Abstract
Epilepsy is one of the most common neurological diseases, which is caused by abnormal brain activity. A wide variety of studies have shown the importance of the phosphatidylinositol-3-kinase (PI3K) signaling pathway in epilepsy pathogenesis. Duvelisib (DUV) is a selective inhibitor of PI3K. The present study investigated the anticonvulsant potential of DUV in a rat model of pentylenetetrazole (PTZ)-induced convulsions. Male Wistar rats (200-250 g, 8 weeks old) were injected intraperitoneally (IP) with DUV at different doses of 5 and 10 mg/kg, or vehicle 30 minutes prior to PTZ (70 mg/kg, IP) treatment. Based on Racine’s scale, behavioral seizures were assessed. The results showed that pretreatment with DUV prolonged the seizure stages according to the Racine scale, significantly decreased the duration of general tonic-clonic seizure and reduced the number of myoclonic jerks (P < .05). In conclusion, we found that PI3K antagonist DUV significantly reduced PTZ-induced seizures, indicating that DUV exerts an anticonvulsant effect by inhibiting PI3K signaling pathway.
Introduction
There is an electrical imbalance between excitatory and inhibitory neurotransmission in the brain that causes epilepsy, a chronic neurological condition characterized by periodic seizures. 1 In addition to the basic excito-inhibitory mechanisms, there are other factors that contribute to epileptic seizures, such as non-synaptic electrical transmission through gap junctions, involvement of non-neuronal glial cells, namely astrocytes and microglia, and metabolic disturbances caused by oxidative stress.2 -4 Conventional antiepileptic drugs (AEDs) act via synaptic neuronal signal transmission by either prolonging excitation or facilitating inhibitory neurotransmission. It remains unclear what the exact epileptogenic mechanism is that causes seizures despite all the research done so far on epilepsy. Furthermore, the available AEDs only provide symptomatic relief and do not provide insights into what causes epilepsy in the first place.5,6 A patient may become resistant to conventional AED therapies due to accumulating seizures along with other associated conditions such as memory impairment, anxiety, depression, and so on, thus decreasing their quality of life. The identification of biomarkers (genetic, structural, functional, electrophysiological, and neuroinflammatory) for the treatment of epilepsy has been recognized as valuable for improving the outcome of the disease condition, but these markers come with their own set of challenges that need to be addressed very pragmatically. 7 To this end, efforts are being made to understand the exact molecular pathways underlying epileptogenic processes so that better drugs can be developed for epilepsy treatment.8,9
Phosphatidylinositol-3-kinase (PI3K) has become an increasingly important molecular target in the pathogenesis of diseases. PI3K belongs to a family of lipid kinases that are involved in many different cellular functions such as growth, proliferation, metabolism, and other characteristics of a cell. 10 Growth factors (epidermal growth factor, insulin-like growth factor, human epidermal growth factor, etc.) or receptor tyrosine kinase can attach to this, causing it to release phosphatidylinositol into the cell. 11 The PI3K phosphorylation of the AKT activates the mTOR pathway and its downstream genes, which then lead to epilepsy progression. 12 A growing body of evidence indicates that mTOR hyperactivation can be suppressed using specific inhibitors. 13 Therefore, inhibition of the mTOR pathway can effectively treat epilepsy. 14 Furthermore, PI3K is activated and subsequently cellular processes occur when it is phosphorylated. 15 Class I PI3Ks (which consist of a catalytic subunit, p110, and a regulatory adaptor subunit, p85) are widely implicated in different diseases that alter normal physiological function. 16
Duvelisib (DUV) is the first-in-class, small-molecule, selective inhibitor of both δ and γ isoforms of PI3K. It is being developed as treatment for various cancer indications. B and T cells are activated, proliferate and survive by modulating PI3K activity. In response to PI3K activation, some intracellular enzymes that control cell proliferation, survival, and motility are activated. 17 Research has suggested that PI3K inhibitors are associated with neuronal death during embryogenesis and apoptosis during primary culture, though PI3K/AKT is critical to brain development and promotes neuroprotection.18,19 Based on the available literature, it is apparent that PI3K inhibitors such as DUV act as both neuroprotectants and apoptosis inducers. However, a potent mTOR inhibitor, such as rapamycin, possessed similar characteristics, such as antiepileptic properties, neuroprotective properties, and pro-apoptotic effects,20,21 thus justifying the use of DUV in our study.
Using pentylenetetrazole (PTZ)-mediated convulsions as a model, the present study explored the anticonvulsant potential of DUV.
Materials and Methods
Drugs and chemicals
PTZ was purchased from Sigma Aldrich, USA. Phenobarbital sodium (PHB) procured from Chemidarou Pharmaceutical Company, Iran and used in this study as a conventional anticonvulsant drug. DUV was obtained from Gilead Sciences, Inc, Canada. PTZ was prepared as a 1% v/w solution in saline. PHB and DUV were dissolved in dimethyl sulfoxide (DMSO) (60 mg/mL) and stored at −20°C.
Animal maintenance and ethical consideration
Male Wistar rats (200-250 g) of 8 weeks old were purchased from the Hamadan University of Medical Sciences and were housed in standard cages at 20 to 24°C and supplied with fresh water and food ad libitum, with light and dark cycles (12:12 hours). All methods were carried out in accordance with relevant guidelines and regulations, the ethics of working with animals were observed throughout the whole study in accordance with the Animal Research Reporting In Vivo Experiments (ARRIVE) guidelines and the research protocol was also approved by the Bu-Ali Sina University’s Research Ethics Committee (IR.BASU.REC.1400.055).
PTZ-mediated seizures in rats
The animals were randomly assigned into 4 groups via simple randomization (n = 5/group) as follow:
Negative control group (NC): In this group, adult male rats administrated with vehicle (dimethyl sulfoxide (DMSO) diluted in distilled water) 30 minutes prior to PTZ (70 mg/kg) exposure.
Positive control group (PHB): In this group, adult male rats administrated with PHB (30 mg/kg) 30 minutes prior to PTZ (70 mg/kg) exposure.
Experimental group 1 (DUV 5): In this group, adult male rats administrated with DUV (5 mg/kg) 30 minutes prior to PTZ (70 mg/kg) exposure.
Experimental group 2 (DUV 10): In this group, adult male rats administrated with DUV (10 mg/kg) 30 minutes prior to PTZ (70 mg/kg) exposure.
Over the course of the study, fresh solutions of vehicle, phenobarbital, and duvelisib were injected intraperitoneally (IP) in a constant volume of 4 mL/kg. The doses of PTZ, PHB, and DUV were chosen based on previous studies on experimental animal models.22 -24 Also, the prolonged administration of DMSO via IP route at this concentration did not result in any detrimental physiological effects as evidenced in previous studies.25,26 An upper cut-off time of 30 minutes was used in measuring behavior after each rat had been exposed to PTZ. PTZ convulsive behavior was graded on a 5-point scale based on Racine’s stages as, Stage 1: ear and facial twitching; Stage 2: head nodding and myoclonic jerks; Stage 3: unilateral forelimb clonus with lorditic posture; Stage 4: bilateral forelimb clonus with rearing and falling; and Stage 5: generalized tonic-clonic seizure (GTCS) with loss of postural tone. 27 Video of rat seizures was captured using camera in the center of the chamber and software (SMART V3.0, Panlab, Barcelona). The latency to each stage (S1-5), 27 number of myoclonic jerks, and GTCS duration was also recorded in a blind fashion with help of 2 independent observer.
Statistical analysis
All the results were expressed as mean ± standard deviation. Data was tested for normality using the Shapiro-Wilk test. Given to the normal distribution of the data the statistical significant difference in behavioral parameters among different groups was examined by one-way analysis of variance followed by Tukey’s post hoc test using GraphPad Prism version 9.4.0 for Windows (GraphPad Software, San Diego, California, USA). Significant results were determined at a P value of <.05.
Results
Effect of DUV on PTZ-mediated seizures in adult rats
The latency to S1 was significantly (P = .006) increased in the rats treated with 10 mg/kg of DUV as compared to NC group [F(3,16) = 32.80, P < .001]. However, insignificant (P = .390) change was observed at 5 mg/kg of DUV as that of NC group (Figure 1).
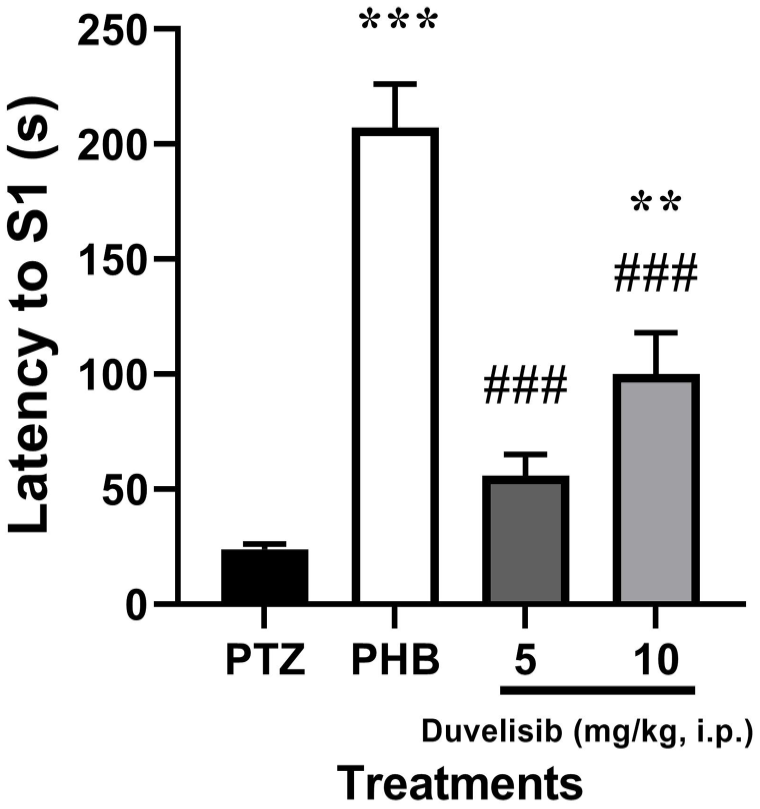
The effect of DUV (5 and 10 mg/kg, i.p.) 30 minutes prior to PTZ exposure (70 mg/kg, i.p.) on latency to Stage 1 (S1) in male Wistar rats. Data are shown as mean ± SD of n = 5 rat/group.
PHB group showed significant (P < .001) increase in latency to S2 as compared to NC group of rats [F(3,16) = 38.13, P < .001]. The latency to S2 was significantly (P = .003) increased in the group preexposed to DUV at dose of 10 mg/kg followed by PTZ (Figure 2).

The effect of DUV (5 and 10 mg/kg, i.p.) 30 minutes prior to PTZ exposure (70 mg/kg, i.p.) on latency of to Stage 2 (S2) in male Wistar rats. Data are shown as mean ± SD of n = 5 rat/group.
The seizure latency to S3 was significantly (P < .001) increased in the rats treated with 10 mg/kg of DUV as compared to NC group [F(3,16) = 59.18, P < .001]. Also, significant (P = .009) change was observed at 5 mg/kg of DUV as that of NC group (Figure 3).

The effect of DUV (5 and 10 mg/kg, i.p.) 30 minutes prior to PTZ exposure (70 mg/kg, i.p.) on latency to Stage 3 (S3) in male Wistar rats. Data are shown as mean ± SD of n = 5 rat/group.
The latency to S4 seizures onset in rats was 42.80 ± 17.60 seconds following 70 mg/kg PTZ exposure in NC group (Figure 2). The latency to S4 was significantly increased in the groups of rats pre-treated with PBH (P < .001), however significantly change was observed at 5 and 10 mg/kg (DUV group) as compared to NC group (P = .030 and P = .001, respectively) [F(3,16) = 47.04, P < .001] (Figure 4).
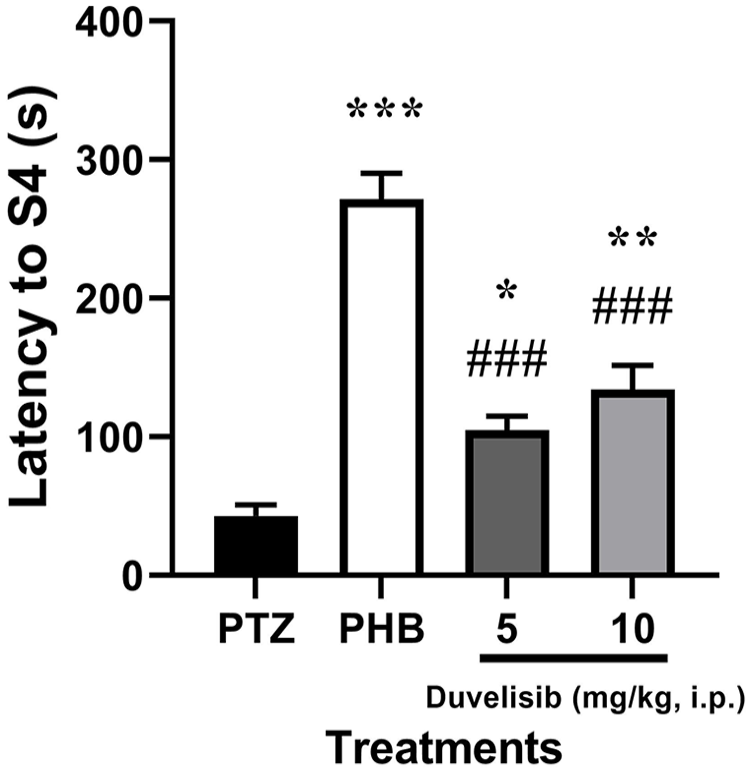
The effect of DUV (5 and 10 mg/kg, i.p.) 30 minutes prior to PTZ exposure (70 mg/kg, i.p.) on latency of to Stage 4 (S4) in male Wistar rats. Data are shown as mean ± SD of n = 5 rat/group.
The latency to S5 was significantly (P < .001) increased following PTZ exposure in rat of PHB group as compared to NC group [F(3,16) = 36.88, P < .001]. The increased latency to S5 was found to be significantly (P = .006) increased in 10 mg/kg DUV pre-incubated group as compared to NC group (Figure 5).
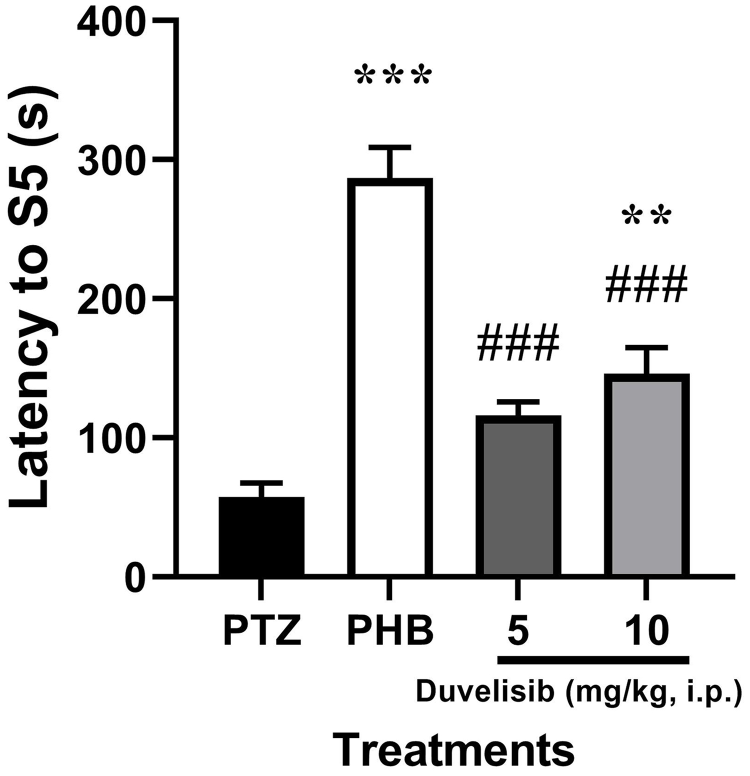
The effect of DUV (5 and 10 mg/kg, i.p.,) 30 minutes prior to PTZ exposure (70 mg/kg, i.p.) on latency to Stage 5 (S5) in male Wistar rats. Data are shown as mean ± SD of n = 5 rat/group.
The number of myoclonic jerks was found to be 54.80 ± 19.27 in NC group (Figure 6). A marked decrease in the number was observed in experimental groups treated with DUV at doses of 5 and 10 mg/kg as compared to NC group (P = .002 and P < .001, respectively) [F(3,16) = 12.84, P < .001].
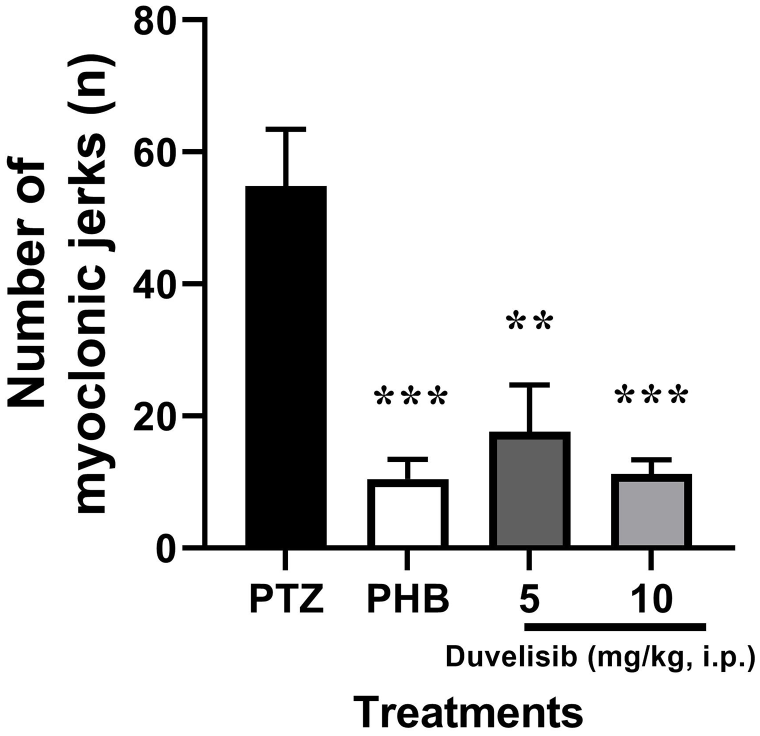
The effect of DUV (5 and 10 mg/kg, i.p.) 30 minutes prior to PTZ exposure (70 mg/kg, i.p.) on number of myoclonic jerks in male Wistar rats. Data are shown as mean ± SD of n = 5 rat/group.
GTCS duration showed a significant change in the tested adult rats between groups [F(3,16) = 12.19, P < .001]. The GTCS duration significantly (P = .019) reduced to 24.20 ± 8.25 seconds in groups treated with 10 mg/kg of DUV, as compared to NC group. However, insignificant (P = .339) change was observed at 5 mg/kg of DUV as that of NC group (Figure 7).

The effect of DUV (5 and 10 mg/kg, i.p.) 30 minutes prior to PTZ exposure (70 mg/kg, i.p.) on GTCS duration in male Wistar rats. Data are shown as mean ± SD of n = 5 rat/group.
Discussion
In the present study, we examined DUV's anticonvulsant properties in an experimental model for the first time. Compared to the NC group, DUV pre-incubation was associated with a significant delay in Racine’s stages. Furthermore, the number of myoclonic jerks was significantly reduced following pretreatment with either 5 or 10 mg/kg of DUV when compared to the NC group. As a result of treatment with 5 or 10 mg/kg of DUV, we also found that GTCS duration in adult rats significantly decreased after treatment.
Using the chemoconvulsant PTZ, rats undergo seizures and bursts of hyperlocomotor activity. Epileptic seizures are induced by hyperlocomotion, behavioural seizures, electrophysiological events (electrical events occurring in the brain as a result of PTZ exposure). 28 The conventional antiepileptic drugs are shown to decrease locomotion in the PTZ model of rats and seizure severity.22,27 In our study, rats treated with 5 or 10 mg/kg of DUV showed an increased latency to Stages 1 to 5 seizures in comparison to those untreated with DUV. Following treatment with DUV, the number of myoclonic jerks and GTCS duration decreased, indicating a decrease in the seizure activity as a result of PTZ. The results of the present study supported DUV's anticonvulsant activity.
There has been increasing interest in the role played by PI3K/AKT/mTOR in a variety of neurological disorders, including epilepsy.29,30 In addition, hyperactivation of this pathway has been shown to produce and propagate further seizures.19,31,32 Certain studies have also suggested PI3K/AKT signaling cascades are neuroprotective in animal models. 18 According to scientific literature, PI3K is necessary for AKT phosphorylation in order to activate mTOR, the master regulator of cellular processes, when activated by growth factors and receptor tyrosine kinases.33,34 In literature, the mTOR pathway is implicated in the propagation of epilepsy, thus making it an important target for controlling epilepsy. 35 Epilepsy therapy interventions involving mTOR inhibitors have been widely reported, especially for acquired epilepsy. It is important to note, however, that the use of these inhibitors, as well as the use of rapamycin, may lead to untoward side effects.5,6,34 As a result, continuous research is being conducted to identify safe and effective mTOR inhibitors for epilepsy treatment that act directly or indirectly. The anticonvulsant effect of DUV in PTZ-mediated convulsions in rats emphasizes the involvement of PI3K/AKT/mTOR pathways in epilepsy. PTZ-induced acute seizures in rats supported these results on mTOR activation induced by acute seizures. 36 As a consequence, seizure activity caused by acute exposure to PTZ increased PI3K expression, which then activated downstream genes AKT and mTOR.
The selective inhibitor of PI3K, DUV, may also have an effect on other molecular targets in the pathway, thereby modifying the central signaling pathway, thereby altering the normal cellular function. 17 The results in this regard need to be consolidated through further studies. Researchers have linked hyperactivation of PI3K/AKT/mTOR in epileptic rodent models to altered gene expression.37,38 We found that inhibitors of PI3K, such as DUV, were effective at inhibiting the development and progression of seizures in mice. It is important to note that the present study has a number of limitations. Since the study was limited to behavioral analysis and did not measure the biochemical and molecular component of the PI3K/AKT/mTOR pathway, which plays an important role in epilepsy pathology, the results of this study could pave the way for further research.
Conclusion
Based on our findings, DUV, a PI3K inhibitor, reduced PTZ-mediated seizures in adult rats. PI3K/AKT/mTOR pathway inhibition might explain DUV’s anticonvulsant effect. Results of our study demonstrated that DUV may be affect the PTZ-mediated seizures and pave the way for further studies. It is still necessary to conduct more studies to determine whether the drug has been proven effective in other epilepsy models.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MA, NM, and AH contribute equally to the conception and design, or analysis and interpretation of the data; the drafting of the paper, revising it critically for intellectual content; and the final approval of the version to be published; and that all authors agree to be accountable for all aspects of the work.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
