Abstract
Chondroitin sulfate proteoglycans (CSPGs), one of the major extracellular matrix components of the glial scar that surrounds central nervous system (CNS) injuries, are known to inhibit the regeneration of neurons. This study investigated whether pleiotrophin (PTN), a growth factor upregulated during early CNS development, can overcome the inhibition mediated by CSPGs and promote the neurite outgrowth of neurons in vitro. The data showed that a CSPG matrix inhibited the outgrowth of neurites in primary cortical neuron cultures compared to a control matrix. PTN elicited a dose-dependent increase in the neurite outgrowth even in the presence of the growth inhibitory CSPG matrix, with optimal growth at 15 ng mL−1 of PTN (114.8% of neuronal outgrowth relative to laminin control). The growth-promoting effect of PTN was blocked by inhibition of the receptor anaplastic lymphoma kinase (ALK) by alectinib in a dose-dependent manner. Neurite outgrowth in the presence of this CSPG matrix was induced by activation of the protein kinase B (AKT) pathway, a key downstream mediator of ALK activation. This study identified PTN as a dose-dependent regulator of neurite outgrowth in primary cortical neurons cultured in the presence of a CSPG matrix and identified ALK activation as a key driver of PTN-induced growth.
Introduction
Chondroitin sulfate proteoglycans (CSPGs) are a major component of the extracellular matrix that surrounds cells of the central nervous system (CNS). 1 CSPGs maintain CNS health by regulating the growth of axons during development and protecting against oxidative stress. 2 After CNS injury, activated glial cells increase the synthesis of CSPGs to form a glial scar that contains the injury site.1,3 The scar reduces lesion growth but also acts as a barrier to the regeneration of neurons that may limit functional recovery.1,4,5 CSPGs are also upregulated in neurodegenerative conditions including Alzheimer’s disease (AD), Pick bodies in Pick’s disease, and Lewy bodies in Parkinson’s disease. 6 Several experimental approaches have been investigated to neutralize the growth inhibitory effect of CSPGs in vivo, including digestion of CSPGs by chondroitinase ABC (ChABC), knockdown of CSPG polymerization enzymes by RNA interference (RNAi), and peptide blocking the signaling of 2 major receptors of CSPGs: protein tyrosine phosphatase sigma (PTPσ) and leukocyte antigen receptor (LAR). 7 Although these strategies have shown positive effects on the growth of neurons in model systems, there are disadvantages that may limit translation to clinical use. Both ChABC and RNAi are exogenous macromolecules that must be delivered directly to the injury site, have short half-lives in vivo, and could induce immunological response due to repeated administration. 8 Potentiating regeneration of neurons without degrading the glial scar may be possible via the heparin-binding growth factor pleiotrophin (PTN). PTN has long half life9,10 and may overcome the disadvantage of repeated administration associated with the use of ChABC and RNAi. PTN is a developmentally regulated protein whose expression peaks (in rats) 3 to 4 weeks after birth. 11 Notably, PTN expression is low in adults but increases transiently after CNS injury. 11 PTN is associated with neuroplasticity including the maturation of new neurons and induction of neurite outgrowth. 12 In Parkinson’s disease, PTN has been shown to reduce nigrostriatal degeneration and improve functional recovery. 6 Notably, PTN has a sustained presence in the tissue when injected into the nervous system directly. In cell culture systems without the presence of inhibitory CSPGs, PTN can drive significant neurite growth in neurons.13,14 Identifying key receptors involved in PTN-induced neuroplasticity may therefore allow for systemic pharmacotherapy.
PTN has several putative cell surface receptors, including receptor protein tyrosine phosphatase ζ (RPTPζ/PTPRZ), syndecans, nucleolin, neuropilin-1, integrin αVβ3 and αMβ2, N-syndecan receptor, glypican 2, neuroglycan-C, and anaplastic lymphoma kinase (ALK).9,11 Moreover, PTN can integrate into the extracellular matrix, limiting CSPG interactions with growth-inhibitory receptor PTPσ for a sustained period. 9 Specifically, PTN can reverse aggrecan (a prominent CSPG) mediated inhibition in vitro by preventing the binding of aggrecan to PTPσ. 9 PTN also interacts with the extracellular component of PTPRζ,1,6 and blocking this interaction reduces neurite growth in the presence of PTN in a cell culture system without CSPGs. 13
This study investigates the potential role of ALK receptor activation by PTN to increase the growth of neurons in the presence of a CSPG matrix. ALK is expressed during early development in CNS, with low expression in the adult CNS15,16 ALK signaling is critical for the differentiation of neuronal progenitor cells to neurons and regulates their survival.12,17 By binding to CSPGs, PTN also reduces phosphatase activity of RPTPζ, which increases activity of ALK. 11 Moreover, in developing neurons PTN signals directly via ALK receptor binding12,18 and in SK-N-SH cells PTN promotes cell and neurite outgrowth. 19 Thus, the evidence suggests that the ALK receptor may facilitate neuroplasticity even in the presence of CSPGs. Here, we directly investigated in vitro the role of PTN signaling via ALK in driving neuron growth in the presence of inhibitory CSPGs. Our data reveals a dose-dependent effect of PTN on neurite outgrowth in the presence of CSPGs. Notably, selective pharmacological inhibition of the ALK receptor attenuated the growth-promoting effect of PTN.
Materials and Methods
Matrices preparation
Coverslips were coated with 100 µg mL−1 Poly-L-Lysine (Sigma-Aldrich, P5899, USA) for 2 hours. After 3 washes with water, the coverslips were coated with either a growth permissive matrix −10 µg mL−1 of laminin (Corning, 354232) or an inhibitory matrix—10 µg mL−1 laminin + 1.25 µg mL−1 of CSPGs (Sigma Aldrich, CC117) for 2 hours and washed 2 times with PBS.
Primary cortical neuronal culture
All animal protocols were conducted in accordance with the Canadian Council on Animal Care Guidelines and approved by the Animal Care and Use Committee: Health Sciences for the University of Alberta. Rat primary cortical neurons were isolated from 0 to 1 day old Sprague Dawley rat pups. The cortices were dissected and digested with TrypLE (Gibco, 12605-028) for 15 minutes at 37°C. The cells were dissociated from tissue by trituration in neurobasal A medium (Thermofisher, 1088802) containing B27 supplement (1:50 v/v) (Gibco, 17504-044), antibiotics and GlutaMAX (Gibco, 35050-061). The cell suspension was seeded at 20 000 cells / well on coverslips with different matrices and incubated at 37°C, 5% CO2 for 1 hour. Following incubation, media was replaced with media containing different concentrations of recombinant human PTN (rhPTN, R&D systems 252 - PL) or the drugs alectinib (Toronto Research Chemicals, C183665) or SC79 (Selleck Chemicals, S7863) and cells were incubated for 72 hours.
Microscopy, fluorescent immunostaining and image analysis
Cells were fixed after 72 hours of treatment with 5% formaldehyde solution for 15 minutes. and washed with PBS. The fixed samples were blocked with 10% normal horse serum in PBS containing 0.1% Tritton-X 100 and washed with PBS. Samples were stained overnight with microtubule-associated protein 2 (MAP 2) antibody (1:500, Sigma Aldrich M9942), ALK (1:500, abcam ab190934) rinse with PBS. The primary antibody MAP 2 was detected with the secondary antibody donkey anti-mouse Alexa 647 (1:500 abcam ab150107) and ALK was detected with donkey anti-rabbit Alexa 488 (1:500 abcam ab150073), nucleus was stained with Hoechst 33342 (1:1000, Invitrogen, 62249). The images were acquired using Leica DMI6000B fluorescent microscope with a Leica DFC365FX camera at an objective magnification of 20× lens with 0.7 numerical aperture. The images were analyzed using Simple Neurite Tracer (SNT) plugin in Fiji. 20 All the neurite outgrowth from soma of each neuron was traced and any neurite not originating from soma was considered as branched neurite. Total neurite outgrowth, branched neurite outgrowth and number of neurons possessing neurite outgrowth for neurons in each treatment condition were manually traced and counted using SNT plugin in Fiji. The data was then normalized to respective laminin control.
Statistics
Statistical analyses of data from fluorescent images were carried out using one-way ANOVA followed by Dunnett’s multiple comparisons test for significance between treatment groups. n represents a single independent experiment (ie, an independent culture preparation) with a minimum of 3 technical replicates. Each technical replicate represents a well in a 24-well culture plate. All statistical analyses were done using Graphpad Prism version 9.1.2.
Results
Pleiotrophin induces neurite outgrowth in the presence of CSPGs
CSPGs are known to inhibit the growth of neurons. To understand the effect of PTN on neurite outgrowth, cortical neurons were cultured on the growth permissive (laminin) matrix or the growth inhibitory (laminin + CSPGs) matrix and treated with different concentrations of PTN (5 ng mL−1-20 ng mL−1) for 72 hours. MAP2 immunofluorescence was used to quantify neurite outgrowth. Neurons showed extensive neurite outgrowth on the laminin matrix (Figure 1(1) to (3)) that was inhibited by CSPGs (Figure 1(4) to (6)). PTN restored neurite extension in a dose-dependent manner (Figure 1(7) to (9)). Neuronal morphology was analyzed using the Simple Neurite Tracker plugin for ImageJ 21 (Figure 1(10)). A dose-dependent effect of PTN on total neurite outgrowth was observed, with PTN counteracting the inhibitory effect of CSPGs at concentrations below 20 ng mL−1 and maximal effect at 15 ng mL−1 concentration (Figure 1(11)). PTN treatment increased the complexity of cortical neuron neurite growth by increasing branched neurite growth, with maximum branch length observed at 15 ng mL−1 PTN (Figure 1(12)), and increased the number of neurons with neurite outgrowth at 10 to 15 ng mL−1 PTN (Figure 1(13)). This data suggests that PTN at specific concentrations potentiates neurite extension from cortical neurons grown in CSPGs matrices.

All the images were acquired at 20× magnification. Neurons cultured on laminin (growth permissive matrix) and stained for (1) Hoechst, (2) MAP 2 (Microtubule associated protein 2) with Alexa fluor 647, (3) overlay. Neurons cultured on laminin + CSPGs (growth inhibitory matrix) and stained for (4) Hoechst, (5) MAP 2 with Alexa fluor 647 (6) overlay. Scale bar: 30 µm. Neurons cultured laminin + CSPGs (L + C, growth inhibitory matrix) and treated with 15 ng mL−1 pleiotrophin (PTN) and stained for (7) Hoechst (8) MAP 2 with alexa fluor 647 (9) overlay. Scale bar: 30 µm. (10) Neurite outgrowth traced using simple neurite tracer plugin (SNT) in Fiji, total neurite growth (highlighted in purple + green), branched neurite outgrowth (highlighted in green) and neurons with no neurite outgrowth (indicated by yellow arrow). (11) PTN induces neurite growth in the presence of CSPGs (ANOVA, F(4,10)=9.028, P = .0024). (12) PTN induced branched neurite growth in the presence of CSPGs (ANOVA, F(4,10)=3.740, P = .0413) (13) PTN increases number of neurons with neurite growth in the presence of CSPGs (ANOVA, F(4,10)=4.560, P = .0235). (11-13) Error bars represent standard error of mean (SEM), symbols *, ** represent P < .05 and .01, respectively, on Dunnett’s multiple comparisons against control. All data are based on 3 independent experiments with a minimum of three technical replicates.
Pleiotrophin signals via ALK receptor on neurons
PTN signals by inactivating the phosphatase activity of PTPRζ. 22 Currently, available pharmacological inhibitors of PTPRζ are quite limited. 6 Thus, identifying other potential receptors for PTN may identify new targets to drive neuron growth. ALK is a putative PTN receptor and is broadly expressed on cortical neurons in vitro (Figure 2(1) to (3)). Alectinib selectively inhibits phosphorylation of ALK and blocks activity of ALK.23-25 Treatment of primary neuronal cultures with alectinib was used to probe the involvement of the ALK receptor in PTN signaling. Alectinib inhibited neurite outgrowth from PTN-treated neurons cultured on a CSPGs matrix (Figure 2(4) to (15)), exhibiting a dose-dependent effect on neurite outgrowth (Figure 2(16)), and on neurite branching (Figure 2(17)). Alectinib also reduced the number of neurons possessing neurite outgrowth in the presence of PTN (Figure 2(18)), suggesting that PTN signaling via ALK receptors is key to its growth-inducing effects in the presence of CSPGs.
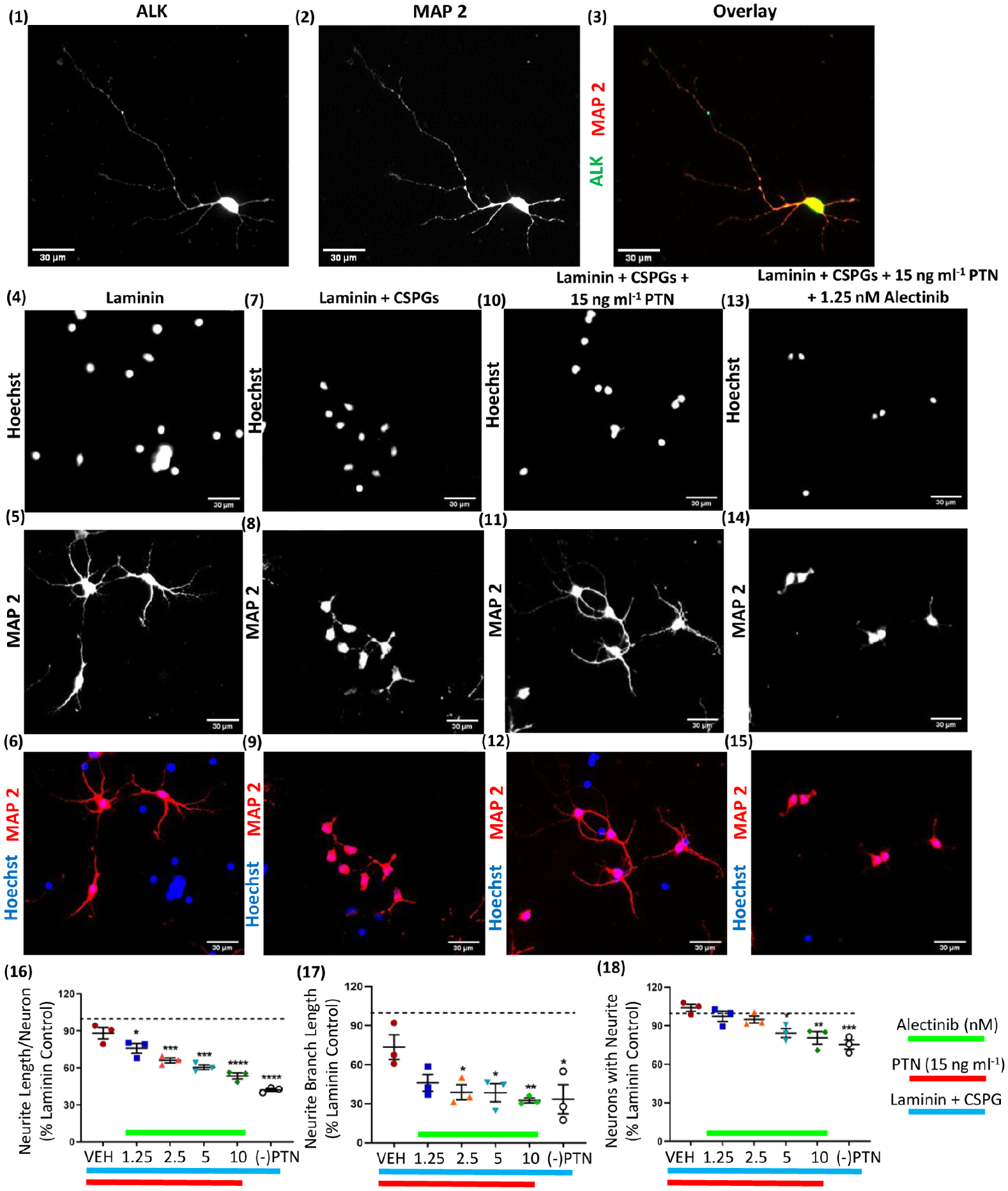
All the images were acquired at 20× magnification. Expression of ALK in cortical neuronal culture and stained for (1) ALK with Alexa fluor 488 (2) MAP 2 with Alexa fluor 647 and (3) overlay. Scale bar: 30 µm. Based on PTN dose-response data from Figure 1 (11, 12, 13) 15 ng mL−1 of PTN concentration showed maximum growth response therefore 15 ng mL−1 PTN was used for alectinib experiments. Cortical neurons cultured on laminin matrix (4,5,6). Cortical neurons cultured on Laminin + CSPGs matrix (7,8,9). Cortical neurons cultured on Laminin + CSPGs matrix and treated with 15 ng mL−1 PTN (10,11,12). Cortical neurons cultured on Laminin + CSPGs matrix and treated with 15 ng mL−1 PTN with 1.25 nM alectinib (13,14,15). After 72 hours incubation neurons were stained for Hoechst (4,7,10,13) and MAP2 (5,8,11,14) with Alexa fluor 647. Scale bar: 30 µm. (16) Neurite growth induced by PTN in the presence of CSPGs is blocked by alectinib (ANOVA, F(5,12)=30.48, P < .0001). (17) PTN-induced branched neurite growth in the presence of CSPGs is blocked by alectinib (ANOVA, F(5,12)=4.078, P = .0214). (18) Alectinib reduced the number of neurons with neurite growth in the presence of PTN in the presence of CSPGs (ANOVA, F(5,12)=9.068, P = .0009). (16-18) Error bars represent standard error of mean (SEM), symbols *, ** and ***/**** represent P < .05, .01, and .001 respectively. All data are based on 3 independent experiments with a minimum of three technical replicates.
Activating the AKT pathway drives neurite growth
The protein kinase B (AKT) pathway is a key downstream transducer of PTN/ALK signaling. 12 To investigate the involvement of this pathway in the regeneration of neurons, cortical neurons cultured on a CSPG matrix were treated with the AKT-activating compound SC79 in the absence of PTN. 12 SC79-treated neurons showed a dose-dependent enhanced neurite outgrowth even in the presence of CSPGs, with 5 µm SC79 inducing growth approaching PTN-induced growth on CSPG matrices (Figure 3).

All the images were acquired at 20× magnification. Cortical neurons cultured on (1,2,3) Laminin matrix, (4,5,6) Laminin + CSPGs (L + C) matrix, (7,8,9) Laminin + CSPGs matrix and treated with 15 ng mL−1 PTN, (10,11,12) Laminin + CSPGs matrix and treated with different concentration of SC79 (AKT activator). After 72 hours incubation neurons were stained for stained MAP2 with Alexa fluor 647 (2,5,8,11) and Hoechst (1,4,7,10). Scale bar: 30 µm. (13) SC79 induced neurite growth in the presence of CSPGs (ANOVA, F(4,10) = 8.222, P = .0033). (14) SC79 increases branched neurite growth of neurons in the presence of CSPGs (ANOVA, F(4,10) = 4.441, P = .0254). (15) SC79 increases the number of neurons with neurite growth in the presence of CSPGs (ANOVA, F(4,10) = 3.699, P = .0425). (13-15) Error bars represent the standard error of mean (SEM), symbols * and ** represent P < .05 and .01, respectively, on Dunnett’s multiple comparisons against vehicle control. All data are based on 3 independent experiments with a minimum of 3 technical replicates.
Discussion
This study examined the effect of PTN on the growth of cultured cortical neurons in the presence of CSPGs. Neurite outgrowth of primary cortical neurons in the presence of CSPGs was enhanced by PTN treatment in a dose-dependent manner, with PTN counteracting the inhibitory effect of CSPGs at concentrations below 20 ng mL−1. These findings support PTN as a candidate to restore neurite extension in CSPG-rich lesion areas after CNS injury and other neurological conditions. This is consistent with cell growth induced by PTN in studies of neurodegenerative disease. PTN has been shown to promote cell survival signal in dopaminergic neurons in vitro and in a mouse model of Parkinson’s disease PTN was shown to promote survival of grafted dopaminergic neurons, thus improving functional recovery of the nigrostriatal pathway6,26
The available literature indicates the involvement of ALK in neuron-like differentiation of PC12 cells 27 and depletion of ALK receptors attenuates neuronal proliferation and neurogenesis. 28 Here, attenuation of neurite growth due to alectinib treatment provides direct evidence for the necessity of ALK activity in neurite extension, identifying a further avenue for investigation for the development of ALK agonists to drive neuroplasticity.
Activating AKT signaling with SC79 induced more moderate neuronal growth relative to PTN treatment in the same set of cultures, with 15 ng mL−1 PTN showing 94.0 % as compared to 5 µM SC79 with 78.4 % increased neurite outgrowth compared to laminin control. The reason for the reduced effectiveness of SC79 relative to PTN incubation could reflect other potential downstream activators of ALK signaling. ALK is known to activate many pathways including phospholipase C γ, Janus kinase (JAK), PI3K-AKT, mTOR and MAPK signaling cascades.22,27 Thus, PTN may activate multiple growth associated pathways via ALK, and blocking ALK activity with alectinib inhibits multiple pathways beyond AKT that may contribute to the significant reduction in neurite growth even at the lowest tested concentrations (Figure 2(8))
Notably, PTN may also modulate the activity of glial cells including OPCs (oligodendrocyte progenitor cells) and microglia. PTN induces differentiation of OPCs to mature oligodendrocytes, thus promoting myelination of developing neurons, 22 and could therefore potentiate remyelination after injury. Microglia increase their release of neurotrophic factors including ciliary neurotrophic factor (CNTF), nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF) after stimulation with PTN. 29 Thus, while the data presented here demonstrate direct actions on neurons via ALK, PTN is a strong candidate to generate an environment favoring neuronal growth and its functionality by actions on multiple cell types. These data suggest PTN signaling may be an exciting approach to enhancing neuroplasticity after CNS injury, though the dose-response relationship for PTN in the presence of CSPGs will be important to verify in vivo. PTN/ALK signaling may therefore be a potential target to induce neuron regeneration after CNS injury or degenerative diseases associated with CSPG upregulation, including Alzheimer’s Disease, Parkinson’s Disease, and multiple sclerosis.
Footnotes
Author Contributions
SJG conceived the study with IRW, designed and analyzed experiments, and wrote the manuscript. MAC, KGT, and IRW assisted with analysis and interpretation of data and co-wrote the manuscript.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Canadian Institutes of Health Research (IRW, CIHR PS 166144), the Natural Sciences and Engineering Research Council (IRW, NSERC RGPIN-2017-05380), and the George Davey Endowment for Brain Research (KGT).
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
