Abstract
Recent evidence documented a protective effect of Class II human leukocyte antigen (HLA) DRB1*13 on brain health across the lifespan including evidence of reduced neural network variability relative to non-carriers. Here, in an extension of those findings, we evaluated the influence of a large number of Class I and Class II HLA alleles on aging-related changes in neural network variability. Cognitively healthy women (N = 178) ranging in age from 28 to 99 years old underwent a magnetoencephalography scan from which neural network variability was calculated and provided a blood sample from which HLA and apolipoprotein E (ApoE) genotype were determined. The primary analyses assessed the dependence of network variability on age in carriers of a specific HLA allele compared to non-carriers. Effects were considered protective if there was a significant increase of network variability with age in the absence of a given HLA allele but not in its presence, and were considered to confer susceptibility if the converse was documented; HLA alleles that did not influence the dependence of network variability on age in their presence or absence were considered neutral. Of 50 alleles investigated, 22 were found to be protective, 7 were found to confer susceptibility, and 21 were neutral. The frequencies of those 50 alleles were not associated significantly with ApoE genotype. The findings, which document the influence of HLA on age-related brain changes and highlight the role of HLA in healthy brain function, are discussed in terms of the role of HLA in the human immune response to foreign antigens.
Introduction
Human leukocyte antigen (HLA)
HLA genes are involved in the human immune response to foreign antigens and are widely implicated in human health and disease 1 with mounting evidence documenting the influence of HLA on brain health. For example, genome wide association studies2 -6 and epidemiological studies7,8 have identified HLA alleles that confer susceptibility or protection against age-related brain diseases, including dementia and Parkinson’s disease. Aging-related brain changes are detectable long before disease onset9,10; thus, identification of brain anomalies in cognitively healthy individuals may permit identification of individuals at risk of cognitive decline. Previous studies have documented that HLA DRB1 genes influence age-related changes in brain structure, 11 function, 12 and cognitive performance13,14 in non-demented individuals, even in the presence of apolipoprotein E4, an allele that is associated with cognitive decline and dementia.,15,16 Those findings suggest that HLA is an important genetic contributor to brain health.
HLA genes play a central role in host health via elimination of non-self (foreign) antigens. 17 The HLA region on chromosome 6 is the most highly polymorphic region of the human genome. 18 Given the role of HLA in protection against foreign antigens, the extremely polymorphic nature of HLA maximizes protection against diverse pathogens that may otherwise contribute to brain deterioration. A given individual carries a total of 12 alleles of classical HLA genes, namely 2 alleles from each Class I gene (A, B, C) and 2 from each Class II gene (DPB1, DQB1, DRB1). These alleles code for cell-surface proteins that are instrumental in the adaptive immune response. Molecules of the 2 classes differ in several respects including their structure, location, and mechanism of action, but they share the same goal—namely, antigen elimination. The success of antigen elimination depends, in part, on the ability of the HLA molecule to bind antigens for presentation to T cells, which is, in turn, determined by the amino acid composition of the peptide binding groove of the HLA molecule where nearly all of the polymorphisms are located. Even single amino acid residue differences alter the binding structure, thereby influencing antigen elimination and, consequently, disease associations. 19 In a number of studies, we have documented the influence of HLA on several conditions affecting the brain and on brain health.7,8,11,12,20 -25
Neural communication
Efficient communication across a highly interconnected neural network is a hallmark of healthy brain functioning. Magnetoencephalography (MEG) provides a high fidelity measure of integrated synaptic activity that can be used to evaluate brain health since synchronous neural interactions (SNI) (zero-lag partial cross-correlations between MEG sensors) are virtually identical across healthy individuals 26 and deviate in characteristic ways for different brain-related conditions.25,27,28 With regard to aging, previous research has documented age-related deviations in SNI characterized by increased network variability that are remarkably absent in individuals carrying HLA-DRB1*13, 12 which confers protection against several immune-related conditions.29,30 Here we extend those findings to evaluate the influence of a large number of HLA alleles on aging-related changes in SNI.
Materials and Methods
Participants
A total of 178 cognitively healthy women (60.3 ± 15.8 years, mean age ± SD; age range 28-99 years) participated in this study focused on women’s healthy brain aging as paid volunteers after providing informed consent, in adherence to the Declaration of Helsinki. Their cognitive status was assessed using the Montreal Cognitive Assessment (MoCA 31 ; N = 166) or Modified Mini-Mental State exam (3MS 32 ; N = 12). All participant’s cognitive scores exceeded the suggested cut-offs indicative of healthy cognitive functioning. Written informed consent was obtained from study participants. All study protocols were approved by the appropriate Institutional Review Boards.
HLA genotyping
DNA isolation was carried out from 3 ml of whole blood drawn in EDTA tubes, using a commercially available kit (ArchivePure cat. 2300730) from 5Prime (distributed by Fisher Scientific or VWR) with an expected yield of 50 to 150 µg of DNA. The purified DNA samples were sent to Histogenetics (http://www.histogenetics.com/) for high-resolution HLA Sequence-based Typing (SBT; details are given in https://bioinformatics.bethematchclinical.org/HLA-Resources/HLA-Typing/High-Resolution-Typing-Procedures/ and https://bioinformatics.bethematchclinical.org/WorkArea/DownloadAsset.aspx?id=6482). Their sequencing DNA templates are produced by locus- and group-specific amplifications that include exon 2 and 3 for class I (A, B, C) and exon 2 for class II (DRB1, DRB3/4/5, DQB1, and DPB1) and reported as Antigen Recognition Site (ARS) alleles as per ASHI recommendation. 33
ApoE genotyping
DNA samples were genotyped using PCR amplification followed by restriction enzyme digestion. 34 Each amplification reaction contained PCR buffer with 15 mmol/L MgCl2 ng amounts of genomic DNA, 20 pmol ApoE forward (5N TAA GCT TGG CAC GGC TGT CCA AGG A 3N) and reverse (5N ATA AAT ATA AAA TAT AAA TAA CAG AAT TCG CCC CGG CCT GGT ACA C 3N) primers, 1.25 mmol/L of each deoxynucleotide triphosphate, 10% dimethylsulfoxide, and 0.25 μL Amplitaq DNA polymerase. Reaction conditions in a thermocycler included an initial denaturing period of 3 minutes at 95°C, 1 minute at 60°C, and 2 minutes at 72°C; followed by 32 cycles of 1 minute at 95°C, 1 minute at 60°C, and 2 minutes at 72°C; and a final extension of 1 minute at 95°C, 1 minute at 60°C, and 3 minutes at 72°C. PCR products were digested with HhaI and separated on a 4% Agarose gel which was stained with Ethidium Bromide. Known ApoE isoform standards were included in the analysis.
MEG data acquisition
All participants underwent a MEG scan. As described previously,27,28 subjects lay supine within the electromagnetically shielded chamber and fixated their eyes on a spot ~ 65 cm in front of them, for 60 seconds. MEG data were acquired using a 248-channel axial gradiometer system (Magnes 3600WH, 4-D Neuroimaging, San Diego, CA), band-filtered between 0.1 and 400 Hz, and sampled at 1017.25 Hz. Data with artifacts (eg, from non-removable metal or excessive subject motion) were eliminated from further analysis.
Data analysis
General
Standard statistical methods were used to analyze the data, including Pearson correlation for assessing pairwise associations and Wilson’s score for assessing one-sample proportions. The following packages were employed: IBM-SPSS statistical package (version 27) and ad hoc FORTRAN computer programs employing the International Mathematics and Statistics Library (IMSL; Rogue Wave Software, Louisville, CO, USA) statistical and mathematical libraries. Prewhitening of the raw MEG series (see below) was performed using programs in Python. 35
MEG data preprocessing
Single trial MEG time series from all sensors (N = 60 000 time samples per series) underwent “prewhitening” 36 using a (50,1,3) ARIMA model 35 to obtain practically white noise innovations (ie, residuals). Given 248 MEG sensors, there were 30 628 pairs possible. All zero-lag pairwise crosscorrelations, r, were computed for valid data (see above) and were analyzed further.
Statistical analyses
Since we were interested in the strength of neural interactions irrespective of its sign, we took the absolute value of
For each participant, the standard deviation of
The possible associations between the frequency distributions of ApoE genotypes and specific HLA alleles were assessed using the chi-square test on a 6 rows × 2 columns table, where the rows contained the frequency (counts) of the 6 ApoE genotypes (ε2/ε2, ε2/ε3, ε2/ε4, ε3/ε3, ε3/ε4, ε4/ε4) occurring in the sample tested and the columns contained the frequency (counts) of the absence/presence of a specific HLA allele.
The IBM-SPSS statistical package (version 27) and ad hoc FORTRAN computer programs employing the International Mathematics and Statistics Library (IMSL; Louisville, CO: Rogue Wave Software) were used for analyses. All P-values reported are two-sided.
Results
HLA alleles
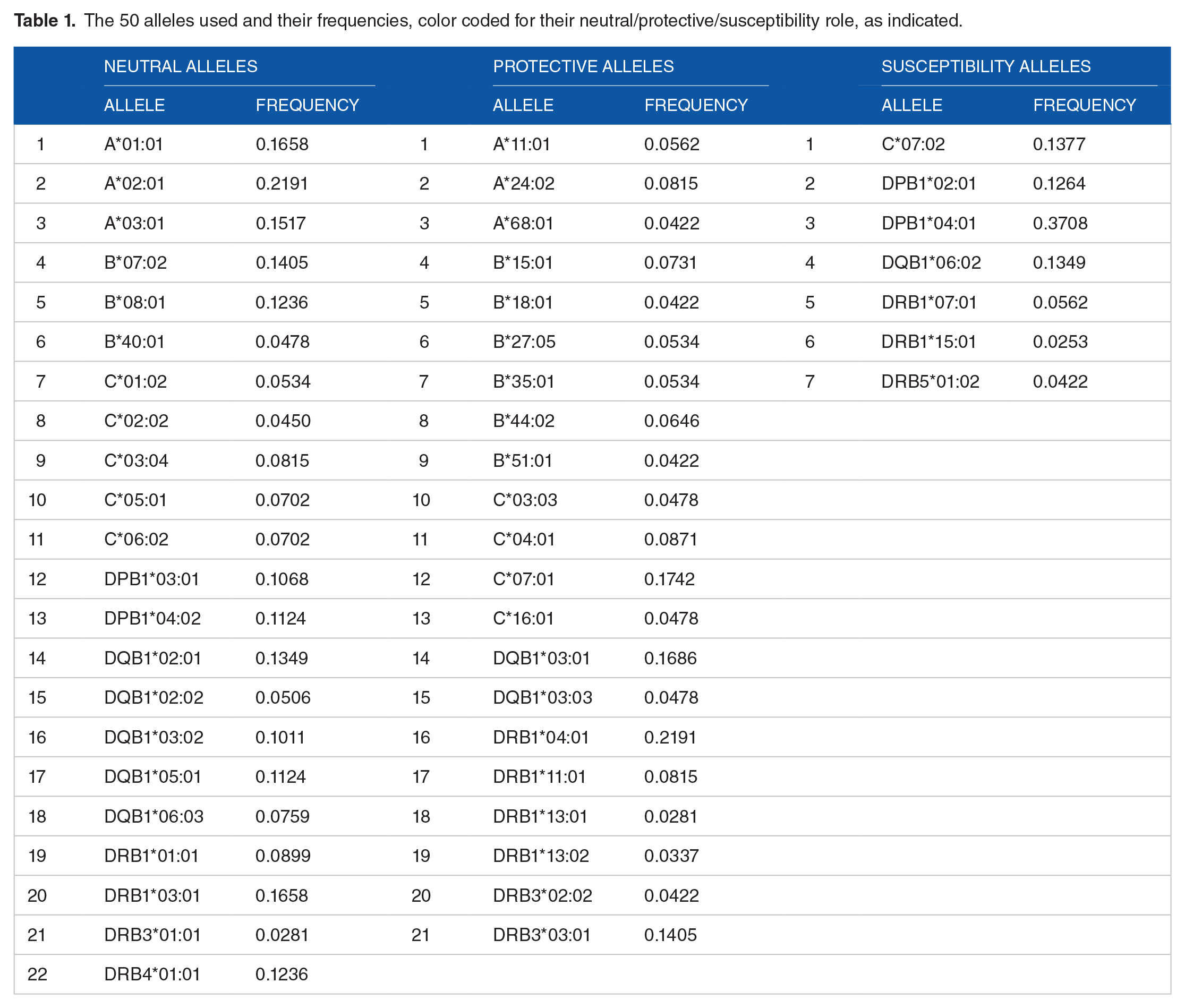
There was a total of 195 distinct alleles in our sample of 178 women. Of those, 50 occurred in at least 15 participants to qualify entering into the regression analysis of
The 50 alleles used and their frequencies, color coded for their neutral/protective/susceptibility role, as indicated.
Effect of HLA alleles on the relation of
versus age
There was a highly significant, positive overall dependence of

The variability of synchronous neural interactions is plotted against age. r = .3, P < .001, N = 178 participants.

An example of a neutral allele. Left panel, HLA A*03:01 absent; r = .230, P = .008, N = 133. Right panel, HLA A*03:01 present; r = .404, P = .006, N = 45. In 62 of 100 bootstrap test runs with N = 45 and A*03:01 absent, there was a statistically significant positive correlation between

An example of a protective allele. Left panel, HLA B*18:01 absent; r = .350, P < .001, N = 143. Right panel, HLA B*18:01 present; r = −.061, P = .830, N = 15. In only 23 of 100 bootstrap test runs with N = 15 and B*18:01 absent, there was a statistically significant positive correlation between

An example of a susceptibility allele. Left panel, HLA DPB1*04:01 absent; r = .251, P = .092, N = 46. Right panel, HLA DPB1*04:01 present; r = .324, P < .001, N = 132. In only 6 of 100 bootstrap test runs with N = 46 and DPB1*04:01 absent, there was a statistically significant positive correlation between
ApoE genotypes
The overall frequency distribution of the 5 ApoE genotypes in the sample of 178 women studied is shown in Table 2.
Frequency distribution of the 6 ApoE genotypes.

Association of protective and susceptibility alleles with the apoE genotype
No statistically significant associations were found (chi-square test).
Discussion
Here we evaluated the influence of HLA alleles on the increase of neural network variability with age in cognitively healthy women. We found that age-related changes in network variability depend on the presence or absence of particular HLA alleles. Furthermore, consistent with our previous findings, 12 the effects of HLA were independent of apoE genotype. We discuss these findings below in more detail and consider the implications for healthy brain aging in light of the role of HLA in antigen elimination.
Healthy brain function depends on efficient communication, a phenomenon that is remarkably consistent across brains of healthy individuals. 26 On the other hand, neural network variability impedes neural network efficiency. Network variability increases with age overall suggesting age-related deterioration of network efficiency. 12 The impact of these disruptions of network efficiency on cognitive functioning remains to be investigated. The women in the present study were determined to be cognitively healthy as determined by cognitive screening measures at the time of MEG scan acquisition. It is possible that network variability may underlie subtle cognitive changes that are not captured in brief cognitive screening measures. Alternatively, we suspect that age-related alterations in network variability may be harbingers of cognitive changes, a supposition that is currently under investigation.
Despite an overall effect of age on network variability, previous findings indicated that the effect is influenced by HLA. 12 More specifically, we documented previously protective effects of HLA-DRB1*13:01 and DRB1*13:02 on network variability even among ApoE4 carriers 12 ; here, we extended those findings, evaluating the influence of 50 HLA alleles. Nearly one-half of the HLA alleles investigated here were protective against age-dependent increase in network variability, as evidenced by the presence of age-related effects on neural network variability in the absence but not in the presence of those alleles. Protective HLA effects were observed for both Class I (13/21) and Class II (8/21) alleles. This is noteworthy in light of important differences between HLA Class I and Class II molecules. HLA Class I molecules are expressed in nucleated cells, bind with and export small endogenous peptides (8-10 amino acid residues) to the cell surface for presentation to cytotoxic CD8 T-cells, signaling cell destruction. On the other hand, HLA Class II molecules are expressed in professional antigen presenting cells, bind with an export larger endocytosed exogenous peptides (15-22 amino acid residues) to CD4+ T-cells, stimulating production of B-cell mediated antibodies and facilitating immunological memory. The present findings suggest that both of these mechanisms influence age-related changes in brain functioning, a finding that is perhaps to be expected since both classes are involved in immune surveillance and work together to promote host protection via elimination of foreign antigens.
Seven of the alleles were found to promote susceptibility to network variability as indicated by a significant dependence of network variability on age in their presence but no significant effect in in the absence of the allele. Notably, all but one of the susceptibility alleles were in Class II, which has been strongly implicated in autoimmune conditions. 38 The present findings are consistent with a role of autoimmunity and inflammation in aging-related changes in brain function that may ultimately result in dementia. 39 Of the susceptibility alleles, DPB1*04:01 was the most frequent, occurring in over one-third of study participants. DPB1*04:01 is protective against several conditions including hepatitis B, 40 celiac disease, 41 and narcolepsy, 42 but has notably been associated with vasculitis 43 which can result in irreversible brain damage.44,45
Taken together, this study documented the influence of HLA on age-related changes in brain functioning that is independent of ApoE. The primary role of HLA involves the immune response to and elimination of foreign antigens which is partially determined by allelic variation in the binding groove. For example, we have documented variability in HLA binding affinity to human herpes viruses 46 which are nearly ubiquitous and have been widely implicated in brain diseases.47 -49 In the absence of high-affinity binding, human herpes virus and other non-self antigens may persist resulting in chronic low-grade inflammation and/or autoimmunity and, ultimately, brain effects. 50 In that light, the present findings suggest that alleles shown to protect against age-related changes in neural network variability may have superior ability to bind to and eliminate foreign antigens whereas susceptibility alleles may confer risk through inability to bind and eliminate foreign antigens, thereby contributing to pernicious brain effects. We have previously opined that persistent antigens result in a neuroimmune cascade in which the influence of apoE is secondary to HLA. 51 ApoE is known to be synthesized in response to neuronal damage to facilitate repair. 52 We have speculated that in the absence of protective HLA, persistent antigens contribute to neuronal damage and inflammation thereby stimulating ApoE synthesis, the effects of which are determined by an individual’s ApoE genotype; however, that cascade is presumably prevented in the presence of HLA that binds with and facilitates elimination of foreign antigens. 51 The present findings indicating that HLA effects are independent of ApoE are consistent with a pivotal early role of HLA in maintaining brain health subsequent to antigen exposure.
This study adds to the literature documenting moderating influences of HLA on age-related brain changes and highlights the role of HLA in healthy brain function. The use of high-resolution HLA genotyping in a relatively large sample of cognitively healthy women bolsters the findings; nonetheless, they must also be considered within the context of several qualifications. First, we investigated the effects of a relatively large number of HLA alleles; however, since HLA is the most highly polymorphic region of the human genome the effects of many HLA alleles remain to be determined. Second, we evaluated the effect of single alleles on age-related network variability, yet each individual possesses 12 alleles (6 from each class) that may differ in terms of their influence on network variability. It is unclear how an individual’s 12 allele HLA composition may influence age-related brain function. Third, while we are confident the women participants were non-demented, the screening instruments used to determine cognitive status may miss subtle cognitive alterations that could be indicative of age-related changes in brain function. Finally, this sample was comprised of cognitively healthy women of primarily European ancestry. As such, it remains to be determined to what extent the present findings may extend to other populations, since HLA varies by geography and ethnicity,53,54 and to men. Finally, protective alleles may be overrepresented in this sample of cognitively healthy women compared to the population overall.
Footnotes
Acknowledgements
We would like to thank Laura Kalipe and Stacy Dolan for their contributions to data collection.
Author Contributions
APG: conceived the research; AFL and LMJ contributed to data acquisition; LMJ and APG wrote the paper; all authors read, edited, and approved the final version of the paper.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Partial funding for this study was provided by the University of Minnesota (the Anita Kunin Chair in Women’s Healthy Brain Aging, the Brain and Genomics Fund, the McKnight Presidential Chair of Cognitive Neuroscience, and the American Legion Brain Sciences Chair) and the U.S. Department of Veterans Affairs. The sponsors had no role in the current study design, analysis or interpretation, or in the writing of this paper. The contents do not represent the views of the U.S. Department of Veterans Affairs or the United States Government.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Data will be available to qualified researchers by the corresponding author on a reasonable request.
Significance
Human leukocyte antigen (HLA) alleles influence age-related neural network variability. HLA is an important genetic contributor to brain health.

