Abstract
Development of neuropathic pain (NP) is one of the major complications associated with spinal cord injury (SCI). While well-established methods such as von Frey mechanical and facial grimace testing are often used to assess SCI-induced NP-like behaviors in animal models, these assays have significant limitations, including experimenter bias and long periods of active testing and analysis. To address these challenges, we aimed to develop a novel open field 2-texture preference test (TTPT) to assess NP-like behaviors following unilateral C5 hemicontusion SCI in mice. To do so, we modified the open field apparatus by introducing both a rough and a smooth texture to different portions of the chamber floor based on the hypothesis that the abrasive rough surface would differentially elicit NP-like avoidance behavior. However, at both pre-injury baseline and following SCI, mice spent more time and traveled a greater distance on the rough compared to smooth surface. Additionally, the TTPT did not show any correlation with von Frey or grimace data obtained from the same animals. While this novel test may be able to provide information pertaining to other components of functional outcome, the assay is not associated with the persistent NP-like phenotype that occurs following SCI.
Introduction
Nearly 1 in 5 adults report chronic pain. 1 Chronic pain reduces quality of life, increases mental health illnesses such as depression, and presents financial burdens to affected individuals and their caregivers.1,2 Neuropathic pain (NP) is a type of chronic pain that occurs after injury or disease to the nervous system and can manifest as hyperalgesia, allodynia, and spontaneous pain. 3 NP after spinal cord injury (SCI) is especially difficult to treat and is prevalent in over 80% of individuals 1 year after injury, even with currently-available treatments.4,5 Thus, it is imperative to develop new pain therapies to better serve the SCI population. One obstacle in the therapeutic pipeline is the disconnect between how pain is reported in the clinic and how it is assessed in the laboratory. Pain is a subjective experience that is determined clinically via self-report.3,6 In contrast, potential analgesics are usually first tested in preclinical rodent models, and pain-like behavior in these animals is often inferred from reflexive paw withdrawals, vocalization, and motor behaviors such as licking and guarding of the affected area.7,8 Well-established behavioral assays for evoked pain-like behavior include the Hargreaves thermal assay and von Frey filament mechanical test. Unfortunately, since both the von Frey and Hargreaves tests rely on experimenter evoked stimuli, these assays lend themselves to experimenter bias and decrease the efficiency of therapeutics discovery.8,9 While blinding to group assignment reduces experimenter bias, phenotypic differences between experimental animals and their controls, such as partial limb paralysis from neuronal injury (as is the case following SCI), can unblind experimenters without their intent.
Additional tests, such as the mechanical conflict avoidance system, quantify the animal response to nociceptive stimuli it encounters across a test chamber and thus decreases chances of experimenter bias. 10 However, these and similar types of tasks require extensive acclimation and training before testing can even occur, which decreases throughput for testing potential therapeutics. 10 Furthermore, other tests measure spontaneous and ongoing pain by analyzing facial features using automated programs that both decrease potential experimental bias and reduce analysis time.11,12 However, these programs are often specific to certain environmental conditions, camera features, and animal strain; they are not universal, unlike manual assessments. 13
To overcome these drawbacks of established sensory behavior assays, our work examined the potential use of a modified open field test to assess NP-like behavior in mice after SCI. We analyzed behavior of animals with and without SCI in an open field chamber which contained a mixture of abrasive and smooth surfaces, allowing the mice to freely choose between textures. We hypothesized that animals with a SCI-induced NP-like phenotype would show increased aversion to the abrasive rough texture compared to uninjured mice. Surprisingly, we found that mice both with and without SCI prefer the rough surface compared to the smooth-textured surface.
Materials and Methods
Animal Care
Female (20-27 g) and male (28-35 g) wildtype C57BL/6 mice were group housed in 12-hour light-dark cycle with food and water ad libitum. Procedures were conducted based on the Guide for the Care and Use of Laboratory Animals and approved by the Thomas Jefferson University IACUC (approval number: 01230).
Surgical Procedures
Mice were randomly assigned to sham laminectomy-only surgery control (n = 6) or SCI (n = 6) groups. Mice with SCI received a unilateral hemicontusion injury at cervical level five (C5), as previously described. 14 Briefly, all mice received hemi-laminectomy, exposing the right dorsal surface of C5 spinal cord. SCI mice received contusion performed at 60-kdyne force with 2 seconds dwell time using the Infinite Horizon Impactor (Precision Systems and Instrumentation, Lexington, KY). Surgical sites were closed in layers with vicryl 5.0 suture (Ethicon Inc., Raritan, NJ), and mice were given subcutaneous prophylactic ampicillin antibiotic and 0.9% saline to prevent dehydration. All mice received buprenorphine (0.08 mg/kg) analgesia for 48 hours post-surgery.
Experimental Design
We conducted the von Frey assay and mouse grimace testing at pre-surgical baseline and at 2 weeks post-surgery. TTPT was conducted at pre-surgery baseline and at a later 4-week post-injury point to provide time for any altered sensitivity induced by the von Frey assay to resolve. Our previously published von Frey and mouse grimace scale results show that both the 2-week and 4-week time points are associated with a robust ipsilesional forepaw allodynic phenotype and also increased grimacing features indicative of spontaneous pain. In addition, we find that forepaw hypersensitivity in the von Frey assay does not show a temporal peak following cervical contusion SCI; instead, the allodynia starts at 1 to 2 weeks after SCI and persists to the same degree for at least several months post-injury. We therefore chose the single 4-week point for the TTPT since it represents a late time after injury, which is relevant to the chronic nature of NP after SCI.
von Frey Filament Test
The von Frey filament test was conducted ipsilateral to surgery on the right forepaw, as described. 15 At baseline and 2 weeks post-surgery, mice were placed on a wire mesh grid (10 × 10 cm) and contained in an acrylic chamber. Monofilaments were applied to the forepaw plantar surface using the up-down method, beginning with the 0.60 g monofilament. Intervals of 2 to 5 minutes between each stimulus application was used with a total of 10 applications per forepaw. A positive withdrawal response was recorded when a quick paw withdrawal in response to the monofilament was observed. Spontaneous movements, those without signs of a supraspinal response (ie, vocalization, licking, and flapping or guarding of the forepaw), were not recorded and the trial was repeated. A cutoff of 2 g of force was implemented to avoid paw injury. The 50% response threshold was determined as the lowest force that elicited withdrawal and accompanying supraspinal responses.
Open Field Two-Texture Preference Test
To create a novel non-evoked and unbiased assay to assess mechanical NP-like behavior, we added material with an abrasive texture that would potentially be selectively aversive to SCI mice with forepaw hypersensitivity (as measured by von Frey testing). To cover half of the floor of an acrylic open field chamber (1600 cm2), coarse-grit skateboard grip tape (Vicious Griptape, Vancouver, BC, Canada) was used, as shown in Figure 1C. Grip tape was used to test the effect of a large particle size that is still considered “coarse” by the Federation of European Producers of Abrasives (FEPA) standards (800-1200 μm particle diameter; 107 particles per 1 cm2). The particle characteristics of this material are similar (though a bit larger) than the ~250 μm diameter of the allodynic von Frey filament. This surface material was chosen to ensure sufficient distance between particles to maximize the chances a single particle comes in contact with the plantar paw surface, allowing maximal force at 1 point of contact with the paw. All animals were checked for signs of abrasion or irritation after each testing session to ensure the material was not causing paw injury. The remaining smooth floor consisted of regular masking tape completely covering the abrasive surface to control for smells associated with the grip tape. The region of rough surface was rotated between test sessions to ensure mice did not habituate to placement of each texture. Swann SWPRO-735CAM (15 Frames Per Second [FPS]; Swann, CA) camera was placed directly over the open field chamber (120 cm from the chamber floor). Video was calibrated at the start of each testing session. For each trial, the mouse was placed in the chamber center and activity was recorded for 5 minutes, a commonly used length of time for open field analysis.16,17 Videos were analyzed via open-source software published for MATLAB (RRID:SCR_001622). 18

Two-texture preference testing did not correlate with established assays of SCI-induced NP-like behavior. Diagram of behavioral tests performed at pre-surgical baseline and post-surgery: two-texture preference test (TTPT), mouse grimace scale assay, and von Frey filament test (A). Illustration of the two experimental groups assessed: unilateral cervical contusion spinal cord injury (SCI); laminectomy-only without SCI as sham-surgery control (B). As shown in the TTPT trial example, the path for each mouse in each trial was tracked (C). Compared to pre-surgical baseline and to laminectomy-only, SCI mice showed a significant decrease in ipsilesional forepaw withdrawal threshold in the von Frey filament assay (D). SCI mice also demonstrated increased facial grimace scores based the mouse grimace scale (E). The proportion of distance or time spent on each surface was determined by calculating the ratio of total distance traveled (F) or time spent (G) on the rough versus smooth surface. Time spent frozen on the rough versus smooth surface (expressed as a portion of total time spent on each surface type) was assessed (H). Ratio of time spent in the outer zone, defined as the outer one-fifth of the open field chamber, was calculated comparing the rough to smooth surface (I)). All ratios were expressed as rough-to-smooth (R:S). Proportions greater than 1 indicate that more distance was traveled or more time was spent on the rough surface for each measurement. When assessing time spent or distance traveled at pre-surgical baseline and post-surgery, the proportion was significantly greater than 1 for some time points when using a one-sample t-test (P < .5). Using a Fisher’s least significant difference (LSD) test, no significant differences were found within a group between baseline and post-surgery, nor were any significant differences found between laminectomy-only and SCI groups at each timepoint (P > .5). Correlation analysis of data obtained from the TTPT and the von Frey or grimace assays was performed via simple linear regression. Ipsilateral forepaw withdrawal threshold after surgery (expressed relative to the pre-surgery baseline value) was assessed versus the TTPT results: total distance (J), total time spent (K), portion time frozen (L), and time spent in the outer zone (M). Grimace score following surgery (expressed relative to pre-surgery baseline value) was analyzed versus the TTPT values: total distance (N), total time spent (O), portion time frozen (P), and time spent in outer zone (Q). For all linear regression analyses, no significant relationships were found. For all statistical comparisons, details of these analyses are presented in Tables 1 to 3.
Mouse Grimace Scale
Image capture and analysis of facial grimace features were conducted as previously described.15,19 At baseline and 2 weeks post-surgery, mice were contained individually in an acrylic chamber atop a wire mesh grid (10 × 10 cm). A GoPro Hero 2018 (60 Frames Per Second [FPS]; GoPro, San Mateo, CA) camera was placed facing the animal in a manner ensuring a clear view of facial features, including eyes, ears, nose, and cheeks. Thirty-minute videos of each mouse were recorded. Five screenshots, with a minimum of 2 minutes apart, were obtained of each mouse by blinded researchers, ensuring each screenshot had a full view of facial features and the mouse was not performing other behaviors such as sleeping or grooming. Images were randomly chosen for blinded grading. The following facial features were scored on a 0 to 2 scale (0 = not present, 1 = somewhat present, 2 = obviously present): orbital tightening, nose and cheek bulge, and ear position. Whisker position was not taken into account since some mice had removed most of their whiskers via excessive grooming. The scores of the 4 facial features were averaged per animal at each timepoint.
Statistical Analysis
Data are represented as mean ± standard error of the mean throughout. We provide details of n’s and results of all statistical analyses (including exact P-values) for each experiment and for all statistical comparisons in the provided Tables. All statistics were calculated using Graphpad Prism 9 (San Diego, CA). Von Frey, mouse grimace scale, and TTPT analyses were conducted using a 2-way repeated measures ANOVA (group x time). Behavioral parameters assessed on rough versus smooth surface were compared to an expected value of 1.0 using a 1-sample t-test. Correlation analyses were performed using simple linear regression.
Transparency, Rigor and Reproducibility Summary
Surgical procedures and behavioral analyses were conducted in a blinded manner. Individual data for all outcome measures are included in graphical representations and are freely available at the Open Data Commons for Spinal Cord Injury (https://odc-sci.org/; RRID:SCR_016673). We have provided Research Resource Identification Initiative (RRID) numbers for relevant reagents.
Results
Both female and male adult wildtype C57BL/6 mice were subjected to pre-surgical baseline behavior testing and were then followed to 4 weeks post-surgery by additional behavioral assessments (Figure 1A). Mice received C5 hemicontusion SCI or laminectomy-only sham procedure (Figure 1B). Throughout the entire experiment duration, all mice displayed use of the plantar surface of all 4 limbs during behaviors such as locomotion, grooming, and eating. Using the TTPT, we assessed 4 behavioral parameters on the rough versus smooth floor surfaces of a modified open field chamber: distance traveled, total time spent, portion of time spent frozen, and time spent near the chamber perimeter. The path of an example SCI mouse in the TTPT chamber over a 5-minute test period is shown with the rough-smooth pattern configured as 4 quadrants (Figure 1C).
To confirm that these cervical contusion SCI mice exhibited a NP-like phenotype using established behavioral assays, we conducted both the von Frey filament test and mouse grimace scale analysis at pre-surgical baseline and at 2 weeks after SCI or laminectomy-only. Compared both to pre-surgical baseline and to uninjured control mice, SCI animals showed mechanical allodynia in ipsilesional forepaw with the von Frey assay as measured by a significantly reduced withdrawal threshold (expressed as a ratio to pre-surgery baseline value; Figure 1D; Table 1). These von Frey assay findings are consistent with our previously reported work in the mouse cervical contusion SCI paradigm.14,15,20,21 In addition, the SCI mice showed a significantly greater non-evoked facial grimace score compared to pre-surgery baseline and to laminectomy-only animals (Figure 1E; Table 1), which is also consistent with our previous work in this mouse cervical contusion SCI model. 15
Statistical analysis details for von Frey filament test and mouse grimace scale.

Results are shown for laminectomy-only sham and SCI groups. An unpaired t-test was used to compare paw withdrawal threshold in laminectomy-only versus SCI animals. An unpaired t-test was also used to compare facial grimace score in laminectomy-only versus SCI animals. For all statistical tests, a 2-tailed P < .05 was considered significant, and any values reaching significance are bolded.
Using the TTPT, we found that mice in the SCI and sham groups generally preferred the rough to smooth surface. Though not statistically significant, sham and SCI groups traveled a greater distance on the rough compared to smooth surface (as indicated by a ratio greater than 1.0) at baseline and 4 weeks post-surgery (Figure 1F; Table 2). Sham and SCI groups also spent more time on rough compared to smooth surface at baseline and 4 weeks post-surgery, though this was not significant (Figure 1G; Table 2). In addition, mice showed a non-significantly increased portion of time freezing on the rough compared to smooth area (Figure 1H; Table 2). We also assessed thigmotaxis by calculating time spent in the outer one-fifth (outer zone) of the chamber; we found no differences in the time spent in the outer zone when comparing all-smooth versus all-rough chambers (Figure 1I; Table 2). These data demonstrate that uninjured mice preferred the rough to smooth surface and also that SCI did not alter rough versus smooth surface discrimination in this TTPT assay. Furthermore, we did not find sex-specific effects with any TTPT measurements when comparing sham and SCI groups (Supplemental Tables 1–3).
Statistical details for 2-texure preference testing.
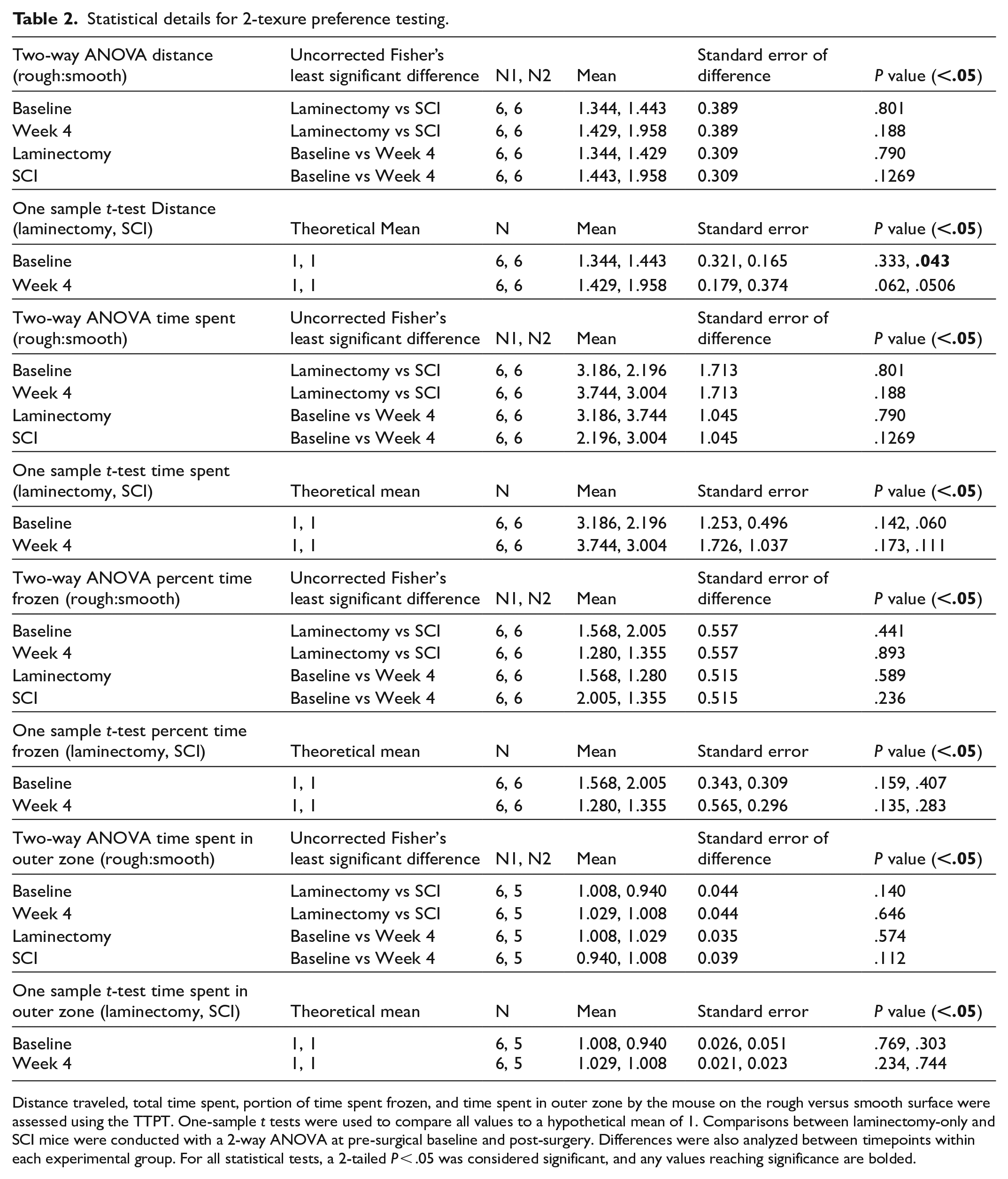
Distance traveled, total time spent, portion of time spent frozen, and time spent in outer zone by the mouse on the rough versus smooth surface were assessed using the TTPT. One-sample t tests were used to compare all values to a hypothetical mean of 1. Comparisons between laminectomy-only and SCI mice were conducted with a 2-way ANOVA at pre-surgical baseline and post-surgery. Differences were also analyzed between timepoints within each experimental group. For all statistical tests, a 2-tailed P < .05 was considered significant, and any values reaching significance are bolded.
In addition, we performed correlation analyses comparing the TTPT results to data obtained from the 2 established assays of NP-like behavior, the von Frey and grimace tests. Linear regression analysis demonstrated that there was no relationship between the TTPT data and von Frey results, including for both uninjured and SCI animals (Figure 1J-M; Table 3). Similarly, we found no relationship between the TTPT and mouse grimace scale results for both uninjured and SCI mice (Figure 1N-Q; Table 3).
Statistical details for linear regression analyses.
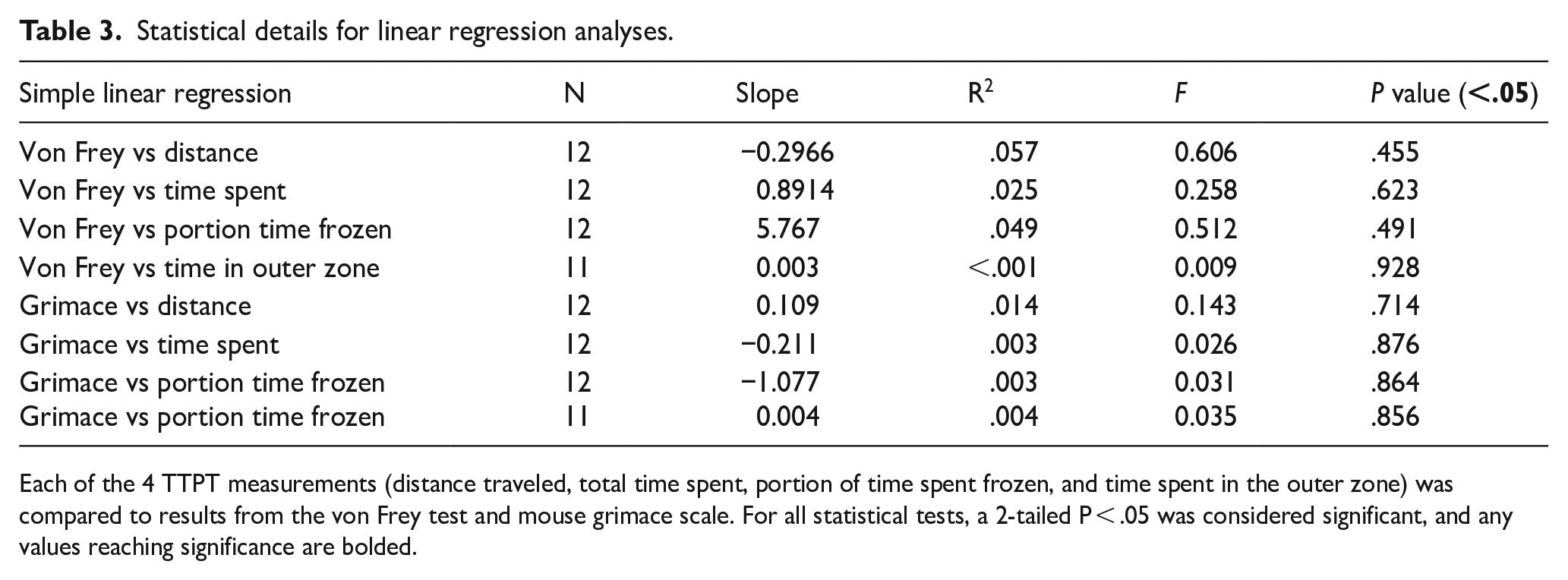
Each of the 4 TTPT measurements (distance traveled, total time spent, portion of time spent frozen, and time spent in the outer zone) was compared to results from the von Frey test and mouse grimace scale. For all statistical tests, a 2-tailed P < .05 was considered significant, and any values reaching significance are bolded.
Discussion
To increase likelihood that therapeutics advance through the translational pipeline, preclinical work should utilize animal models and behavioral assessments that best emulate human conditions, 22 allow high throughput screening, and are resistant to experimenter bias.8,9 SCI-induced NP in patients is predominantly determined by self-report questionnaires rather than by quantitative sensory threshold testing. 23 In contrast, the majority of assessments of NP-like behavior in experimental SCI and others chronic pain models focus on sensory thresholds in response to applied stimuli. Previous work in rodent SCI has used the open field to assess outcomes such as motor function.24,25 Open field testing serves as a basis for our novel TTPT assay since it can be used for objective measurements based on unevoked responses and requires minimal acclimation and active experimenter involvement.26,27 Added to this, many programs have been generated to vastly increase throughput of video analysis, and much of this software is open-source and therefore cost effective.19,20
We investigated whether open field TTPT could be a sensitive measure of NP-like behavior after SCI. While our current and previous work show that established sensory assays reveal that mouse cervical contusion produces marked allodynia14,21 and increased grimace, 15 we found no SCI effects on TTPT. Moreover, there was no correlation between TTPT data and the von Frey or grimace outcomes assessed in the same animals.
Because SCI mice demonstrating NP-like behavior with von Frey and grimace assays showed no difference in texture preference, our findings suggest that the rough surface was not adequate to differentially elicit an aversive response. Interestingly, we found that, rather than avoiding the rough surface, mice from all groups spent more time and traveled greater distance on the rough area. It is possible that SCI mice showed this preference because the added friction impacted locomotor performance, 28 though this may not explain why uninjured mice also preferred the rough portion. Previous work using a 2-texture preference assay has shown that some strains of wild-type mice prefer a rough surface, while other mouse strains show no preference. 29
In conclusion, our study demonstrates that the TTPT is not associated with the persistent NP-like phenotypes (observed using multiple established behavioral assays) that occur after mouse cervical contusion SCI. Despite these findings that our TTPT may not be a useful tool in its present form to assess NP-like behavior after SCI, there is still a need to improve the arsenal of behavioral tests for measuring NP. Future experiments could, for example, increase aversiveness of the rough surface, assess more subtle behaviors in open field analysis, change timing of the trials post-injury, or adjust the pattern of rough and smooth surfaces to tease out potential differences between SCI mice and control animals. While we focused on the cervical contusion model since it results in robust NP-like behaviors and also because cervical SCI is the most common location of spinal cord trauma in the clinical population, it will also be important in future work to assess the TTPT using additional rodent models of chronic pain such as peripheral nerve injury and inflammatory pain, as well as other SCI paradigms that vary in anatomical location, injury severity, and type of lesion (ie, contusion, crush, tractotomy).
Supplemental Material
sj-docx-1-exn-10.1177_26331055251361442 – Supplemental material for Open Field Two-Texture Preference Testing Is Not Associated With the Neuropathic Pain-Like Phenotype That Occurs Following Mouse Cervical Contusion Spinal Cord Injury
Supplemental material, sj-docx-1-exn-10.1177_26331055251361442 for Open Field Two-Texture Preference Testing Is Not Associated With the Neuropathic Pain-Like Phenotype That Occurs Following Mouse Cervical Contusion Spinal Cord Injury by David A. Jaffe, Megan A. Lyttle and Angelo C. Lepore in Neuroscience Insights
Footnotes
Abbreviations
C5: cervical level 5; NP: neuropathic pain; SCI: spinal cord injury: TTPT: two-texture preference test
Ethical Considerations
All animal procedures were conducted based on the NIH Guide for the Care and Use of Laboratory Animals and approved by Thomas Jefferson University Institutional Animal Care and Use Committee.
Author Contributions
Conceptualization: DA Jaffe, AC Lepore. Data curation: DA Jaffe, AC Lepore. Formal analysis: DA Jaffe, MA Lyttle. Funding acquisition: AC Lepore. Investigation: DA Jaffe, MA Lyttle. Project administration: AC Lepore. Supervision: AC Lepore. Validation: DA Jaffe. Visualization: DA Jaffe, MA Lyttle. Roles/Writing – original draft: DA Jaffe, AC Lepore. Writing – review & editing: DA Jaffe, AC Lepore.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Neurological Disorders and Stroke (R01NS110385 to ACL; R01NS079702 to ACL) and by the Yant Family Spinal Cord Regeneration Fund (to ACL).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
