Abstract
The compressive strength of sulfoaluminate cement-based grouting materials (CBGMs) cannot meet the requirement of repair and reinforcement engineering in the presence of high water-to-binder ratio (w/b) of 0.5-1.2. Using nanotechnology, the mechanical property of the CBGM paste at different ages can be improved and our previous experimental results showed that lithium aluminum layered double hydroxides (LiAl-LDHs) with nanostructure can significantly increase the compressive strength of CBGM paste. In addition to the characteristics of nanomaterials, the performance of nanomaterials is also related to w/b of paste. In this work, the effect of w/b ratio on the compressive strength, hydration process, and products of calcium sulphoaluminate cement (CSA) CBGMs with and without LiAl-LDH was studied. Results indicate that with the increase of w/b from 0.6 to 1.0, the growth rate of compressive strength of CBGM paste with LiAl-LDH enhanced. Moreover, LiAl-LDH enhanced total hydration heat more effectively at a higher w/b and as the w/b increased from 0.6 to 1.0, the growth rate of hydration product boosted, which is the reason why the enhancement ratio of compressive strength of the paste was larger at high w/b.
Introduction
Cement-based grouting materials (CBGMs) are usually applied in repair and consolidation and the suspension consists of cement, admixture, water, and possibly, gypsum or anhydrite. 1,2 The properties of grouting material are very important in grouting engineering. The ideal grouting material should have good fluidity during the pumping process and high compressive strength at the place need to be reinforced. The water-to-binder ratio (w/b) of CBGM for consolidation and repair is usually in the range of 0.5–1.5. 3 However, using high w/b (≥0.5), the early compressive strength of CBGM develops slowly, which cannot meet the requirements of special projects such as tunnels reinforcement, rock prestressing anchors and applications 4,5 Therefore, the early mechanical properties of CBGMs need to be further improved.
As a sulfoaluminate cement accelerator, lithium salt has been studied by many researchers. 6 –8 The results of the previous research studies indicate that the addition of lithium carbonate can significantly improve the early compressive strength but weaken the compressive strength at later ages. 9
Now, significant concerns have been raised about the effect of ultrafine powder on the performance of cement-based material. Due to the crystal nucleus and filler effect, a lot of nanomaterials were added to improve the mechanical property both in the portland cement-based materials 10 –12 and sulfoaluminate cement-based materials. 13,14
Based on the theory of crystal growth,
15
superfine powder with the same structure as hydration product of cement paste has better ability to promote the formation of hydration product. Layered double hydroxide (LDH), a class of layered anionic clay, has been investigated for many years.
16,17
The formula of the LDH can be generalized to [M1−
The w/b is a crucial parameter in grouting engineering. The w/b ratio influences mainly the porosity of the hydrated cementitious materials, 21 which influences the mechanical properties. And the difference in w/b 22 can lead to a different hydration rate and microstructure, which may affect the nucleation efficiency of nanomaterials.
In this study, the effect of w/b on the compressive strength of CBGM with and without LiAl-LDH was investigated, and a possible origin of the enhanced performances tested was suggested based on the results of hydration heat, X-ray diffraction (XRD), thermogravimetric and differential thermal analysis (TG-DTA), and Fourier transform infrared spectroscopy (FTIR).
Materials and methods
Materials
In this article, sulfoaluminate cement clinker, anhydrite, and quicklime were taken as binder materials. Sulfoaluminate cement clinker and anhydrite were purchased from Huayan cement plant and the quicklime was bought from Taihang lime factory, Henan Province of China. The calcium oxide content in quicklime was 65.3 wt%, including 15.5 wt% calcium carbonate. Blaine fineness of sulfoaluminate clinker and anhydrite was 350 and 365 m2 kg−1, respectively, as reported in our previous study. 2 The mineralogical, chemical composition of sulfoaluminate cement clinker and anhydrite determined X-ray fluorescence is provided in Tables 1 and 2, respectively. Naphthalene water reducer and sodium gluconate were purchased from Dinghao Technology Co. Ltd (Henan Province, China). LiAl-LDH was synthesized according to our previous experiment and the shape of LiAl-LDH was elliptical and the particle size was about 90 nm. 23
Mineralogical composition of the sulfoaluminate clinker/wt%.

Chemical composition of the CSA clinker and anhydrite/wt%.
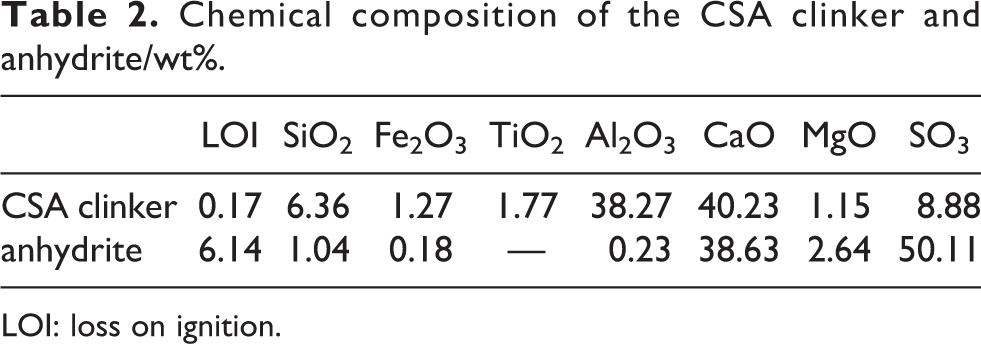
LOI: loss on ignition.
Preparation of CBGM paste
The mix formulas of component A and component B, as listed in Table 3, had been demonstrated optimum by our previous experimental work. 1 Slurry A and B were separately prepared by blending with water through a mixer for cement paste with a speed of 250 r min−1 for 15 min. The CBGM paste was prepared by mixing the slurry A and slurry B and the mass ratio of slurry A and B was 1:1. The w/b used in the study was 0.6, 0.7, 0.8, 0.9, and 1.0. LiAl-LDH was added to replace CBGM by 2.0 wt% to cast the specimens of cement composite.
Components of sulfoaluminate cement-based grouting material/wt%.

NAW: naphthalene water reducer; SMG: sodium gluconate.
Experimental procedure
The compressive strength of CBGM was tested according to Chinese National Standards GB/T 7897-2008. Specimens were cured in moist air at 20 ± 1°C with more than 90% relative humidity. Twenty-four cubic specimens were prepared for each mixture, each four of which were cured until 4 h, 1 day, 3 days, 7 days, 28 days, and 60 days, respectively. LiAl-LDH, 2 wt% of CBGM, was added to CBGM. The specimens less than 5 mm collected from the crushed piece of the compressive strength test were ended hydration by immersing in the absolute alcohol for several days, dried in a chamber at 20 ± 2°C until the weight is no longer lost and tested by XRD, FTIR, and TG-DTA. Crystallized structure was analyzed by XRD in the range 5–60° 2
Results and discussion
Compressive strength
The compressive strengths of CBGM pastes without and with LiAl-LDH are illustrated in Figure 1. At 4 h, both of the compressive strength of neat pastes and CBGM pastes with LiAl-LDH were decreased as the increase of w/b from 0.6 to 1.0. The 1-day 3-day, 7-day, 28-day, and 60-day compressive strengths of CBGM pastes presented a similar tendency with the compressive strength at 4 h. The result of Chen and Wu 24 also found that an increase in the water-to-cement ratio increases the total porosity and decreases the compressive strength. Compared to the control paste, the compressive strength of CBGM pastes with 2 wt% LiAl-LDH at 4 h was enhanced by 48.5%, 62.1%, 72.3%, 80.1%, and 89. 8% when the w/b ratio was increased from 0.6 to 1.0. A similar trend was also found at the 1-day, 3-day, 7-day, 28-day, and 60-day. In other words, the addition of LiAl-LDH enhanced the compressive strength of CBGM and the enhancement ratio of compressive strength of CBGM increased with the increment of w/b, as shown in Figure 2.

Compressive strength of CBGM cement paste with and without LiAl-LDH: (a) control sample and (b) CBGM paste with LiAl-LDH. CBGM: cement-based grouting material.

Enhancement ratio of compressive strength of CBGM cement paste with LiAl-LDH to that of control CBGM cement paste at different w/b ratios. CBGM: cement-based grouting material.
The reason that LiAl-LDH improved the compressive strength may be because LiAl-LDH served as the crystal seeds provide the nucleation sites, which prompt the formation of hydration products of cement. It is also inferred that the different compressive strength value of CBGM paste with different w/b is caused by the different amount of hydration products at different w/b.
Hydration released heat
The hydration heat evolution process of CBGM paste with and without LiAl-LDH is shown in Figure 3. The addition of LiAl-LDH had little effect on the time of first hydration heat peak of CBGM paste but decreased the heat flow rate of pastes with the increase of w/b from 0.6 to 1.0. Compared to the neat paste, LiAl-LDH shortened the time of second hydration released heat peak. And no matter whether the LiAl-LDH was added in the CBGM paste or not, the time of second hydration released heat peak was shortened as the w/b ratio increased. The effect of LiAl-LDH on the heat released from CBGM paste at the first 20 h was shown in Figure 3(b). It could be found that the addition of LiAl-LDH enhanced the heat release of CBGM paste at early ages. With the increase of w/b from 0.6 to 1.0, the hydration heat released from CBGM was firstly decreased and then increased. At 20 h, compared to the control paste, the total amount of hydration heat of CBGM paste with LiAl-LDH with 0.6, 0.8, and 1.0 w/b ratio was increased by 1.6%, 3.9%, and 14.9%, respectively.

Heat flow rate and hydration heat of CBGM paste with and without LiAl-LDH at different w/b ratios: (a) heat flow rate and (b) hydration heat. CBGM: cement-based grouting material.
In general, LiAl-LDH accelerates the hydration released rate and hydration heat of CBGM paste. Moreover, LiAl-LDH enhanced hydration heat more effectively at a higher w/b ratio.
XRD, FTIR, and TG-DTA
Figure 4(a) shows the XRD patterns of the CBGM paste at 1 day. The main hydration product was ettringite and C3AH6 whether the LiAl-LDH was added in the CBGM or not. Diffraction peaks of thaumasite around 9.2°, 16.0°,18.5°, 19.3°, 23.5°, 26.0°, and 28.0° were discovered. 25,26 Diffraction peaks of calcite can be found in XRD patterns, and the reason is that it is a part of the cement from the beginning and its increase is a result of the carbonation reactions. The structure of thaumasite was similar to that of ettringite. The results of Juel et al. 27 had shown that thaumasite is unstable in the presence of AFm phases and can form stable phase of thaumasite using the model based made up of CaCO3, Fe2O3, SiO2, CaSO4, MgO, Al2O3, CaO, and H2O.

XRD and FTIR patterns of CBGM with and without LiAl-LDH at 1 day: (a) XRD and (b) FTIR. CBGM: cement-based grouting material; XRD: X-ray diffraction; FTIR: Fourier transform infrared.
The hydration product of aluminum hydroxide was not observed because it may be amorphous. The incorporation of LiAl-LDH was not beneficial to form a new phase. The intensity of ettringite increased and unhydrated yeelimite decreased when adding 2% LiAl-LDH at different w/b ratio, indicating that LiAl-LDH prompted the hydration of CBGM paste.
In general, LiAl-LDH accelerated the hydration of CBGM paste and did not get the new hydration products formed at different w/b.
The FTIR spectra of the CBGM pastes with and without LiAl-LDH are shown in Figure 4(b). Vibration frequency characteristics located at 3635 and 3485 cm−1 were related to the free and associated OH bands of aluminum hydroxide, respectively 28 Hydration products of ettringite with the [SO4]2− vibrations and OH bands seated at 1115 29,30 and 3635 cm−1 were discovered on the spectra. The Al–OH stretching vibrations were found at 987 cm−1. 31 The double band generated by the [CO3]2− v3 vibrations was seated at 1423 cm−1 indicating the existence of calcite in the pastes. The vibration frequencies located at 630 and 522 cm−1 were associated with SiO6 group of thaumasite. 32,33 The addition of LiAl-LDH at different w/b ratios was not beneficial to form a new hydration product, which is an agreement with the analysis result of XRD.
Figure 5 illustrates the TG-DTA patterns of CBGM paste at w/b ratio of 0.6, 0.8, and 1.0. At 1 day, the endothermic peaks located at 110°C, 260°C, and 730°C are associated with the dehydration of the lattice water of ettringite, the dihydroxylation of aluminum hydroxide, and the decomposition of calcite. Endothermic peak of thaumasite sited at 145°C was also observed at the patterns. Matschei and Glasser 34 had found that the thaumasite dehydrated the lattice water at about 140°C. The mechanism of thaumasite formation is still under discussion. 35,36 The exothermic peak at 520°C is mainly caused by the dehydration of tricalcium aluminate hydrate. 37 With the increment of w/b ratio from 0.6 to 1.0, the weight loss of the CBGM paste was increased. When adding 2 wt% LiAl-LDH, compared to the neat paste, the weight loss of the CBGM paste was increased. At w/b ratio of 0.6, the weight loss of ettringite of CBGM paste without and with LiAl-LDH was 6.1% and 6.2%, and the enhancement ratio of ettringite was 1.3%. Similarly, the enhancement ratio of ettringite at w/b of 0.8 and 1.0 was 25.3% and 32.6%. It can be seen that the addition of LiAl-LDH improved the amount of hydration product more effectively under higher w/b ratio, which is the reason that LiAl-LDH can improve the compressive strength of CBGM paste more effectively under higher w/b ratio.

TG-DTA patterns of CBGM with and without LiAl-LDH with w/b of (a) 0.6, (b) 0.8, and (c) 1.0 at 1 day. CBGM: cement-based grouting material; TG-DTA: thermogravimetric and differential thermal analysis.
Conclusions
In this work, the effect of w/b on the properties of CSA CBGMs with and without LiAl-LDH was investigated. Some conclusion was shown as follows: The w/b ratio affected the enhancement performance of LiAl-LDH in the CBGM paste. Compared to the control paste, the growth rate of compressive strength of CBGM paste with LiAl-LDH was enhanced with the increase of w/b ratio from 0.6 to 1.0. The addition of LiAl-LDH improved the hydration of CBGM paste at early ages. With the increase of w/b ratio, the hydration heat released from CBGM was firstly decreased and then increased, and LiAl-LDH enhanced hydration heat more effectively at a higher w/b ratio. With incorporating LiAl-LDH, there is no new phase formed in the cement paste. Due to seed effect, the addition of LiAl-LDH increases the amount of hydration products. Compared to the control paste, the growth rate of hydration product was boosted with the increase of w/b ratio, which is the reason why the enhancement ratio of compressive strength of the CBGM paste was larger with higher w/b ratio.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Key Research and Development Program of China (2017YFC0603004).
