Abstract
The present report offers a survey, with re-examination and extension of data, of previously published intermediate papers on the functional recovery of manual dexterity in non-human primates following primary motor cortex (M1) lesion, either spontaneous or enhanced with 2 distinct therapies. For each monkey, the M1 lesion volume was derived from histological sections. As a result of data extension, spontaneous recovery is represented here by individual data points derived from 14 monkeys, while data points from 8 monkeys represent therapy enhanced recovery. One therapy consisted of the administration immediately post-lesion of an anti-Nogo-A antibody (n = 4); the other treatment was a delayed autologous cellular therapy (ANCE; n = 4). The manual dexterity performance was longitudinally assessed during several years with the modified Brinkman Board task, testing the ability to grasp small food pellets from horizontal or vertical slots, using the precision grip. After M1 lesion, the grasping score dropped to zero, followed by a spontaneous, incomplete recovery in untreated monkeys, characterized by a unique plateau of recovery. In the majority of treated monkeys (n = 6), there was an additional second plateau of recovery, reflecting the effect of the treatment. Statistical non-parametric comparisons between the 14 untreated versus the 8 treated data points showed a statistically significant difference between the 2 subpopulations, based both on univariate statistics and bivariate statistics taking into account the lesion volume. This was true for recovery expressed as a degree of recuperation expressed in percent (post-lesion score/pre-lesion score × 100) as well as for the post-lesion scores only. The difference between the 2 subpopulations was more pronounced for the most difficult horizontal slots than for the less challenging vertical slots. In conclusion, the treatments appear to be efficient to promote functional motor recovery, even when considering post-lesion behavioral readouts only, as this would be the case in clinical trials.
Keywords
Introduction
The exquisite ability to finely control finger movements in primates, corresponding to manual dexterity, depends to a large extent on the primary motor cortex (M1), via its corticomotoneuronal projection directly contacting the cervical and thoracic motoneurons1 -6; but see Refs.7 ,8 Over the last 3 decades, our laboratory investigated in Macaques the effect on manual dexterity of unilateral lesion of the hand representation in M1.9 -19 These studies can be shortly summarized as follows:
A lesion of the hand representation of M1 provokes a dramatic loss of manual dexterity of the contralesional hand (Figure 1).
After a few weeks, a spontaneous functional recovery progressively takes place, reaching a plateau, corresponding to an incomplete recovery (Figure 1A), whose extent depends in part on the M1 lesion volume.
Following a lesion of M1 at neonatal stage, a “new” hand area was observed later at adult stage adjacent to the lesion in M1, at a location normally devoted to more proximal motor territories (wrist, elbow, shoulder).
Following a large lesion of the hand area in M1 at adult stage, the functional recovery is not mainly supported by a rearrangement of territories adjacent to the lesion in M1, but rather by the intact ipsilesional premotor cortex, while other cortical and subcortical areas may contribute as well.
The administration of a treatment, either anti-Nogo-A antibody or an autologous cellular therapy, enhanced the functional recovery in the form in most cases of a second plateau of manual dexterity performance, added to the first plateau of spontaneous recovery (Figure 1B and C).

Cartoons representing typical time courses of manual dexterity behavioral data obtained from monkeys subjected to a unilateral M1 lesion (vertical purple line) and for the contralesional hand. The manual dexterity score is in ordinate and time (months) in abscissa: (A) In 8 control monkeys subjected to the M1 lesion, without any treatment, there was a dramatic drop of manual dexterity after the lesion, followed by a progressive recovery until reaching a plateau, reflecting an incomplete spontaneous functional recovery. The vertical arrow represents the post-lesion recovered score, (B) In 4 monkeys subjected to M1 lesion, an anti-Nogo-A antibody treatment was administered immediately after the lesion. In 2 of them (dashed red curve), a single plateau of functional recovery was reached. In the other 2 monkeys, there was a first plateau of functional recovery (black curve), on to which a second plateau emerged subsequently (solid red curve), presumably as a result of the treatment, and (C) In 4 monkeys subjected to M1 lesion, an autologous cellular therapy (ANCE) was applied 2 to 3 months after the lesion. In all of them, a single plateau of functional recovery was reached first (black curve). Later, in all 4 monkeys, a second plateau emerged subsequently (green curve), presumably as a result of the cellular therapy. The black vertical arrow represents the spontaneous post-lesion score, before the ANCE treatment exerted its effect. The green vertical arrow toward up represents the ANCE enhanced recovered score.
However, as it is often the case for studies conducted on non-human primates, previous reports from our laboratory (see above) were issued as restricted projects, dispersed at various time points over 3 decades and based on a limited number of monkeys. The goal of the present re-examination article is to offer here a comprehensive re-examination and survey of these data by assembling them altogether, as well as introducing an original extension of the number of data points, in order to provide more robust quantitative behavioral results.
Material and Methods
Macaque Monkeys
The present report includes an overall collection of 16 adult Macaques (Macaca fascicularis; Table 1), all subjected to a permanent unilateral M1 lesion at adult stage, assembled over consecutive studies conducted in our laboratory.10 -20 The methodological and ethical aspects of these studies were reported in detail in the original intermediate reports and are therefore not repeated here, including the M1 chemical lesion procedure (infusion of the excitotoxic ibotenic acid).10 -20 Eight monkeys were true control animals as they were not subjected to any treatment (Treatment “None” in Table 1), therefore illustrating the spontaneous functional recovery from the M1 lesion (as depicted in Figure 1A). Four monkeys (Treatment “Anti-Nogo-A” in Table 1) were treated with an anti-Nogo-A antibody, to neutralize the inhibition of neurite growth in adults due to the presence of Nogo-A.21,22 Four other monkeys (Treatment “ANCE” in Table 1) were subjected to an autologous cellular therapy (ANCE).12,19,23 -27 Precise and extensive descriptions of these 2 treatments applied to our monkeys were reported previously.11,16,17,19,28 -31 These 2 subgroups of “treated” monkeys were pooled together for further quantitative analysis, based on a rationale presented in the discussion section. The data points extracted from these 16 M1 lesioned monkeys were extended to 22 overall, as follows. As depicted in Figure 1B and C and Table 1, 6 out of the 8 “treated” monkeys exhibited a “two plateau” functional recovery of manual dexterity time course: a first plateau (black curves in Figure 1B and C) reflecting spontaneous recovery, followed by a second plateau (red or green curves in Figure 1B and C), reflecting the treatment enhanced functional recovery. In order to extend our “control” data points (spontaneous recovery), the present report considers the first plateau of these 6 “treated” monkeys as a “control-like” data point, representative of the spontaneous recovery before the treatment exerted its effect. These 6 additional “control-like” data points are listed as such in Table 1 (Treatment “Control-like”). As a result of this extension, there are overall 22 data points (Table 1): 14 “control” data points versus 8 “treated” data points.
List of the Adult Monkeys Subjected to a Primary Motor Cortex (M1) Lesion, With Their ID (Leftmost Column) and the Volume of the Lesion (Second Column).

Note. The Third Column from the Left Indicates the Corresponding Therapy or Treatment. The Fourth Column from the Left Indicates the Absence (N) or Presence (Y) of a Second Plateau of Functional Recovery of Manual Dexterity; In Case of the Presence of a Second Plateau of Recovery, the First Plateau was Considered as Spontaneous Recovery, Thus Yielding an Additional “control-like” (ie, No Treatment) Data Point for the Corresponding Monkey. Data Points in the Lines from “Mk-BI” to “Mk-LO – first pl” thus Represent Spontaneous Functional Recovery, While Data Points in the 8 bottom Lines Represent Treatment Enhanced Functional Recovery (anti-Nogo-A Antibody or ANCE, Respectively). The 6 Rightmost Columns List the 6 Behavioral Parameters Considered in the Analysis (See Text). In these 6 Columns Headings, V is for Vertical Slots and H is for Horizontal Slots. In the 2 Rightmost Columns, “post” Is for “post-lesion” While the Score Corresponds to the Median Value at First or Second Plateau. The Monkeys’ IDs are the Same as in Previous Reports from This Laboratory. The sex of the monkeys is indicated by the small letters m (male) or f (female) next to the ID in the first column. The age of most monkeys at time of the lesion ranged from 3.5 to 5 years old, except the older monkeys Mk-DG (9.5 years), Mk-DI (9.5 years), Mk-AN (14 years), Mk-CA (11 years) and Mk-LO (11.5 years).
The original behavioral data curves (manual dexterity scores as a function of time over several consecutive years), as depicted in Figure 1, were published previously for all monkeys.10 -12,14,16 -20,32 This is also the case for illustrations of the precise location and extent of the M1 lesion, as they appear on a lateral view of the brain.10 -20 The original experimental studies were covered by the official veterinary authorizations No FR 24/95/1; FR 44/92/3; FR 157/01, FR 157/03, FR 166-03, FR-166-05, FR 157/04, FR 156/04, FR 156/06, FR 157e/06; FR 206/08, FR 185-08, FR 192/07, FR 192/07E, 19017, 22010, 2014-FR-42E, FR 17-09, FR 18-10, all delivered by the Cantonal (Fribourg) and Federal (Swiss) veterinary authorities. Note that, in Switzerland, the animal experiments are not ethically assessed by an internal Institutional Review Board (IRB), but by specialized and independent ethics approval committees under the supervision of state (Canton of Fribourg: “commission cantonale d’expérimentation animale” and “vétérinaire cantonal”) and federal (Switzerland: “office vétérinaire fédéral”) authorities. The veterinary authorizations listed above by their specific and official numbers covered all experimental interventions conducted on the 16 monkeys comprised in the present report (see Table 1), including the procedure to produce the lesion of M1 using infusion of ibotenic acid in each monkey.
Manual Dexterity Behavioral Task
In the present report, manual dexterity was quantified exclusively with our standard behavioral test, the “modified Brinkman Board” task, described in detail on the methodological point of view earlier 14 ; see also Refs.9,10,11,12,14,17,19,20,28,30,32 -37 Pictures (Figure 2) and a video sequence (Video 1 in supplemental data attached to the present report) also illustrate a typical extract of a daily session of the modified Brinkman Board task executed by a monkey before the M1 lesion. Previous reports also display video sequences showing the manual dexterity behavior, before the M1 lesion, during the acute phase post-lesion, as well as when the first plateau and second plateau were reached.12,17,19 The time point (experimental session day) at which a plateau was reached, either pre- or post-lesion, was defined based on a quantitative criterion, as previously reported.12,17 -20,34
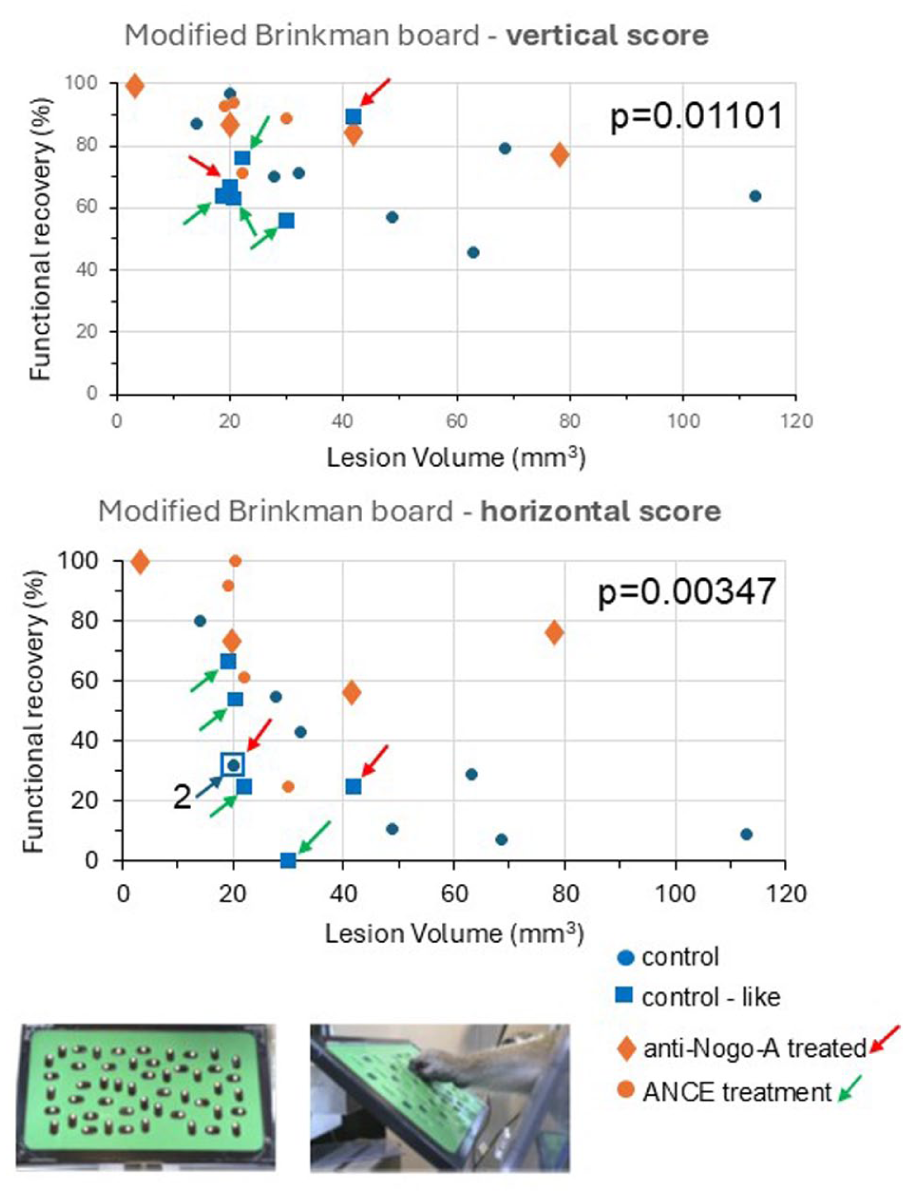
Assessed with the modified Brinkman Board task, the functional recovery of manual dexterity (%) in ordinate was plotted as a function of M1 lesion volume (mm3) in abscissa, for the 22 data points listed in Table 1. Vertical slots are in the top graph and horizontal slots in the bottom graph. The 22 data points are subdivided in the 4 subgroups as in Table 1: blue circles for the control (spontaneous) functional recovery, blue squares for the “control-like” (spontaneous) functional recovery, brown diamonds for the anti-Nogo-A antibody treated functional recovery and brown circles for the cellular therapy functional recovery. As the blue squares represent the first plateau of recovery in monkeys in which a treatment or therapy was applied, red or green arrows point to a blue square data point associated to a brown diamond or a brown circle symbol on the same fictive vertical line, corresponding to the second plateau of recovery in the same monkey. The gray arrow with the “2” number indicates that 2 data points are superimposed (see Table 1). The P-values provide the results of a graphical bivariate non-parametric comparison with the Mann and Withney test between the blue versus brown data points (see also Table 2 and Supplemental Figure 1). The 2 insets in the bottom part of the Figure illustrate the modified Brinkman Board, with the distribution of the 25 horizontal slots and the 25 vertical slots on the left and, on the right, a lateral view of a pellet grasped with the right hand of a monkey.
Statistical comparisons (P values).

Note. List of P-values obtained from

Graphs displaying functional recovery (%) of manual dexterity using the same 22 data points as in Figure 2, but combining in 2 different ways the vertical slots and the horizontal slots. In the top graph, the % of recovery for vertical slots and the % for the horizontal slots were averaged. In the bottom graph, these same two% were multiplied. Otherwise, same conventions as in Figure 2. The corresponding data points are listed in Table 1 (seventh and eighth columns from left). The P-values provide the results of a graphical bivariate non-parametric comparison with the Mann and Withney test between the blue versus brown data points (see also Table 2 and Supplemental Figure 1). In addition, tendency curves based on a power function were added, with the corresponding formulas and coefficients of determination (below each graph).
The modified Brinkman Board task aims at quantifying separately for each hand the monkey’s ability to grasp small food pellets from spatially restricted slots, requesting to perform the precision grip defined as an opposition of the thumb and index finger. The modified Brinkman Board comprises 25 vertically and 25 horizontally oriented wells, a version that was elaborated from the original Brinkman Board.38 -40 For the vertical slots the hand was in its neutral posture whereas, for the horizontal slots, a wrist/arm rotation (ulnar or radial deviation) was needed in addition to the precision grip. 34 The horizontal slots were thus more challenging than the vertical slots. The daily manual performance in the modified Brinkman Board task was expressed as a score given by the number of pellets retrieved during the 30 first seconds of the task. The score was computed separately for the vertical and horizontal slots.
The analysis of the functional recovery of manual dexterity after M1 lesion was conducted based on the median score values, established pre-lesion and post-lesion at plateau (as previously published). These 2 median score values were used to derive the percentage of functional recovery, given by dividing the post-lesion median score by the pre-lesion median score, multiplied by 100 (fifth and sixth columns in Table 1). These percentages of functional recovery established for the vertical (% V) and horizontal (% H) slots were combined for further analysis: (i) average of %V and %H (seventh column of Table 1).; (ii) %V × %H (eighth column of Table 1). Finally, in order to mimic a “clinical trial” without knowing the pre-lesion performance, the post-lesion scores only were also considered (ninth and tenth columns in Table 1). All these behavioral parameters were represented graphically in plots (Figures 2-4; Supplemental Figure 1), as a function of the monkey’s respective lesion volume (second column in Table 1). Moreover, the data displayed in the 6 rightmost columns of Table 1 were represented in the form of bar graphs in the Supplemental Figures 2 and 3, without taking into account the M1 lesion volumes. Finally, the variability of the behavioral performance was assessed by calculating the standard deviations (SDs) derived from the daily sessions at the different plateaus of performance (Supplemental Figure 4 and Supplemental Table 1).

Graphs displaying the manual dexterity median scores observed post-lesion with the modified Brinkman Board task, for the vertical slots (top graph) and for the horizontal slots (bottom graph), as a function of M1 lesion volume. Otherwise, same conventions as in Figure 2. The corresponding data points are listed in Table 1 (2 rightmost columns). The P-values provide the results of a graphical bivariate non-parametric comparison with the Mann and Withney test between the blue versus brown data points (see also Table 2 and Supplemental Figure 1).
Statistical Analysis
The statistical analysis aimed at comparing the 2 subpopulations of data points, corresponding to “control” and “control-like” data points (n = 14) on one hand versus “treated” data points on the other hand (n = 8). The comparison was made based on the non-parametric Mann and Withney test, using 2 approaches. First, a standard univariate comparison, restricted to the percentage or score values, without taking into account the lesion volume. Second, a graphical bivariate comparison, tentatively integrating the lesion volume impact on the post-lesion performance. This second approach is illustrated in the Supplemental Figure 1 and was used in our previous study on cervical cord injury. 30 Table 2 lists the P-values derived from these 2 approaches, for all 6 behavioral parameters mentioned above.
Further statistical analysis was conducted to compare the manual dexterity performance for pellets’ retrieval from the vertical slots versus the horizontal slots. As the data are paired, that is, a comparison within each monkey, the non-parametric Wilcoxon signed-rank test was applied. Statistical calculations (Mann and Withney test, Wilcoxon signed-rank test, coefficient of correlation) were performed using the free software “www.socscistatistics.com.”
Results
The percentages of functional recovery after M1 lesion were plotted as a function of the lesion volume, for the 22 data points (Table 1) and separately for the vertical and horizontal slots in Figure 2. The 2 graphs confirm the notion that functional recovery tends to decrease for increasing lesion volumes, a tendency more prominent for horizontal slots than for vertical slots. The red and green arrows allow a direct comparison between the first plateau recovery and the second plateau recovery in the 6 treated monkeys characterized by the presence of a second plateau (Table 1). Two different colors (blue and brown) were used to confront the “control” and “control-like” data points on one hand with the “treated” data points on the other hand (Figure 2). These 2 subpopulations were used to apply a graphical bivariate non-parametric statistical test, as illustrated in Supplemental Figure 1. For both the vertical slots and the horizontal slots, the result of the bivariate statistical test (Mann and Withney) was that the 2 subpopulations are significantly different, with a P value of .01101 and .00347, respectively (Table 2). In other words, the “treated” monkeys recovered better than the “control” monkeys, including the control-like data points in the latter subpopulation. Based on an univariate statistical test (Mann and Withney test also), similar conclusions are met, with however slightly lower P values (Table 2). The numbers of behavioral daily sessions from which the median score data were derived at each plateau are indicated in the Supplemental Table 1. The median numbers of daily behavioral sessions pre-lesion were 16 and 14.5 in the “untreated” and “treated” subpopulations, respectively. Post-lesion, the corresponding median numbers of sessions were 20.5 and 25.5, respectively.
In 20 out of 22 data points (Table 1), the percentage of functional recovery was higher for the vertical slots than for the horizontal slots, an observation consistent with the higher difficulty to retrieve pellets from horizontal slots than from vertical slots (see also the last paragraph of the results’ section for a statistical comparison between vertical slots vs horizontal slots). However, the differences in percentage of functional recovery between vertical and horizontal slots within the same monkey was quite variable, ranging from 0% to 72%. Due to this large interindividual variability with respect to the slot orientation, in order to yield a more representative functional recovery figure across monkeys, a combination of the two% of functional recoveries within each monkey may be appropriate. In Figure 3, the individual percentages of functional recovery for vertical and horizontal slots were combined in 2 ways: in the top graph, the percentages were averaged, while in the bottom graph the percentages were multiplied, again segregating the 4 subgroups with different symbols (as in Figure 2). Applying the same 2 statistical tests as above for Figure 2, Table 2 shows that the 2 subpopulations (“untreated” in blue and “treated” in brown) are significantly different, with P values somewhat smaller than those observed for the individual percentages of functional recovery when considering the 2 slot orientations separately (Table 2). In Figure 3, the 2 subpopulations (blue and brown) were tentatively fitted with a power function. In the top graph, the coefficient of correlation for the blue data points (r = .7299) was statistically significant (P = .00304), while for the brown data point (r = .605) it was not statistically significant (P = .11204). In the bottom graph, similarly the coefficient of correlation for the blue data points (r = .8064) was statistically significant (P = .00049) while there was no statistically significant correlation for the brown data points (r = .6148; P = .10479).
In the data reported above, the functional recovery was expressed as a percentage of recovery, yielded by dividing the post-lesion score by the pre-lesion score, a possibility offered by the present non-human primate model. In clinical research, the pre-lesion data for the same behavioral task used post-lesion are not available. As a result, only the post-lesion scores of the patients are available for the statistical analysis. The post-lesion only scores data were plotted here for our monkeys as a function of M1 lesion volume (Figure 4), using the same conventions as in Figures 2 and 3. The relationships between scores and lesion volumes observed in Figure 4 are consistent with those exhibited in Figures 2 and 3, namely a general decrease of post-lesion score for increasing M1 lesion volumes. The important issue here is the statistical comparison between the 2 subpopulations (blue and brown symbols; Figure 4). As indicated as inset on the top right of each plot, the bivariate statistics including the impact of the M1 lesion volume yielded different results depending on the slot orientation: the difference between the 2 subpopulations was not statistically significant for the vertical slots (P > .05; see also Table 2), while it was statistically significant for the horizontal slots (P = .00187). However, usually in clinical trials when patients are still alive, the information about the lesion volume may not be available, except in case of systematic MRI acquisition. For this reason, it may also be appropriate to consider an univariate statistics, considering the scores data only, independently of the lesion volume. The univariate non-parametric statistics yielded a statistically significant difference between the 2 subpopulations (Table 2), with P values of .03836 for the vertical slots and .00187 for the horizontal slots. The behavioral data represented in Figures 2 to 4 as a function of the M1 lesion volume are illustrated in the form of bar graphs in Supplemental Figures 2 and 3. These graphs further display the difference between the “untreated” and “treated” subpopulations, especially for the horizontal slots or when the latter were combined with the vertical slots.
Back to the slots’ orientation, the difference in manual dexterity performance for retrieving pellets from vertical slots versus horizontal slots was tested statistically based on the Wilcoxon signed-rank test, applied to the data listed in Table 1. First, the percentages of functional recovery were compared in the subpopulation of “untreated” monkeys, yielding a statistically significant difference between vertical slots versus horizontal slots (P < .01), supporting the observation of higher recovery in vertical slots than in horizontal slots. In the “treated” monkeys’ subpopulation, the percentages of functional recovery were also significantly higher for vertical slots than for horizontal slots (P < .05). Second, the statistical comparison was conducted on the scores post-lesion (2 rightmost columns of Table 1). In all monkeys, the vertical score was larger than the horizontal score, thus yielding statistically significant differences (P < .01) in both monkeys’ subpopulations (in “untreated” and in “treated” monkeys).
The data reported above were based on the median score values assessed at the different plateaus (Figures 2-4). It is also of interest to investigate the dispersion of the behavioral score data derived from the modified Brinkman Board task. To this aim, the standard deviations (SDs) were calculated for the vertical and horizontal scores separately, at pre-lesion and at post-lesion plateaus. These SDs data are listed in Supplemental Table 1, and represented graphically in Supplemental Figure 4. Overall, the SDs ranged from 0.59 to 3.27 pellets recovered in 30 seconds across the 16 monkeys. For both vertical scores and horizontal scores (Supplemental Figure 4: panels A and B, respectively), the distributions of SDs post-lesion as a function of SDs pre-lesion are comparable in the 2 subpopulations “untreated” and “treated.” The SDs for the vertical slots were confronted to the SDs for the horizontal slots in panels C and D of Supplemental Figure 4. Pre-lesion, the SDs for the 2 slots orientations were significantly correlated in both the “untreated” and “treated” subpopulations (panel C). In contrast, post-lesion, the SDs for the 2 slots orientations were significantly correlated in the “treated” subpopulation (panel D), but not in the “untreated” subpopulation.
Discussion
Re-examination of previously published data, consisting mainly here in extending the number of data points (see Table 1: particularly the number of “control” ones), provided a confirmation that the 2 treatments considered and cumulated here for the statistical analysis (anti-Nogo-A antibody and ANCE cellular therapy) indeed enhanced the functional recovery of manual dexterity after a M1 lesion affecting the hand area, in line with previous intermediate reports from this laboratory.12,15,17,19 It is noticeable that among the 8 “true” control monkeys (M1 lesion, untreated) none of them exhibited a second plateau of recovery following an initial first plateau of recovery. In sharp contrast, 6 out of 8 treated monkeys exhibited a second plateau of functional recovery. In particular, a second plateau was observed in all 4 ANCE treated monkeys see also Refs.12,19 In the 4 anti-Nogo-A antibody treated monkeys, 2 exhibited a second plateau (Mk-VA and Mk-MO); a third monkey of this subgroup (Mk-LA) turned out to have received such a small M1 lesion (Table 1), that the initial plateau reached 100% recovery, leaving no possibility for the treatment to further improve the recovery; the fourth monkey (Mk-SL) showed a considerable recovery at first plateau already, in spite of a rather large M1 lesion, possibly explaining why the treatment did not provide any further significant improvement see also Ref. 17 Nevertheless, there is still a contrast between the 2 subpopulations (“control” vs “treated”) with respect to the absence/presence of a second plateau, most likely reflecting an enhancement effect of the corresponding treatment.
Beyond the difference reflected by the absence/presence of a second plateau of recovery, the statistical analyses summarized in Table 2 clearly show that the 2 subpopulations (“untreated” vs “treated”) are statistically significantly different, for the 6 parameters considered and irrespectively of the statistical approach (univariate vs bivariate). The only exception is the median post-lesion score for the vertical slots (P = .0548). The aim of introducing here a bivariate approach was an attempt to take into account the impact of the lesion. Surprisingly, as compared to the univariate approach, the bivariate comparisons did not yield very different statistical results, the P values being fairly close (Table 2), with a general tendency toward smaller P values derived from the univariate approach. This observation is encouraging for the clinical trials having limited access to the lesion volume after stroke for instance (unless a systematic MRI investigation is conducted), as it suggests that a univariate statistical comparison based on post-lesion behavioral scores only may be sufficient to test the validity of a treatment. However, the results displayed in Table 2 suggest that the behavioral readout used to evaluate the degree of functional recovery needs to be sufficiently challenging, as indicated by clearly lower P values derived from the horizontal slots than from the vertical ones. The introduction of even more challenging behavioral tests may be considered. 32
As far as the anti-Nogo-A antibody treatment is concerned, the present report extending previous intermediate reports from this laboratory15 -17 does reinforce the notion that such a treatment may improve functional recovery of manual dexterity after a lesion affecting the primary motor cortex. Although evidence for such anti-Nogo-A antibody treatment benefit after cortical lesion was provided earlier in rodents,41 -47 the present report remains original as far as non-human primates are concerned, together with its associated intermediate reports from this laboratory.15 -17 Changes in Nogo-A receptors or upregulation of Nogo-A on reactive astrocytes following cortical lesion were reported in non-human primates.48,49 A benefit of treatments blocking Nogo-A inhibitory action was already reported earlier in non-human primates following spinal cord injury.28,30,50,51 Another antibody treatment targeting myelin-associated glycoprotein was reported to facilitate functional recovery following focal cortical ischemic infarct. 52
The autologous ANCE cellular therapy also provided a benefit for the recovery of manual dexterity following M1 lesion, as compared to a large number of control animals (Figures 2-4), confirming previous intermediate reports.12,19 Beneficial cellular therapies, but heterologous, in Macaques after motor cortex lesion were previously reported.53 -56 Other therapeutic approaches were reported to enhance functional recovery after motor cortex lesion, such as electrical stimulation,57 -59 administration of inosine, 60 or motor rehabilitative training.57,61 -64
The present observations related to recovery of manual dexterity following a lesion of M1 may be impacted by the sex of the monkeys, 65 as well as by their age.66,67 However, as discussed earlier in detail, 19 the distributions of monkeys within our 2 subpopulations of monkeys (“control” vs “treated”) with respect to sex and age are largely comparable (Table 1), making a significant impact of these 2 parameters unlikely, or minor. The panel B of the Supplemental Figure 2 indicates on the bar graph the sex of each individual animal. It appears that females and males are well intermingled, without obvious bias for instance of males predominantly located at low values and females at high values. Such a bias, not present, would be problematic as females were shown to recover to a larger extent than males after motor cortex lesion. 65 As far as age is concerned, a mild deficit of motor function was reported in aged monkeys (15-26 years old).66,67 Our monkeys were younger, actually much younger in 13 out of 16 of them (see Table 1), suggesting that age did not strongly impact on the present behavioral data.
As reported earlier, 19 irrespective of “untreated” or “treated” subpopulations, our monkeys largely recovered post-lesion a pattern of grasping close to the original one pre-lesion, a restoration condition which favors better recovery than compensatory movements and/or strategies of substitution. 62 This is also true for the anti-Nogo-A antibody treated monkeys, which were not included in the study of Roux et al. 19 Only very minor changes in a minority of (“control”) monkeys were observed post-lesion see Ref. 19
In case of neonatal M1 lesion 9 or tiny lesion in adults,68 -70 a reorganization of the territory immediately adjacent to the lesion takes place, in the form of a new hand area to the detriment of intact sectors originally devoted to more proximal muscle representations (eg, wrist, elbow). After larger M1 lesion affecting the entire hand representation or even additional territories, mechanisms involving neuroplasticity of non-primary motor cortical areas (eg, PM, SMA) have been proposed,10,16,55,58,63,71 -85 among others. The anti-Nogo-A antibody treatment typically promotes post-lesion fiber sprouting from both severed (restorative sprouting) and intact (compensatory sprouting) axons.20,21,29 For the ANCE treatment, as other cellular therapies, mechanisms of recovery are multiple and very diverse: for instance, establishment of new connections, stimulation of the reconstruction of local neural circuits, reduction of secondary damage, promotion of cellular and/or synaptic plasticity, modulation of inflammation, enhancement of local angiogenesis, and/or delivery of trophic factors, to cite a few of them.12,31,37,53,55,56,59,60,82,85 -100 Following a lesion of M1, the multiplicity of fronto-parietal spinal command modules,101 -109 with some redundancy among them, offers a large palette of tools and scenarios in order to supply the loss of M1. The mechanisms underlying the enhancement of functional recovery of manual dexterity after treatment are different for the anti-Nogo-A antibody treatment and for the ANCE treatment, with more diverse and complex mechanisms for the latter as compared to the former (see above). These involved mechanisms have been reported and discussed in detail earlier, for the anti-Nogo-A antibody treatment15 -17,21,22,28 -30 and the ANCE treatment,12,19,25 -27,31 and are therefore not repeated here.
Mainly for statistical purposes, in order to reach a sufficient number of data points in the “treated” subpopulation, the anti-Nogo-A antibody treated monkeys and the ANCE monkeys were pooled. The rationale for such a pooling is not only based on statistics’ purposes, but also because these 2 subgroups share some common properties. First, the gain of functional recovery is comparable, in the order of 20% to 40% across monkeys.12,17,19 Second, both treatments favored the occurrence of a second plateau of functional recovery, especially in the ANCE subgroup. A second plateau was never observed in “control” monkeys (n = 8; M1 lesioned monkeys without treatment).12,17,19,20,32 Third, the delay between the onset of the treatment and the occurrence of its effect is consistent for the 2 subgroups (around 3 months). Fourth, the time course of the second plateau from its onset to its stable level is also comparable.12,17,19 Fifth, as a result of M1 lesion, the connectional changes observed for the efferent corticoreticular and corticorubral projections originating from the ipsilesional PM are comparable for both subgroups as well. 85 All these comparable properties between the anti-Nogo-A antibody and ANCE treatments support the present strategy to pool the monkeys subjected to one or the other of these 2 treatments.
The present study is based on behavioral data derived from the modified Brinkman Board task, which has proven to be pertinent to assess manual dexterity in non-human primates. However, this motor test has some limitations. Facing 25 vertically and 25 horizontally oriented slots, the monkeys may be tempted to introduce some bias by choosing to favor the vertical slots, which are less challenging (see results). As a consequence, in particular in the post-lesion phase, the number of horizontal slots visited may be low. The modified Brinkman Board task may also be considered as moderately challenging, especially the vertical slots. For this reason, a much more challenging version of the task was introduced, in the form of a “Brinkman Box” task, executed in absence of visual control as well as in a restricted space, limiting the degree of freedom for executing the grasping of pellets.14,32 The Brinkman Box task without vision yielded results consistent with the present ones, with “treated” monkeys outperforming “control” monkeys, though on a smaller pool of monkeys. 32 Moreover, the modified Brinkman Board task does not test quantitatively the ability of the monkeys to develop different levels of force when opposing the index finger and the thumb. To this aim, an additional test was introduced in our laboratory, namely the “reach and grasp drawer” task, measuring the load force and the grip force to pull open a drawer with the index finger and thumb exerting these forces on the drawer’s knob.14,110 Finally, the modified Brinkman Board task is a static motor task in the sense that the objects to be manipulated by the monkeys are static, not testing the ability to anticipate the displacement of the target object. For this reason, a mobile version of the task was developed with the “rotating Brinkman Board” task.14,33 More recently, new quantitative manual dexterity tasks combining reach, grasp, grip pressure and pull force were introduced, using a robot to present the target object to the monkey.111,112 In sum, to fill the gap represented by these limitations, it is recommended to complement the modified Brinkman Board task with other tests aimed at assessing specific motor attributes not covered by the modified Brinkman Board task. However, the latter task remains a basic behavioral approach to assess manual dexterity, as it challenges the motivation of the monkeys less than the additional tasks mentioned above, especially post-lesion.
Conclusion
The present re-examination and extension of the M1 lesion data in adult Macaque monkeys supports the conclusion that the 2 treatments (anti-Nogo-A antibody or ANCE cumulated in the present analysis) promote a better functional recovery of manual dexterity, as compared to “control” monkeys (control and control-like data points). In addition, behavioral data limited to post-lesion scores also yield statistically significant differences between “control” versus “treated” data points. These conclusions are encouraging in the context of putative clinical application, although clinical trials remain a considerable challenge, as shown by an ongoing anti-Nogo-A antibody study following cervical spinal cord lesion in human subjects. 113
Supplemental Material
sj-docx-1-exn-10.1177_26331055261421348 – Supplemental material for Re-Examination and Extension of Manual Dexterity Behavioral Data in M1 Lesioned Adult Macaque Monkeys: A Survey of Therapies Induced Enhancement of Functional Recovery
Supplemental material, sj-docx-1-exn-10.1177_26331055261421348 for Re-Examination and Extension of Manual Dexterity Behavioral Data in M1 Lesioned Adult Macaque Monkeys: A Survey of Therapies Induced Enhancement of Functional Recovery by Eric M. Rouiller in Neuroscience Insights
Supplemental Material
sj-docx-2-exn-10.1177_26331055261421348 – Supplemental material for Re-Examination and Extension of Manual Dexterity Behavioral Data in M1 Lesioned Adult Macaque Monkeys: A Survey of Therapies Induced Enhancement of Functional Recovery
Supplemental material, sj-docx-2-exn-10.1177_26331055261421348 for Re-Examination and Extension of Manual Dexterity Behavioral Data in M1 Lesioned Adult Macaque Monkeys: A Survey of Therapies Induced Enhancement of Functional Recovery by Eric M. Rouiller in Neuroscience Insights
Supplemental Material
sj-jpg-3-exn-10.1177_26331055261421348 – Supplemental material for Re-Examination and Extension of Manual Dexterity Behavioral Data in M1 Lesioned Adult Macaque Monkeys: A Survey of Therapies Induced Enhancement of Functional Recovery
Supplemental material, sj-jpg-3-exn-10.1177_26331055261421348 for Re-Examination and Extension of Manual Dexterity Behavioral Data in M1 Lesioned Adult Macaque Monkeys: A Survey of Therapies Induced Enhancement of Functional Recovery by Eric M. Rouiller in Neuroscience Insights
Supplemental Material
sj-jpg-4-exn-10.1177_26331055261421348 – Supplemental material for Re-Examination and Extension of Manual Dexterity Behavioral Data in M1 Lesioned Adult Macaque Monkeys: A Survey of Therapies Induced Enhancement of Functional Recovery
Supplemental material, sj-jpg-4-exn-10.1177_26331055261421348 for Re-Examination and Extension of Manual Dexterity Behavioral Data in M1 Lesioned Adult Macaque Monkeys: A Survey of Therapies Induced Enhancement of Functional Recovery by Eric M. Rouiller in Neuroscience Insights
Supplemental Material
sj-jpg-5-exn-10.1177_26331055261421348 – Supplemental material for Re-Examination and Extension of Manual Dexterity Behavioral Data in M1 Lesioned Adult Macaque Monkeys: A Survey of Therapies Induced Enhancement of Functional Recovery
Supplemental material, sj-jpg-5-exn-10.1177_26331055261421348 for Re-Examination and Extension of Manual Dexterity Behavioral Data in M1 Lesioned Adult Macaque Monkeys: A Survey of Therapies Induced Enhancement of Functional Recovery by Eric M. Rouiller in Neuroscience Insights
Supplemental Material
sj-jpg-6-exn-10.1177_26331055261421348 – Supplemental material for Re-Examination and Extension of Manual Dexterity Behavioral Data in M1 Lesioned Adult Macaque Monkeys: A Survey of Therapies Induced Enhancement of Functional Recovery
Supplemental material, sj-jpg-6-exn-10.1177_26331055261421348 for Re-Examination and Extension of Manual Dexterity Behavioral Data in M1 Lesioned Adult Macaque Monkeys: A Survey of Therapies Induced Enhancement of Functional Recovery by Eric M. Rouiller in Neuroscience Insights
Footnotes
Acknowledgements
The author thanks former collaborators who contributed to the collection of already published data (re-evaluated here, compared and confronted to original data): Y. Liu, M. Kaeser, E. Schmidlin, A. Wyss, A. Hamadjida, S. Bashir, A. Belhaj-Saif, J.F. Brunet, J. Bloch, A.D. Gindrat, J. Savidan, C. Roux, M. Schwab, A. Mir.
The author thanks the excellent technical assistance of Mrs C. Roulin and Mrs F. Tinguely (histology and behavior), of Mrs V. Moret (histology, behavior, illustration with video sequences), of Mrs C. Marti and Mrs G. Fischer (histology), of Mr J. Corpataux, Mr B. Morandi, Mr L. Bossy and Mr J. Maillard (animal care taking), Mr A. Gaillard (mechanics), Mr B. Aebischer and Mr A. Francovich (electronics), and Mr L. Monney (informatics).
Author Contributions
The author designed the present re-examination and extension study. Based on previously available data collected by the author and former collaborators (see acknowledgments), the author re-analyzed the data, created the figures and drafted the manuscript.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by: Swiss National Science Foundation, Grants No 31-61857.00, 310000-110005, 31003A-132465, 310030B-149643 (EMR), No 320030-160229 (to E. Schmidlin), the National Center of Competence in Research (NCCR) on “Neural plasticity and repair”; the Novartis Foundation; the Christopher Reeves Foundation (Springfield, NJ, USA); the Swiss Primate Competence Center for Research (SPCCR).
Declaration of Conflicting Interests
The author declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: the anti-Nogo-A antibody was provided by Novartis AG.
Data Availability Statement
The raw data are accessible on request via the corresponding author.*
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
