Abstract
Previous studies have indicated that the infralimbic (IL) and prelimbic (PL) subdivisions of the medial prefrontal cortex (mPFC) serve as critical modulators of fear suppression and expression. Although significant research has been conducted on the extinction of conditioned fear, the mechanisms underlying contextual fear discrimination learning, a form of contingency judgment learning, remain inadequately understood. Our investigation aimed to explore the influence of epigenetic regulation associated with cyclic AMP-response element binding protein (CREB)-dependent long-term memory encoding within the IL and PL on contextual fear discrimination. Our prior and current findings illustrate that epigenetic hypofunction induced by a CREB-Binding Protein (CBP) mutant, which is deficient in histone acetyltransferase activity (CBPΔHAT), within the mPFC leads to compromised contextual fear discrimination while not affecting contextual fear conditioning in these mutants. Unexpectedly, the effect was not noticeable when the hypofunction was constrained to the infralimbic (IL) area; however, the hypofunction of the prelimbic (PL) network led to considerable impairment in fear discrimination. The findings indicate that learning fear discrimination involves differential encoding across the specialized networks of the mPFC. These data suggest that the IL network is not essential for encoding during the acquisition and discrimination of fear or that the PL network may compensate for the IL’s inability to encode new information. Furthermore, these results emphasize the importance of histone acetylation in the mPFC as a crucial physiological mechanism for learning contingency judgment.
Keywords
Introduction
The process of contingency judgment learning (CJL) involves interpreting cues and transforming unreliable cue-outcome relationships into reliable ones. It is noteworthy that both humans1 -3 and animals4,5 exhibit an ability to detect contingency and its variations accurately. Furthermore, impairments in CJL can be effectively studied in murine models and have significant clinical relevance. For example, post-traumatic stress disorder (PTSD) presents with impaired contextual fear processing across the hippocampus - the basolateral amygdala (BLA) - mPFC circuit, resulting in misjudgment of cue-safety versus cue-danger relationships. Impaired contextual fear inhibition and overgeneralized fear are postulated to be markers for PTSD risk and treatment outcome as individuals with PTSD feel acutely threatened by generalized cues.6 -9 The hippocampus,10,11 the basolateral amygdala, 12 the medial prefrontal cortex (mPFC), and the dorsal anterior cingulate cortex 8 have been implicated in contextual fear inhibition and PTSD risk. 9 Both rodent and human studies implicated prefrontal regions in contextual fear acquisition, expression, generalization, inhibition, and discrimination (reviewed in Liberzon and Abelson, 7 Ressler et al, 9 Rozeske et al, 13 Plas et al, 14 and Maren et al 15 ).
The neurobiology of fear acquisition and extinction has been extensively studied; however, contextual fear discrimination learning needs to be more understood. Fear discrimination learning is a type of CJL that involves learning the distinction between contextual conditioned stimulus CS+ predicting danger (i.e., paired with an unconditioned aversive stimulus, US) and a second contextual stimulus CS− (not paired with US) that share non-trivial similarity with CS+. Initially, responses to CS− are generalized (i.e., similar to CS+), but after repeated exposures to CS+ paired with US and CS− without reinforcement, subjects can distinguish between CS+ and CS−, which defines fear discrimination learning. The medial prefrontal cortex (mPFC) has been implicated in fear discrimination learning.16,17
Fear behavior is controlled by discrimination, generalization, and extinction.18 -21 The PL and IL subdivisions of mPFC and their long-distance interaction differentially regulate fear behavior22 -25 via distinct connectivity with the amygdala,26,27 including BLA pathways 28 and the amygdala intercalated neurons inhibitory relay.29 -32 The modulation of fear during fear discrimination learning depends on the integrity of the amygdala, hippocampus, and mPFC. Contextual information encoded in the hippocampus is believed to be projected to the BLA directly from the ventral hippocampus or indirectly through the medial entorhinal cortex.14,33 In addition, mPFC receives direct projection from the ventral hippocampus. 34 During fear conditioning, the paired aversive and neutral stimuli converge in the amygdala, causing synaptic changes that constitute fear memory. 35 Hypofunction of the mPFC does not impair fear conditioning17,36,37; however, it does impair fear modulation, including fear extinction recall32,37 and fear discrimination learning.17,36,38
The IL and PL subdivisions of the mPFC have been shown to modulate fear behaviors differentially (reviewed in Gonzalez and Fanselow
39
). Studies have implicated IL and PL as critical modulators of fear suppression and expression. Inactivation of IL, but not PL, impairs consolidation and recall of fear extinction,
40
while microstimulation of PL promotes fear expression and prevents fear extinction.41,42 The activity of these 2 regions is not static throughout fear discrimination learning.
Learning-induced cAMP response element binding protein (CREB)-dependent gene expression program plays a pivotal role in converting short-term to long-term memory across species.46 -51 CREB does not function alone, and its activation requires phosphorylation-depended recruitment of CREB Binding Protein (CBP),52,53 which is critical for chromatin remodeling enzymatic activity referred to as HAT (histone acetyltransferase).54,55 Evidence indicates that with the intrinsic acetyltransferase activity, CBP is a critical component of the neural signaling underlying learning and memory.56 -64
While there is evidence that IL and PL play significant roles in fear acquisition and the extinction of conditioned fear,14,24,39 -42,45 little is known about the specific roles of mPFC subdivisions in mediating fear discrimination learning. Genetic tagging studies showed differential PL and IL cortices engagement during fear discrimination learning. Still, it is unclear how and if these local circuits are required to learn the task. 43 Utilizing viral vectors to induce CBP acetyltransferase-dependent hypofunction in PL or IL regions, we tested the functional requirement of these 2 subdivisions of the mPFC in learning to disambiguate between threat and safety. These data confirm that histone acetylation-based mechanism in neuronal populations within mPFC is required for fear discrimination learning and suggests differential roles for IL and PL in CJL.
Materials and Methods
Subjects
The experiments used male C57BL/6J mice from Taconic. The mice were housed 3 to 4 per cage with their littermates, maintained on a 12-hour light/dark cycle, and had ad libitum access to food and water. Sterilized bedding was changed every week. Mice used in the experiments were separated from their littermates and housed with an ovariectomized female companion mouse during the surgery recovery period and behavioral assay. All procedures were approved by the UC Riverside Institutional Animal Care and Use Committee and complied with NIH guidelines for the care and use of laboratory mice.
Surgery
To express marker or CBPΔHAT in precise neuroanatomical locations, a StereoDrive robotic stereotaxic frame (Neurostar) was used to inject the virus according to the following procedure. Anesthesia was induced in an induction box via exposure to 3.5% isoflurane at a 1 LPM flow rate. Once the subject failed to respond to a toe or tail pinch, it was transferred to the stereotaxic apparatus, where anesthesia was maintained with 1.0% to 1.5% isoflurane at a 0.4 LPM flow rate. Breathing was constantly monitored, and the depth of anesthesia was adjusted accordingly. A heating pad was placed under the subject to maintain a core body temperature of 37°C as measured by a rectal probe. Zygoma ear bars were used to secure the head, a bite bar was situated in the mouth, and a nose cone was firmly secured to maintain isoflurane delivery and further stabilize the head. Ophthalmic ointment was applied to the eyes to prevent drying, and carprofen and buprenorphine were injected subcutaneously to minimize pain during and immediately after surgery. About 0.9% saline was also injected subcutaneously to prevent dehydration during surgery. The head was shaved and disinfected with 3 rounds of washing with betadine followed by 70% ethanol. A midline incision was made, and the scalp was pulled laterally with skin hooks. The skull was cleaned and dried with a brief application of hydrogen peroxide. The beveled tip of a Hamilton 10 µl syringe was used to mark the coordinates of lambda and bregma to level the skull. Two points 2 mm lateral from the midline were also recorded to correct for lateral tilt. Once the skull was level and bregma set as the reference point, a small craniotomy was made above the injection site using a dental drill.
The virus was loaded into the syringe, and the needle tip was slowly (0.1 mm/s) lowered through the craniotomy into the brain until the tip was at the desired coordinate. The virus was injected at 0.02 µl/min, and once the injection was finished, the needle was left in the tissue for 10 minutes to allow the virus to diffuse before it was slowly (0.1 mm/s) removed from the brain.
The craniotomy was sealed with bone wax, and the scalp was brought together and sutured. Triple antibiotic ointment was applied to the wound to prevent infection. The subject was then removed from the stereotaxic instrument, placed back in the induction box, and supplied with 100% oxygen until it recovered. Once the subject regained awareness, it was transferred back to its home cage, which had been warmed with a heating pad. After surgery, subjects were administered carprofen once daily for 2 days, and ibuprofen was dissolved in their drinking water to minimize inflammation and pain.
Virus injection coordinates were based on the Paxinos and Franklin 65 mouse atlas. For PL injections, 0.3 µl was injected per hemisphere at a coordinate of 2.1 mm rostral to bregma, ±0.37 mm lateral to the midline, and 2.1 mm ventral to bregma, at a 15-degree angle relative to the dorsoventral axis. For IL injections, 0.2 µl was injected per hemisphere at a coordinate of 1.7 mm rostral to bregma, ±0.37 mm lateral to the midline, and 2.8 mm ventral to bregma at a 30-degree angle relative to the dorsoventral axis. Viruses and titers used were as follows: AAV1.CaMKII.GFP-Cre at a 2 × 1012 GC/ml titer, HSV.hEF1a.LS1L.CBPΔHAT.mCherry at a 5 × 108 GC/ml titer, and HSV.hEF1a.LS1L.mCherry at a 5 × 108 GC/ml titer. Surgical procedures were tailored to standardize the variability of AAV and HSV viral injections. Identical stereotaxic coordinates were used for all PL injections and separate but identical coordinates for IL injections. Injected virus volume was also kept the same for all PL injections, and separate but identical volumes were used for the IL injections. As previously described, CBPDHAT and mCherry were cloned into the HSV amplicon and packaged using a replication-defective helper virus.66,67
Behavioral assay
The contextual fear discrimination (Figure 1A) assay is composed of the following phases: habituation (Days −1 & 0), conditioning (Day 1), exposure to NS (Days 3 & 4), fear memory recall (Day 5) and fear discrimination (Days 6-7). During the two habituation days, mice were exposed to the 3 experimental contexts (context A, B, and C) for 10 minutes per context, with no US administration. Next, on the fear conditioning day, animals were exposed to 3 US (foot shock, 2 seconds, 0.75 mA) presentations during a single 420 seconds exposure to Context A, starting after a 180 second baseline, with a 90 seconds inter-trial interval and 60 seconds post-stimulus period. Two days after fear conditioning, mice were exposed 4 times to neutral stimulus (NS) Context C (Day 3 and 4) for 360 seconds, once in the morning and once in the afternoon. Four days after the fear conditioning (Day 5), mice were subjected to the fear memory recall test, in which they were exposed first to Context C (NS) to assess the fear level of a neutral stimulus and then to Context A (CS+) to assess fear memory recall. On the same day, the mice were exposed to a similar context B (CS−) to determine a level of generalized fear. During the fear discrimination test, individual mice were exposed once per day to each tested context stimuli (NS, CS−, and CS+) for 2 days (Days 6 and 7) with an alternative order of CS+ and CS− stimuli presentations each day. During the CS+ trial, mice were placed in the CS+ context for 180 seconds, received a 0.75 mA, 2 seconds foot shock, and were left for another 60 seconds inside the chamber. During NS and CS− trials, mice were exposed to stimuli for 242 seconds and received no foot shock. The level of fear was monitored during the first 180 seconds.
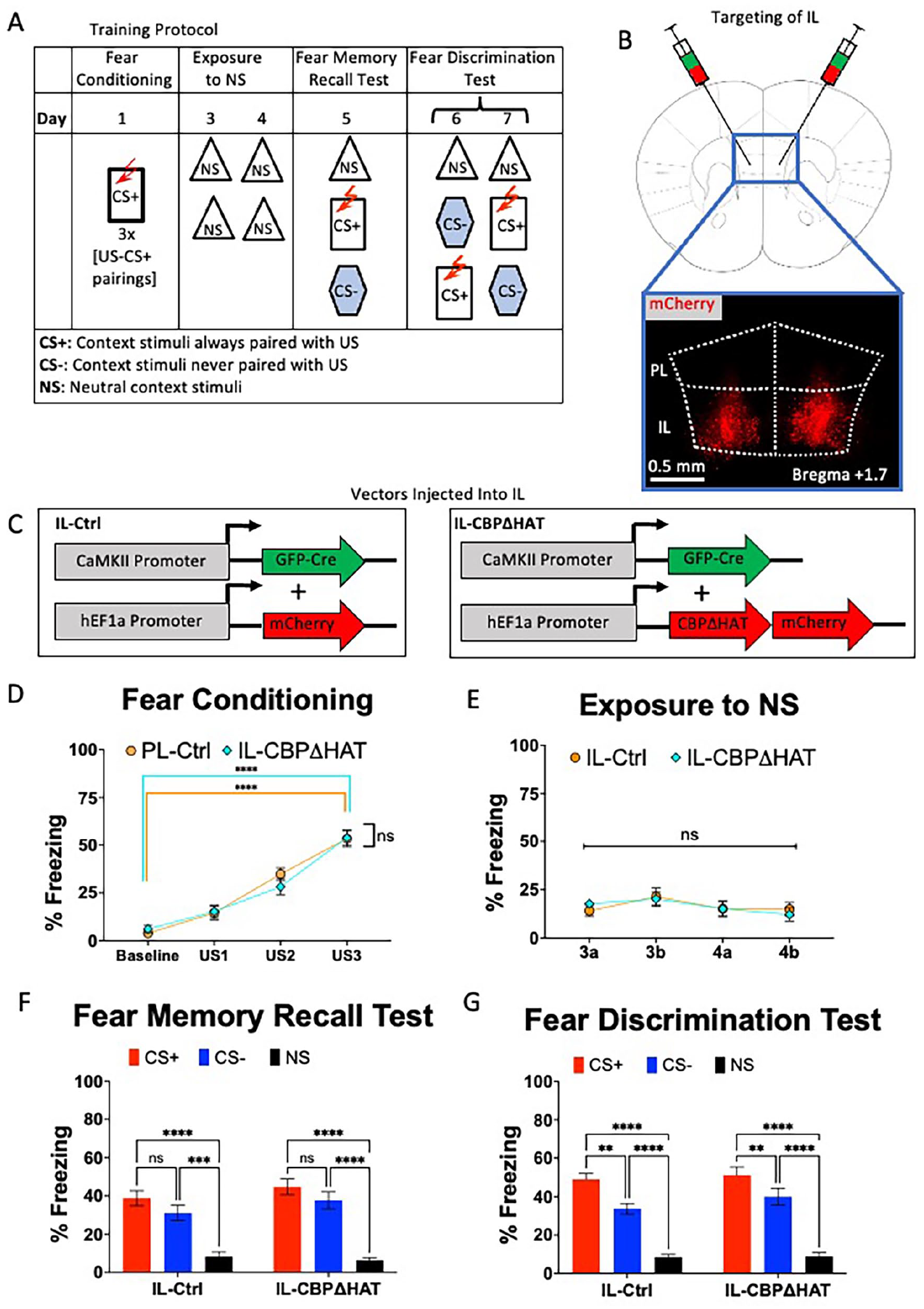
Effect of expression of CBPΔHAT in IL on fear discrimination. (A) Experimental design for contextual fear discrimination assay. During Fear Conditioning (Day 1), mice are placed in the CS+ context, which consists of level bars, lemon scent, normal chamber, and tone. About 180 seconds after baseline recording, 3 foot-shocks (2 seconds, 0.75 mA) are delivered with a 90-second inter-trial interval. After conditioning, mice undergo repeated exposures to NS (Days 3-4), where they are placed in a home cage analog twice daily for 360 seconds each trial. Fear memory recall (Day 5) was performed 72 hours after fear conditioning. During the fear discrimination test (Days 6-7), mice are exposed to the NS context for 242 seconds, and then an alternating presentation of CS+ (where they receive 1 shock) and CS− (where they are not shocked) for 242 seconds each. (B) Viral injection targeting of IL cortex. About 0.2 µl of a 1:1 mixture of a GFP-Cre virus and a Cre-dependent mCherry virus was injected bilaterally into the IL cortex at a 30° angle. (C) Dual viral-vector system for expressing CBPΔHAT in IL cortex. An AAV1.CamKII.Cre-GFP virus was mixed in a 1:1 ratio with either HSV.hEF1a.LS1L.mCherry for Ctrl group or HSV.hEF1a.LS1L.CBPΔHAT.mCherry for the CBPΔHAT group and injected into IL. (D) Performance on the fear conditioning test. Both IL-Ctrl and IL-CBPΔHAT groups display equivalent performance on the fear acquisition and the 72-hour fear memory test. (E) Both IL-Ctrl and IL-CBPΔHAT groups display low freezing to NS context. (F) Performance on the fear memory recall test. Both IL-Ctrl and IL-CBPΔHAT groups display equivalent performance on the 72-hour fear memory test. (G) Performance on the fear discrimination test. Both IL-Ctrl and IL-CBPΔHAT could discriminate between CS+ and CS− contexts on the block trial (ie, days 6 and 7).
All parts of the behavior assay were carried out in a 12.0″ L × 10.2″ D × 12.0″ H plexiglass fear conditioning chamber (Lafayette Instrument Co.) with overhead IR-filtered video camera, speakers, IR light, chamber light, shock grid, and ventilation fan, all coordinated via FreezeFrame 4 (Actimetrics). Each conditioning chamber is placed within an opaque sound-attenuating box, with 4 chambers/boxes running simultaneously. Before and after each trial, the chambers were cleaned with 70% EtOH and DI water. Chambers were cleaned with Quatricide after each testing day. The chambers were modified to present distinct visual, olfactory, tactile, and auditory cues, collectively representing distinct contexts. Context A had no visual changes to the base conditioning chamber, a lemon-extract scent, auditory cue, and a shock grid floor consisting of a single layer of parallel metal bars. Context B consisted of black and white vertical-striped wall inserts for all 4 walls of the conditioning chamber, a vanilla extract olfactory cue, a constant auditory tone, and a shock grid floor consisting of 2 staggered levels of parallel metal bars. Context C consisted of an open-topped 9″ L × 6″ D × 6.5″ H plastic box with a layer of Sani-Chips bedding (new bedding for each subject), placed within the unmodified conditioning chamber, with no additional visual, auditory, or olfactory cues. Animals were placed within this plastic box and could not leave the plastic box and contact the metal grids during Context C trials. Contexts A and B were counter-balanced as to which context was assigned as the CS+ and CS− for each subject.
Freezing performance was analyzed semi-automatically using FreezeFrame 4 (Actimetrics) software. FreezeFrame analyzed 30 FPM video data to calculate the frame-by-frame pixel difference for each trial. Freezing was defined as a period of at least 1 second in which the subject’s movement value remains below a manually set threshold. All reported “% freezing” values were calculated as the percentage of time spent freezing during the first 180 seconds of a context exposure (for fear conditioning, 90 seconds ITI and 60 seconds poststimulus freezing were also calculated).
Image acquisition and processing
An Olympus FV1000 laser scanning confocal microscope was controlled using FluoView software to image virus expression and acetylation. GFP, mCherry, and Alexa-647 were imaged using 473, 587, and 647-nm lasers, respectively. Multiple brain sections were imaged using identical microscope settings within FluoView. Stitching together images and intensity quantification was performed using the Fiji package of ImageJ and the MosaicJ plugin. Ten optical sections were acquired from a 30 μm Z-stack within a 40 μm coronal section. The extent of viral vector spread and centroid of expression was determined by analyzing the cloud of fluorescence through a range of coronal mPFC slices (~2.3-1.5 mm anterior to bregma). Determination of IL and PL boundaries was accomplished by imaging cytoarchitectural differences between IL and PL regions and using GIMP (GNU Image Manipulation Program) to overlay a mouse reference atlas 65 on the actual image of the specimen coronal section (Supplemental Figure 1).
Histology
To quantitatively measure the acetylation effects of CBPΔHAT, a previously described IHC procedure was used. 17 Mice were anesthetized using Nembutal (200 mg/kg, i.p. injection) and transcardially perfused with PBS and 4% PFA. The extracted brain was soaked in 4% PFA overnight and then transferred to PBS until histological sectioning.
In this study, 40 µm-thick sections of the mPFC were obtained using a cryostat (Leica) and placed in a 24-well plate for free-floating immunohistochemistry (IHC). The sections were washed 3 times for 10 minutes in a wash buffer (PBS, 0.3% Triton X-100, 0.02% NaN2) followed by a 1-hour incubation in blocking buffer (5% normal goat serum in washing buffer), followed by a 10-minute incubation in the wash buffer. The sections were incubated overnight at 4°C with primary antibodies: chicken anti-GFP (Molecular Probes, 1:1000), anti-acetyl-Histone H3 (Millipore, 1:2000), or anti-acetyl-Histone H4 (Millipore, 1:2000). After 3 washes with the wash buffer, the sections were incubated with secondary antibodies (Alexa647-goat anti-mouse IgG, Alexa488-goat anti-chicken IgG, Alexa647-goat anti-rabbit IgG; Molecular Probes, 1:1000), in blocking buffer for 4 hour at room temperature. The sections were rewashed 3 times with the wash buffer before mounting for viewing. After immunostaining, the tissue was mounted directly onto glass slides, covered, and sealed with nail polish before imaging.
Statistical analysis
Data are expressed as the means + SEM. N indicates the number of animals unless stated otherwise. Statistical analysis was performed using Prism (GraphPad). The
Results
CBPΔHAT-induced hypofunction of IL spares fear discrimination
In light of the recognized function of the infralimbic (IL) cortex in the modulation of fear, particularly regarding the extinction of conditioned fear responses,40,68,69 our initial objective was to evaluate the effects of IL hypofunction on contextual fear discrimination learning. Our behavioral protocol commences with a fear conditioning process involving 3 pairings of foot shock (unconditioned stimulus, US) paired with the conditioned stimulus (CS+) context. This is followed by multiple exposures to neutral context stimuli to eliminate a potential generalized fear to general experimental conditions (ie, testing room and personnel) and a fear memory recall test conducted 4 days after the acquisition of fear. During the final 2 days (Days 6 and 7) of the behavioral protocol, the subjects were subjected to the fear discrimination test, which entails exposure to neutral stimuli (NS) followed by presentations of the conditioned stimulus (CS+) and the non-conditioned stimulus (CS−) in an alternative sequence (as illustrated in Figure 1A). Fear acquisition is operationally defined as an increase in freezing behavior in response to the conditioned stimulus (CS+) during the fear conditioning phase, while fear discrimination performance is characterized by the differential freezing responses to CS+ compared to CS− throughout the discrimination learning phase.
Prior studies indicated that acute expression of the mutated CBP with eliminated acetyltransferase activity, called CBPΔHAT, in adult mouse excitatory neurons, impaired long-term memory consolidation.17,57 To assess the role of IL in fear discrimination, we tested the effects of the expression of CBPΔHAT in IL. We used a dual viral-vector Cre-dependent system to conditionally express mCherry in IL neurons for the IL-Ctrl group, or CBPΔHAT and mCherry in IL neurons for the IL-CBPΔHAT group (Figure 1B and C, Supplemental Figure 1). IL-Ctrl and IL-CBPΔHAT mice showed similar freezing levels during fear conditioning (Figure 1D, two-way RM ANOVA, US × Group:
Following the assessment of fear memory recall, mice were subjected to fear discrimination testing on days 6 and 7, with an alternative order of CS+ and CS− stimuli presentations. IL-Ctrl and IL-CBPΔHAT groups showed no apparent differences in the performance during the fear discrimination test (Figure 1G, RM two-way ANOVA Group × Context,
CBPΔHAT-induced hypofunction of PL resulted in fear discrimination impairment
The prelimbic cortex (PL) is located next to the infralimbic cortex (IL) and is linked to fear modulation rather than acquisition
70
and fear discrimination learning.
71
Thus, we tested the effect of PL hypofunction in the same behavioral paradigm shown in Figure 1 to compare IL versus PL effects on contingency judgment. Using bilateral stereotaxic injection, we targeted the expression of CBPΔHAT within PL (Figure 2A) using mCherry expression in PL neurons for the PL-Ctrl group and CBPΔHAT-mCherry in PL neurons for the PL-CBPΔHAT group (Figure 2B). Both PL-Ctrl and PL-CBPΔHAT mice displayed similar freezing levels throughout contextual fear conditioning (Figure 2C, two-way RM ANOVA, US × Group:
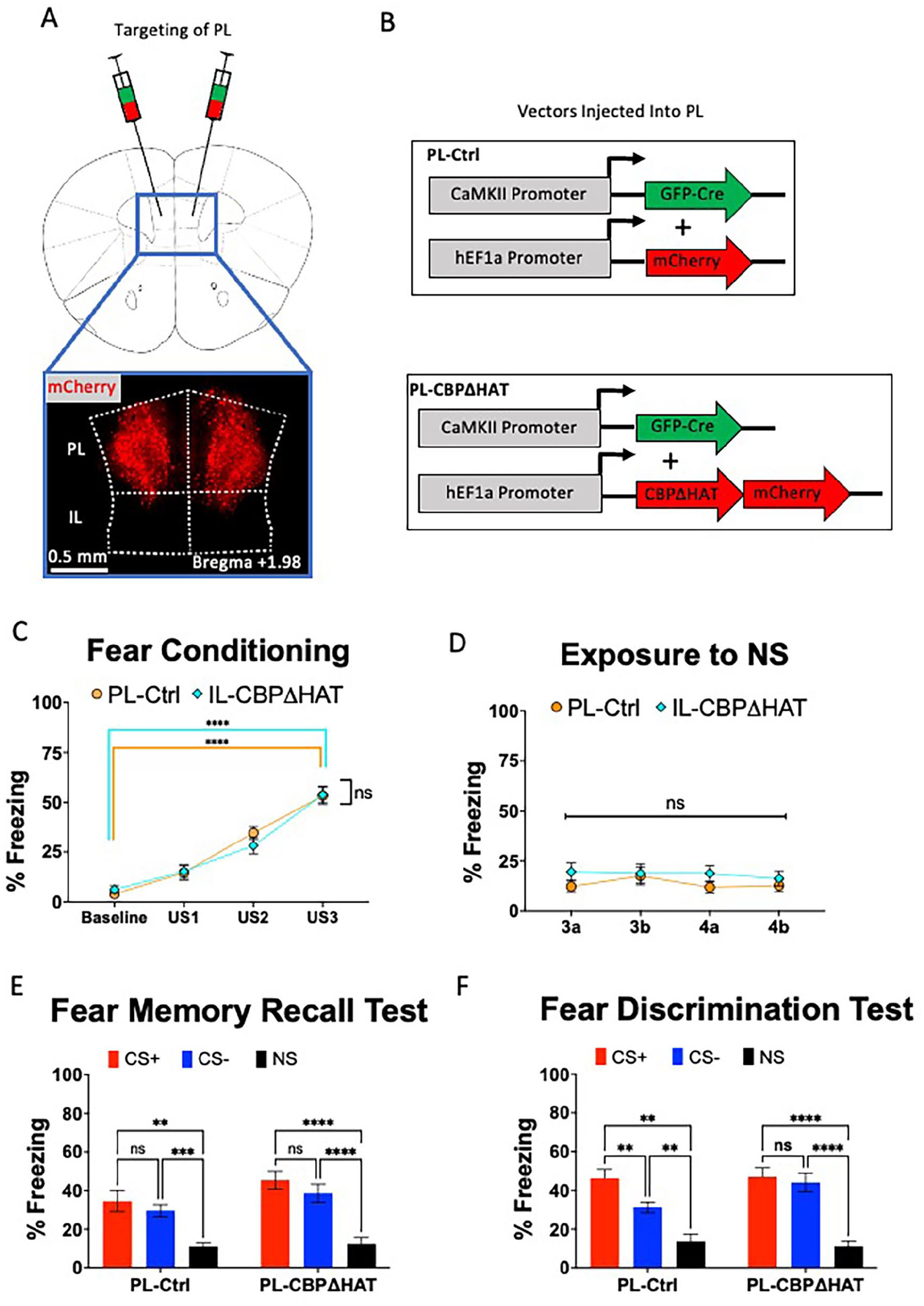
Expression of CBPΔHAT in PL impairs discrimination learning. (A) Viral injection targeting of PL cortex. About 0.2 µl of a 1:1 mixture of a GFP-Cre virus and a Cre-dependent mCherry virus was injected bilaterally into PL cortex, at a 15° angle. (B) Dual viral-vector system for expressing CBPΔHAT in PL cortex. An AAV1.CamKII.Cre-GFP virus was mixed in a 1:1 ratio with either HSV.hEF1a.LS1L.mCherry for Ctrl group, or HSV.hEF1a.LS1L.CBPΔHAT.mCherry for CBPΔHAT group and injected into PL. (C) Performance on the fear conditioning test. Both PL-Ctrl and PL-CBPΔHAT groups display equivalent performance on the fear acquisition and the 72-hour fear memory test. (D) Both PL-Ctrl and PL-CBPΔHAT groups display low freezing to NS. (E) Performance on the fear memory test. Both PL-Ctrl and PL-CBPΔHAT groups display similar level of fear during 72-hour fear memory test. (F) Performance on the fear discrimination test. PL-Ctrl mice discriminate between CS+ and CS− contexts; however, PL-CBPΔHAT mice do not discriminate between CS+ and CS− contexts during the fear discrimination test.
Analysis of the performance on the fear memory recall test applied 4 days after fear acquisition revealed substantially, but equally, elevated levels of fear in response to CS+ compared to NS in both PL-Ctrl and PL-CBPΔHAT groups (Figure 2E. two-way RMANOVA, Group × Context:
PL-Ctrl and PL-CBPΔHAT groups showed apparent differences in performance during the fear discrimination test (Figure 1G, RM two-way ANOVA Group × Context, Group × Context:
In summary, CBPΔHAT expression-triggered hypofunction in PL and IL does not affect fear acquisition or memory recall. However, the ability for fear discrimination was substantially impaired when CBPΔHAT was expressed in PL, but the abnormal fear discrimination phenotype was spared in IL-CBPΔHAT mice.
Targeting PL neurons with CBPΔHAT reduces histone acetylation
cAMP response element-binding protein (CREB)-binding protein (CBP) is a transcription coactivator, directing epigenetic gene expression programs. CBP executes its epigenetic functions via protein-protein interactions and intrinsic histone acetylation activity, which are critical to ensure precise temporal and unique genome expression patterns.72,73 The epigenetic functions of CBP have been studied across different biological systems. A mutant version of this molecule called CBPΔHAT retains regular protein-protein interactions but has deficient histone acetyltransferase activity and has been very effective in studies of neural mechanisms.
55
Acute expression of the CBPΔHAT mutant in adult mouse excitatory neurons results in impaired long-term memory consolidation and reduced histone acetylation without impacting short-term memory or network interactions.17,57 We tested the effects of the expression of CBPΔHAT on the H3 and H4 histone acetylation in PL neurons. Figure 3A shows the AAV and HSV viral vectors used to generate targeted CBPHAT hypofunction in PL neurons (ie, AAV1.CaMKII.GFP-Cre and HSV.hEF1a.LS1L.CBPΔHAT.mCherry) were both targeted to the same neuronal populations (Figure 3A). In addition, Figure 3B confirmed that the expression of CBPΔHAT was targeted to cells expressing neuronal marker NeuN. Figure 3C shows immunochemistry for detecting H3 and H4 histone acetylation levels in neurons CBPΔHAT in PL-CBPΔHAT mutant mice. PL-CBPΔHAT mice displayed reduced acetylation of histone H3 (

Expression of CBPΔHAT in neurons decreases H3 and H4 acetylation. (A) Representative images of viral GFP and mCherry expression. About 2 weeks after injection of dual viral vectors, GFP is expressed in nuclei, with a subpopulation of GFP+ cells also expressing mCherry. (B) Staining mouse brain slices with NeuN, a marker specific for neurons, shows that all mCherry+ cells are NeuN+. (C) Expression of CBPΔHAT reduces acetylation of histones H3 and H4. CBPΔHAT+ neurons have reduced acetylation of histone H3 (Ac-H3, left panel) and H4 (Ac-H4, right panel) compared to controls.
The current data indicate that successful contextual fear discrimination learning relies on the integrity of mPFC. Notably, the hypofunction of PL yielded learning impairment. The acetylation of H3 and H4 histones mediates this learning deficit. Mice expressing CBPΔHAT show an imbalance of acetylation and deacetylation, with reduced acetylation due to lower acetylase activity.
Discussion
These data demonstrate the differential roles of IL and PL in contextual fear discrimination learning and the critical role of the CBP’s acetyltransferase activity in contingency judgment learning to gage appropriate fear responses. An epigenetic hypofunction triggered by the expression CBPΔHAT mutant in PL cortices led to an impairment in contextual fear discrimination learning. Still, the performance of IL-targeted CBPΔHAT mutant mice was spared. Performance on fear conditioning was spared in both IL- and PL-targeted mutant mice. CBPΔHAT expression in PL reduced global histone acetylation levels in prefrontal neurons, which is consistent with prior studies17,55,57 and aligned with the idea that CBP’s acetyltransferase function is involved in the neural epigenetic signaling that underlies long-term synaptic plasticity and memory consolidation.57,59,60
CBPΔHAT expression in IL had a negligible effect on the performance in contextual fear discrimination learning. This is an unexpected result. IL has been strongly considered to be involved in fear suppression and extinction of conditioned fear.39,74,75 Indeed, the IL cortex seems to be a critical modulatory region for reducing fear behavior during fear extinction learning, underscored by IL’s anatomical and functional connectivity with the amygdala. IL is likely to negatively control fear via polysynaptic pathways to the central nucleus of the amygdala. IL projects to the amygdala intercalated neurons inhibitory relay29 -32 and shares reciprocal projections with the basolateral amygdala (BLA).44,75,76 BLA itself projects to the central nucleus of the amygdala via monosynaptic projections and polysynaptic pathways via the amygdala intercalated neurons relay,29 -32,77 the main output for fear behavior, so modulation of BLA activity via the IL-BLA pathway could reduce downstream CeA output, leading to decreased fear behavior. 78 However, IL is not the only region within mPFC that has anatomical and functional connectivity with the BLA; PL also has robust connectivity with BLA, which is associated with fear expression 79 and discrimination. 80 For this reason, we wanted to compare the effects of CBPΔHAT-induced hypofunction in PL and IL on contextual fear discrimination learning.
Surprisingly, the discrimination impairment seen in PL-targeted mice (Figure 2F) was not reproduced in IL-targeted mice (Figure 1G). Research regarding cognitive processes post-brain injury suggests that brain functionality operates in a dynamic manner. In the context of Pavlovian fear conditioning, it is evident that contextual memories may develop independently from the conventional neural pathways identified as responsible for processing and storing environmental information in animals. 81 These observations imply that alternative systems can produce contextual fear responses, but their efficacy may not be as strong. For example, the prefrontal microcircuit can compensate for the loss of the hippocampus in contextual learning. 82 Consequently, the data indicate that it is plausible for the PL to mitigate the deficit of IL in the process of encoding new memories; however, it is evident that the contrary is not feasible.
While previous work has proposed differential roles of fear expression and fear inhibition for the PL and IL regions, respectively, 41 recent studies provide evidence for a critical role of PL in fear extinction safety learning. 45 Not only is there reciprocal anatomical connectivity between PL and IL, 44 but there is also evidence that PL and IL have specific and opposing roles to support new learning together flexibly. 44 Moreover, PL input to IL is required to mediate fear extinction learning. 45 Our data complement these findings and provide strong evidence for the necessity of PL in fear discrimination learning that relies on learning opposing cue-outcome relationships. It also suggests that the IL network is not critical for coding during fear discrimination learning or that the PL network can compensate for IL’s inability to code new information.
Conclusions
These results align with recent work investigating PL functions in fear discrimination learning. 43 However, the role of IL has not been confirmed. Previous work from our lab used Arc-based activity tagging to assess neuronal ensemble activity associated with contextual discrimination learning. We found that in early discrimination, PL displays elevated activity in both CS+ and CS− during the early- and late- phases of fear discrimination learning. In addition, the PL cortex appears to be a locus for encoding memory representations for both CS+ and CS−, while the IL network encodes CS− memory representation. 43 However, in the absence of the IL network, the PL network may adequately compensate for any dysfunction associated with the IL system. It is also possible that the IL network does not code any new information during fear discrimination learning and serves as a relay between PL and the Amygdala during contingency judgment learning to gage fear.
Supplemental Material
sj-jpg-1-exn-10.1177_26331055241305378 – Supplemental material for The Neuronal Hypofunction of Subdivisions of the Prefrontal Cortex Shows Differential Effects on Contingency Judgment Learning to Gauge Fear Responses
Supplemental material, sj-jpg-1-exn-10.1177_26331055241305378 for The Neuronal Hypofunction of Subdivisions of the Prefrontal Cortex Shows Differential Effects on Contingency Judgment Learning to Gauge Fear Responses by Tyler W Bailey, John H Speigel, Johannes Mayer and Edward Korzus in Neuroscience Insights
Footnotes
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: DoD/ARL grant W911NF-23-1-0145 (EK). National Institutes of Health grant R01 MH106617 (EK). Brain and Behavior Research Foundation grant (EK). We thank Richard Nguyen, Justin Pastore, Ashley Burroughs, and Kritee Sekhon, whose tireless efforts contributed to this project’s success. The research was sponsored by the Army Research Office and was accomplished under Grant Number W911NF-23-1-0145. The views and conclusions contained in this document are those of the authors and should not be interpreted as representing the official policies, either expressed or implied, of the Army Research Office or the U.S. Government. The U.S. Government is authorized to reproduce and distribute reprints for Government purposes, notwithstanding any copyright notation herein.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
