Abstract
Identification of early influences on cognitive decline is of paramount importance in order to stem the impacts of decrements in cognitive functioning and to potentially intervene. Thus, here we focused on 132 healthy adult women (age range 26-98 years) to (a) determine whether factors circulating in serum may exert neurotoxic effects in vitro, (b) evaluate associations between serum neurotoxicity and cognitive performance, and (c) assess the influence of human herpes virus (HHV) seroprevalence and other factors on apoptosis and cognitive performance. The results documented that the addition of serum from healthy adult women to neural cell cultures resulted in apoptosis, indicating the presence of circulating neurotoxic factors in the serum. Furthermore, apoptosis increased with age, and was associated with decreased cognitive performance. Stepwise regression evaluating the influence of 6 HHVs on apoptosis and cognitive function revealed that only HHV5 (cytomegalovirus; CMV) seropositivity was significantly associated with apoptosis and cognitive decline, controlling for age. These findings document neurotoxic effects of serum from healthy women across the adult lifespan and suggest a unique detrimental influence associated with CMV seropositivity.
Introduction
Humans are exposed to myriad natural and synthetic neurotoxins, several of which have been implicated in neurodegeneration.1 -8 Neurotoxins circulate in the blood and, therefore, are readily detected in blood and serum samples. 9 Under healthy conditions, the brain is protected from exposure to neurotoxins via the blood-brain barrier (BBB); however, breakdown of the BBB enables entry of harmful substances into the brain, ultimately contributing to neuronal dysfunction and neurodegeneration.10,11 Several factors including sex, age, temperature, physical exercise, neurotransmitters, inflammatory substances, and inflammatory markers, among others, influence BBB permeability.12,13 In addition, infection with neurotropic viruses results in structural and functional changes in several components of the BBB, thereby affecting permeability and permitting entry of viruses and other pathogens into the brain. 14
Numerous viruses are neurotropic and therefore capable of entering the nervous system and causing immediate or delayed neuropathological changes and disease. 15 Human herpes viruses (HHVs) are among the most common viruses worldwide and are associated with numerous and varied human health conditions.16 -19 After initial infection, typically in childhood, HHVs establish latency and may be periodically reactivated by various factors including stress, fever, and UV exposure, as well as age-related immunological changes and conditions and/or treatments associated with immunosuppression.20,21 HHVs vary in terms of cell tropism. Some HHVs are neurotropic and establish latency within the nervous system; several others establish latency elsewhere (eg, B cells, monocytes) but are capable of entering the brain via the bloodstream or peripheral nerves. Consequently, several HHVs have been documented in brain tissue,15,22 -26 and increasing evidence points to a role of HHVs in conditions affecting the brain.19,25,27 -37 HHVs are ubiquitous; consequently, nearly 100% of adult humans are seropositive for at least 1 type of HHV. Five HHVs are most commonly implicated in human disease—namely, herpes simplex virus type 1 (HSV/HHV1) and 2 (HSV/HHV2), varicella zoster virus (VZV/HHV3), Epstein-Barr virus (EBV/HHV4), cytomegalovirus (CMV/HHV5). 21 Burgeoning evidence also links HHV6 and HHV7 to neurodegenerative diseases.26,38 -41
Here, we bridge these separate lines of research implicating neurotoxins and HHVs in neuropathology by assessing the effects of serum obtained from healthy adult women on neural cell cultures and with regard to HHV seropositivity. Previous research in our lab and elsewhere has documented neurotoxic effects of serum from patient populations 42 and older adults 43 on neural and hippocampal progenitor cell cultures, respectively. It is not clear whether findings documenting the presence of harmful substances circulating in the blood in those populations extend to younger, healthy samples; however, since subtle neuropathological changes are evident decades before symptom onset and clinical diagnoses, 44 it is important to assess for the presence of potentially neurotoxic factors in healthy individuals across the lifespan. Thus, we first evaluated whether the addition of serum from healthy adult women to neural cell cultures resulted in apoptosis, and assessed the correspondence between apoptosis and cognitive performance. Then, in light of immense literature implicating HHVs in conditions affecting the brain, we evaluated apoptosis and cognitive performance with regard to seropositivity of neurotropic HHVs. Since apolipoprotein E and body mass index (BMI) have been shown to influence cognitive function,45,46 we also evaluated and controlled for their influence in this sample.
Materials and Methods
Participants
A total of 132 healthy women participated in the study as paid volunteers. This data was obtained as part of an ongoing longitudinal study involving annual data acquisition; consequently, the number of annual visits varied for participants as follows: 60 participants completed 1 annual visit, 56 completed 2 annual visits, and 16 completed 3 annual visits, for a total of 220 visits. Women were excluded from participation if they had been diagnosed at any point in their lifetime with a neurological disorder, any autoimmune disorder associated with neurocognitive dysfunction (eg, systemic lupus erythematous, rheumatoid arthritis), any major medical condition affecting brain function (eg, brain cancer, head injury with cognitive sequelae), serious psychiatric diagnoses (eg, bipolar disorder, schizophrenia, any history of psychiatric hospitalization), or any recent/current medication or treatment known to affect brain functioning (eg, radiation, chemotherapy). Written informed consent was obtained from study participants. The institutional review board and relevant committees of the Minneapolis VA Health Care System approved the study protocol.
Cognitive assessment
The Montreal Cognitive Assessment (MoCA) 47 was administered to participants to screen for cognitive impairment. The MoCA assesses several domains of cognitive function including executive function, memory, language, and abstract reasoning, among others. Scores for each domain are added to reflect a total score ranging from 0 to 30; total scores greater than 25 are considered normal. 47 At recruitment, all participants had a total MoCA score (without the education point) of greater than 25. MoCA assessment was obtained during all but 1 visit, for a total of 219 MoCA assessments.
ApoE genotyping
DNA samples were genotyped using PCR amplification followed by restriction enzyme digestion as per recommendations. 48 Known apoE isoform standards were included in the analysis.
HHV seropositivity assays
We determined the seroprevalence of IgG antibodies against HHV1, HHV2, HHV3, HHV4, HHV5, and HHV6; we could not obtain reliable results from kits for HHV7 and HHV8. We employed standard method using commercially available indirect Enzyme-Linked Immunosorbent Assays (ELISA). The ELISA tests were performed as per the manufacturer’s instructions and recommendations. The specimens were processed using a mini-automated 5-in-1 workstation (Crocodile cat. 84024-01; Berthold Technologies, Oak Ridge, TN, USA). The workstation includes the ELISA microtiter plate reader which was read at dual wavelengths for absorbance at 450 and 620 nm as reference wavelengths. Details of the virus-specific ELISA kits are as follows. (a) HHV1: Human Anti-Herpes simplex virus Type 1 IgG ELISA Kit (HSV1), Abcam Inc., Boston, MA USA, cat. ab 108737; (b) HHV2: Human Anti-Herpes simplex virus Type 2 IgG ELISA Kit (HSV2) Abcam cat. ab 108739; (c) HHV3: Human Anti-Varicella-Zoster virus IgG ELISA Kit (VZV) Abcam cat. ab 108782; (d) HHV4: Human Anti-Epstein Barr virus IgG ELISA Kit (EBV-VCA) Abcam cat. ab 108730; (e) Human Anti-Cytomegalovirus IgG ELISA Kit (CMV) Abcam cat. ab 108724; (f) Human Herpesvirus 6 IgG ELISA Kit cat. KA1457, Abnova, Taiwan. Seroprevalence for 1 or more HHVs were missing for 7 women.
Body mass index (BMI)
BMI was calculated from anthropometric measurements. Weight was not able to obtained for a small number of acquisitions (n = 7), precluding BMI calculations in those instances; BMI was available for 213 acquisitions and was used as a continuous measure in analyses.
Neural cell culture
Neuro-2A neuroblastoma (N2A) cells were cultured in Eagle’s minimal essential medium (EMEM, ATCC, VA, USA) containing 10% fetal bovine serum (ThermoFisher Scientific, Waltham, MA) poly-D-lysine coated, 24-well plates at a concentration of 30 000 to 50 000/well for 48 to 72 hour. The medium was then changed to Neurobasal containing N2 supplement and L-glutamine (ThermoFisher Scientific, Waltham, MA), in the absence (medium control) or presence of human serum. For all experiments, human serum (10%) was added and the cells were cultured for 2 more days. All experiments were carried out in quintuplicate.
Cell apoptosis with terminal deoxynucleotidyl transferase mediated dUTP Nick End Labeling assay (TUNEL) assay
The extent of cell apoptosis of Neuro-2A cells incubated with women’s serum was examined. All samples were examined using 4- and 8-chamber glass slides (ThermoFisher Scientific, Waltham, MA) coated with poly-D-lysine at 50 μg/ml as mentioned above. N2A cells were seeded at a concentration of 50 000 to 100 000 cells per chamber, in 1 ml of Neurobasal/N2/ L glutamine medium for 2 days. In sequence, 10% of each sample of women’s serum, incubated for 60 minutes at 37°C. At the end of the incubation period, the cells were examined for apoptosis. Apoptotic cells were detected using the In Situ Cell Death Detection Kit, TMR red (Terminal deoxynucleotidyl transferase (TdT) enzyme and fluorochrome labeling solution), according to the manufacturer’s protocol. Briefly, the cells were fixed in ice-cold methanol for 10 minutes at room temperature, rinsed with PBS and permeabilized with 0.1% Triton X-100 in PBS for 3 minutes on ice. The cells were then incubated with 150 μl of TUNEL reaction mixture for 60 minutes at 37°C in the dark (Insitu Cell Death Detection Kit, TMR red, ThermoFisher scientific, Waltham, MA). The cells were then washed 3X with PBS and Diamond AntiFade mounting medium with 4′,6-diamidino-2-phenylindole (DAPI) stain (ThermoFisher Scientific, Waltham, MA) was used for visualization of nuclei, using the EVOS FL Cell Imaging System (ThermoFisher Scientific, Waltham, MA) or an Olympus 3000 confocal microscope. The 8 to 10 images were obtained from different fields from a minimum of 2 experiments with each different experimental condition. Apoptosis was then calculated with ImageJ software by measuring the number of TUNEL-labeled cells (red nuclei) relative to the total cell number (DAPI-stained nuclei). Since experiments were run in quintuplicate, there were 5 Media and 5 Test apoptosis values per run (
Data Analysis
MoCA
MoCA values are counts and hence were square-root transformed, a transformation appropriate for the statistical analysis of counts 49 :
All MoCA-related statistical analyses were performed using sMoCA.
ApoE
With respect to apoE genotype, 3 apoE isoforms (E4, E3, and E2) are the result of cysteine-arginine interchanges at 2 sites: there are zero interchanges in E4, 1 interchange in E3, and 2 interchanges in E2. The resulting 6 apoE genotypes (E4/4, E4/3, E4/2, E3/3, E3/2, E2/2) yield 5 groups with respect to the number of cysteine residues per mole, as follows. ApoE4/4 contains zero cysteine residues per mole (0-CysR/mole), E4/3 contains 1 (1-CysR/mole), E4/2 and E3/3 each contain 2 (0-CysR/mole), E3/2 contains 3 (0-CysR/mole), and E2/2 contains 4 (0-CysR/mole). The use of the number of cysteine residues per mole to characterize the apoE molecule converts the categorical apoE genotype scale, consisting of 6 distinct genotypes above, to a 5-point continuous scale (0-4 cysteine residues per mole), a conversion that allows the use of statistical analyses suitable for continuous variables (eg, regression) to quantify the relations between various variables and apoE.50,51 Therefore, we used the number of cysteine residues per mole (CysR/mole) as a covariate to evaluate the possible association of apoE with MoCA and apoptosis. In addition, as a measure complementary to CysR/mole above), we computed a binary variable, E4, based on the presence [apoE4(+) = 1] or absence [apoE4(−) = 0] of the apoE4 allele in the participant’s apoE genotype and used it as a fixed group factor in statistical analyses below.
Implementation of statistical analyses
The IBM-SPSS Statistical package (version 29) was used to analyze the data, including Pearson correlation, partial correlation, and analysis of covariance (ANCOVA). All P-values reported are 2-tailed.
Results
Age, MoCA, apoE, BMI
The mean (±SEM) age of the study participants was 70.4 ± 0.79 years, (range 25.7-97.8 years, N = 220), the mean of the MoCA was 27.25 ± 0.16 (range 17-30, N = 219), and the mean BMI was 28.28 ± 0.44 (N = 213). The frequency distributions of age, MoCA, and BMI are shown in Figure 1. The frequency distribution of the apoE genotypes and the number of apoE CysR/mole are given in Tables 1 and 2, respectively; there were no E2/E2 homozygotes. Analyses evaluating associations between sMoCA, apoE, BMI, and age revealed several significant associations. Age was associated with decreased sMoCA (Pearson r = −.308, P < .001, N = 219; Figure 2A) and decreased BMI (r = −.373, P < .001, N = 213). BMI was not significantly associated with sMoCA, controlling for age (partial correlation r = − 0.117, P = .097, N = 213). Finally, sMoCA increased significantly with the number of apoE CysR/mole, controlling for age (sMoCA vs CysR/mole, partial correlation = .154, P = .024, N = 218) and was significantly higher in the apoE4(+) than the apoE4(−) group (P = .042, F-test in ANCOVA, where sMoCA was the dependent variable, apoE4 was a (binary) fixed factor, and age was a covariate).
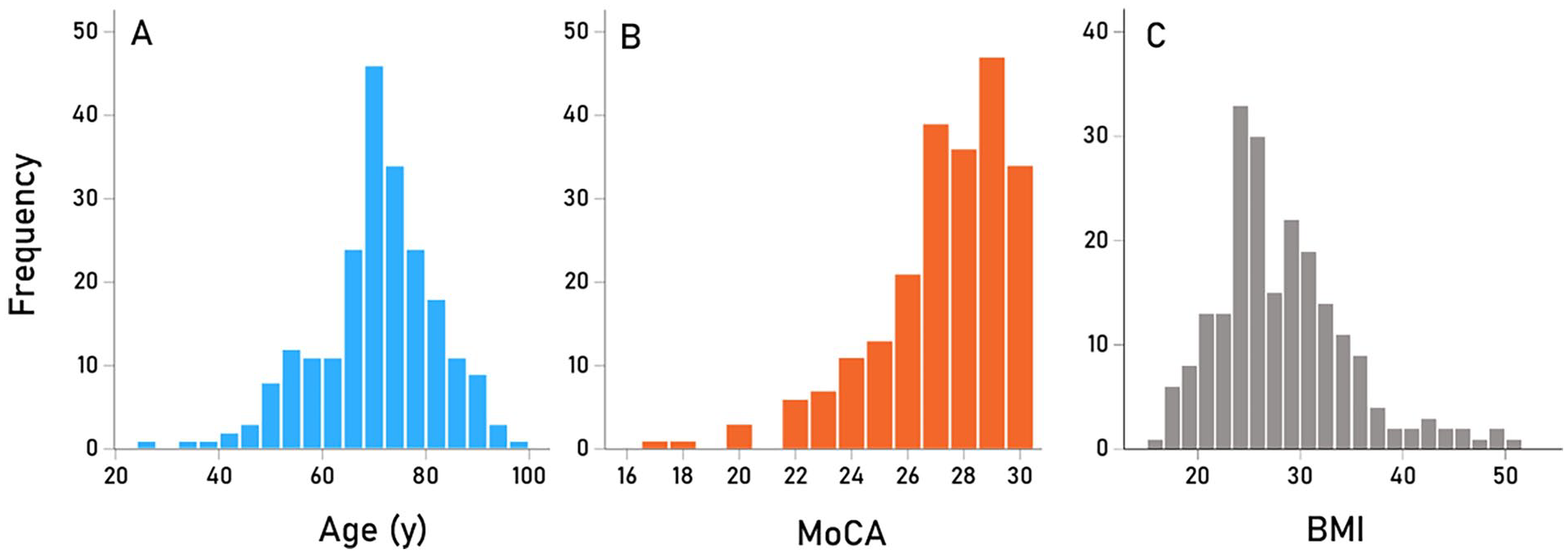
(A) Frequency distribution of ages at time of test. N = 220 participants. (B) Frequency distribution of MoCA scores at time of test. N = 220 participants. (C) Frequency distribution of BMI. N = 213 participants.
Frequency distribution of apoE genotypes.

Frequency distribution of number of apoE CysR/mole.


(A) mean (±SEM) MoCA scores are plotted against age. N = 220. (B) % apoptosis
Apoptosis
Next we evaluated the associations between sMoCA, apoE, BMI, and age with apoptosis. We found that apoptosis increased significantly with increasing age (Partial correlation
HHV
The seroprevalence of HHV1-6 are given in Table 3. The effect of HHV seroprevalence on apoptosis was evaluated using a stepwise multiple linear regression, where
Prevalence of seropositivity for the 6 HHV strains investigated. Numbers in the column Total indicate the number of tests that gave unambiguous results in the ELISA.

Results of the multiple regression analysis described in the text.

VIF, variance inflation factor. Bold denotes significant effect on HHV seroprevalence on apoptosis.
The HHV5 effect was quantified by performing an ANCOVA where

Mean % apoptosis
Discussion
In this study we evaluated the effect of serum from healthy women on neural cell cultures and with regard to cognitive performance and HHV seroprevalence. The findings documented the presence of neurotoxic factors, unrelated to BMI or apoE, that resulted in apoptosis and influenced cognitive function. Previous studies have documented serum neurotoxicity in older adults 43 and in certain patient populations 42 ; here, we show neurotoxic factors resulting in in vitro apoptosis and decreased cognitive performance in healthy women across the adult lifespan. This finding highlights deleterious effects of neurotoxic factors circulating in the blood. Since HHVs have been widely implicated in conditions affecting the brain, we then evaluated the influence of seroprevalence of 6 HHVs, BMI, and apoE on apoptosis and cognitive performance. The findings documented a unique influence of HHV5/CMV seropositivity on increased apoptosis and decreased cognitive performance. Notably, apoE CysR/mole was associated with protective effects on cognitive performance in CMV seronegative women but not those who were seropositive. Taken together, the findings of this study point to neurotoxic factors in serum of healthy women that may be associated with CMV exposure, adding to the literature regarding the potential for harmful downstream effects of CMV infection on brain health.
CMV is a common infection worldwide 52 that is transmitted congenitally or through direct contact with body fluids. The seroprevalence of CMV increases with age such that approximately half of women of childbearing age and two-thirds of women age 70 to 79 are seropositive.53,54 Like other human herpes viruses, CMV establishes lifelong persistence characterized by periods of latency and reactivation. 20 Age-related increases in chronic reactivation of CMV, 55 coupled with increased CMV viral load, 56 and CMV-specific adaptive immune system dysregulation (eg, inflation of memory T cells and low or inverted CD4/CD8 ratio)57,58 exhaust the immune system over time. 57 These effects are thought to contribute to low-grade chronic inflammation (ie, “inflammaging”), 59 immunosenescence, 60 epigenetic aging, 61 and increased mortality.62,63 To that end, CMV seropositivity has been associated with various chronic health conditions including atherosclerosis 64 and vascular dysfunction, 65 autoimmune disorders, 66 and cancer, 67 as well as age-related frailty 68 (cf Matheï et al 69 ), functional impairment, 70 and cognitive decline.71,72
Here, we showed correspondence between neurotoxic effects of serum from CMV seropositive women and decrements in cognitive performance. These findings suggest that the presence of harmful agents circulating in the blood of CMV seropositive individuals may impact brain function. Previous studies have identified CMV as a risk factor for cognitive impairment and dementia71 -75 (cf, Matheï et al 69 ). Furthermore, CMV is present in a high proportion of brains from vascular dementia patients, 76 and recently, CMV has been shown to induce tau pathology, a hallmark of Alzheimer’s dementia. 77 The extent to which the neurotoxic effects of CMV seropositive serum observed in these healthy women is a predictor of future cognitive dysfunction remains to be determined and is an ongoing area of investigation in our lab.
These novel findings implicating CMV in the association between serum neurotoxicity and cognitive function must be considered within the context of study limitations. First, we used neural cell cultures as a proxy for the brain and it is not clear to what extent the deleterious in vitro effects correspond with in vivo effects in the human brain. Remarkably, the in vitro effects were associated with cognitive function suggesting some correspondence between in vitro and in vivo effects. Second, the present study focused exclusively on women. Since previous studies have documented sex-specific neurotoxic effects, 78 it is unknown whether the present findings generalize to men. Third, our analyses were limited to presence or absence of HHV antibodies, precluding evaluation of effects of titer concentration on apoptosis and cognitive performance. Finally, it is worth noting that the present findings do not preclude involvement of other potentially neurotoxic factors; nonetheless, the findings document serum neurotoxicity and decrements in cognitive performance in CMV seropositive women. Consequently, the development of CMV vaccines 79 may be beneficial in preventing CMV-related cognitive decline.
Footnotes
Acknowledgements
We would like to thank Laura Kalipe, Briana Pomonis, and Stacy Dolan for their contributions to data collection.
Author Contributions
APG: conceived the research; LMJ, EPT, and EJW contributed to data acquisition; APG, LMJ, and EPT wrote the paper; all authors edited and approved the final version of the paper.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Partial funding for this study was provided by the University of Minnesota (the Anita Kunin Chair in Women’s Healthy Brain Aging, the Brain and Genomics Fund, the McKnight Presidential Chair of Cognitive Neuroscience, and the American Legion Brain Sciences Chair) and the U.S. Department of Veterans Affairs. The sponsors had no role in the current study design, analysis or interpretation, or in the writing of this paper. The contents do not represent the views of the U.S. Department of Veterans Affairs or the United States Government.
Data Availability
Data will be available to qualified researchers by the corresponding author on a reasonable request.

