Abstract
In this study, LiAl-layered double hydroxides Lithium aluminum hydrotalcite (LiAl-LDH) with different specific surface area were prepared by the separate nucleation and aging steps (SNAS) method and then were employed to prepare calcium sulfoaluminate cement-based grouting material (CBGM) paste. The influence of LiAl-LDH slurries on fresh and hardened properties of the CBGM paste was investigated in terms of fluidity, stability, setting time, and compressive strength. Additionally, the hydration process and hydration products of the CBGM paste were characterized by hydration heat, X-ray diffraction, differential thermal analysis–thermogravimetry, and Fourier transform infrared analyses. The acquired results illustrated that LiAl-LDH with larger specific surface area led to a faster hydration rate at early age, a lower fluidity, a shorter setting time, and a higher stability. Furthermore, due to the crystal nucleation effect, the addition of LiAl-LDH slurries did not cause a new phase to form but changed the morphology and increased the amount of hydration products, yielding higher compressive strength.
Introduction
Grouting materials have been used in various engineering areas, such as tunnel and underground coal mine engineering, primarily for consolidation and repair. 1 The performance of grouting materials are crucial in grouting engineering. A preferred grouting material should have many characteristics, such as high fluidity and stability during pumping process and short setting time and high compressive strength at the reinforcement site. Double liquid grouting materials-based calcium sulfoaluminate (CSA) cement clinker–quicklime–anhydrite are widely employed 2 –4 in grouting engineering due to its high early strength, rapid setting, impermeability, and corrosion resistance. For the repair and consolidation of masonry structures, the water-to-binder (w/b) ratio of cement-based grout has a range between 0.5 and 1.5. 5 For a high w/b ratio (w/b ≥ 0.5), the setting time and compressive strength of the CSA cement-based materials, however, cannot satisfy the demands of projects such as reinforcement of large sections of coal roadways and surrounding rocks, indicating that accelerators must be added to the complex systems.
Currently, new strategies of incorporation of superfine particles in cement-based materials have been widely studied to improve their physical and mechanical properties. It has been reported that superfine particles can act as nuclei, accelerating the cement hydration at early age and as a filler that densifies the microstructure of the cement-based materials 6 and improve the strength at early and late ages. For instance, the addition of nano-SiO2 7 shortened the setting time and increased the compressive strength of the cement-based grouting material (CBGM) while improving the cohesiveness of the mixes in the fresh state. Li and Xu 8 investigated the effects of nanosilica (NS) on the fresh properties of CBGM in the portland–sulfoaluminate composite system. The results showed that the fluidity decreased with the increased NS content, while the influence of particle sizes on flow properties was still uncertain.
Layered double hydroxides (LDHs), also known as hydrotalcite-like materials, are a large class of anionic lamellar functional materials. Taylor 9 described these phases as being structurally relevant to brucite the same way that AFm phases are to portlandite. The general formula of LDH is [M1 x 2+ M x 3+(OH)2] x +[(A n −) x /n ·mH2O] x −, where M2+ can be Ca2+, Mg2+, Zn2+, Ni2+, or Fe2+, M3+ can be Al3+, Co3+, Cr3+, or Fe3+, and A n − can be NO3 −, OH−, Cl−, SO4 2−, or CO3 2−. LiAl2(OH)6 +A− is also a case of a LDH and is made up of sheets of lithium and aluminum atoms octahedrally surrounded by hydroxyl groups. 10 The results of our previous study 11 have indicated that LiAl-layered double hydroxide (LiAl-LDH) powder with 3D micro- and nano structure (1−15 µm) can greatly accelerate the hydration rate and enhance the value of compressive strength of CSA cement clinker by 178% at 1 day and 19% at 28 days, and LiAl-LDH with bigger specific surface area results in faster hydration rate and higher compressive strength.
To obtain better performance of CBGM paste, it is necessary to prepare LiAl-LDH with bigger specific surface area. Nevertheless, the main limitations of superfine powder are their dispersion performance. Therefore, it is important for the performance that nanomaterials disperse effectively. Our previous study 12 has shown that, compared to superfine powder with solid necks among the particles, LiAl-LDH without drying process named LiAl-LDH slurry has better dispersion stability. In this article, three LiAl-LDH slurries with different specific surface areas were synthesized by the separate nucleation and aging steps (SNAS) method, and then the effects of LiAl-LDH slurries on properties of CSA CBGM have been investigated in terms of setting time, fluidity, compressive strength, and stability. Furthermore, the hydration and microstructure development of CBGM are further analyzed by techniques including isothermal calorimetry, differential thermal analysis–thermogravimetry (TG-DTA), Fourier transform infrared (FTIR), and X-Ray diffraction (XRD).
Experiment
Materials
Sulfoaluminate cement clinker, anhydrite, and quicklime which are commercially available in local market were selected and used as binders for grouting materials. The calcium oxide content in quicklime was 70.3%, and the calcium carbonate content was 7.35%. The mineralogical/chemical compositions of CSA cement clinker and anhydrite provided by the manufacturer are presented in Tables 1 and 2. Chemical reagents, such as sodium carbonate (Na2CO3), lithium nitrate (LiNO3), aluminum nitrate nonahydrate (Al(NO3)3·9H2O), carboxyl methyl cellulose, sodium gluconate, and sodium hydroxide (NaOH), were of analytical purity. Sodium bentonite and naphthalene series water-reducing agent (NSA) were purchased from the local market.
Chemical composition of CSA clinker and anhydrite (wt%).
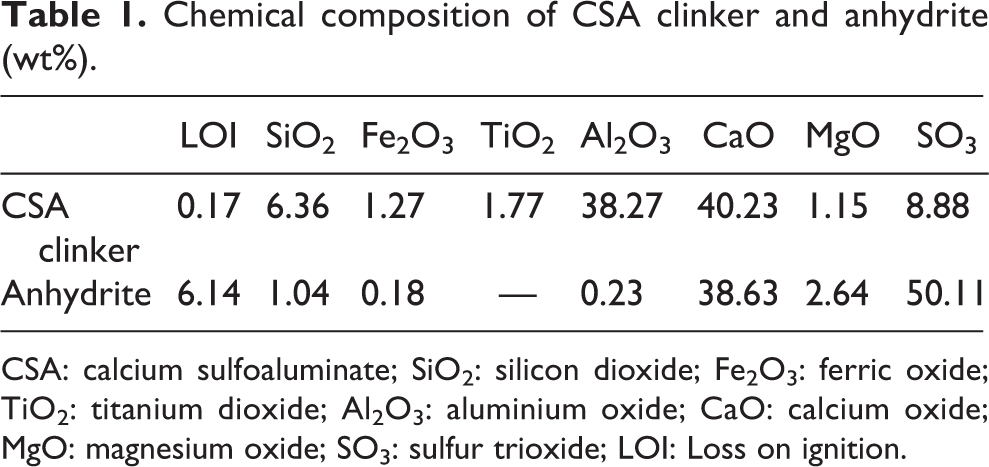
CSA: calcium sulfoaluminate; SiO2: silicon dioxide; Fe2O3: ferric oxide; TiO2: titanium dioxide; Al2O3: aluminium oxide; CaO: calcium oxide; MgO: magnesium oxide; SO3: sulfur trioxide; LOI: Loss on ignition.
Mineralogical composition of CSA clinker (wt%).

C4A3S¯M: calcium sulfoaluminate; β-C2S: β-dicalcium silicate; C4AF: calcium aluminoferrite; SO3: sulfur trioxide; CaO: calcium oxide; TiO2: titanium dioxide.
Synthesis of LiAl-LDH
LiAl-LDH were synthesized by SNAS method, 13 but with some procedures modified. Firstly, two solutions were prepared: solution A consist of Al(NO3)3 and LiNO3 dissolved in 250 mL of solvent with a Li+/Al3+ molar ratio of 4 and Li + concentration of 1.6 mol L−1; solution B was prepared by dissolving NaOH and Na2CO3 in the same solvent as solution A. The concentrations of NaOH and Na2CO3 were 1.6 mol L−1 and 0.1 mol L−1, respectively. Both solution A and solution B were then simultaneously added to a colloid mill with a rotor speed of 3000 r min−1 and vigorously mixed for 2–3 min. The resulting slurry was taken out of the colloid mill and aged at 368 K for 5 h. Lastly, the precipitate was accessed by filtration and washing with deionized water until the washing reached a pH of 7.0. The solvents mentioned above were actually ethanol–water solutions which were prepared with different concentrations (66.7 mL H2O/33.3 mL C2H5OH, 75 mL H2O/25 mL C2H5OH, and 100 mL H2O). Correspondingly, the formed LDHs in these solvents were hereafter denoted by SLDH-1, SLDH-2, and SLDH-3, respectively. LiAl-LDH were then redispersed in 1.0% NSA solution resulting in a stock suspension of 40 g L−1, followed by 5-min ultrasonic treatment at a power of 100 W. The dispersed LiAl-LDH slurries were then employed for further studies.
Preparation of CBGM paste
Based on the mix formulas listed in Table 3, slurry A and slurry B which proved optimum by our preliminary experimental work were separately prepared; and then the CSA CBGM was prepared by blending the slurry A and slurry B. The binder was replaced by LiAl-LDH slurries at a level of 2.0%.
Component of the CSA cement-based grouting material (wt%).

CMC: carboxyl methyl cellulose; NW: naphthalene water reducer; SG: sodium gluconate; w/b: water-to-binder ratio.
Performance assessment
Stability, setting time, and rheological test
Stability of slurry A and slurry B was evaluated by determining the accumulation of bleed water at the surface of fresh paste, according to ASTM C940-98a. A well-mixed slurry (1 L) was collected and placed in a 1000-mL graduated cylinder for setting, and the volume of bleed liquid on top of the grout compared to the total volume of the suspension were recorded after setting for 30 min and 1 h. The cylinder was graduated and placed on a plane surface that was free of vibrations and covered by a thin film to prevent the evaporation of bleeding water. Values for the volume of the sample and the time at which the reading was made were approximated to the closest 1 mL and 1 min, respectively. The setting time of the blended slurries of A and B was determined by conducting a Vicat needle test in accordance with the Chinese National Standard GB/T1346. Rheological measurements of slurry A and slurry B were performed at the constant temperature of 25°C by using a rotary viscometer with NXS-11B type coaxial cylinders (Chengdu Instrument Factory, Chengdu, China) and a thermostat system. Slurry properties were approximated from the flow curves that were obtained for increasing and decreasing values within the shear stress scope from 15.5 s−1 to 996.1 s−1. Plastic viscosities and yield stress values were determined from the Bingham model. 14
Compressive strength
The compressive strength of CBGM with and without LiAl-LDH slurries was examined according to Chinese National Standards GB/T 7897-2008. Then, the paste was placed into a 2 × 2 × 2 cm3 mold for vibrating. The specimens were subsequently cured in moist air at 20 ± 1°C with over 90% relative humidity for several hours, followed by demolding. Next, the specimens were cured in water until the required age. Thirty cubic specimens were prepared for each mixture, and six of each type of specimen were cured until 4 h, 1 day, 3 days, 7 days, and 28 days, respectively.
Hydration heat
The CSA CBGM with and without LiAl-LDH were analyzed for their hydration heat flow development. A TAM air isothermal calorimeter (TAM air, TA Instruments, New Castle, DE, USA) was used to measure the exothermic hydration rate within 72 h at 20°C.
Structure characterization
Characterization of LiAl-LDH
Powder XRD patterns were collected on a Rigaku SmartLab (Rigaku Tokyo, Japan) X-ray diffractometer with copper K α radiation (λ = 0.154 nm) in the 2θ scope of 5–70°, with a step size of 0.02°. Elemental analysis for Al and Li was performed by inductively coupled plasma (ICP) with a PerkinElmer Optima 7000DV (PerkinElmer, Waltham, Massachusetts, USA). The specific surface areas were acquired by the Brunauer–Emmett–Teller (BET) method that was based on the nitrogen adsorption isotherms at 77 K, the data being collected with an analyzer (ASAP 2050, Micromeritics, Norcross, GA, USA). The pretreatment conditions are 105°C and vacuum for 12 h.
Characterization of hardened paste
The specimens were obtained from the fragments of failure sample after compressive strength testing. The hydration of these specimens was terminated by using absolute alcohol, followed by drying in a vacuum desiccator and analyzing with FTIR, TG-DTA, and XRD. Powder XRD patterns were recorded on a Rigaku SmartLab X-ray diffractometer with copper K α radiation (λ = 0.154 nm) in the 2θ range of 5–50° with a step size of 0.02°. IR spectral analysis was collected with potassium bromide (KBr) disks employing a Bruker V70 IR spectrometer (Bruker, Karlsruhe, Germany) in the range of 400−4000 cm−1. The samples were prepared using KBr that was pressed into a disk, which can give a further decrease in scattering. The TG-DTA curves were tested using a Hengjiu synchronous thermal instrument at 10°C min−1 under the thermal range from room temperature up to 900°C.
Results and discussion
XRD, BET, and ICP results of LiAl-LDH
Figure 1 shows the XRD patterns of these samples prepared with different solvents. The diffraction peaks [002][004][006][110][016][330] of the three samples are consistent with the hexagonal phase of LiAl-LDH which is illustrated in JCPDS No. 42-0729. The sharp and strong diffraction peaks indicate that the hydration products are highly crystalline.
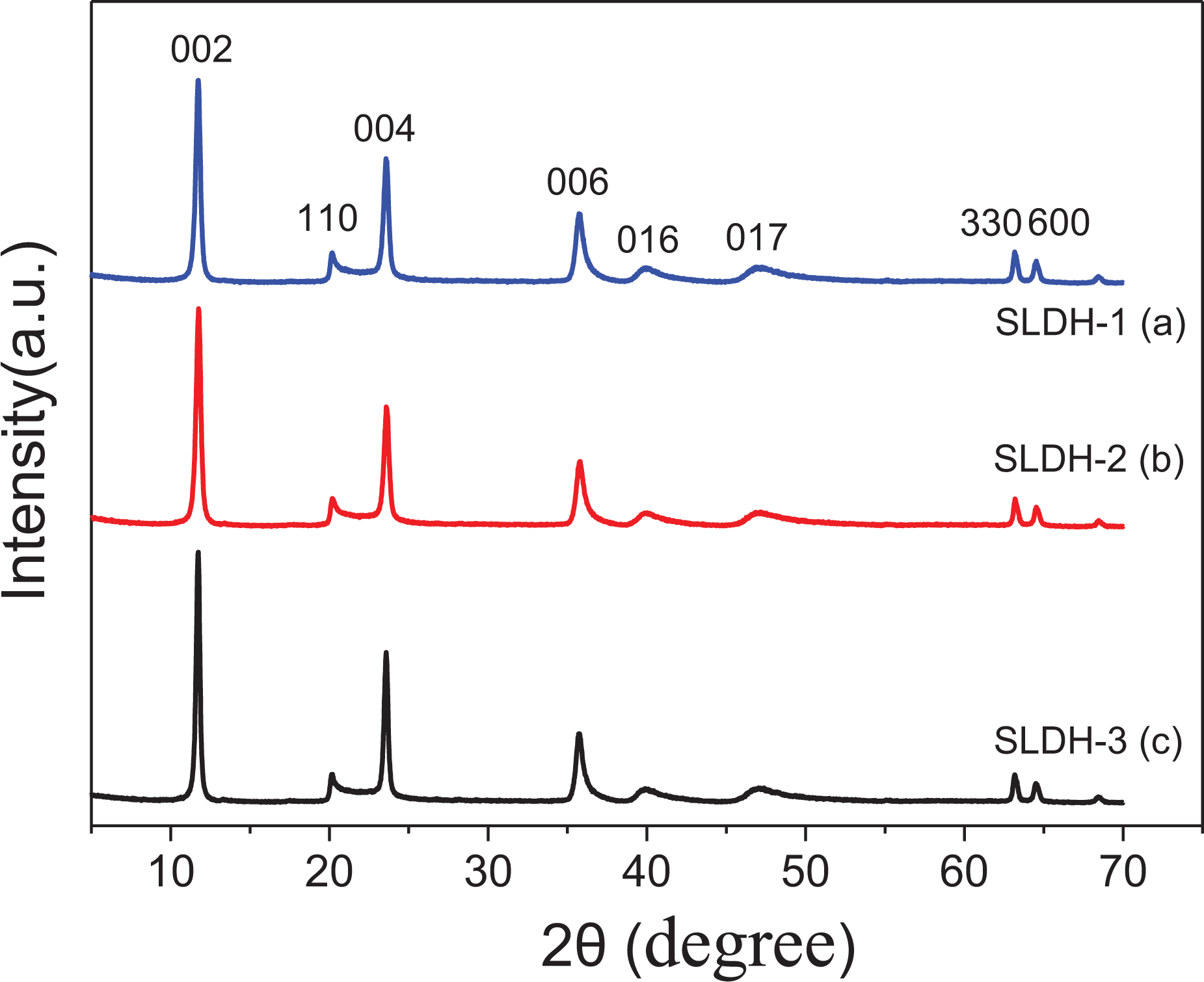
XRD patterns of products prepared at different water/ethanol ratios: (a) 66.7:33.5 mL, (b) 75:25 mL, and (c) 100:0 mL. XRD: X-ray diffraction.
The BET surface area of SLDH-1, SLDH-2, and SLDH-3 are 68.759 m2 g−1, 52.145 m2 g−1, and 34.518 m2 g−1, respectively, indicating a higher water to ethanol volume ratio resulted in a smaller specific surface area. Owing to the different viscosity and surface tension of water compared to ethanol, the solvent properties could impact the diffusing behavior of the regents and intermediates.11 Controlling the ion diffusion rate in the solvent and changing the ethanol-to-water volume ratio could enable the formation of LiAl-LDH particles with different specific surface area or size during synthesis. 15,16
The results of the ICP test are presented in Table 4, the Li+/Al3+ molar ratios of SLDH-1, SLDH-2, and SLDH-3 are 0.5098, 0.5230, and 0.5162, respectively, indicating that three LiAl-LDH samples had similar Li+/Al3+ molar ratio.
ICP and BET results of LiAl-LDH.
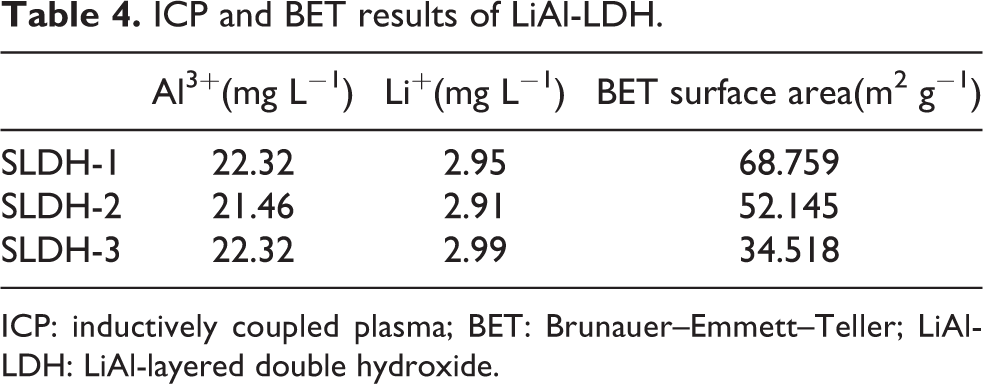
ICP: inductively coupled plasma; BET: Brunauer–Emmett–Teller; LiAl-LDH: LiAl-layered double hydroxide.
Properties of CSA CBGM
Rheology, stability, and setting time
The rheological curves of slurry A, slurry B, slurry B with SLDH-1, SLDH-2, and SLDH-3 are shown in Figure 2. Plastic viscosity and yield stress are two key parameters that should be fitted to study the rheological behavior of the slurries A and B. The Bingham law, 17 given by Equation (1), can be adapted to obtain plastic viscosity and yield stress.

Shear stress versus shear rate curves fitted to the Bingham model at 8 min.

where τ is the shear stress (Pa), τ0 is the yield stress (Pa), μ is the plastic viscosity (Pa s), and γ is the shear rate (s−1).
The yield stress of slurry A, slurry B, and slurry B with SLDH-3, SLDH-2, and SLDH-1 at 8 min were 1.337 Pa, 0.126 Pa, 1.009 Pa, 1.247 Pa, and 1.401 Pa, respectively (Table 5). The yield stress of slurry B was increased by LiAl-LDH slurries addition, and higher yield stress was given by slurry B with SLDH-1 than by slurry B with SLDH-3 or SLDH-2. Namely, LiAl-LDH with larger specific surface area resulted in a higher yield stress. The plastic viscosities of slurry A, slurry B, and slurry B with SLDH-3, SLDH-2, and SLDH-1 at 8 min were 0.008 Pa s, 0.009 Pa s, 0.010 Pa s, 0.011 Pa s, and 0.011 Pa s, respectively. The change in plastic viscosity of slurry B without and with LiAl-LDH slurries was very small. In general, the LiAl-LDH slurries addition had a significant impact on the yield stress but a small impact on the plastic viscosity of slurry B. Generally, cement-based pastes incorporating superfine particles with higher specific surface area need more water to maintain the workability of the paste. 18 It was believed that LiAl-LDH slurries promoted the particle packing of paste, which decreased the volume between the particles and reduced the amount of free water. Therefore, the internal friction between solid particles was higher, which contributed to an increase in torque. 19 Thus, yield stress values were seriously affected by the specific surface area of LiAl-LDH.
Results of stability and rheology.

LDH: layered double hydroxide.
The stability (bleeding rate and bleeding ability) of slurry is extremely important, such that the low stability of a slurry causes the particles to settle rapidly and clog the lines. 20 The bleeding rates observed in slurry A, slurry B, and slurry B with LiAl-LDH slurries are presented in Table 5. It can be seen that, after 30-min setting, slurry A had a bleeding rate of 1.88% while slurry B had a bleeding rate of 3.75%, which was reduced to 0.63%, 0.87%, and 1.08% with the addition of 2 wt% of SLDH-1, SLDH-2, and SLDH-3, respectively. After 1 h, the bleeding rate of the slurry B with SLDH-1, SLDH-2, and SLDH-3 decreased compared with the control paste (without LiAl-LDH) by approximately 83.45%, 77.76%, and 72.42%, respectively. The addition of LiAl-LDH slurries reduced the bleeding of slurry B at a w/b ratio of 1, and LiAl-LDH slurries with larger specific surface area caused a lower bleeding rate. In general, except for slurry B, the bleeding rate of slurry A and the bleeding rates of slurry B with LiAl-LDH slurries at 30 min and 60 min were less than 5% and could be regarded as stable.
The addition of LiAl-LDH slurries can shorten the CBGM initial and final setting time, as shown in Figure 3. By employing 2 wt% LiAl-LDH, the initial setting times of the CBGM paste with SLDH-3, SLDH-2, and SLDH-1 decreased by approximately 41.1%, 42.8%, and 43.9%, respectively, compared with the control paste (without LiAl-LDH). Meanwhile, the final setting time of the CBGM paste was also decreased from 408 min, while the substitute was at 0% to 240, 208, and 202 min when the substitute was at 2 wt% SLDH-3, SLDH-2, and SLDH-1, respectively. The SLDH-1 provided a shorter setting time for CBGM paste than the SLDH-3 and SLDH-2 did. Namely, employing LiAl-LDH slurries with a larger specific surface area shortened the CBGM paste setting time much more. Furthermore, the interval was shortened by the LiAl-LDH slurries between the initial setting time and the final setting time. The reduction of the setting times of the CBGM paste with the LiAl-LDH slurries might be associated with the crystal nucleation effect of the LiAl-LDH accelerating cement hydration (discussed in “Hydration heat” section).
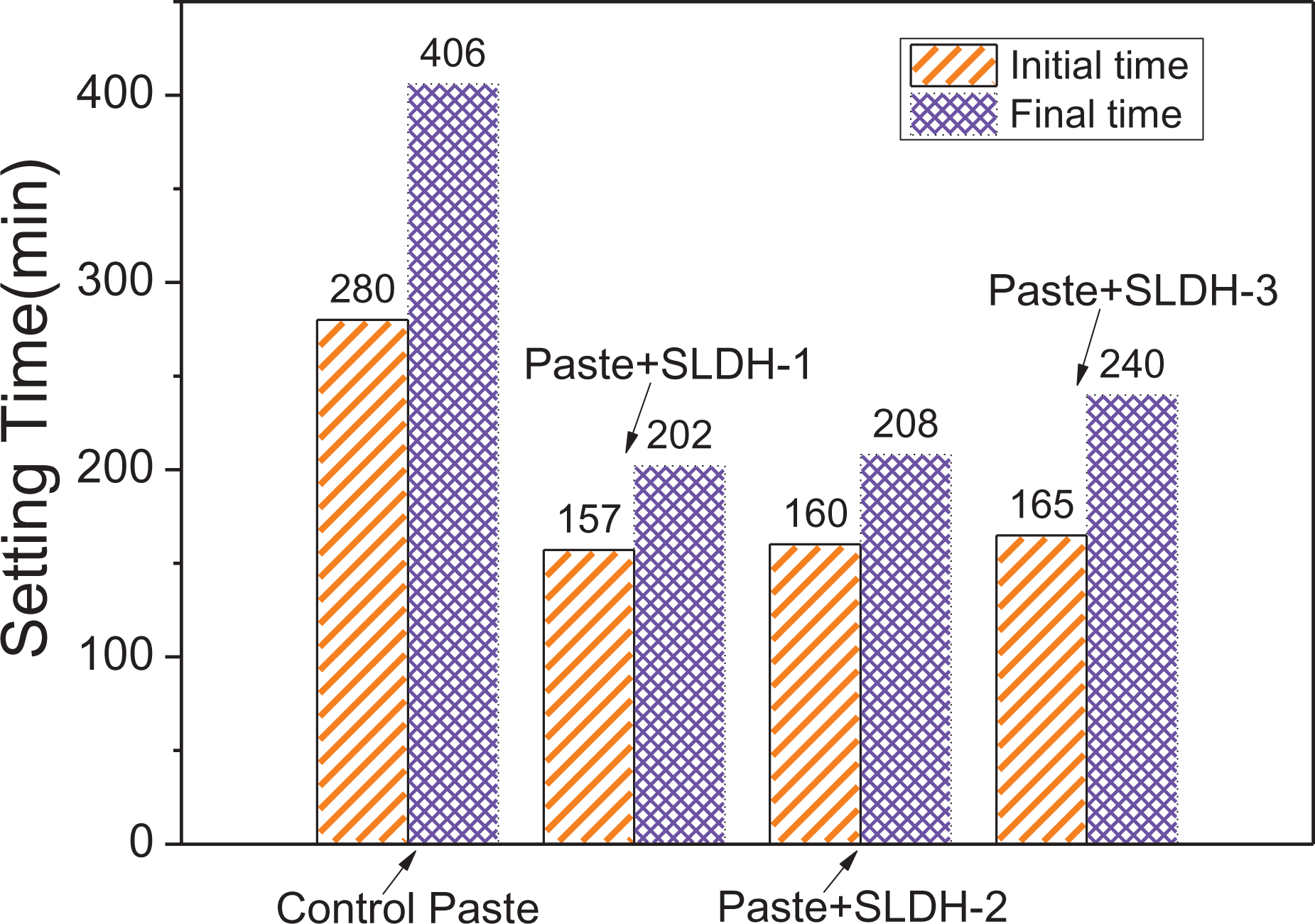
Effect of LiAl-LDHs on the setting time of the CBGM pastes. LiAl-LDHs: LiAl-layered double hydroxides.
Compressive strength
Figure 4 shows the compressive strengths of the CBGM paste with LDH at all ages. Compared with the control paste, the addition of LiAl-LDH slurries improved the compressive strength of the CBGM paste at all ages. When adding 2 wt% SLDH-1, the compressive strength at 4 h, 1 day, 3 days, 7days, and 28 days were 5.8 MPa, 7.7 MPa, 7.9 MPa, 8.8 MPa, and 15.7 MPa, and the increase in compressive strength value was at the ages of 4 h, was 112%, 104%, 91%, 76%, and 54%, respectively. At 4 h, the compressive strength of the CBGM paste with SLDH-1, SLDH-2, and SLDH-3 were 5.975 MPa, 4.822 MPa, and 4.371 MPa, and increased by 112%, 71%, and 55% compared with the control paste. Apparently, utilizing LiAl-LDH slurries with a larger specific surface area raised the compressive strength of the CBGM paste much more at 4 h. The 1-day, 3-day, 7-day, and 28-day compressive strengths of the CBGM paste were similarly increased with an increase in the specific surface area of LiAl-LDH. This could be explained by the crystal nucleus effect to promote hydration and form more products which can be seen by hydration heat and TG-DTA.

Compressive strength of the CSA cement-based grouting material pastes with and without LiAl-LDH. CSA: calcium sulfoaluminate; LiAl-LDH: LiAl-layered double hydroxide.
Isothermal conduction calorimetry
Figure 5 presents the effect of LiAl-LDH on the heat flow and the total heat released during the first 72 h hydration of the CBGM paste. The hydration heat flow of the CBGM paste with LiAl-LDH in comparison with the control paste is shown in Figure 5(a). The onset of the hydration resulting in the first exothermic peak seemed to be barely affected by the addition of LiAl-LDH. However, the intensity of first exothermic peak became sharp with the increase of specific surface area of LiAl-LDH. There was a rapid development of a heat peak culminating within the first 1–10 min, which was attributed to the effects of the cement wetting heat and the free lime hydration (equation (2)). In the presence of sufficient calcium sulfate and calcium oxide, 21 the main contribution to the first peak appears to be the hydration of ye’elimite to ettringite (equation (3)).
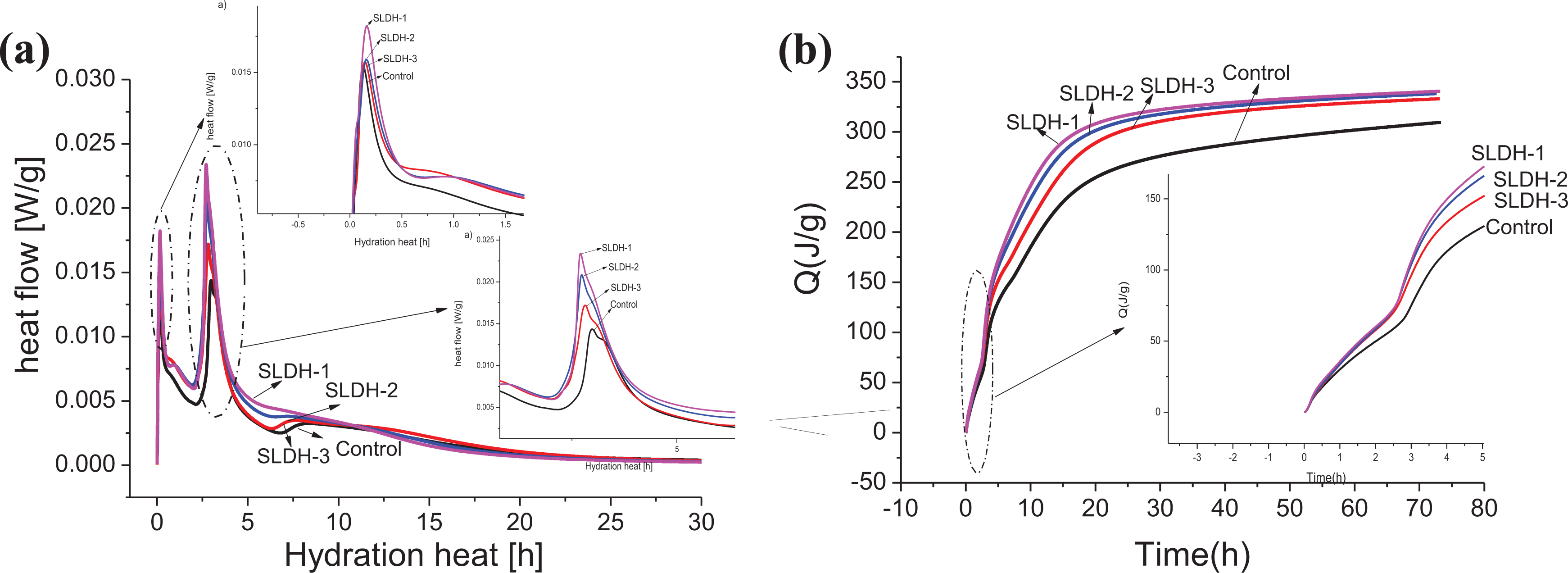
Hydration heat of CBGM paste with and without LiAl-LDHs: (a) hydration heat flow and (b) total heat released. CBGM: cement-based grouting material; LiAl-LDHs: LiAl-layered double hydroxides.



It also can be observed that the second exothermic peak for the CBGM pastes with LiAl-LDH appeared slightly earlier than that for the control CBGM paste. The contribution to the second peak appears to the hydration of ye’elimite to ettringite (equation (3)) and alumina gel to ettringite in the presence of calcium sulfate and calcium oxide (equation (4)). The reaction resulting in the second heat flow peak also tended to proceed more rapidly in samples with addition of LiAl-LDH, which can be deduced from the sharper form of the recorded maxima. With the increase of specific surface area, the rate of heat release of the second heat flow peak of the CBGM paste was faster. The effect of LiAl-LDH on the heat released from the CBGM paste is shown in Figure 5(b). The cumulative heat released after 72 h in CBGM paste was higher than the control paste. At the very early age (<5 h), the larger specific surface area of LiAl-LDH resulted in a higher total heat released in CBGM paste, which might account for the shortened setting time and the declined fluidity. The enhancement of cement hydration by active or inert ultrafine powder has been showed by some studies. 22,23 The addition of LiAl-LDH improved the hydration rate and total hydration heat may be ascribed to the crystal nucleation effect of LiAl-LDH.
XRD, FTIR, and TG-DTA
The XRD patterns of the CBGM pastes are shown in Figure 6(a) and (b). At 1 day and 7 days, the main hydration product formed was ettringite. Alumina gel was not detected in the XRD spectrum, potentially because it was amorphous. The unhydrated anhydrite phase intensity decreased and ettringite phase intensity increased when incorporating 2 wt% LiAl-LDH slurries. Larger specific surface area of the LiAl-LDH resulted in lower unhydrated anhydrite phase intensities and higher ettringite phase intensity. The results indicated that the addition of LiAl-LDH slurries promoted the hydration of the CBGM paste, and a larger LiAl-LDH specific surface area resulted in faster hydration of the CSA CBGM paste. Figure 7 illustrates the FTIR spectra of the CBGM pastes that contain LiAl-LDH slurries and the control paste at 7 days. The vibrational frequency characteristics of aluminum hydroxide, 24 with associated and free OH bands located at 3636 cm−1 and 3485 cm−1, respectively, were detected in the spectra. Hydration products such as C3A·3CS·32 H also demonstrate a signal that is characteristic of OH bands at a frequency of 3636 cm−1. The gibbsite Al–OH stretching vibration and the [CO3]2 − v3 vibration were detected at 1022 cm−1 and 1500 cm−1, respectively. 25 The sharp and strong peak at 1115 cm−1 resulted from [SO4]2 − vibrations 26 due to the formation of C3A·3CS·32 H. 24 The Si–O stretching vibration occurred at 522 cm−1. The CBGM paste with and without LiAl-LDH slurries possessed the same peak locations at the same ages, indicating that no new phases were produced.

Patterns of the CBGM pastes with and without 2 wt% LiAl-LDHs: (a) XRD patterns at 1 day and (b) XRD patterns at 7 days. XRD: X-ray diffraction; CBGM: cement-based grouting material; LiAl-LDHs: LiAl-layered double hydroxides.

FTIR spectra of CBGM pastes with and without 2 wt% LiAl-LDHs at 7 days. FTIR: Fourier transform infrared spectroscopy; CBGM: cement-based grouting material; LiAl-LDHs: LiAl-layered double hydroxides.
The TG-DTA curves of the CBGM paste that contained control paste and LiAl-LDH slurries are illustrated in Figure 8. The endothermic peaks at 110°C and 270°C are associated with the dehydration of the lattice water of ettringite and the dehydroxylation of Al(OH)3. 27 As shown in Figure 8(a), a larger specific surface area of LiAl-LDH resulted in a greater endothermic peak area. Because the quantity of hydration products is proportional to the endothermic peak area, this result demonstrates that larger LiAl-LDH specific surface area result in a faster rate of the CBGM paste hydration. This observation was the same at 7 days.
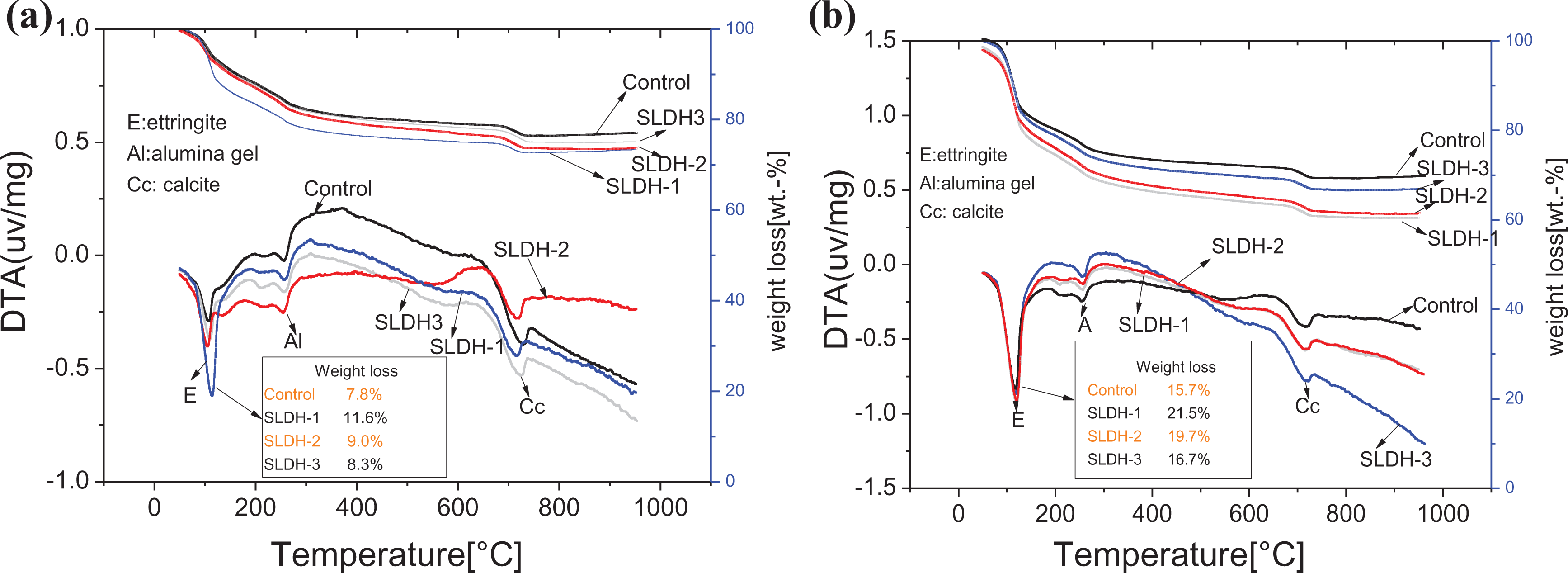
TG-DTA curves of the CBGM pastes with and without LiAl-LDHs of 2% by weight: (a) 1 day and (b) 7 days. LiAl-LDHs: LiAl-layered double hydroxides; TG-DTA: differential thermal analysis–thermogravimetry.
As shown in Figure 8(a), the weight loss of ettringite in the control paste was 7.8%, while the weight loss of the CBGM pastes with 2 wt% SLDH-1, SLDH-2, and SLDH-3 were 11.6%, 9.0%, and 8.3%, respectively. As the specific surface area of the LiAl-LDH particles increased, the weight loss of ettringite increased. Figure 8(b) shows the TG curves for the CBGM pastes with LiAl-LDH relative to the control paste at 7 days. The weight loss of the ettringite in the CBGM pastes without LiAl-LDH and with 2 wt% SLDH-1, SLDH-2, and SLDH-3 were 15.7%, 16.7%, 19.7%, and 21.5%, respectively. The results also illustrate that LiAl-LDH with larger specific surface area resulted in more hydration products, which explains why the compressive strengths of the pastes increased at all ages.
Conclusions
Based on the results of the investigation, the following conclusions can be drawn.
Three LiAl-LDHs slurries were successfully prepared through SNAS method. The specific surface area of three LiAl-LDHs slurries were 68.759 m2 g−1, 52.145 m2 g−1, and 34.518 m2 g−1 and increased with the decrease of volume ratio of water/ethanol in the solution. The specific surface area of LiAl-LDH directly affected the fluidity, setting time, bleeding, and compressive strength of CBGM pastes. LiAl-LDH with larger specific surface area led to poor fluidity, shorter setting time, and smaller bleeding rate of CBGM pastes under fresh condition and also yielded higher compressive strength of CBGM paste at both early and later age. The hydration process of CBGM paste can be accelerated by the addition of LiAl-LDH at early ages. There were no new hydration products detected in CBGM paste. With larger specific surface area, LiAl-LDH led to more hydration products, which is the reason why the compressive strength of CBGM paste was enhanced at all ages.
Footnotes
Acknowledgements
The authors would like to thank Dr Zhan from Department of Civil and Environmental Engineering, Hong Kong Polytechnic University (Hong Kong, China) for helping with proofreading.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research and Development Program of China (2017YFC0603004) and Doctoral Program of Henan Polytechnic University (No. B2019-39).
