Abstract
Neuro-inflammation is associated with the production of cytokines, which influence neuronal and glial functions. Although the proinflammatory cytokines interferon-γ (IFN-γ) and interleukin-1Beta (IL-1β) are thought to be the major mediators of neuro-inflammation, their role in brain injury remains ill-defined. The objective of this study was to examine the effect of IFN-γ and IL-1β on survival of cortical neurons in stab wound injury in mice. A stab wound injury was made in the cortex of male BALB/c mice. Injured mice (I) were divide into IFN-γ and IL-1β treatment experiments. Mice in I + IFN-γ group were treated with IFN-γ (ip, 10 µg/kg/day) for 1, 3 and 7 days and mice in I + IL-1β group were treated with 5 IP injection of IL-1β (0.5 µg /12 h). Appropriate control mice were maintained for comparison. Immunostaining of frozen brain sections for astrocytes (GFAP), microglia (Iba-1) and Fluoro-Jade B staining for degenerating neurons were used. Western blotting and ELISA for brain-derived neurotrophic factor (BDNF) were done on the tissues isolated from the injured sites. Results showed a significant increase in the number of both astrocytes and microglia in I + IFN-γ and I + IL-1β groups. There were no significant changes in the number of astrocytes or microglia in noninjury groups (NI) treated with IFN-γ or IL-1β. The number of degenerating neurons significantly decreased in I + IFN-γ and I + IL-1β groups. GFAP and BDNF levels were significantly increased in I + IFN-γ and I + IL-1β groups. Interferon-γ and IL-1β induce astrogliosis, microgliosis, enhance the secretion of BDNF, one of the many neurotrophic factors after brain injury, and promote the survival of cortical neurons in stab wound brain injury.
Introduction
Central nervous system (CNS) trauma can trigger a cascade of events, which results in a robust inflammatory reaction. Glial activation, proliferation of microglia, leukocyte recruitment, and upregulation and secretion of mediators such as cytokines, all which characterize this inflammatory response. 1 The trauma-induced inflammatory responses show sexual dimorphism, where in trauma cause an enhanced microglia/macrophage response in male mice with an activated microglia producing both pro-(IL-1β and TNFα) and anti-inflammatory (TGFβ) cytokines. 2 The neuro-inflammatory response after CNS injury may be harmful or beneficial.3,4 However, the role played by inflammation in the course of degeneration and regeneration is not completely understood, and CNS trauma is often associated with poor recovery. 5 The overall effect of immune cells on injury, as assessed by depletion or knockout studies, and other studies appears to be generally beneficial.6-8 A component of the neuro-inflammatory response is the secretion of cytokines that seem to play a beneficial role in the acute phase of traumatic brain injury (TBI), setting the stage for proper tissue recovery.9-12
There are two sources of cytokines within the brain. Those that are produced by peripheral immune cells and cross the blood-brain-barrier, and those that are produced by CNS resident cells, namely the neurons and glial cells.13-16 Cytokines produced by neurons and glial cells within the brain may participate in the complex autonomic, neuroendocrine, metabolic, and behavioral responses to infection, inflammation, ischemia, and other brain injuries.16-18 The physiological importance of proinflammatory cytokines such as interferon-γ (IFN-γ) lies in its function in regulating immune and inflammatory events. 19 Interferon-γ is mainly produced by T-lymphocytes and natural killer cells and in the CNS; it is produced by astrocytes, microglial cells, as well as neurons.20,21 The functions of IFN-γ include regulation of leukocyte attraction as well as directing growth, maturation, and differentiation/inducing of many cell types including astrocytes and microglia.22,23 In addition, it has antiviral, antimicrobial, and antitumor activities among many other functions.24,25 Disruption of IFN-γ production in experimental models have shown to increase in autoimmune diseases, suggesting its important function in immune regulation even in the healthy CNS.26,27 In another study, the absence of IFN-γ expression in the spinal cord lead to neuronal degeneration, again suggesting a neuroprotective role of this cytokine under physiological conditions. 28
Similar to IFN-γ, IL-1β has been shown to be associated with a proinflammatory role in CNS disease and in stimulation of astrogliosis and neovascularization.29-32 It is generally believed that the inflammatory processes stimulated by IL-1β are detrimental and can aggravate the primary damage caused by infections of the CNS.33,34 The mechanisms by which IL-1β exerts its activities are not totally understood. In several different inflammatory and degenerative conditions of the CNS, an increase in the expression of IL-1β has been extensively documented.35-37
Among the most important effects of inflammatory cytokines, the regulation of neurotrophic factors is the main one. The interactions of cytokines and neurotrophic factors in the brain have important consequences for neuronal survival and function.38,39 Neurotrophins are a family of growth and survival factors regulating the development and the maintenance of functional phenotype of neuronal cells. 39 They are important in neuronal survival, differentiation, and myelination. In addition, they play a critical role in synaptic plasticity and in cognitive functions.40-42 Among the neurotrophins, brain-derived neurotropic factor (BDNF) is the most abundant and widely distributed neurotropic factor in the adult brain.43,44 The interactions of cytokines and neurotrophic factors in the brain have important consequences for neuronal survival and function. For example, it has been shown that IL-6 modulates BDNF levels by acting on adenosine receptors through a complex cross-talk. 38 The biological actions of BDNF are achieved through the specific binding and activation of its membrane-bound tropomyosin-related kinase (Trk) receptors or a single pan-neurotrophin receptor, p75NTR. 45 Changes in BDNF expression after different types of brain insults are related to neuroprotection, stimulation of axonal sprouting, and synaptic reorganization.
Although the proinflammatory cytokines IFN-γ and IL-1β are thought to be major mediators of neuro-inflammation, their role in case of brain injury remains ill-defined. The objective of this study was to examine the effect of IFN-γ and IL-1β on the cortical neuron survival in stab wound injury and also to examine their role in modulating BDNF expression.
Methods and Materials
Animals
BALB/c male mice (2 months old) were used in this study. Mice were maintained in the Central Animal Research Facility of Faculty of Medicine, Kuwait University. Mice were fed with food and water ad-libitum. Animals were maintained in 12:12 dark:light cycle and room temperature was maintained at 22°C ± 2°C. This study was carried out in accordance with recommendations of NIH Guidelines and Guide for the Care and Use of Laboratory animals. The protocol was reviewed and approved by Institutional Animal Care and Use Committee of Kuwait University, 13110. (Permit [Grant] no. MA01/14). Anesthetic cocktail of ketamine (40 mg/kg) and xylocaine (5 mg/kg) mixture was used to anesthetize the mice during surgical procedure and during sacrifice. The analgesic agent xylazine (2%) was injected (0.2 mL) around the suture every 8 h for 24 h for perioperative pain relief. Antiseptic betadine solution was applied on stab wound to prevent infection.
Experimental animal groups, stab wound injury, and treatment
This study consisted of two experiments. Experiment 1: Effects of IFN-γ on survival of neurons in stab wound injury and experiment 2: Effects of IL-1β on survival of neurons in stab wound jury. In both experiments, there were 4 groups of mice. In experiment 1, the groups were (1) stab wound injury + IFN-γ-treated group (I + IFN-γ), (2) stab wound injury + phosphate-buffered saline (PBS)-treated group (I + PBS1), (3) no injury + IFN-γ-treated group (NI + IFN-γ), (4) no injury + PBS-treated group (NI + PBS1). In experiment 2, the groups were (1) stab wound injury + IL-1β-treated group (I + IL-1β), (2) stab wound injury + PBS-treated group (I + PBS2), (3) no injury + IL-1β-treated group (NI + IL-1β), (4) no injury + PBS-treated group (NI + PBS2). Male BALB/c mice (2 months old) were randomly divided into above 8 groups (n = 54 in each group). A stab wound injury was made in cerebral cortical region of mice in injury + treatment groups as described earlier. 46 Briefly, mice were anesthetized with a ketamine (40 mg/kg) and xylocaine (5 mg/kg) mixture (Sigma Chemicals, St. Louis, USA). A stab wound was made by inserting 21-gauge sterile needle into the right of the frontal cerebral cortex (3 mm depth from skull surface) and 2 mm to the right side of the midline. Mouse head was secured horizontally in the stereotaxic frame and skull was exposed by a midline skin incision. Sterile needle (21G) held vertically in the stereotaxic needle holder was lowered into the brain slowly through the skull (1 mm/min speed), to a depth of 3 mm from skull surface. The needle was remained in the brain for 1 min before being retracted, at the same speed at which it was inserted. Skin was sutured and the analgesic agent xylazine (2%) was injected (0.2 mL) around the suture every 8 h for 24 h for perioperative pain relief. Antiseptic betadine solution was applied on stab wound to prevent infection. Mice in I +IFN-γ group received IP injection of IFN-γ (10 µg/kg/day, Sigma Chemicals, St. Louis, USA), for 1, 3, and 7 days commencing 1 h after stab wound injury (day 0). 47 A stock solution of IFN-γ at a concentration of 3 µg/mL PBS was prepared and injected based on their body weight. Mean body weight of the mice was 30 g. A mouse weighing 30 g was injected with 0.1 mL of IFN-γ solution (0.3 µg IFN-γ). Whereas, group I + PBS1 received IP injection of equivolume PBS (0.1 mL) every day for 1, 3, and 7 days. Mice in group I + IL-1β received 5 IP injections of IL-1β (0.5 µg/12 h, Sigma Chemicals, St. Louis, USA), commencing 1 h after stab wound injury (day 0). 34 A stock solution of I + IL-1β at a concentration of 0.15 µg/mL PBS was prepared and injected based on their body weight. A mouse weighing 30 g was injected with 0.1 mL of I + IL-1β solution (0.015 µg I + IL-1β). The I + PBS2 group of mice received 5 IP injections of equivolume PBS (0.1 mL/12 h) for 5 times. The dose and duration of treatment with IFN-γ and IL-1β is based on earlier publications.47,48
Animals were sacrificed following the treatment period. Mice in all groups were perfused with saline followed by 4% paraformaldehyde for morphological studies (Immunostaining for glial fibrillary acidic protein [GFAP; marker for astrocytes], Ionized calcium binding adaptor molecule 1 [Iba1; marker for microglia], and Flouro-Jade B staining [FJB, for staining degenerating neurons]) or perfused with cold saline for biochemical studies (Western blot analysis of GFAP and ELISA analysis of BDNF in the tissue around the injury site).
Three independent experiments were done with 18 mice/group. The number of animals used for IFN-γ and IL-1β experiments were summarized in Tables 1 and 2, respectively. In each IFN-γ and IL-1β experiments there were 4 groups, each group with 54 mice as shown in Tables 1 and 2, respectively. Of these 54 mice 18 mice were for morphological studies (6 each for 1 day, 3 days and 7 days group) and 36 for biochemical studies (6 each for 1 day, 3 days and 7 days group for Western blot analysis of GFAP and 6 each for 1 day, 3 days and 7 days group for ELISA analysis of BDNF). Since there were 4 groups, each group with large number of mice, (n = 54), we repeated the experiment (injury and treatment) 3 times (3 independent experiment), each time taking 18 mice/group. All mice in first experiments were perfused and used for morphological study (processed for cryosectioning, FJB staining, immunostaining and quantification of astrocytes and microglia). All mice in the second experiment were perfused with cold saline and fresh tissue around the injury site was collected for Western blot analysis of GFAP. All mice in the third experiment were perfused with cold saline and fresh tissue around the injury site was collected for ELISA analysis of BDNF. Data from 6 mice/group/study parameter (morphological, Western blot and ELISA) was collected and analyzed statistically.
Number for mice used for morphological and biochemical studies at first, third, and seventh day after treatment in different groups in IFN-γ experiment.

Abbreviations: BDNF, brain-derived neurotrophic factor; ELISA, enzyme-linked immunosorbent assay; FJB, Flouro-Jade B; GFAP, glial fibrillary acidic protein; Iba1, ionized calcium binding adaptor molecule-1; IFN, interferon; PBS, phosphate buffered saline.
Number for mice used for morphological and biochemical studies at first, third, and seventh day after treatment in different groups in IL-1β experiment.

Abbreviations: BDNF, brain-derived neurotrophic factor; ELISA, enzyme-linked immunosorbent assay; FJB, Flouro-Jade B; GFAP, glial fibrillary acidic protein; Iba1, ionized calcium binding adaptor molecule-1; IL, interleukin; PBS, phosphate buffered saline.
Tissue fixation and processing for histological and immunostaining
The mice in all groups were euthanized with CO2, at the end of the experiment and brain tissue was fixed and processed for histological and immunostaining as described earlier. 46 Briefly, heart was exposed and perfused with 50 mL of heparinized saline followed by 100 mL of 4% paraformaldehyde (in 0.1 M PBS, pH 7.4). The brain was dissected and postfixed for 48 h in the same fixative. Tissues were cryoprotected in 10%, 20%, and 30% sucrose solution for 24 h each. Then the tissues were embedded in optimal cutting temperature (OCT) compound (Sigma Chemicals, St. Louis, USA). Thirty micron (30 μm) thick serial, coronal cryostat sections were taken and stored in 24-well culture plate filled with PBS. Serial brain sections were stained for Fluoro-Jade B staining for assessing the degenerating neurons. Furthermore, sections were also immunostained for GFAP and Iba-1 for assessing the astrogliosis and microglial responses, respectively.
Fluoro-Jade B staining
Brain sections from injured brains were mounted on gelatin-coated slides and air-dried overnight at room temperature. Slides were rehydrated and treated with 0.06% potassium permanganate (Sigma Chemicals, St. Louis, USA) for 20 min, washed in distilled water and incubated in 0.004% FJB (Histo-Chem, Inc. Jefferson, Arkansas, USA) solution in 0.1% acetic acid for 30 min at 25°C. The sections were rinsed in distilled water, dehydrated and mounted with DPX. Number of FJB-stained degenerating neurons were counted in 6 randomly selected field/section (photographs taken with 40× objective in an Olympus fluorescence microscope). Finally, the number of degenerating neurons/mm3 tissue was calculated for each mouse as detailed below.
GFAP and Iba1 immunostaining and quantification
Serial, coronal frozen sections were immunostained for GFAP and Iba1 as described earlier. 46 Sections were treated with 3% hydrogen peroxide to reduce the endogenous peroxidase activity. The sections were then incubated with polyclonal rabbit anti-GFAP (1:100, Dako, Denmark) or polyclonal rabbit anti-Iba-1 (1:500, Abcam, Cambridge, MA, USA) antibodies overnight at 4°C. The sections were incubated with biotinylated anti-rabbit IgG (1:20, Vector Labs, Burlingame, CA, USA) for 1 h, followed by incubation with ExtrAvidin peroxidase (1:20, Sigma Chemicals, St. Louis, USA) for another 1 h. The color was developed using DAB (Vector Labs, Burlingame, CA, USA) as chromogen. Sections were mounted on gelatin-coated slides, air dried, dehydrated in ethyl alcohol, cleared in xylene, and mounted with DPX. All the brain sections were examined and immunostained cells were counted by Olympus BX51 TF upright transmitted light microscope using 40× objective (aperture is 0.75).
Double immunofluorescence labeling for GFAP and Iba-1; GFAP and FJB
A set of sections were double labeled for astrocytes (GFAP) and microglia (Iba-1). Sections were incubated in a cocktail of polyclonal rabbit anti-GFAP antibody (1:100, Dako) and monoclonal mouse anti-Iba-1 (1:200, Abcam) overnight, then the sections were washed and treated with a cocktail of rhodamine-conjugated donkey anti-rabbit IgG (1:100, Jackson Laboratories) and fluorescein-conjugated donkey antimouse IgG (1:100, Jakson Laboratories) for 45 min. Another set of sections were double-labeled with GFAP and FJB. The sections were first immunosatined for GFAP with polyclonal rabbit anti-GFAP antibody and rhodamine-conjugated donkey anti-rabbit IgG. Sections were further processed for fluoro-Jade B staining as described above. In all the above immunostaining, for negative control, the primary antibodies were omitted in the immunostaining protocol.
Quantification of degenerating neurons and immunostained astrocytes and microglia
Number of Fluoro-Jade B stained degenerating neurons, GFAP stained astrocytes and Iba-1 stained microglia were counted in 6 randomly selected fields (photographs taken with 40× objective) in each section around the lesion site in stab wound injury. From each mouse, 6 sections, (60µm apart) were selected for quantification. Finally, number of FJB-stained degenerating neurons, astrocytes, or microglia/mm3 tissue were calculated for each mouse as described earlier. 49 Briefly, total number of degenerating neurons, astrocytes, or microglia/mm3 tissue were quantified using the data collected from a total of 36 sections/mice (n = 6/group) using the formula: T = N*V/t, where N is the numerical cell density, V is the volume of the lesioned tissue around the lesion site used for quantification, and t is section thickness (30 µm). The numerical cell density (N) was calculated by measuring the area the section used for quantification with NIS-Elements software (NIS-Elements-D2.20) and total cell counts in 6 randomly selected fields. The volume (V) was calculated by measuring the area of the section used for quantification with NIS-Elements software (NIS-Elements-D2.20) and multiplying by section thickness (30 µm), intersection distance (60 µm) and number of sections (6).
Polyacrylamide gel electrophoresis and immunoblotting
Animals were perfused with 50 mL of cold saline. Tissue around the injury site (5 mm3) was removed and snap frozen in liquid nitrogen and stored at −80°C until Western Blot analysis as described earlier. 46 Briefly, tissue was thawed and incubated in ice cold radioimmunoprecipitation assay (RIPA) lysis buffer, with sodium orthovanadate (0.5 mM), and the protease inhibitors, phenylmethanesulfonyl fluoride (PMSF; 1 mM), aprotinin (10 µg/mL), leupeptin (1 µg/mL)] for 10 min. Tissue was homogenized in cold, in a tissue homogenizer for 3 to 5 min, homogenate was centrifuged at 14 000 r/min at 4°C for 5 min to collect the supernatant. The protein concentration in the samples was determined using a spectrophotometer (Bioteck). All samples (75 µg protein/well) were analyzed electrophoretically on a 10% SDS-PAGE gel. 50 The proteins in the gel were transferred to a nitrocellulose membrane. 51 After transfer, the membranes were incubated for 1 h with 5% skim milk in Tris-buffered Saline-Tween 20 (TBST). The immunoblots were probed with rabbit anti-GFAP antibody and rabbit anti-Glyceradehde-3-phosphate dehydrogenase (GAPDH, Sigma Chemicals, St. Louis, USA) antibody diluted in 5% skimmed milk in TBST. Glyceradehde-3-phosphate dehydrogenase was used as endogenous sample loading control. This was then followed by incubation with affinity-purified goat anti-rabbit IgG conjugated to horse-radish peroxidase (1:100, Sigma, Chemicals, St. Louis, USA). Immunoreactive bands were visualized using an enhanced chemiluminescence system (ECL, Santa Cruz Biotechnology, Inc Dallas, Texas USA). The difference in the band intensities on exposed films were determined by densitometric scanning. Intensity of bands were quantified in the Image-J image analysis software and normalized to density of the GAPDH band.
ELISA for BDNF
Tissue samples (5 mm3 tissue around injury site) were collected weighed immediately to get the wet weight of the samples, snap frozen in liquid nitrogen and stored at −80°C until analysis. Brain-derived neurotrophic factor content in the tissues was analyzed as described earlier. 46 Each sample was transferred to 250 µL of ice-cold homogenization buffer and homogenized for 1 min in a tissue homogenizer. Composition of the homogenization buffer was 100 mM Tris/HCl, pH 7, 2% bovine serum albumin (BSA), 1 M NaCl, 4mM EDTA. Na2, 2% Triton X-100, 0.1% sodium azide and the protease inhibitors (5 µg/mL aprotinin, 0.5 µg/mL antipain, 157 µg/mL benzamidine, 0.1 µg/mL pepstatin A and 17 µg/mL phenylmethyl-sulphonyl fluoride). The lysate from each sample was centrifuged at 14 000g for 30 min at 4°C and the supernatant solutions were collected. The supernatant from each sample was frozen for subsequent measurements of BDNF using ChemiKine BDNF sandwich ELISA kit (Merck Millipore, Billerica MA, USA) following the protocol provided in the kit. Briefly 100 µL of standards or 100 µL samples were added into each flat-bottom wells, precoated with mouse anti-Human BDNF monoclonal antibody and incubated at 4°C overnight on a shaker. Wells were washed 3 times with 250 µL of diluted wash buffer. diluted biotinylated mouse anti-BDNF monoclonal antibody (100 µL; 1:1000 in sample diluent) was added to each well and incubated at room temperature for 3 h on a shaker. Wells were washed again 3 times with 250 µL of diluted wash buffer. Diluted streptavidin-HRP conjugate solution (100 µL; 1:1000, in sample diluent) was added to each well and incubated at room temperature for 1 h on a shaker. Wells were washed 3 times with 250 µL of diluted wash buffer. About 100 µL of TMB substrate (3,3,′5,5′-tetramethylbenzidine) was added to each well and incubated at room temperature for 15 min. Reaction was stopped by adding 100 µL of stop solution to each well. The wells were read immediately in a ELISA plate reader at 450 nm. Optical density (OD) of standard solution was plotted against known concentration of the standards to get the standard curve. Unknown concentration of the BDNF in the samples was calculated by plotting their OD values into the standard curve. Finally, data were expressed as pg/mg tissue and group mean was determined.
Statistical analysis
The number of GFAP-stained astrocytes, Iba-1-stained microglia and FJB-positive degenerating neurons were counted in 6 randomly selected fields (photographs taken with 40× objective) in each section around the injury site. From each mouse 6 sections, (60 µm apart) were selected for quantification. Six mice were used in each subgroup. Finally, number of astrocytes or microglia or degenerating neurons/mm3 tissue were calculated for each mouse. 49 Three independent experiments were done and the data were expressed as mean ± standard error of mean (SEM). Intensity of the immunoblot bands were normalized to density of the GAPDH band and expressed as mean ± SEM. Concentration of BDNF was determined by plotting OD values in the standard curve and mean ± SEM data were calculated. All data were analyzed by unpaired two-tailed Student’s t-test or way analysis of variance (ANOVA), followed by Bonferroni’s post-test. P values < .05 were considered statistically significant.
Results
Effect of IFN-γ and IL-1β on number of degenerating neurons
Stab wound resulted in a time-dependent degeneration of neurons around the injury sites. However, neurodegeneration was less extensive both in I + IFN-γ and I + IL-1β groups compared to respective injury only groups (I + PBS1 and I + PBS2; Figures 1A and B and 2A and B). Quantification of the degenerating neurons showed significantly decreased number of degenerating neurons both in I +IFN-γ and I + IL-1β groups compared to respective injury only groups at all-time points studied (Student’s t-test, I + IFN-γ: first day: t = 23.23, df-10, P < .001, third day: t = 16.97, df-10, P < .001, seventh day: t = 16.23, df-10, P < .001; I + IL-1β: first day: t = 3.56, df-10, P< .05, third day: t = 8.48, df-10, P < .001, seventh day: t = 6.36, df-10, P < .001; Figures 1C and 2C). There were no FJB-positive cells found in no-injury groups (NI + IFN-γ, NI + PBS1 [Figure 3A] and NI + IL-1β, NI + PBS2 [Figure 3B]). FJB/GFAP double-labeling did not show any co-localization, suggesting that the degenerating cells are not astrocytes, but neurons (Figure 4).

(A) Representative low magnification photomicrograph of brain sections stained by Fluoro-Jade B, showing degenerating neurons around the stab wound in injured and treated with IFN-γ (I + IFN-γ) or PBS (I + PBS1) at third day (one side of injury site only shown). IS (needle track), is bounded by interrupted line. Scale bar = 80 µm. (B) Photomicrograph showing degenerating neurons (arrows) around the stab wound in injured and treated with IFN-γ (I + IFN-γ) or PBS (I + PBS1) at first, third, and seventh day. Note the decreased number of degenerated neurons in I + IFN-γ group compared to I + PBS1 group. Scale bar = 50 µm.

(A) Representative low magnification photomicrograph of brain sections stained by Fluoro-Jade B, showing degenerating neurons around the stab wound in injured and treated with IL-1β (I + IL-1β) or PBS (I + PBS2) at third day (one side of injury site only shown). IS (needle track) is bounded by interrupted line. Scale bar = 80 µm. (B) Photomicrograph showing degenerating neurons (arrows) around the stab wound in injured and treated with IL-1β (I + IL-1β) or PBS (I + PBS2) at first, third, and seventh day. Note the decreased number of degenerated neurons in I + IL-1β group compared to I + PBS2 group. Scale bar = 50 µm. (C) Graph showing the number of degenerated neurons/mm3 tissue. Note significant decrease in the number of degenerated neurons in I + IL-1β group at all-time points studied compared to I + PBS2 group (P < .05 and .001). IL indicates interleukin; IS, injury site; PBS, phosphate-buffered saline.

Photomicrograph of the brain sections (cortical region, at 3 days, corresponding to the injury site in stab wound injured mice) of non-injured mice, stained by Fluoro-Jade B. (A) Noninjured and treated with IFN-γ (NI + IFN-γ) or PBS (NI + PBS1), (B) Noninjured and treated with IL-1β (NI + IL-1β) or PBS (NI + PBS2). Note there were no degenerated neurons in both IFN-γ and IL-1β treated groups. Similar result was found at 1 day and 7 days also. Scale bar = 50 µm. IL indicates interleukin; PBS, phosphate-buffered saline.

(A) Double immunofluorescence staining of FJB/GFAP from a representative mouse. (B) Magnified view of the region shown in the rectangular box in A. FJB/GFAP double-labeling did not show any co-localization suggesting that the degenerating cells are not astrocytes, but neurons. Note that the degenerating neurons (green colored, indicated by arrowheads) are localized close to injury site (IS, injury site/needle track) whereas the astrocytes (red colored, indicated by arrows) are localized away from injury site. Scale bar = 100 µm in A, 50 µm in B. FJB indicates Flouro-Jade B; GFAP, glial fibrillary acidic protein; IS, injury site.
Effect of IFN-γ and IL-1β on astrocytes
Stab wound lesion increased the astrocytes around the lesion site with time in all injury groups both in IFN-γ and IL-1β experiments (Figure 5A and B). However, astrocytes population densities were very extensive both in I + IFN-γ and I + IL-1β groups compared to respective lesion only groups (I + PBS1, I + PBS2; Figures 6A and 7A). Quantification of the number of astrocytes showed significant increase in the number of astrocytes/mm3 tissue in both I + IFN-γ and I + IL-1β groups compared to respective injury only (I + PBS1 and I + PBS2) groups at all-time points studied (One-way ANOVA, I + IFN-γ: first day- F = 175 df-3,20, P < .01; third day F = 276, df-3,20, P < .001; seventh day F = 636, df-3,20, P < .001. I + IL-1β: first day F = 97.71 df-3,20, P < .001; third day F = 243.19, df-3,20, P < .001; seventh day F = 358.82, df-3,20, P < .001, Figures 6B and 7B). Treatment with both IFN-γ and IL-1β in noninjury mice (NI + IFN-γ and NI + IL-1β) groups did not increase the number of astrocytes, Figures 6B, 7B, 8 and 9). Western blot analysis for GFAP showed significantly enhanced GFAP content in both IFN-γ and IL-1β treated subgroups (I + IFN-γ, I + IL-1β) compared to respective injury only groups (I + PBS1, I + PBS2; (Student’s t-test, I + IFN-γ: first day: t = 0.079, df-4, P > .05, third day: t = 8.28, df-4, P < .01, seventh day: t = 198.05, df-4, P < .0001; I + IL-1β: first day: t = 57.48, df-4, P < .0001, third day: t = 39.59, df-4, P < .0001, seventh day: t = 16.76, df-4, P < .0001; Figures 10 and 11).

Low magnification photomicrograph of brain sections (immunostained for GFAP) of stab wound injured mice showing the distribution of astrocytes around the stab wound injury site (IS/needle track) at third day. (A) injured and treated with IFN-γ (I + IFN-γ) or PBS (I + PBS1), (B) injured and treated with IL-1β (I + IL-1β) or injured and treated with PBS (I + PBS2). Note the dense distribution of astrocytes in both IFN-γ and IL-1β-treated groups. Similar result was found at 1 day and 7 days also. Scale bar = 100 µm. GFAP indicates glial fibrillary acidic protein; IFN, interferon; IL, interleukin; IS, injury site; PBS, phosphate-buffered saline.

(A) Photomicrograph of brain sections immunostained for astrocytes, showing GFAP-positive astrocytes (arrows) around the stab wound in injured and treated with IFN-γ (I + IFN-γ) or PBS (I + PBS1) at first, third, and seventh day. Note the increased population of astrocytes in I + IFN-γ group compared to (I + PBS1) group. Scale bar = 50 µm. (B) Graph showing the number of astrocytes/mm3 tissue around the injured site in all groups. Note significant increase in the number of astrocytes in I + IFN-γ group at all-time points studied compared to I + PBS1 group (P < .01 to P < .001) and there is no significant difference between the noninjured (NI + IFN-γ and NI + PBS1) groups. GFAP indicates glial fibrillary acidic protein; IFN, interferon; PBS, phosphate-buffered saline.

(A) Photomicrograph of brain sections immunostained for astrocytes, showing GFAP-positive astrocytes (arrows) around the stab wound in injured and treated with IL-1β (I + IL-1β) or PBS (I + PBS2) groups at first, third, and seventh day. Note the increased population of astrocytes in I + IL-1β group compared to (I + PBS2) group. Scale bar = 50 µm. (B) Graph showing the number of astrocytes/mm3 tissue around the injured site in all groups. Note significant increase in the number of astrocytes in I + IL-1β group at all-time points studied compared to I + PBS2 group (P < .001), and there is no significant difference between the noninjured (NI + IL-1β and NI + PBS2) groups. GFAP indicates glial fibrillary acidic protein; IL, interleukin; PBS, phosphate-buffered saline.

Photomicrograph of brain sections (corresponding to the injury site in the injured group) immunostained for astrocytes, showing GFAP-positive astrocytes (arrows) in noninjured and treated with IFN-γ (NI + IFN-γ) or PBS (NI + PBS1) groups. Note the uniform, normal distribution of astrocytes in mice both treated with IFN-γ, and PBS (PBS1)*treated groups at all days. Scale bar = 50 µm. GFAP indicates glial fibrillary acidic protein; IFN, interferon; PBS, phosphate-buffered saline.
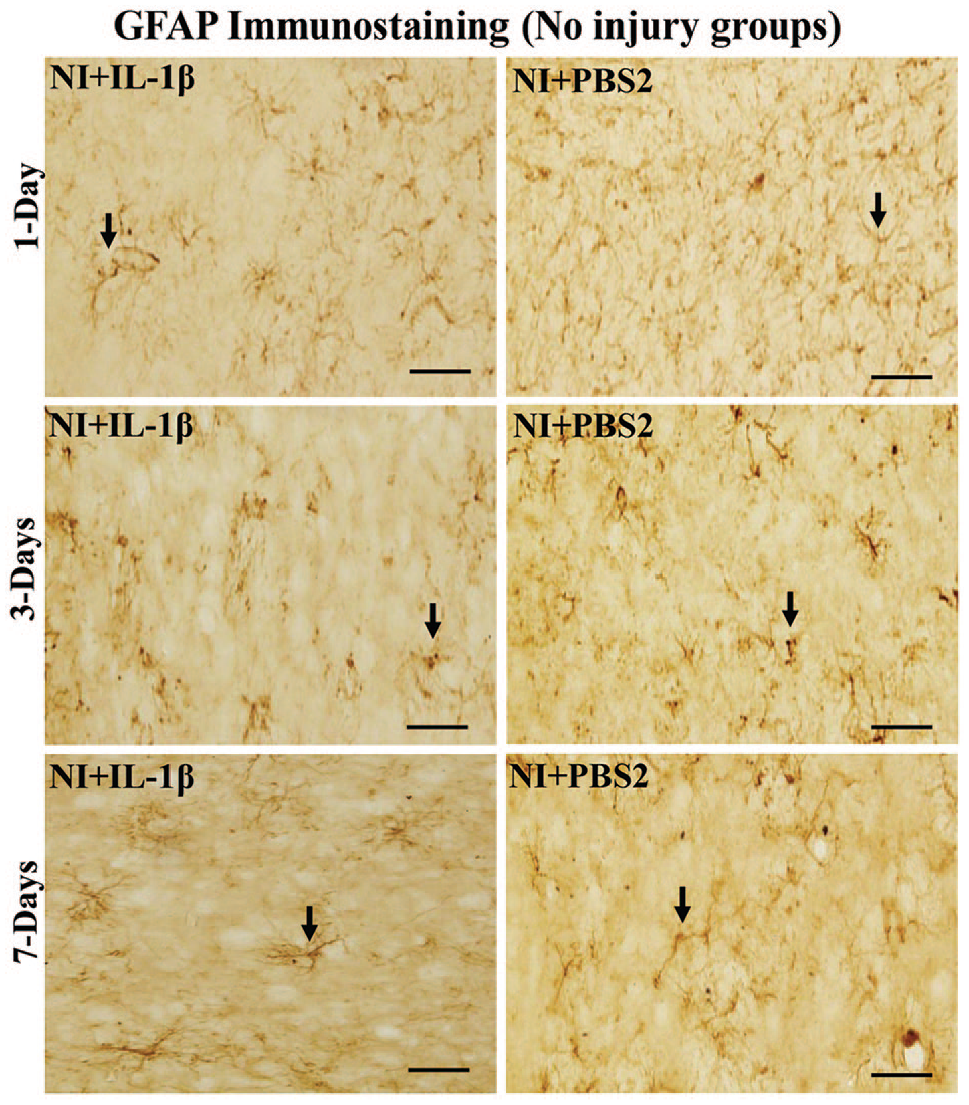
Photomicrograph of brain sections (corresponding to the injury site in the injured group) immunostained for astrocytes, showing GFAP-positive astrocytes (arrows) in noninjured and treated with IL-1β (NI + IL-1β) or PBS-treated (NI + PBS2) groups. Note the uniform, normal distribution of astrocytes in mice treated with both IL-1β, and PBS (PBS2)-treated groups at all days. Scale bar = 50 µm. GFAP indicates glial fibrillary acidic protein; IL, interleukin; PBS, phosphate buffered saline.

Immunoblotting of GFAP from first, third, and seventh postinjury day tissue from injured mice, treated with IFN-γ or I + PBS1 (A) and from noninjured mice treated with IFN-γ or PBS1 (C), stained with antibodies to GFAP and GAPDH. (B) and (D) Graph showing the mean gray value (GFAP/GAPDH) of the immunoblots. Note significantly increased GFAP content in I + IFN-γ compared to I + PBS1 at third and seventh days (P < .01 and .0001), and there is no significant difference in the GFAP content in NI + IFN-γ compared to NI + PBS1 at all days. GAPDH indicates anti-Glyceradehde-3-phosphate dehydrogenase; GFAP, glial fibrillary acidic protein; IFN, interferon; PBS, phosphate-buffered saline.
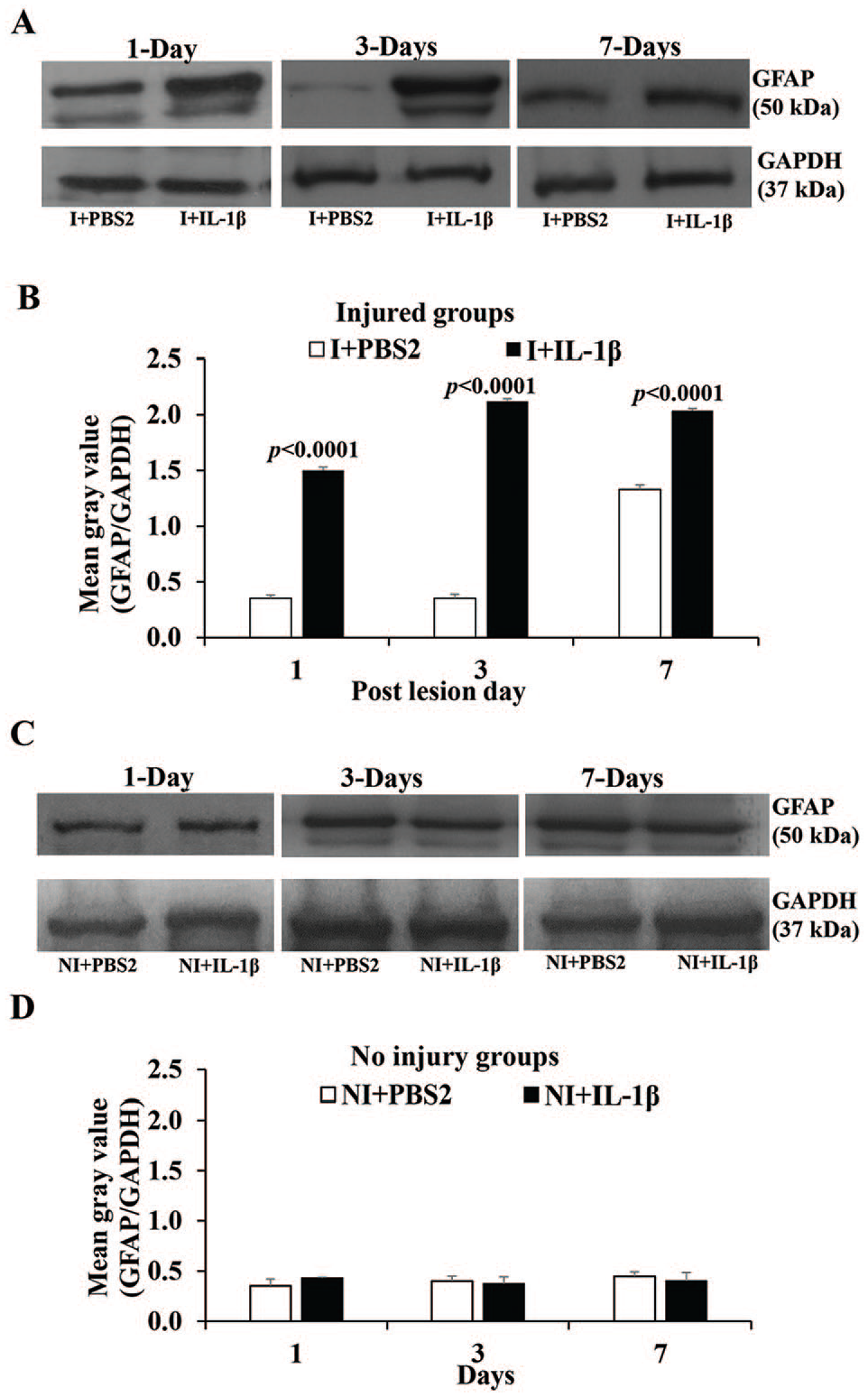
Immunoblotting of GFAP from first, third, and seventh postinjury day tissue from injured mice, treated with IL-1β or PBS2 (A) and from noninjured mice treated with IL-1β or PBS2 (C), stained with antibodies to GFAP and GAPDH. (B) and (D) Graph showing the mean gray value (GFAP/GAPDH) of the immunoblots. Note significantly increased GFAP content in I + IL-1β compared to I + PBS2 at all-time points studied (P < .0001), and there is no significant difference in the GFAP content in NI + IL-1β compared to NI + PBS2 at all-time points studied. GAPDH indicates anti-Glyceradehde-3-phosphate dehydrogenase; GFAP, glial fibrillary acidic protein; IL, interleukin; PBS, phosphate buffered saline.
Effect of IFN-γ and IL-1β on microglia
Stab wound lesion increased the microglia around the injury site in a time-dependent manner in both injury only (I) and injury and treated with both IFN-γ and IL-1β (I + IFN-γ and I + IL-1β) groups (Figure 12). However, the number of microglia were very extensive in both I + IFN-γ and I + IL-1β groups compared to respective lesion only (I + PBS1, I + PBS2) groups (Figures 13A and 4A). Quantification of the microglia showed significant increase in the number of microglia/mm3 tissue both in IFN-γ and IL-1β treated groups (I + IFN-γ and I + IL-1β) compared to respective lesion only (I + PBS1, I + PBS2) groups at all-time points studied (one-way ANOVA, I + IFN-γ: first day F = 221.56 df-3,20, P < .001; third day F = 193.67, df-3,20, P < .0.001; seventh day F = 512.25, df-3,20, P < .001. I + IL-1β: first day F = 116.33, df-3,20, P < .001; third day F = 197.14, df-3,20, P < .001; seventh day F = 491.95, df-3,20, P < .001, Figures 13B and 14B). Treatment with both IFN-γ and IL-1β in noninjury mice (NI + IFN-γ and NI + IL-1β) groups did not increase the number of microglia, Figures 13B and 14B). Treatment with both IFN-γ and IL-1β in noninjury mice groups (NI + IFN-γ and NI + IL-1β) groups did not increase the number of microglia compared to control groups (NI + PBS1, NI + PBS2; Figures 13B, 14B, 15 and 16).

Low magnification photomicrograph of brain sections (immunostained for Iba-1) of stab wound injured mice showing the distribution of microglia around the stab wound injury site at third day. (A) Injured and treated with IFN-γ (I + IFN-γ) or treated with PBS (I + PBS1), (B) Injured and treated with IL-1β (I + IL-1β) or treated with PBS (I + PBS2). Note the dense distribution of microglia in IFN-γ and IL-1β treated groups. Similar result was found at 1 day and 7 days also. Scale bar = 100 µm. Iba-1 indicates ionized calcium-binding adaptor molecule-1; IFN, interferon; IS, injury site; PBS, phosphate-buffered saline.
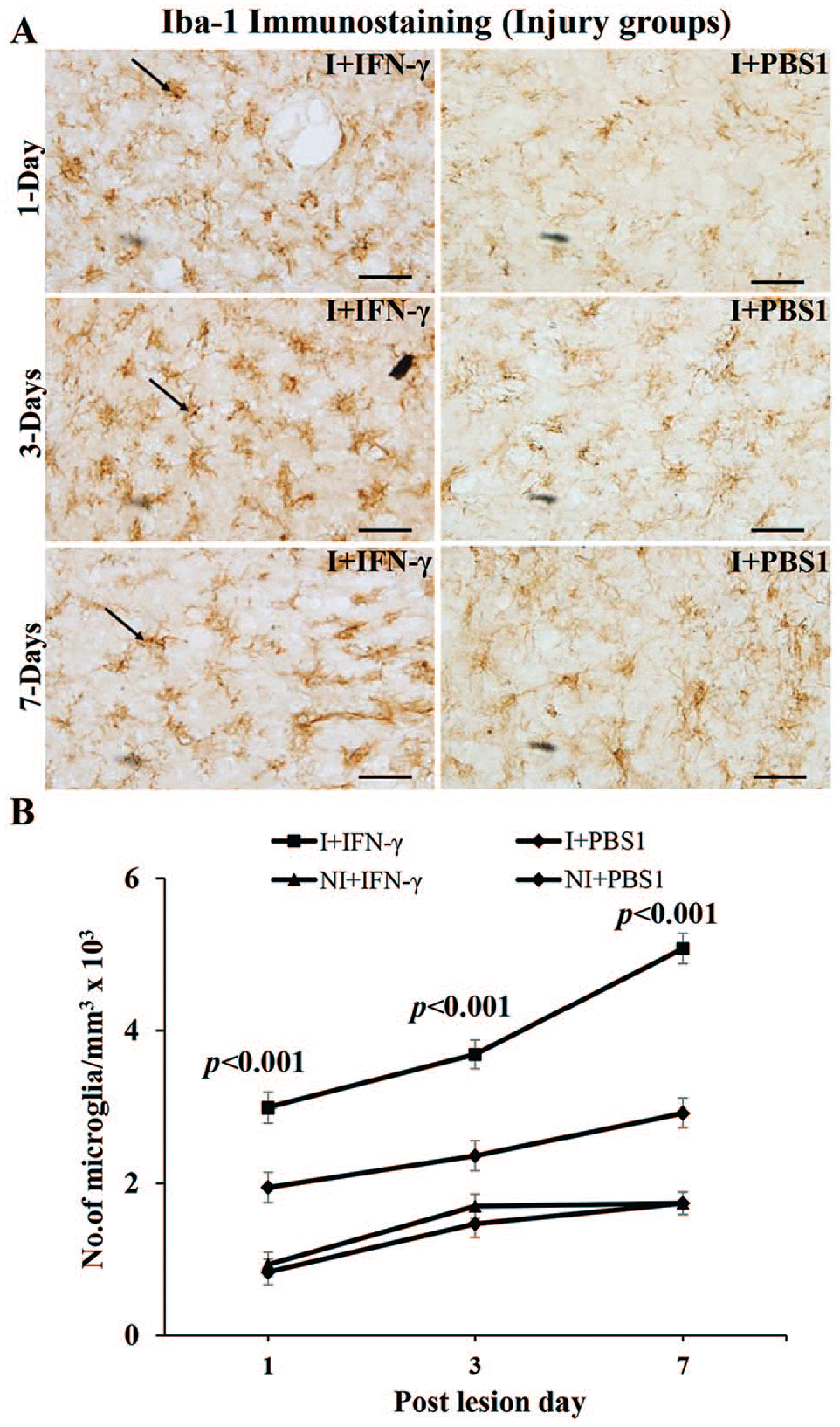
(A) Photomicrograph of brain section immunostained for Iba-1, showing Iba-1-positive microglia (arrows) around the stab wound in I + IFN-γ and I + PBS1 groups. Note the increase in the population of the microglia in I + IFN-γ group compared to I + PBS1 group at all-time points studied. Scale bar = 50 µm. (B) Graph showing the number of microglia/mm3 tissue in all groups. Note significant increase in the number of microglia with time in I + IFN-γ group at all time-point studied compared to I + PBS1 group (P < .001), and there is no significant difference between the noninjured (NI + IFN-γ and NI + PBS1) groups. Iba-1 indicates ionized calcium-binding adaptor molecule-1; IFN, interferon; PBS, phosphate-buffered saline.

(A) Photomicrograph of brain section immunostained for Iba-1, showing Iba-1-positive microglia (arrows) around the stab wound in I + IL-1β and I + PBS2 groups. Note the increased population of microglia in I + IL-1β compared to I + PBS2 at all-time points studied. Scale bar = 50 µm. (B) Graph showing the number of the microglia/mm3 tissue around the lesion site in I + IL-1β and I + PBS2 groups. Note significant increase in the number of the microglia with time in I + IL-1β group at all time-point studied compared to I + PBS2 group (P < .001), and there is no significant difference between these noninjured (NI + IL-1β and NI + PBS2) groups. Iba-1 indicates ionized calcium-binding adaptor molecule-1; IL, interleukin; PBS, phosphate-buffered saline.

Photomicrograph of brain sections (corresponding to the injury site in the injured group) immunostained for microglia, showing Iba1-positive microglia (arrows) in noninjured and treated with IFN-γ (NI + IFN-γ) or noninjured and treated with PBS (NI + PBS1) groups. Note the uniform, normal distribution of microglia in mice treated with both IFN-γ, and PBS (PBS1) at first, third, and seventh day. Scale bar = 50 µm. Iba-1 indicates ionized calcium-binding adaptor molecule-1; IFN, interferon; PBS, phosphate-buffered saline.
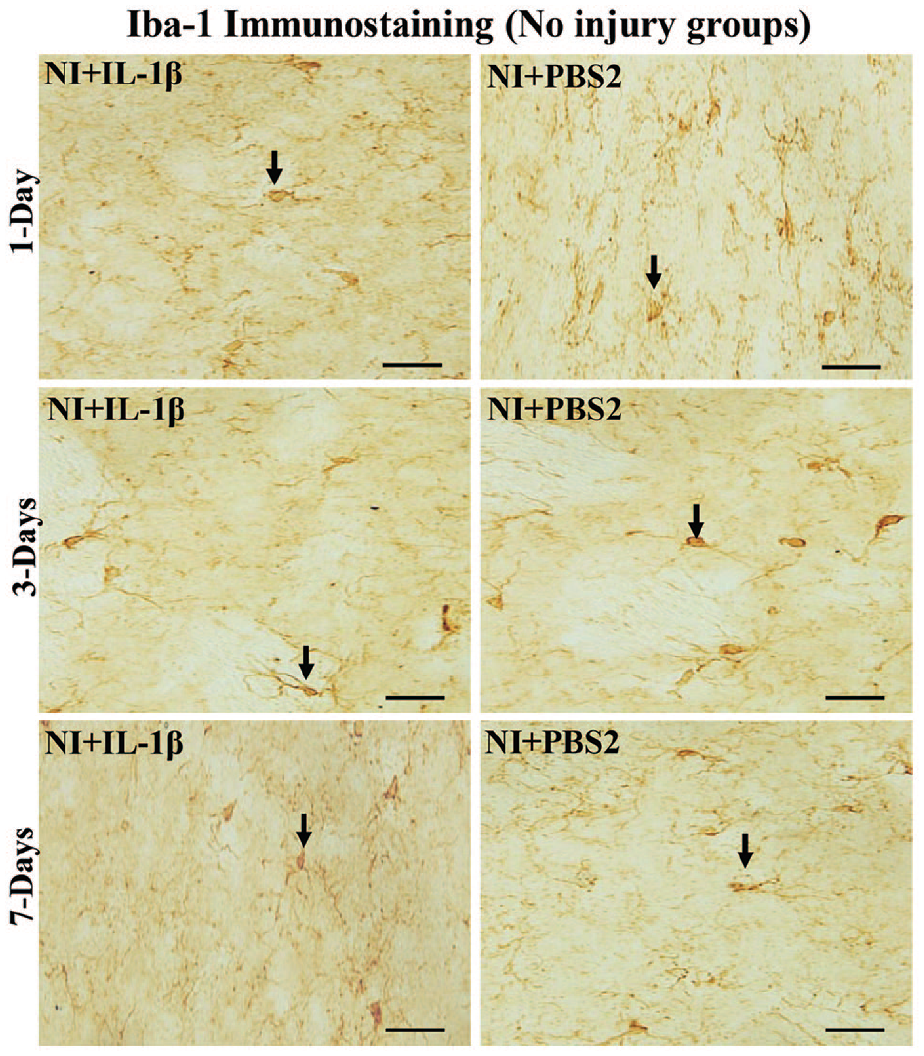
Photomicrograph of brain sections (corresponding to the injury site in the injured group) immunostained for microglia, showing Iba1-positive microglia (arrows) in noninjured and treated with IL-1β (NI + IL-1β) or PBS (NI + PBS1) groups. Note the uniform, normal distribution of microglia in mice treated with both IL-1β, and PBS (PBS1) at first, third, and seventh day. Scale bar = 50 µm. Iba1 indicates ionized calcium-binding adaptor molecule-1; IL, interleukin, PBS, phosphate-buffered saline.
Double-labeling immunostaining for Iba-1 and GFAP showed normal distribution of both astrocytes and microglia around the injury site without any specific pattern of distribution (Figure 17).

Double immunofluorescence (Astrocytes [Red] and Microglia [Green]) staining of brain sections from a representative brain section of an injured and treated with IL-1β mice, 1 day after treatment. Double immunofluorescence staining showed normal distribution of both astrocytes and microglia around the injury site without any specific pattern of distribution. Scale bar = 30 µm. GFAP indicates glial fibrillary acidic protein; IL, interleukin.
Effect of IFN-γ and IL-1β on expression of BDNF
Brain-derived neurotrophic factor level assessed by ELISA method was significantly increased in the mice injured and treated with IFN-γ and IL-1β (I + IFN-γ and I + IL-1β) groups compared to mice injured and treated with PBS (I + PBS1, I + PBS2) groups at all postinjury days (Student’s t-test, I + IFN-γ: first day: t = 20.68, df-10, P < .001, third day: t = 27.77, df-10, P < .001, seventh day: t = 17.03, df-10, P < .001; I + IL-1β: first day: t = 20.63, df-10, P < .001, third day: t = 33.35, df-10, P < .001, seventh day: t = 14.18, df-10, P < .001; Figure 18A and B, P < .001). Treatment with both IFN-γ and IL-1β in noninjury groups (NI + IFN-γ and NI + IL-1β) did not increase the BDNF content compared to control groups (NI + PBS1, NI + PBS2; Figure 18A and B).
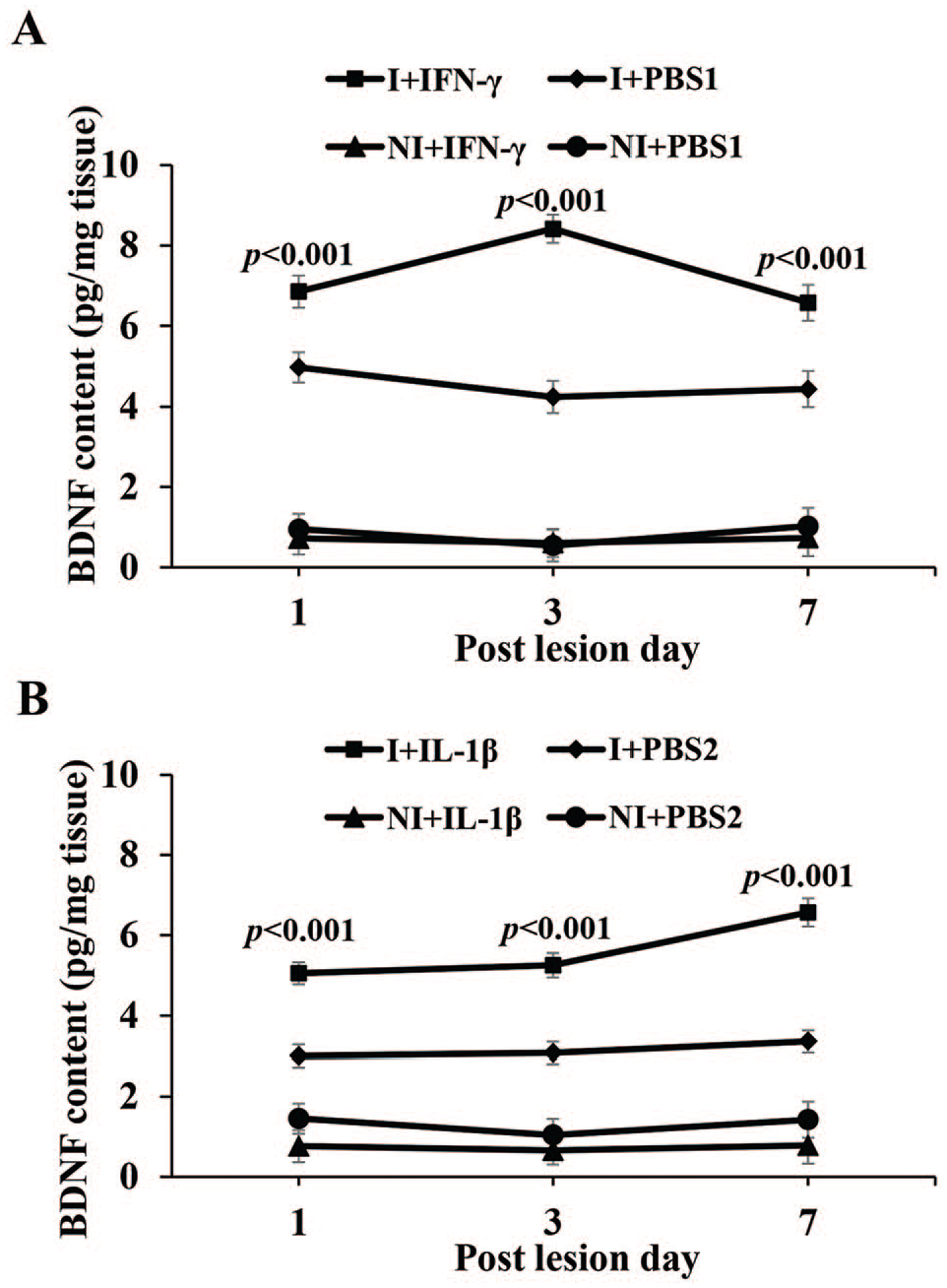
(A) Graph showing the concentration of BDNF as assessed by ELISA method in the tissue around the injured site in injured and treated with IFN-γ or PBS1 and in non-injured and treated with IFN or PBS2 (in the tissue collected from the region corresponding to the injury site in injured group). (B) Graph showing the concentration of BDNF in the tissue around the injury site in injured and treated with IL-1β and PBS2, and noninjured and treated with IFN-γ or PBS2 (in the tissue collected from the region corresponding to the injury site in injured group). Note significantly increased BDNF content in both I + IFN-γ and I + IL-1β groups compared to I + PBS1 and I + PBS2 groups, respectively, at all-time points studied. There is no significant difference in the BDNF content in NI + IFN-γ and NI + IL-1β compared to NI + PBS1 and NI + PBS2 groups, respectively, at all-time points studied. BDNF indicates brain-derived neurotrophic factor; ELISA, enzyme-linked immunosorbent assay; IFN, interferon, IL, interleukin; PBS, phosphate buffered saline.
Discussion
In this study, we have demonstrated that both IFN-γ and IL-1β increase the number of astrocytes and microglia in a stab wound model of brain injury as well as an increase in the expression of GFAP and BDNF. These results are consistent with those observed in all types of TBI, where there is a consistently increased astroglia and microglia population around the injury site. In addition, IFN-γ and IL-1β have a neuroprotective role demonstrated by the decreasing numbers of degenerated neurons in the stab wound brain injuries. The proposed mechanism through which this occurs may be by increasing BDNF secretion by activated astrocytes and microglia. Interferon-γ and IL-1β are able to increase secretion of BDNF through astrocytes and microglial activation.
Fluoro-Jade B is an anionic fluorescein derivative. It is used for the histological staining of neurons undergoing degeneration. 52 There are 3 fluoro-jade dyes-Fluoro-Jade, Fluoro-Jade B, and Fluoro-Jade C. 53 Fluoro-Jade B, and Fluoro-Jade Care extensively used for identifying and quantifying degenerated neurons in neurotrauma studies.54-59 In this study, we used Fluoro-Jade B to stain and quantify the degenerating neurons in the stab wound injury. Results showed large number of degenerating neurons around the injury site in injured + treated with PBS (I + PBS1 and I + PBS2 groups) and injured + IFN-γ or IL-1β treated groups (I + IFN-γ and I + IL-1β) a day after injury, with a significant difference between PBS-treated and IFN-γ- or IL-1β-treated groups. FJB/GFAP double-labeling did not show any co-localization, suggesting that the degenerating cells are not astrocytes, but neurons (Figure 4). Number of degenerating neurons found to decrease gradually both in I + PBS, I + IFN-γ and I + IL-1β group on third and seventh postinjury days. The pattern of neurodegeneration observed is consistent with our earlier study using the stab wound injury model and intracerebroventricular kainic acid model of brain injury and several other studies.46,55-57,60 Gradual decrease in the number of degenerating neurons in injured and treated with PBS groups from first day to seventh day postinjury may be due to several neuroprotective compensatory mechanisms triggered around the injury site such as enhanced release of neuroprotective neurotrophic factors. Indeed, in our earlier study and in this study, we showed a progressive increase in number of astrocytes, microglia, and BDNF levels from first day through seventh day in I + PBS group. 46 Similar enhancement of microglial/astrocytes population after traumatic injury has been shown in several studies.54-56,61 Gradual decrease in the number of degenerating neurons in injured and treated with IFN-γ (I + IFN-γ) and IL-1β (I + IL-1β) groups from first day to seventh day postinjury may be due to specific trophic neuroprotective effects of IFN-γ and IL-1β directly or through enhancement of microglia and astrocytes, thereby enhancing the neurotrophic factors such as BDNF as observed in this study, which may be one of the many neurotrophic factors provided neuroprotection. We have reported similar enhancement of astrocytes, microglia and BDNF by dibutyryl cyclic adenosine monophosphate (dBcAMP), a synthetic analogue of cAMP. 46
Astrocytes are the major supportive cells in the brain, with important functions including providing nutrients and regulating neuronal activities. 62 Following all types of injuries to the CNS, a homotypic response of astrocytes known as reactive astrogliosis occurs.63,64 This process involves heterogeneous and progressive changes of gene expression, morphology, proliferative capacity, and function of astrocytes.65-67 This astrocytic reaction is directly or indirectly produced by proinflammatory cytokines. Among these cytokines IFN-γ and IL-1β are key ones. The production of IFN-γ and IL-1β as a consequence of nervous tissue inflammation leads to microgliosis, astrogliosis, and immune cell recruitment, which all affect neuronal survival and repair. 68 Previous studies have suggested that IL-1β acts as an astroglia growth factor during brain development and induces astrogliosis in tissue culture69,70 and after brain injury.29-31,71,72 This study also demonstrated that IFN-γ and IL-1β induce astrogliosis after brain injury but not in the normal uninjured brain. This indicates that astrogliosis induced by trauma and other factors associated with trauma can be aggravated by IFN-γ or IL-1β.
Reactive astrocytes produce proteins that can have beneficial and also have detrimental effects on neurons. The effect depends on many factors, for example, the type and extent of injury as well as the relative interaction between cytokines and BDNF. 73
In addition to the role of astrocytes in brain injury, microglia also play a critical role in neuro-inflammation following brain injury.74-76 Microglia play a dual role in promoting beneficial and detrimental effect on neurons by producing neuroprotective factors or cytotoxic mediators, respectively, depending on the functional response.77-82 Microglia-neuron interaction plays an important role in maintenance of homeostasis of the CNS. Phagocytosis of neuronal debris by microglia helps to maintain neuronal networks. Deficiency of the phagocytic function of microglia is associated with an inadequate regenerative response. 83 The dual role of microglia activation in promoting beneficial (production of neurotrophic factors, anti-inflammatory cytokines, and clearing cellular debris) and detrimental (production of high levels of proinflammatory and cytotoxic mediators) effects on neurons may be accounted for their polarization state and functional response after injury and stage of the disease. An improved understanding of regulatory mechanisms that control microglia phenotypic shift may advance our knowledge of postinjury recovery and repair and provide opportunities for the development of novel therapeutic strategies for brain injury.81,84-87 In our study, the neuroprotective effect of activated microglia may be due to their increased phagocytic activities 88 and enhanced level of BDNF (one of the many neurotrophic factors to play role in neuroprotection) which has been demonstrated in this study.
The effect of IFN-γ on neurons in traumatic CNS injury in the literature is controversial, with some studies describing a protective function and others a degenerative role. Absence of IFN-γ expression in spinal cord leads to neuronal degeneration suggesting its role in neuroprotection as we have demonstrated in this study. 28 The cytokines secreted during the neuro-inflammatory response seem to play a beneficial role in acute phase of TBI, setting an environment for proper tissue recovery.11,12 Most studies on the subject assumes a pathological role for neuro-inflammation occurring after a TBI and therefore by increasing IFN-γ, being a proinflammatory cytokine, must have a detrimental effect. However, recent evidence is challenging a previous study by Roselli et al. 89 Intraperitoneal injections of IFN-γ to a spinal cord contusion model in mice resulted in an increase in astrocytes activation as well as faster motor performance recovery. 90
In another experiment, intracerebral injection of IFN-γ inhibited astrocyte proliferation following brain injury in rats contrary to our results. 91 It is important to emphasize the differences in experimental methodology used. In the aforementioned study, IFN-γ was administered directly into the cerebrum, whereas in this study on brain injury and pervious study on spinal cord injury, IFN-γ was administered through intraperitoneal injections. 90 This may explain the contradicting results. This suggests that the effect of IFN-γ on brain injury could be dependent on the method of its administration.
In other forms of CNS injury, IFN-γ has been shown to have contradicting roles. This emphasizes the complex and numerous roles of this cytokine that are dependent on many factors, including context. A recent study showed a direct association between IFN-γ signaling and stroke induced neurodegeneration. Inhibition of IFN-γ signaling resulted in a reduction in infarct volume. 92 In the experimental autoimmune encephalomyelitis (EAE) model, (which represents the multiple sclerosis model in animals), there was no consistency in results with regard to the role of IFN-γ. 92 Some studies reported dual role of IFN-γ on CNS cells, being protective for some and harmful to others as well as a stage specific role, having different functions early and late in the disease process.93-97
Like IFN-γ, IL-1β is also secreted by activated astroglia and microglia in brain, where it exerts a diverse range of activities on immune function and coordination of many aspects of the acute phase response to trauma and infection.32,98 Inflammation is the key host-defense response to infection and injury, but is also thought to be a major contributor of a diverse range of diseases. It is generally believed that the inflammatory processes stimulated by IL-1β is detrimental and can aggravate the primary damage caused by the CNS infections. 34 A previous study showed antenatal suppression of IL-1β protects several tissues from inflammation-induced injury and improves neonatal developmental outcomes in mice. 99 In addition, increased IL-1β level is detected in the peripheral blood with idiopathic and structural epilepsy in dogs suggesting an involvement of inflammation in pathophysiology of epilepsy. 100 The mechanisms by which IL-1β exerts its activities are not clearly understood. In addition, the role of IL-1β in neuroprotection is investigated by treating JunD siRNA mice with an anti-IL-1β monoclonal antibody and suppression of IL-1β was shown to cause brain injury. 101
As discussed above, our present study demonstrated that both IFN-γ and IL-1β increased the number of astrocytes and microglia around the stab wound brain injury, increased the expression BDNF in the tissue around the injury which provide neuroprotection. Reaction of astrocytes and microglia to the injury and treatment with these cytokines is comparable to that observed in several other studies using contusion, penetration, diffusion and mild repetitive brain injury models.102-112 Mild TBI was shown to increase the IL-1β protein within 6 h and 1 day after injury. Furthermore, it induced both proinflammatory, anti-inflammatory and immunomodulatory microglial phenotypes. 102 Both moderate and severe contusion injury in experimental animals and humans, IL-1β, IL-6, and tumor necrosis factor-alpha (TNF-α) levels were found to be significantly increased in the brain tissue.103,104
A moderate lateral fluid percussion brain injury in rats showed enhanced astrogliosis, microgliosis and increased levels of TNF-α, IL-1β and IL-6. 105 In a diffuse TBI (moderate midline fluid percussion injury), morphological profiles of glia showed a low, persistent level of inflammation with enhanced expression of GFAP and IL-1β in astrocytes and IL-1β in microglia after thirty days postinjury. 106 Similar type of brain injury resulted in a significant increase in several chemokines, monocyte chemoattractant proteins, macrophage inflammatory proteins, cytokines (IL-1α, IL-1β, IL-4, IL-6, IL-10, IL-13, IL-17α, IL-18, IFN-γ, TNF]-α), and growth factors. 107
Analysis of gene expression during the acute phase of injury (penetrating ballistic brain injury) in rats showed a peak increase in the cytokines (TNF-α, IL-1β, and IL-6) and the several cellular adhesion molecules. Furthermore, in the regions immediately surrounding the lesion, strong immunoreactivity for activated astrocytes and prominent microglial reactivity was observed. 108 Another study with penetrating ballistic-like brain injury in rats, showed up-regulated IL-1β, TNF-α, E-selectin and IL-6 mRNA during the acute phase of injury and enhanced the levels of the proinflammatory cytokines—IL-1β, TNF-α and IFN-γ in the injured brain, but did not affect anti-inflammatory cytokine IL-6 levels. 109
Early postnatal exposure to intermittent hypoxia increased cerebellar IFN-γ and IL-1β at postnatal day13. 110 Repetitive TBI in mice led to widespread astrogliosis, and microgliosis in white matter and significantly increased the levels of TNF-α and IL-1β. 111 Another study with repetitive mild TBI in rats showed neurodegeneration and induced astrocytosis in the hippocampal region. Inflammatory cytokines, such as IL-1β, IL6, and TNF-α were significantly increased in the brain tissues. 112
Several treatment modalities after brain injuries have shown changes in the glial cells and cytokines levels. Treatment with SRS 16-86(ferroptosis inhibitor) was shown to enhance astrogliosis and neuronal survival, decrease the inflammatory cytokine (IL-1β, TNF-α, and ICAM-1) levels in a contusion injury of the spinal cord. 113 Treatment with hypertonic saline after TBI was shown to decrease IL-1β mRNA and protein levels. 114 Luteolin, a member of the flavonoid family, was shown to decrease the mRNA and protein expressions of proinflammatory factors IL-1β and TNF-α and reduce the neuronal degeneration. 115 Doxycycline and tocopherol alone and in combination treatment in experimental TBI, has proved to reduced neuro-inflammatory markers (IL-6, IL-1β, and TNF-α). 116
Thus, in general, injury enhances gliosis and cytokines levels. As discussed earlier, cytokines have both beneficial and harmful effects. In our experiment, they showed beneficial effects. We have injected cytokines IL-1β or IFN-γ through the systemic route. This extraneous and intrinsic tissue released cytokines together enhanced gliosis further, which in turn augmented the secretion of trophic factors (BDNF in particular), enhancing the neuronal survival.
In the healthy noninjured brain, BDNF levels are very low. 75 Nonetheless, it is well known that following injury to the CNS, glial cells increase the expression of growth factors, including BDNF. 117 Brain-derived neurotrophic factor is essential for neuronal differentiation and survival. It is produced by neurons and astrocytes. 118 Astrocytes secrete neurotrophins on demand as proneurotrophins. They are then proteolytically cleaved to mature neurotrophins in the synaptic cleft and rapidly degraded. 119 Brain-derived neurotrophic factor is essential for neuronal differentiation and survival and circulating BDNF has diagnostic value in TBI. 120 Hence we have studied the expression of BDNF as an important neurotrophic factor among the many neurotrophic factors in the brain relesed after brain injury.
This study indicates that both IFN-γ and IL-1β induce astrogliosis and promote the survival of cortical neurons in stab wound brain injury. IFN-γ and IL-1β activates astrocytes and microglia and both can contribute to neuronal survival by increasing the secretion of BDNF. This study supports the strategies of preserving the activating astrocytes and microglia in the planning of future therapies to promote neuronal regeneration in brain injury.
Further studies should be conducted to evaluate the effect of IFN-γ and IL-1β in other forms of CNS injuries including contusion and autoimmune inflammation as the inflammatory response is affected by context among other factors. The long-term effect of IFN-γ and IL-1β needs to be explored because reactive astrogliosis while it increases BDNF and hence neuroprotection, it results over time in a gliotic scar. Those scars are thought to be undesirable for CNS regeneration and might hinder full CNS repair after the injury. 120
In conclusion, our study elucidates that IFN-γ and IL-1β induce astrogliosis, microgliosis, enhance the secretion of BDNF and promote the survival of cortical neurons in stab wound brain injury.
Footnotes
Acknowledgements
The authors acknowledge Dr Muhammad Mostafa Abd-El-Barr, Assistant Professor, Department of Neurosurgery, Duke University, North Carolina, USA, for his valuable suggestions, critical reading, and editing of the manuscript. They acknowledge the Animal Resources Center (ARC) for providing mice and animal house facility. They also thank and Mrs Josily Joy and Mrs Amna Najem for their technical assistance.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by Kuwait University (grant no: MA01/14) and OMICS Research Unit Project No: SRUL02/13.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
EMAB conceived, designed the experiment and wrote the manuscript, AA executed the experiment, EMAB and MSR analyzed the data. MSR did the statistical analysis, artworks and reviewed the manuscript.
Significance statement
This study indicates that both IFN-γ and IL-1β induce astrogliosis, microgliosis, enhance the secretion of BDNF, and promote the survival of cortical neurons in stab wound brain injury. Hence, the study supports the strategies for planning of future therapies to promote neuronal regeneration in brain injury using these molecules.
