Abstract
In the wild, animals face different challenges including multiple events of food scarcity. How they overcome these conditions is essential for survival. Thus, adaptation mechanisms evolved to allow the development and survival of an organism during nutrient restriction periods. Given the high energy demand of the nervous system, the molecular mechanisms of adaptation to malnutrition are of great relevance to fuel the brain. The blood-brain barrier (BBB) is the interface between the central nervous system (CNS) and the circulatory system. The BBB mediates the transport of macromolecules in and out of the CNS, and therefore, it can buffer changes in nutrient availability. In this review, we collect the current evidence using the fruit fly, Drosophila melanogaster, as a model of the role of the BBB in the adaptation to starvation. We discuss the role of the Drosophila BBB during nutrient deprivation as a potential sensor for circulating nutrients, and transient nutrient storage as a regulator of the CNS neurogenic niche.
Introduction
Undoubtedly, one key trait of life is the ability to constantly adapt to environmental changes. Animals are permanently challenged by their surroundings, including predators, climate, and food availability. All these external cues are accompanied by “survival responses” that can adjust animal behavior, and may also trigger metabolic changes. There is a vast consensus that the nutritional status of an animal influences several aspects of its development and physiology. In the wild, animals grow and develop with constant changes in the food supply, and therefore, a proper adaptation to different nutritional contents and caloric intakes is essential for survival. Upon food scarcity, the animal response varies in different organs. For instance, while the liver and adipose tissue mobilize nutrients during caloric restriction, the nervous system’s need for high energy demand should be maintained. However, the nervous system is indeed affected by the new nutritional condition, and different molecular mechanisms are in place that mediates its adaptation.
Unlike most organs, the mammalian nervous system is strictly separated from the circulating blood. A simplistic view of a barrier would be a physical fence that isolates a tissue, however, barriers may also work as organizing centers and, therefore, they could actively sense the environment, regulate transport, and perhaps function as nutritional buffering units. This seems to be the case for the blood-brain barrier (BBB). The BBB selectively regulates the transport of molecules into and out of the CNS working as “the gatekeeper.” The isolation provided by this barrier allows for maintaining a strict metabolic and ion homeostasis of the nervous system that is fundamental for synaptic function and information processing. 1 From an evolutionary perspective, animals that developed complex behaviors and advanced cognitive functions require precise regulation of the neuronal environment, including ion homeostasis, that could work as a selective pressure for the emergence of the BBB.2,3 Although the evolutive origin of the BBB is not fully agreed upon, it is proposed that the BBB has independently evolved multiple times during evolution, however, its function is highly conserved between vertebrates and invertebrates.4 -6
Given the isolation from the circulation that the BBB confers to the CNS, changes in the nutritional intake must be firstly sensed by the BBB. Whether the BBB can influence or trigger an adaptive response that permits the proper function of the CNS, is not completely clear. In this review, we discuss the current understanding of the molecular mechanisms by which the BBB responds to malnutrition, focusing on its role as a nutritional protector of the brain and its function as part of the neurogenic niche during the development of Drosophila melanogaster. We also address the potential function of the insect BBB as a nutritional sensor and energy storage for the CNS, and finally, we propose the future perspectives that the fruit fly has to offer for the understanding of the BBB role during adaptation to malnutrition.
The Blood-Brain Barrier Function is Highly Conserved From Flies to Mammals
In the mammalian central nervous system (CNS), the BBB is formed by a selectively permeable endothelium of the blood vessels. Endothelial cells, together with pericytes, basement membrane, glial cells (astrocytes), and neurons, are part of a functional structure known as the Neurovascular Unit 7 (Figure 1A). Pericytes maintain the integrity of the BBB, while astrocytic end-feet contribute to the modulation of its function. 1 Endothelial cells prevent paracellular diffusion of molecules by the formation of tight junction adhesions, allowing the active control of metabolites transport. Loss of the BBB integrity results in vascular leakage, affecting the chemical composition of the interstitial fluid and allowing plasma and blood cell infiltration causing inflammation that is detrimental to neuronal function. In addition to controlling molecule transport across the CNS, the neurovascular unit also works as a neurogenic niche in the adult brain, controlling neural stem cell (NSC) self-renewal, proliferation, migration, and differentiation.8,9

The human and Drosophila blood-brain barrier. (A) The neurovascular unit in mammals is composed of endothelial cells (EC), pericytes (P), astrocyte end-feet (AEF), neurons, and basement membrane (BM). Tight junctions (TJs) seal the microvascular endothelium in the brain to prevent paracellular diffusion of blood cells and macromolecules. (B) The Drosophila blood-brain barrier is formed by 2 types of glial cells: supberineurial and perineurial glia (SPG and PG respectively). Subperineurial glia form septate junctions (SJs) to block paracellular diffusion, while perineurial glia sits below a dense extracellular matrix called neural lamella (NL). Inside the brain, cortex glia (CG) encase the cell body of neurons (N).
The comprehension of the function of the CNS has benefited from studies in model organisms, including invertebrates such as the fruit fly. 70% of the genes responsible for human diseases have homologs in Drosophila melanogaster, which offers unparalleled powerful genetic tools for gain and loss of function experiments.10,11 Insects have an open circulatory system in which the hemolymph, the insect blood, baths all organs and tissues. Hence, the BBB engulfs the entire nervous system, isolating it from the circulating hemolymph. Similar to mammals, the Drosophila BBB restricts paracellular diffusion, controlling molecule transport and maintaining ion balance of the CNS12,13; and therefore being the functional analog structure to the mammalian neurovascular unit. Interestingly, as in primitive vertebrates, the fly BBB is formed exclusively by glial cells structured in 2 consecutive layers, the subperineurium and perineurium, and a dense extracellular matrix (neural lamella) that faces the hemolymph12 -15 (Figure 1B).
In Drosophila, subperineurial glial cells (SPG) form the layer in direct contact with the nervous tissue, generating a secondary epithelial monolayer barrier encapsulating the nervous system. Subperineurial glial cells are large, thin, and flat, preventing paracellular diffusion by the formation of specific occluding cell-cell adhesions known as septate junctions. During development, the subperineurial barrier is fully formed at the end of embryogenesis, 16 and cells grow by endoreplication in coordination with larval brain development. 17 Furthermore, subperineurial glia is coupled to each other by gap junctions, which synchronize calcium waves across the subperineurial layer, and are essential for neurogenesis during early larval development.18,19 Subperineurial glia regulates the influx of metabolites via many specifically localized transporter proteins.20 -24 Transcriptomic analysis of the BBB glial cells (generically referred to as surface glia) showed the expression of different nutrient transporter families, including highly conserved solute carrier transporters (SLC) for carbohydrate (SLC2), monocarboxylate (SLC16), neutral amino acids (SLC1), monoamine (SLC18) and organic anions (SLC22); and LDL-family lipoprotein receptors. 22 In addition to nutrient transporters, the efflux of xenobiotic molecules, including drugs and insecticides, is carried out by the action of ATP-binding cassette (ABC) transporters that can expel lipophilic molecules to the hemolymph25 -27
Sitting above the subperineurial layer, perineurial glial cells (PG) build the outermost cell layer contacting the neural lamella and the hemolymph. Although perineurial glial cells do not form a tight barrier, they participate in the metabolic regulation of the Drosophila BBB. Similar to subperineurial glia, the perineurial layer also expresses several nutrient transporters.21,22,24,28 It has been proposed that perineurial glial cells form gap junctions between them and with the subperineurial layer, coupling ion homeostasis across the BBB, and regulating the animal circadian rhythm with the BBB efflux activity.29 -31 Additionally, perineurial glial cells secrete extracellular matrix components22,32,33 and metalloproteases34,35 are required for remodeling the neural lamella during CNS development. In this manner, the neural lamella provides structural support for the CNS, maintaining its shape and stiffness by the action of focal adhesion with the perineurial layer.34,36 -38 In addition to this structural support, extracellular matrix components secreted by the perineurial glia promote neurogenesis, forming part of the neural stem cell niche.33,39
The Drosophila Blood-Brain Barrier Acts as the Nutritional Protector of the Brain
Drosophila melanogaster, as a holometabolous insect, transitions across 4 main developmental stages: embryo, larva, pupa, and imago (Figure 2A). The embryonic development is highly stereotyped, regulated by maternally deposited mRNAs in the oocyte, which can be influenced by the nutritional condition of the mother. 40 The embryonic CNS is formed from a relatively constant number of neural stem cells, named neuroblasts, which divide asymmetrically to self-renew and differentiate into neurons and glial cells.41,42 At the end of neurogenesis, neuroblasts shrink and enter into quiescence, ending the primary neurogenesis.43 -45 After hatching, larvae resume tissue growth only in response to an amino acid-rich diet, 46 which initiates a secondary wave of neurogenesis in the CNS. Importantly, the BBB receives information about the nutritional status of the animal from the fat body and then signals to the neuroblasts to re-activate their proliferation. This signaling mechanism requires Insulin-like peptides (dIlps, mainly dIlp6) that are released by the subperineurial glial cells47,48 and involve the coordinated propagation of calcium waves 18 by gap junctions. dIlps in turn activate the highly conserved PI3K/Akt and Tor pathways in neuroblasts, restarting the progression of the cell cycle47,48 (Figure 2B). Although the nutritional signal derived from the fat body is unknown, 46 and very likely composed of several secreted factors, the Drosophila BBB acts as a true NSC niche, linking the nutritional status of the animal with neurogenesis during larval development.
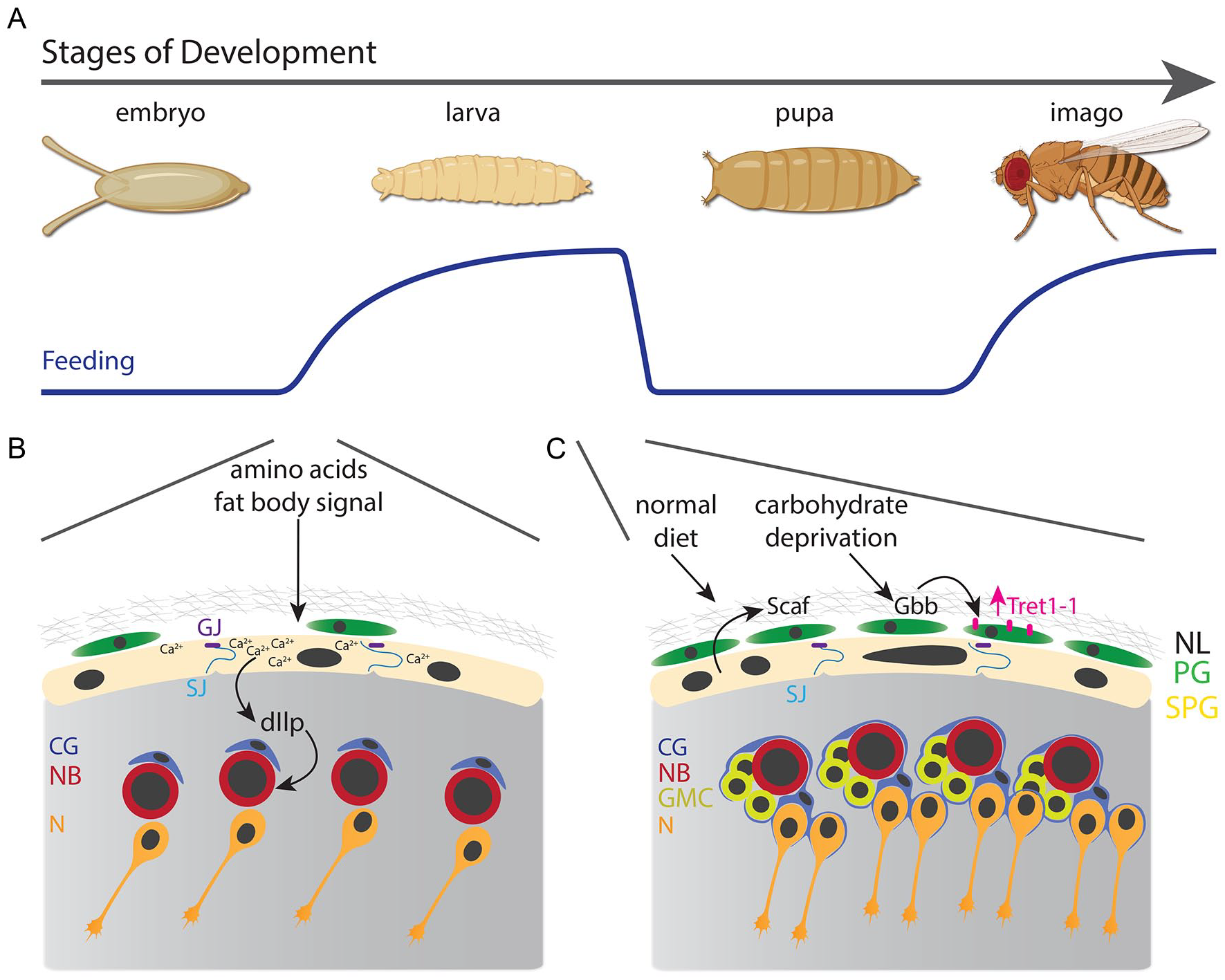
Nutritional effect on the Drosophila Blood-brain barrier. (A) The development of Drosophila melanogaster transitions across 4 stages: embryo, larva, pupa, and imago (adult). Larval and adult stages depend on animal nutrition. (B) During early larval development (first instar), diet amino acids induce fat body cells to secrete an unknown signal that triggers calcium waves in subperineurial glia that release insulin-like peptides, reactivating neuroblast proliferation. (C) At the third instar larval stage, a normal diet induces the expression of Scaf in subperineurial glia to balance the growth of the blood-brain barrier with neurogenesis; meanwhile, carbohydrate deprivation increases the levels of the TGFβ ligand Gbb that induces the expression of the carbohydrate transporter Tret1-1.
After restarting proliferation, the development of the CNS continues mostly independent of animal nutrition. Upon larval starvation, most tissues stop growing, 46 so development is arrested while the animal finds a new source of food. This famine period comes with a systemic response involving glycogen breakdown and fat mobilization to maintain nutrient levels in the hemolymph,49,50 however, changes in hemolymph protein content have also been described. 51 Despite the starvation period, larval neurogenesis is partially maintained in a phenomenon that resembles human intrauterine growth restriction, in which the brain is preferentially protected or spared from the lack of nutrients.46,52,53 Larval neuroblasts keep proliferating by the action of the Anaplastic lymphoma kinase receptor (Alk), activated by the glia-derived ligand Jelly Belly (Jeb), independently of the nutritional condition of the animal. 52
How the larval CNS responds to nutrient deprivation to maintain neurogenesis is not fully understood. Recently, we challenged this question by analyzing, in a cell-type specific manner, the binding of Polymerase II of third instar larvae after 24 hours of nutrient restriction, using the targeted DamID technique. 54 The most significant gene expression changes were found in glial cells of the BBB, while neuroblasts presented a decrease in some metabolism-related genes. 55 Although subperineurial glial endoreplication and perineurial glial proliferation are severely reduced by starvation, neuroblast growth and proliferation are unaffected, suggesting that the development of the BBB is not spared by nutrient scarcity as neurogenesis is. The expression of the serine protease homolog encoded by the gene scarface (scaf) is sensitive to starvation and its function is required for balancing the growth of the BBB with neuroblast proliferation 55 (Figure 2C). Although the molecular targets of Scaf are unknown in the BBB, they may be related to the modulation of one or more growth factors secreted from the BBB during development.
In a more sophisticated approach, the group of Simon Sprecher used single-cell RNA sequencing (scRNA-seq) to profile gene expression of larval brains under normal diet and starvation. 56 For this experiment, they used 12 hours after hatching larvae that were fully starved for 4 hours. This profile comprised all cell types of the larval brain, including glial cells of the BBB. These glial cells showed an increase in the expression of fatty acid metabolism genes, such as a carnitine O-palmitoyltransferase (withered, whd), the long-chain-fatty-acid-CoA ligase (pudgy, pdgy), and the Lipase 4 (Lip4); while the fatty acid synthase, 1 (FASN1) was downregulated after starvation. 56 Thus, it seems that during the first instar stage, glial cells, including the BBB, shift their metabolism to use lipids as a source of energy after a short period of starvation.
Similar to the role of the BBB as a neurogenic niche, the BBB also needs to adapt nutrient transport to food scarcity periods. Under full starvation, perineurial glia increases the levels of the carbohydrate transporter Tret1-1. 57 However, under a sucrose-only diet, Tret1-1 levels are similar to in a normal diet, suggesting that BBB carbohydrate transport is sensitive to larval glycemia. 57 Indeed, using a FRET-based glucose sensor, Hertenstein et al 57 showed that under starvation, glucose uptake in the BBB is increased compared to fed conditions, and this increase depends on the upregulation of Tret1-1. Thus, the larval BBB may promote neurogenesis, and therefore brain sparing, by enhancing carbohydrate uptake from the hemolymph in a starvation condition, while other tissues stop developing (Figure 2C).
This protective mechanism could also be extrapolated to other sources of energy. For instance, the monocarboxylate transporter Chaski (Chk) is enriched in glial cells, including subperineurial glial cells. chk mutant animals, although viable, are more sensitive to adult starvation than wild-type animals. 23 This sensitivity to starvation depends on the glial expression of chk, 23 however, it is not clear whether Chk at the BBB plays a role in survival during nutrient restriction periods. Supporting this notion is the fact that lactate transport in glial cells is reduced in chk mutant animals, 58 suggesting that monocarboxylate transporters may also be regulated in the BBB to adapt to starvation periods. On the other hand, amino acid transporters could follow a similar trend. The amino acid transporter SLC36/Pathetic (Path) is expressed in glial cells, including the BBB, 59 and also in neuroblasts. 60 Although path is necessary for neuroblasts for neurogenesis, it is only required in glial cells for brain growth during nutrient restriction. 60 It remains to be demonstrated whether Path maintains brain growth by enhancing amino acid transport through the BBB under nutrient deprivation. Nevertheless, all data point to a putative role of nutrient transporter at the BBB in the molecular strategy for malnutrition adaptation.
Energy Storage at the Drosophila Blood-Brain Barrier
Given that one of the main roles of the BBB is controlling the flux of nutrients across the CNS, it is sensible to speculate that glial cells of the BBB could store energy for a rapid response to nutrient deprivation. Metabolic coupling of neurons and glial cells has been shown for both mammals and flies.28,61 Therefore, subperineurial or perineurial glia could be playing an energy buffering role for the entire CNS in response to starvation. In animals, 2 major energy storage molecules are present: triglycerides and glycogen. During larval development, cortex glia, as well as perineurial and subperineurial glia store neutral lipids in the form of lipid droplets.62,63 Brain lipid droplets are reduced during pupal development and undetected in adult animals, 63 suggesting they provide energy or lipids during a stage of development that relies only on nutrient reservoirs. In fact, during larval starvation, lipid droplets increase in glial cells, suggesting they are not a source of energy during larval malnutrition. 62 Similarly, lipid droplet synthesis is enhanced during larval hypoxia, this can be interpreted as a protective mechanism to prevent the peroxidation of polyunsaturated fatty acids, particularly 4-hydroxy-2-nonenal (4-HNE), an unsaturated aldehyde that forms covalent adducts with proteins and inhibits neuroblasts proliferation. 62
On the other hand, in Drosophila, although glycogen is stored mainly in the fat body and muscle, it is also found in the larval and adult CNS.49,64 -66 In mammals, while neurons have been described to contain glycogen, 67 glial cells, mostly astrocytes that are part of the neurovascular unit, are the main cells storing carbohydrates in the form of glycogen.68,69 It is not clear whether subperineurial or perineurial glia accumulates glycogen, however, electron microscopy analyses suggest the presence of glycogen in the cockroach BBB70,71 as well as in Drosophila cortex glia. 63 Moreover, it was reported that glycogen phosphorylase, the enzyme that catalyzes glycogen breakdown, is present in the honeybee BBB 72 supporting the notion that insects store glycogen in glial cells of the BBB. Importantly, upon Drosophila larval starvation, it is mainly fat body glycogen that is metabolized to maintain carbohydrate consumption in the tissues, while CNS glycogen seems to be unaffected by nutrient restriction. 49 Following this idea, knockdown of glycogen phosphorylase in all glial cells, under a normal diet, does not affect larval viability and pupariation. 73 Therefore, it is plausible that larval CNS glycogen is not used as a source of energy during larval development, but like fat storage, it may be required to fuel the brain later during metamorphosis. In the adult stages, it needs to be assessed whether energy storage at the BBB is utilized for maintaining the CNS energy demand.
Is the Drosophila Blood-Brain Barrier a True Nutrient Sensor?
Similar to mammals, the Drosophila BBB is the first contact with the circulating blood (hemolymph). Hence, it is tempting to propose that alongside working as a neurogenic niche, the BBB could also be acting as a nutritional sensor for the CNS. This idea would fit with a fast response of BBB glial cells to changes in the concentration of energy sources in the hemolymph, especially considering the brain-sparing phenomenon during development and the preferential protection of the CNS over other tissues during starvation. 52
In the case of the carbohydrate transport in Drosophila, tret1-1 expression is regulated by the action of the TGFβ ligand Glass-bottom boat (Gbb), which is increased in the BBB upon larval starvation. 57 Although the source of Gbb has not been shown, it likely corresponds to autocrine or paracrine signaling at the BBB. One alternative is that the decrease of glucose or trehalose in the perineurial glia triggers the expression of Gbb and so the levels of the Tret1-1 transporter. Alternatively, a relay mechanism may be in play, in which the larval fat body could sense a decrease in sugar, signaling to the BBB to express Gbb, or directly secreting Gbb, and as consequence increases the carbohydrate intake in the developing CNS. Thus, ex vivo assays isolating the CNS could be performed, modifying the levels of carbohydrates or amino acids, to evaluate whether the BBB can act as a nutrient sensor.
By this plausible role of the BBB as a nutrient sensor, mammalian brain endothelial cells can increase the levels of the glucose transporter isoform 1 (GLUT1) at the plasma membrane after glucose depletion, in a mechanism that involves sensing cellular energy by the AMP-dependent kinase (AMPK).74,75 This regulation of carbohydrate transport in the BBB could be a conserved mechanism for a fast response to changes in nutrient availability.
Conclusions and Future Directions
During the evolution of species, the need of developing complex behaviors and advanced cognitive abilities has pushed animals for the isolation of the CNS by the BBB. Although from an evolutionary and anatomical perspective, the arthropod BBB is different from the one found in most vertebrate taxa, the functionality of the BBB presents great similarities across all species. This conservation can be exemplified by the molecular homology of the components of the invertebrate and vertebrate BBB. Hence, nutrient, ion, and xenobiotic transporter families are highly conserved between insects and mammals. A simple explanation for this is that the ancestors for all these transporter families appeared much earlier in evolution than the requirement of an isolated CNS. Therefore, comprehending the molecular mechanisms that regulate the Drosophila BBB function can indeed give invaluable information for the understanding of the human neurovascular unit.
How these mechanisms are regulated by nutrition is just starting to be unveiled. Drosophila melanogaster provides a very simple and powerful system to experimentally assess these molecular mechanisms. We envision that in the near future scRNA-seq experiments will provide further genetic insights into the transcriptional response of the BBB to malnutrition periods. Although Brunet Avalos et al 56 tried this approach, by sequencing all larval brain cells, the number of BBB glia was limited. Thus, a challenge to overcome is the isolation of BBB glial cells to enrich the population of cells to be sequenced. On the other hand, the targeted DamID approach we utilized reflects the accumulated binding of Polymerase II during the entire period of starvation, having the caveat of mixing early and late transcriptional responses. 55 Alternatively, the BBB adaptation to starvation can also be mediated in a transcription-independent manner. It is plausible that the levels of transporters at the plasma membrane are affected by endocytosis, trafficking, or degradation among others. Similarly, molecule transport can be regulated by posttranslational modifications that modulate transport activity. Therefore, studying the transport across the BBB of energy molecules, such as glucose or ketone bodies, is also important for understanding the adaptive response of the BBB to changes in food availability. For this goal, the development of new and improved fluorescent reporters will permit visualizing transport across the BBB with high temporal and spatial resolution.
Importantly, the BBB response to nutritional changes may be different depending on the diet. While most of the studies have been performed using inanition conditions, a more accurate reflection of human dietary changes would be different types of malnutrition, including high sugar or high-fat diets. Furthermore, the molecular response to malnutrition may change according to the developmental stage of the animal. Clear evidence for this is the metabolic shift that transitions the fat body response to starvation from consumption to conservation of energy storage after commitment to pupariation. 50 Therefore, adaptation to nutrient restriction could be, in principle, different in the larval BBB compared to adult stages.
Footnotes
Acknowledgements
Author Contributions
E.G.C. wrote the initial draft and J.S. proofread it. All authors proofread and approved the final manuscript.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received and acknowledged research funding from the following grants: ANID Becas Chile N°74200032 (E.G.C.), FONDECYT Regular 1210586 (J.S.).
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
