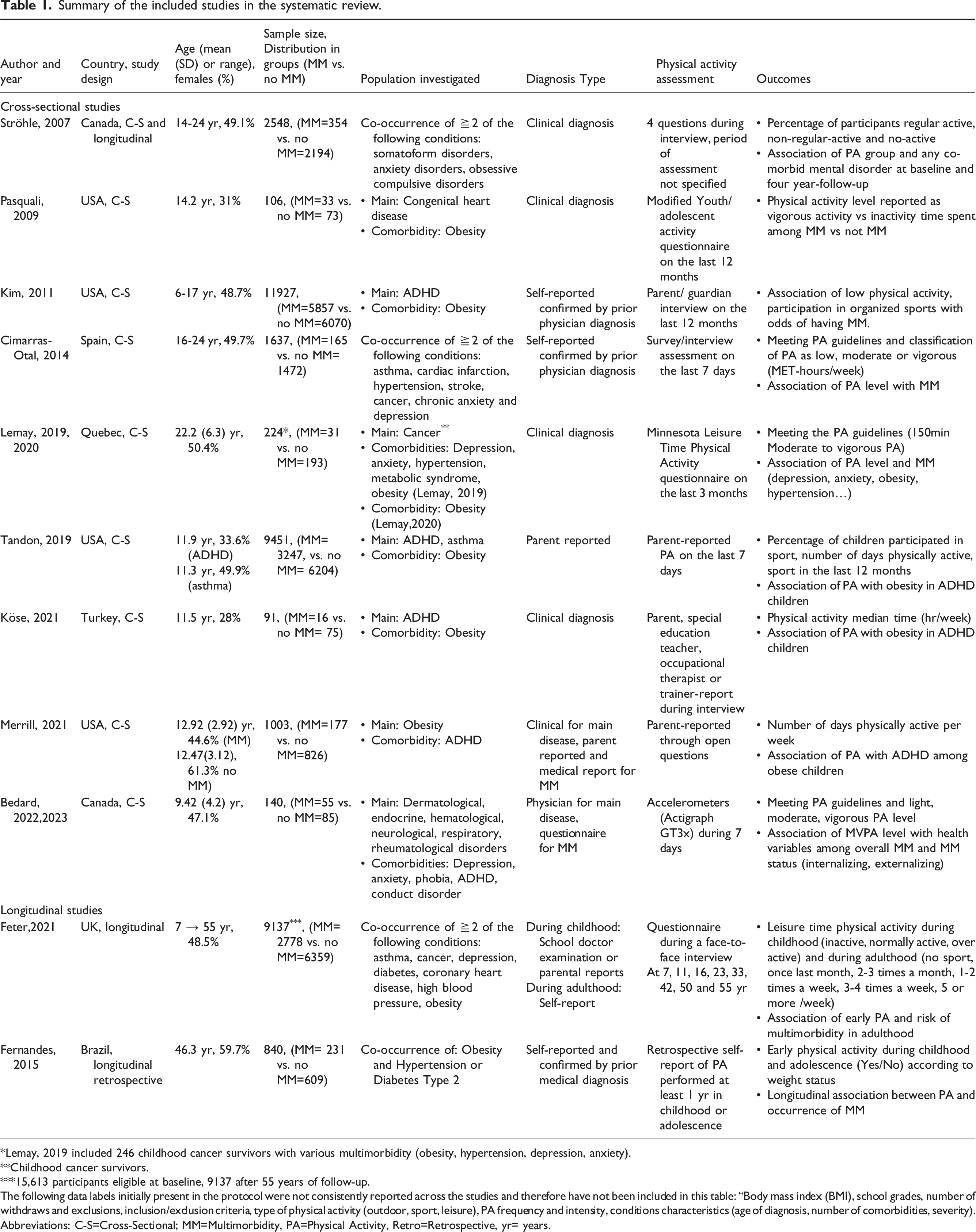
Lemay, 2019 included 246 childhood cancer survivors with various multimorbidity (obesity, hypertension, depression, anxiety).
Childhood cancer survivors.
15,613 participants eligible at baseline, 9137 after 55 years of follow-up.
The following data labels initially present in the protocol were not consistently reported across the studies and therefore have not been included in this table: “Body mass index (BMI), school grades, number of withdraws and exclusions, inclusion/exclusion criteria, type of physical activity (outdoor, sport, leisure), PA frequency and intensity, conditions characteristics (age of diagnosis, number of comorbidities, severity).
Abbreviations: C-S=Cross-Sectional; MM=Multimorbidity, PA=Physical Activity, Retro=Retrospective, yr= years.
Narrative synthesis of results
Primary outcome: The risk of developing multimorbidity in adulthood between physical active versus physically inactive children and adolescents
Narrative synthesis of the results.

Notes: In their study, Ströhle et al., 2007 examined the association at two different time points. Inactive children were at higher risk of having MM after twelve-months (Ströhle et al., 2007a) and four years of follow-up (Ströhle et al., 2007b).
Abbreviations: MM=Multimorbidity, PA=Physical Activity.
Summary of findings for each outcome.

Notes: A single pooled effect estimate was not available for the three outcomes of interest; therefore, we provided a narrative synthesis of the evidence. We did not strongly suspect publication bias because both negative and positive trials were published, and the search for studies was comprehensive.
Commonly used symbols to describe certainty in evidence in evidence profiles: high certainty ⊕⊕⊕⊕, moderate certainty ⊕⊕⊕O, low certainty ⊕⊕OO and very low certainty ⊕OOO.
Definition of multimorbidity used: Occurrence of at least 2 or more chronic conditions.
aDowngraded: The type of chronic condition and assessment of the condition (outcome) were different between studies.
bDowngraded: Method of assessment of PA (exposure) and study design were not comparable. The number of studies on this outcome (n=2) remains low.
cDowngraded: Direction and magnitude of the results between studies were wide and inconsistent (statistical measures of heterogeneity are not available) Abbreviations: MM=Multimorbidity, PA=Physical Activity.
Secondary outcome: Physical activity in children with and without multimorbidity
Three42,44–46 out of the five studies included for this outcome reported a lower physical activity level in children with multimorbidity and two reported no statistically significant difference.39,40,50 (Table 2) However, given the inconsistency of the findings as well as the heterogeneous participant characteristics the certainty of these findings is low (Table 3).
Secondary outcome: The association between physical activity in children and adolescents and multimorbidity
Seven cross-sectional studies were included for this outcome. Two studies42,51 reported that lower physical activity was associated with greater risk of multimorbidity in children and adolescents. Five studies reported that physical activity was not associated with multimorbidity in the entire cohorts (Table 2). Further, two out of the five studies reporting no cross-sectional association reported that the association may vary depending on sex 48 and type of conditions.44,45 Overall, the certainty of these findings is low (Table 3).
Risk of bias and overall certainty of the evidence
We used the Newcastle-Ottawa scale tool to assess risk of bias for each of the included studies (Supplemental material S1B and S1C). Table S1D summarizes these assessments. Overall, most of the studies were deemed at low risk of bias (8 out of 11 studies).
Among the cross-sectional studies, three had some concerns regarding bias, while six were deemed to have no serious risk of bias. The selection domain was scored lower (69%) than comparability and outcomes domains (81%). Notably, in the selection domain, the lowest score was observed for justified sample size (64%) and comparability between respondent’s and non-respondent’s (54%).
For the two longitudinal studies, scores were judged as no serious risk of bias, with the lowest score for demonstration that outcome of interest was not present at start of study (selection domain) and in the outcomes assessment (outcome domain).
Despite, these evaluations, it is essential to acknowledge certain limitations that may affect the quality of evidence. These limitations include a few studies and the heterogeneity in the types of multimorbidity investigated. As a result, because of inconsistency and indirectness, the overall certainty of evidence (GRADE) was deemed low for all the outcomes (Table 3).
Discussion
We included 11 studies reported in 13 papers, using different study designs and physical activity level assessments, and investigated different combinations of chronic conditions. Our main results suggest that physical activity during childhood and adolescence might protect from the developing of multimorbidity in adulthood. Additionally, the level of physical activity might be lower in children and adolescents with multimorbidity compared to those without multimorbidity while the cross-sectional association between physical activity and multimorbidity is unclear. However, the certainty of evidence was deemed low for all outcomes, suggesting that future studies may change the current results and thereby the conclusion.
Results in context
Physical activity in childhood and adolescence might have a protective effect on the development of multimorbidity in adulthood, which aligns with previous studies investigating the impact of early-life physical activity on the development of single chronic conditions or health risk factors later in life.17–19,52–54 A possible reason for these findings is that an active lifestyle during early ages may contribute to higher levels of physical activity in adult life, 55 which in turns may contribute to reduce risk of developing multimorbidity.56,57 The mechanisms by which physical activity prevent the onset of chronic conditions are multifactorial. In fact, a combination of physiological, behavioral, and social determinate seems all to play a role. 27
Physical activity level of children and adolescents with multimorbidity seems to be even lower than children without multimorbidity. Environmental barriers, such as limited access to a suitable physical activity, lack of knowledge about multimorbidity and how to adapt physical activity to different combinations of chronic conditions could be factors that contribute to explain this finding.58,59 Physicians and parents may also contribute to physical activity restrictions through their concerns about the safety of physical activity in children with chronic conditions.60,61 Moreover, youth with multimorbidity may experience reduced physical activity due to lower self-efficacy and higher perceptions of barriers to exercise. 62 Furthermore, lower cardiorespiratory and muscular fitness in children with multimorbidity could further hinder their engagement in physical activity.63,64
It is unclear whether physical activity is associated with multimorbidity during childhood and adolescence (cross-sectional association). One possible explanation for this, is the differences in how the exposure (physical activity) has been assessed across studies, with some using accelerometers and others relying on questionnaires. Indeed, self-reporting often overestimates physical activity in children and adolescents. 65
The other potential explanation for these findings is the diversity in the combination of chronic conditions included in the studies. Although physical activity has similar benefits for children and adolescents with different single chronic conditions, it is unknown whether this is true also for those with different combinations of multiple chronic conditions.
Strength and limitations of our systematic review
This systematic review has been conducted according to the recommendations for performing systematic reviews in the Cochrane Handbook 23 and reported according to PRISMA guidelines, 24 ensuring methodological rigor and transparency. Our goal of inclusivity was achieved by using a comprehensive definition of multimorbidity that considers a wide range of chronic disease combinations. We included all observational studies that used different assessment methods for physical activity (questionnaires, accelerometers, interviews). However, our review has several limitations. The overall certainty of evidence was graded as low due to heterogeneity on exposure assessment (self-reported and objectively), lack of repeated physical activity measurements throughout childhood and adulthood, 66 included chronic conditions and the limited number of studies (two studies for the primary outcome). Moreover, several population characteristics were poorly reported in the included studies. This prevented us from further investigation of the findings' heterogeneity via sub-group analyses and sensitivity analyses limiting the generalizability of the results.
Implications for researchers
This systematic review highlights the need for high-quality cross-sectional and longitudinal studies to evaluate the association between physical activity and multimorbidity in childhood and later in adult life. Despite inherited retention challenges in longitudinal studies, to improve the confidence in these results, we suggest future studies to both focus on retention strategies, 67 as well as including participants with different combinations of conditions to better represent the global multimorbid population. This is in line with the findings of a scoping review by Romano et al., 68 highlighting that despite a rapid increase in studies published over the past decade on children and multimorbidity, the conditions investigated are mainly mental conditions. Additionally, we suggest future studies to specify whether they focus on multimorbidity or comorbidity. As the two terms have different meaning, multimorbidity is defined as the co-occurrence of at least two conditions where no single condition holds priority over any of the co-occurring conditions from the perspective of the patient and the health care professional, versus, comorbidity which is defined as the combined effects of additional condition in reference to an index chronic condition.69,70 It is important to clarify which concept is investigated to better inform clinical practice. Finally, future longitudinal research in this field should adopt this definition and promote the assessment of domain-specific physical activity, encompassing its type, frequency, and duration, as well as utilizing device-based measurements, such as accelerometers, to enhance methodological rigor. 71
Implication for clinicians
Although the limited evidence for the association between physical activity and multimorbidity in childhood, our results support the importance of assessing physical activity in clinical practice. Additionally, clinicians should emphasize advocating for adherence to the WHO guidelines on physical activity for children and adolescents. This recommendation is in line with WHO plan 2018-2030 that aims at a 15% reduction in global prevalence of physical inactivity 72 . Therefore, it is essential to focus on developing comprehensive interventions that encourage physical activity, in different settings, including schools. 9
By incorporating these insights into early-stage care strategies, clinicians can play a pivotal role in mitigating physical activity decline in children and adolescents with multimorbidity, and therefore improved their life expectancy. 73
Conclusion
Previous studies have indicated that engaging in physical activity at a young age provide benefits in adulthood for specific single chronic disease. Our systematic review highlights that the evidence for the association between physical activity during childhood and adolescence and multimorbidity later in life is limited. There might be a potential protective effect of physical activity during childhood and adolescence against development of multimorbidity (the presence of two or more chronic conditions in a single individual) later in adult life, however, there is a pressing need for additional high-quality longitudinal studies to confirm this.
Supplemental Material
Supplemental Material - Childhood and adolescence physical activity and multimorbidity later in life: A systematic review
Supplemental Material for Childhood and adolescence physical activity and multimorbidity later in life: A systematic review by Luc Souilla, Anders C. Larsen, Carsten Juhl, Søren T. Skou, and Alessio Bricca in Journal of Multimorbidity and Comorbidity
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: STS is an associate editor of the Journal of Orthopaedic & Sports Physical Therapy; has received personal fees from Munksgaard, Nestlé Health Science and TrustMe-Ed outside the submitted work; and is a cofounder of GLA:D, a not-for-profit initiative hosted at the University of Southern Denmark aimed at implementing clinical guidelines for osteoarthritis in clinical practice. AB has received personal fees from PhisioVit S.r.l. outside of the submitted work. None of the authors had other financial relationships or activities that might have an interest in the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: LS has obtained a mobility grant EXPLORE #4 by Montpellier University of Excellence and additional mobility grant support by Institut Saint-Pierre, France. AB is supported by the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme (MOBILIZE, grant agreement No 801790). STS is currently funded by a program grant from Region Zealand (Exercise First) and 2 grants from the European Union’s Horizon 2020 Research and Innovation Program, one from the European Research Council (MOBILIZE, grant agreement 801790) and the other under grant agreement 945377 (ESCAPE). The funding source had no involvement in any aspect of this systematic review.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
